ASTM E2526-22
(Test Method)Standard Test Method for Evaluation of Cytotoxicity of Nanoparticulate Materials in Porcine Kidney Cells and Human Hepatocarcinoma Cells
Standard Test Method for Evaluation of Cytotoxicity of Nanoparticulate Materials in Porcine Kidney Cells and Human Hepatocarcinoma Cells
SIGNIFICANCE AND USE
5.1 Assessing the propensity of a nanomaterial to cause cytotoxicity to the cells of a target organ can assist in preclinical development.
5.2 The standard historical cytotoxicity testing of materials and extracts of materials has used fibroblasts and is well documented in Practice F813, Test Method F895, and ISO 10993-5. The use of macrophages and micron size particles has also provided information on cytotoxicity and stimulation using Practice F1903.
5.3 This test method adds to the cytotoxicity test protocols by using target organ cells. Two quantitative assays measuring LDH leakage and MTT reduction are used to estimate cytotoxicity.
5.4 This test method may not be predictive of events occurring in all types of nanomaterial applications, and the user is cautioned to consider the appropriateness of the test for various types of nanomaterial and their applications. This procedure should only be used to compare the cytoxicity of a series of related nanomaterials. Meaningful comparison of unrelated nanomaterials is not possible without additional characterization of physicochemical properties of each individual nanomaterial in the assay matrix.
SCOPE
1.1 This test method provides a methodology to assess the cytotoxicity of suspensions of nanoparticulate materials in porcine proximal tubule cells (LLC-PK1) and human hepatocarcinoma cells (Hep G2), which represent potential target organs following systemic administration.
1.2 This test method is part of an in vitro preclinical characterization cascade.
1.3 This test method consists of a protocol utilizing two methods for estimation of cytotoxicity, 3-(4,5-dimethylthiazolyl-2)-2,5-diphenyltetrazolium bromide (MTT) reduction and lactate dehydrogenase (LDH) release.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Jun-2022
- Technical Committee
- E56 - Nanotechnology
- Drafting Committee
- E56.03 - Environment, Health, and Safety
Relations
- Effective Date
- 01-Oct-2018
- Effective Date
- 01-Oct-2016
- Refers
ASTM F895-11(2016) - Standard Test Method for Agar Diffusion Cell Culture Screening for Cytotoxicity - Effective Date
- 01-Apr-2016
- Effective Date
- 01-Oct-2012
- Refers
ASTM F895-11 - Standard Test Method for Agar Diffusion Cell Culture Screening for Cytotoxicity - Effective Date
- 01-Oct-2011
- Refers
ASTM F1903-10 - Standard Practice for Testing For Biological Responses to Particles <i>In Vitro</i> - Effective Date
- 01-Jun-2010
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Feb-2007
- Refers
ASTM F895-84(2006) - Standard Test Method for Agar Diffusion Cell Culture Screening for Cytotoxicity - Effective Date
- 01-Mar-2006
- Effective Date
- 01-Nov-2005
- Effective Date
- 01-Nov-2005
- Refers
ASTM F1903-98(2003) - Standard Practice for Testing for Biological Responses to Particles In Vitro - Effective Date
- 01-Nov-2003
- Effective Date
- 10-Apr-2003
- Effective Date
- 10-Oct-2001
- Effective Date
- 10-Oct-2001
Overview
ASTM E2526-22 is the internationally recognized standard test method for evaluating the cytotoxicity of nanoparticulate materials in porcine kidney cells (LLC-PK1) and human hepatocarcinoma cells (Hep G2). Developed by ASTM Committee E56 on Nanotechnology, this standard provides a reproducible laboratory protocol for assessing the cytotoxic potential of nanomaterials using in vitro cell cultures. Its purpose is to support preclinical development by establishing whether nanomaterials could pose a risk to specific human organs after systemic exposure.
This test method addresses a critical need in nanotechnology and toxicology, offering guidance for comparing the safety profiles of related nanoparticulate materials. The method employs two quantitative assays - MTT reduction and lactate dehydrogenase (LDH) leakage - for reliable estimation of cytotoxicity.
Key Topics
- Cytotoxicity of Nanoparticles: Offers a standardized assay to determine the potential toxic effects of nanoparticles on kidney and liver cells.
- In Vitro Models: Utilizes porcine proximal tubule (LLC-PK1) and human hepatocarcinoma (Hep G2) cell lines to represent key target organs.
- Quantitative Testing: Measures cell membrane integrity (LDH assay) and cell viability (MTT assay) to quantitatively assess cytotoxicity.
- Comparative Analysis: Designed for use in direct comparison of a series of related nanomaterials, strengthening the preclinical evaluation process.
- Standardized Protocol: Emphasizes aseptic techniques, appropriate controls, and procedural consistency for reproducible results across laboratories.
Applications
- Preclinical Safety Assessment: The standard is widely used by researchers and regulatory bodies for evaluating the cytotoxicity of candidate nanomaterials in the early stages of biomedical product development.
- Medical Devices and Drug Delivery: Applicable to the assessment of nanomaterials intended for use in medical device coatings, drug delivery vehicles, and other biomedical applications where organ-specific toxicity is a concern.
- Toxicology Research: Enables toxicologists to investigate nanoparticle interactions with biological systems, aiding in the development of safer nanotechnologies.
- Comparative Research: Supports research and development activities where various nanomaterial formulations need to be benchmarked against each other for cytotoxic effects on target organs.
- Compliance: Provides a protocol aligned with international safety and regulatory expectations, facilitating compliance for organizations seeking global acceptance of their nanomaterial products.
Related Standards
- ASTM F813 - Direct Contact Cell Culture Evaluation of Materials for Medical Devices
- ASTM F895 - Agar Diffusion Cell Culture Screening for Cytotoxicity
- ASTM F1903 - Testing for Cellular Responses to Particles in vitro
- ISO 10993-5 - Biological Evaluation of Medical Devices: Tests for in vitro Cytotoxicity
Practical Value
ASTM E2526-22 offers clear, step-by-step methodologies for laboratories, ensuring that cytotoxicity evaluations are consistent and scientifically valid. By focusing on biologically relevant organ cell models, this standard delivers meaningful insights vital for nanomaterial safety assessment. While the method is intended for comparative analysis within related groups of nanomaterials, it underscores the need for additional characterization when dealing with unrelated materials.
Keywords: ASTM E2526-22, cytotoxicity testing, nanoparticulate materials, in vitro, porcine kidney cells, human hepatocarcinoma cells, LDH assay, MTT assay, nanomaterial safety, toxicology standards, biomedical safety, nanotechnology standards.
Buy Documents
ASTM E2526-22 - Standard Test Method for Evaluation of Cytotoxicity of Nanoparticulate Materials in Porcine Kidney Cells and Human Hepatocarcinoma Cells
Frequently Asked Questions
ASTM E2526-22 is a standard published by ASTM International. Its full title is "Standard Test Method for Evaluation of Cytotoxicity of Nanoparticulate Materials in Porcine Kidney Cells and Human Hepatocarcinoma Cells". This standard covers: SIGNIFICANCE AND USE 5.1 Assessing the propensity of a nanomaterial to cause cytotoxicity to the cells of a target organ can assist in preclinical development. 5.2 The standard historical cytotoxicity testing of materials and extracts of materials has used fibroblasts and is well documented in Practice F813, Test Method F895, and ISO 10993-5. The use of macrophages and micron size particles has also provided information on cytotoxicity and stimulation using Practice F1903. 5.3 This test method adds to the cytotoxicity test protocols by using target organ cells. Two quantitative assays measuring LDH leakage and MTT reduction are used to estimate cytotoxicity. 5.4 This test method may not be predictive of events occurring in all types of nanomaterial applications, and the user is cautioned to consider the appropriateness of the test for various types of nanomaterial and their applications. This procedure should only be used to compare the cytoxicity of a series of related nanomaterials. Meaningful comparison of unrelated nanomaterials is not possible without additional characterization of physicochemical properties of each individual nanomaterial in the assay matrix. SCOPE 1.1 This test method provides a methodology to assess the cytotoxicity of suspensions of nanoparticulate materials in porcine proximal tubule cells (LLC-PK1) and human hepatocarcinoma cells (Hep G2), which represent potential target organs following systemic administration. 1.2 This test method is part of an in vitro preclinical characterization cascade. 1.3 This test method consists of a protocol utilizing two methods for estimation of cytotoxicity, 3-(4,5-dimethylthiazolyl-2)-2,5-diphenyltetrazolium bromide (MTT) reduction and lactate dehydrogenase (LDH) release. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Assessing the propensity of a nanomaterial to cause cytotoxicity to the cells of a target organ can assist in preclinical development. 5.2 The standard historical cytotoxicity testing of materials and extracts of materials has used fibroblasts and is well documented in Practice F813, Test Method F895, and ISO 10993-5. The use of macrophages and micron size particles has also provided information on cytotoxicity and stimulation using Practice F1903. 5.3 This test method adds to the cytotoxicity test protocols by using target organ cells. Two quantitative assays measuring LDH leakage and MTT reduction are used to estimate cytotoxicity. 5.4 This test method may not be predictive of events occurring in all types of nanomaterial applications, and the user is cautioned to consider the appropriateness of the test for various types of nanomaterial and their applications. This procedure should only be used to compare the cytoxicity of a series of related nanomaterials. Meaningful comparison of unrelated nanomaterials is not possible without additional characterization of physicochemical properties of each individual nanomaterial in the assay matrix. SCOPE 1.1 This test method provides a methodology to assess the cytotoxicity of suspensions of nanoparticulate materials in porcine proximal tubule cells (LLC-PK1) and human hepatocarcinoma cells (Hep G2), which represent potential target organs following systemic administration. 1.2 This test method is part of an in vitro preclinical characterization cascade. 1.3 This test method consists of a protocol utilizing two methods for estimation of cytotoxicity, 3-(4,5-dimethylthiazolyl-2)-2,5-diphenyltetrazolium bromide (MTT) reduction and lactate dehydrogenase (LDH) release. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2526-22 is classified under the following ICS (International Classification for Standards) categories: 07.100.10 - Medical microbiology. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2526-22 has the following relationships with other standards: It is inter standard links to ASTM F1903-18, ASTM F1877-16, ASTM F895-11(2016), ASTM F813-07(2012), ASTM F895-11, ASTM F1903-10, ASTM F1877-05(2010), ASTM F813-07, ASTM F895-84(2006), ASTM F1877-05, ASTM F1877-05e1, ASTM F1903-98(2003), ASTM F1877-98(2003)e1, ASTM F813-83(1996)e1, ASTM F813-01. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2526-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2526 − 22
Standard Test Method for
Evaluation of Cytotoxicity of Nanoparticulate Materials in
Porcine Kidney Cells and Human Hepatocarcinoma Cells
This standard is issued under the fixed designation E2526; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope F895TestMethodforAgarDiffusionCellCultureScreening
for Cytotoxicity
1.1 This test method provides a methodology to assess the
F1877Practice for Characterization of Particles
cytotoxicity of suspensions of nanoparticulate materials in
F1903Practice for Testing for Cellular Responses to Par-
porcine proximal tubule cells (LLC-PK1) and human hepato-
ticles in vitro
carcinoma cells (Hep G2), which represent potential target
organs following systemic administration. 2.2 ISO Standard:
ISO10993-5BiologicalEvaluationofMedicalDevices:Part
1.2 This test method is part of an in vitro preclinical
5 Tests for in vitro Cytotoxicity
characterization cascade.
1.3 This test method consists of a protocol utilizing two
3. Terminology
methods for estimation of cytotoxicity, 3-(4,5-
3.1 Abbreviations:
dimethylthiazolyl-2)-2,5-diphenyltetrazolium bromide (MTT)
3.1.1 APAP—acetaminophen- positive control
reduction and lactate dehydrogenase (LDH) release.
3.1.2 DMSO—dimethyl sulfoxide
1.4 The values stated in SI units are to be regarded as
3.1.3 DMEM—Dulbelcco’s modified eagles media
standard. No other units of measurement are included in this
standard.
3.1.4 FBS—fetal bovine serum
1.5 This standard does not purport to address all of the
3.1.5 Hep G2—human hepatocarcinoma cells
safety concerns, if any, associated with its use. It is the
3.1.6 LDH—lactic dehydrogenase
responsibility of the user of this standard to establish appro-
3.1.7 LLC-PK1—porcine proximal tubule cells
priate safety, health, and environmental practices and deter-
3.1.8 LPS—lipopolysacchride, bacterial endotoxin
mine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accor-
3.1.9 MTT—3-(4,5-dimethylthiazolyl-2)-2,5-diphenyltetra-
dance with internationally recognized principles on standard-
zolium bromide
ization established in the Decision on Principles for the
3.1.10 PBS—phosphate buffered saline
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
4. Summary of Test Method
Barriers to Trade (TBT) Committee.
4.1 Nanoparticulate test materials in suspension in cell
culture media and appropriate controls are added to cell
2. Referenced Documents
cultures. The release of LDH indicates membrane damage and
2.1 ASTM Standards:
thediminutionofMTTreductionindicateslossofcellviability.
F813Practice for Direct Contact Cell Culture Evaluation of
These are quantitative indicators of cytotoxicity. Aseptic pro-
Materials for Medical Devices
cedures are required.
5. Significance and Use
This test method is under the jurisdiction of ASTM Committee E56 on
Nanotechnology and is the direct responsibility of Subcommittee E56.03 on
5.1 Assessing the propensity of a nanomaterial to cause
Environment, Health, and Safety.
cytotoxicity to the cells of a target organ can assist in
CurrenteditionapprovedJuly1,2022.PublishedJuly2022.Originallyapproved
preclinical development.
in 2008. Last previous edition approved in 2013 as E2526 – 08(2013), which was
withdrawn in April 2022 and reinstated in July 2022. DOI: 10.1520/E2526-22.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
the ASTM website. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2526 − 22
2+ 2+
5.2 The standard historical cytotoxicity testing of materials 6.2.13 Sterile Ca /Mg -free phosphate buffered saline
and extracts of materials has used fibroblasts and is well (PBS).
documented in Practice F813, Test Method F895, and ISO 6.2.14 Distilled or deionized water.
10993-5.Theuseofmacrophagesandmicronsizeparticleshas
6.3 Cell Lines:
also provided information on cytotoxicity and stimulation
6.3.1 LLC-PK1(porcineproximaltubulecell)(ATCCprod-
using Practice F1903.
uct #CL-101).
5.3 This test method adds to the cytotoxicity test protocols 6.3.2 Hep G2 (human hepatocarcinoma)(ATCC product #
by using target organ cells.Two quantitative assays measuring HB-8065).
LDH leakage and MTT reduction are used to estimate cyto-
6.4 Equipment:
toxicity.
6.4.1 Plate reader capable of reading 96-well plates.
5.4 This test method may not be predictive of events 6.4.2 Plate centrifuge set at 700 g to 800 g. See Note 2.
occurringinalltypesofnanomaterialapplications,andtheuser
NOTE2—Therelationshipbetweencentrifugationspeedandtherelative
is cautioned to consider the appropriateness of the test for
centrifugal force (RCF, measured in multiples of g, the force of gravity at
various types of nanomaterial and their applications. This
the Earth’s surface) depends on the radius (r) of the rotor, where RCF =
(rpm/1000)2 × 1.12 × r (mm).
procedure should only be used to compare the cytoxicity of a
series of related nanomaterials. Meaningful comparison of
6.4.3 Cell culture microscope.
unrelated nanomaterials is not possible without additional
6.4.4 Vortex mixer.
characterization of physicochemical properties of each indi-
6.4.5 Hemacytometer or cell counter.
vidual nanomaterial in the assay matrix.
6.4.6 Orbital plate shaker.
6. Reagents and Materials
7. Experimental Procedure
6.1 Purity of Reagents—Reagent grade chemicals shall be
7.1 Aseptic precautions are required.
used in all tests. Unless otherwise indicated, it is intended that
7.2 Positive Control Preparation:
all reagents conform to the specifications of the Committee on
7.2.1 LLC-PK1 Acetaminophen (APAP) positive control:
Analytical Reagents of theAmerican Chemical Society where
25 mmol/LAPAP in M199 cell culture media.
such specifications are available. Other grades may be used,
7.2.2 Hepatocyte Acetaminophen (APAP) positive control:
provided it is first ascertained that the reagent is of sufficiently
20 mmol/LAPAP in RPMI 1640 cell culture media.
high purity to permit its use without lessening the accuracy of
7.2.3 Triton X-100 is diluted to a volume fraction of 1% in
the determination.
cell culture medium. This is the positive control for the LDH
6.2 Reagents and supplies (aseptic procedures are needed
assay.
andcareshouldbetakentousesterilereagentsandsuppliesas
7.3 MTT Assay Reagents:
necessary). See Note 1.
7.3.1 MTT solution, 5mg⁄mLMTT in PBS, store for up to
NOTE 1—Commercial sources are indicated for informational purposes
one month at 4°C in the dark.
only to aid laboratories initiating these test procedures. This does not
7.3.2 Glycine buffer, 0.1mol⁄L glycine (75.07 g/mol), 0.1
indicate endorsement by ASTM. Other equivalent sources may be
available. mol/LNaCl(58.44g/mol),pH10.5,storeatroomtemperature.
6.2.1 MTT (3-(4,5-dimethylthiazolyl-2)-2,5- diphenyltetra-
7.4 Biovision LDH-Cytotoxicity Assay Kit Reagents:
zolium bromide).
7.4.1 Reconstitute catalyst in 1 mL dH 0 for 10 min and
6.2.2 Acetaminophen.
vortex (stable for 2 weeks at 4°C).
6.2.3 Dimethyl sulfoxide.
7.4.2 Reaction mixture (for one 96-well plate): add 250 mL
6.2.4 Glycine.
of reconstituted, catalyst solution to 11.25 mL of dye solution
6.2.5 Sodium chloride.
(stable for 2 weeks at 4°C).
6.2.6 Medium 199 cell culture media.
7.4.3 For other LDH Cytotoxicity assay kits, follow their
6.2.7 Triton X-100.
instructions.
6.2.8 LDH-CytotoxicityAssay Kit (Biovision Cat. # K311-
7.5 Cell Culture:
400 was used in developing this test method).
7.5.1 LLC-PK1 Cell Preparation:
6.2.9 96-well flat bottom cell culture plates.
7.5.1.1 Harvest cells from flasks prepared from cryopre-
6.2.10 Roswell Park Memorial Institute (RPMI) 1640 me-
served cells according to the instructions from the supplier
dium.
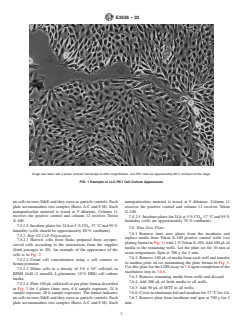
(limit passages to 20). An example of the appearance of the
6.2.11 L-glutamine.
cells is in Fig. 1.
6.2.12 Fetal bovine serum (FBS).
7.5.1.2 Count cell concentration using a cell counter or
hemocytometer.
7.5.1.3 Dilute cells to a density of 2.5 × 10 cells/mol in
Reagent Chemicals, American Chemical Society Specifications, American
Chemical Society, Washington, DC. For Suggestions on the testing of reagents not
M199 (3% FBS) cell culture media.
listed by the American Chemical Society, see Analar Standards for Laboratory
7.5.1.4 Plate100µLcells/wellasperplateformatdescribed
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
in Fig. 3 for 4 plates (time zero, 6 h sample exposure, 24 h
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
MD. sample exposure, 48 h sample exposure).The format indicates
E2526 − 22
Image was taken with a phase contrast microscope at 225× magnification. LLC-PK1 cells are approximately 80 % confluent at this stage.
FIG. 1 Example of LLC-PK1 Cell Culture Appearance
no cells in rows D&E and they serve as particle controls. Each nanoparticulate material is tested at 9 dilutions. Column 11
plate accommodates two samples (Rows A-C and F-H). Each receives the positive control and column 12 receives Triton
nanoparticulate material is tested at 9 dilutions. Column 11 X-100.
receives the positive control and column 12 receives Triton
7.5.2.5 Incubateplatesfor24hat5%CO ,37°Cand95%
X-100.
humidity (cells are approximately 70% confluent
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...