ASTM C1502-16
(Test Method)Standard Test Method for Determination of Total Chlorine and Fluorine in Uranium Dioxide and Gadolinium Oxide
Standard Test Method for Determination of Total Chlorine and Fluorine in Uranium Dioxide and Gadolinium Oxide
SIGNIFICANCE AND USE
5.1 The method is designed to show whether or not the tested materials meet the specifications as given in either Specification C753, C776, C888 or C922.
SCOPE
1.1 This test method covers the determination of chlorine and fluorine in nuclear-grade uranium dioxide (UO2) powder and pellets, nuclear grade gadolinium oxide (Gd2O3 ) powder and gadolinium oxide-uranium oxide (Gd2O3-UO2) powder and pellets.
1.2 With a 2 gram UO2 sample size the detection limit of the method is 4 µg/g for chlorine and 2 µg/g for fluorine. The maximum concentration determined with a 2 gram sample is 500 µg/g for both chlorine and fluorine. The sample size used in this test method can vary from 1 to 10 grams resulting in a corresponding change in the detection limits and range.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 14-Jan-2016
- Technical Committee
- C26 - Nuclear Fuel Cycle
- Drafting Committee
- C26.05 - Methods of Test
Relations
- Effective Date
- 15-Jan-2016
- Effective Date
- 01-Jan-2024
- Effective Date
- 01-Jun-2018
- Effective Date
- 01-Feb-2016
- Effective Date
- 15-Jun-2014
- Effective Date
- 15-Jan-2014
- Effective Date
- 01-Jan-2014
- Effective Date
- 01-Jun-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Jun-2011
- Effective Date
- 01-Nov-2010
- Effective Date
- 01-Aug-2010
- Effective Date
- 01-Feb-2010
- Effective Date
- 15-Feb-2009
- Effective Date
- 01-Dec-2008
Overview
ASTM C1502-16 is the internationally recognized standard test method for the determination of total chlorine and fluorine in nuclear-grade uranium dioxide (UO2) powder and pellets, gadolinium oxide (Gd2O3) powder, and gadolinium oxide-uranium oxide (Gd2O3-UO2) powder and pellets. Published by ASTM International, this standard is vital for quality control in the nuclear fuel cycle, ensuring that critical materials meet required specifications for halide content. Adherence to this test method helps prevent contamination and maintains the integrity of nuclear-grade materials as defined by related ASTM specifications, such as C753, C776, C888, and C922.
Key Topics
- Purpose: The method confirms whether uranium dioxide, gadolinium oxide, and their mixtures comply with strict nuclear-grade requirements for chlorine and fluorine content.
- Detection Limits: With a 2 g UO2 sample, the method can detect as little as 4 µg/g of chlorine and 2 µg/g of fluorine, up to a maximum of 500 µg/g; detection limits and range vary with sample size.
- Methodology: Halogens are separated from the material by pyrohydrolysis at 900°C to 1000°C. Chloride and fluoride are volatilized as acids, absorbed, and then measured using ion-selective electrodes.
- Precision and Bias: The standard includes procedures for calculating results and assessing the method’s precision, with typical use involving blanks, spikes, and controls, as per laboratory quality assurance plans.
- Units: All measurements are provided in SI units, ensuring consistency in international applications.
- Safety: The document emphasizes the user’s responsibility for safety and regulatory compliance.
Applications
This standard plays a critical role in:
- Nuclear Fuel Quality Control: Ensuring nuclear-grade uranium dioxide and gadolinium oxide materials do not exceed acceptable levels of chlorine and fluorine, which is essential to minimize corrosion and material degradation in nuclear reactors.
- Materials Compliance: Verifying conformance with ASTM C753, C776, C888, and C922 specifications as part of the procurement and acceptance process for nuclear materials.
- Analytical Laboratories: Routine analysis of nuclear material batches to confirm purity and adherence to international standards.
- Regulatory and Auditing Requirements: Supporting documentation for regulatory authorities and audits related to nuclear fuel manufacturing and processing.
- Research and Development: Facilitating material development and process optimization in advanced nuclear technology sectors.
Related Standards
For comprehensive quality control and material specification in nuclear applications, reference the following ASTM standards:
- ASTM C753: Specification for Nuclear-Grade, Sinterable Uranium Dioxide Powder
- ASTM C776: Specification for Sintered Uranium Dioxide Pellets
- ASTM C888: Specification for Nuclear-Grade Gadolinium Oxide (Gd₂O₃) Powder
- ASTM C922: Specification for Sintered Gadolinium Oxide-Uranium Dioxide Pellets
- ASTM D1193: Specification for Reagent Water (relevant to analytical procedures)
Keywords: ASTM C1502, chlorine determination, fluorine analysis, uranium dioxide, gadolinium oxide, nuclear-grade materials, pyrohydrolysis, ion-selective electrodes, nuclear fuel quality, international standard, ASTM nuclear standards.
Buy Documents
ASTM C1502-16 - Standard Test Method for Determination of Total Chlorine and Fluorine in Uranium Dioxide and Gadolinium Oxide
REDLINE ASTM C1502-16 - Standard Test Method for Determination of Total Chlorine and Fluorine in Uranium Dioxide and Gadolinium Oxide
Get Certified
Connect with accredited certification bodies for this standard

DNV
DNV is an independent assurance and risk management provider.

Lloyd's Register
Lloyd's Register is a global professional services organisation specialising in engineering and technology.

DNV Energy Systems
Energy and renewable energy certification.
Sponsored listings
Frequently Asked Questions
ASTM C1502-16 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Total Chlorine and Fluorine in Uranium Dioxide and Gadolinium Oxide". This standard covers: SIGNIFICANCE AND USE 5.1 The method is designed to show whether or not the tested materials meet the specifications as given in either Specification C753, C776, C888 or C922. SCOPE 1.1 This test method covers the determination of chlorine and fluorine in nuclear-grade uranium dioxide (UO2) powder and pellets, nuclear grade gadolinium oxide (Gd2O3 ) powder and gadolinium oxide-uranium oxide (Gd2O3-UO2) powder and pellets. 1.2 With a 2 gram UO2 sample size the detection limit of the method is 4 µg/g for chlorine and 2 µg/g for fluorine. The maximum concentration determined with a 2 gram sample is 500 µg/g for both chlorine and fluorine. The sample size used in this test method can vary from 1 to 10 grams resulting in a corresponding change in the detection limits and range. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 5.1 The method is designed to show whether or not the tested materials meet the specifications as given in either Specification C753, C776, C888 or C922. SCOPE 1.1 This test method covers the determination of chlorine and fluorine in nuclear-grade uranium dioxide (UO2) powder and pellets, nuclear grade gadolinium oxide (Gd2O3 ) powder and gadolinium oxide-uranium oxide (Gd2O3-UO2) powder and pellets. 1.2 With a 2 gram UO2 sample size the detection limit of the method is 4 µg/g for chlorine and 2 µg/g for fluorine. The maximum concentration determined with a 2 gram sample is 500 µg/g for both chlorine and fluorine. The sample size used in this test method can vary from 1 to 10 grams resulting in a corresponding change in the detection limits and range. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM C1502-16 is classified under the following ICS (International Classification for Standards) categories: 27.120.30 - Fissile materials and nuclear fuel technology. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM C1502-16 has the following relationships with other standards: It is inter standard links to ASTM C1502-09, ASTM C859-24, ASTM C888-18, ASTM C753-16, ASTM C859-14a, ASTM C859-14, ASTM C888-03(2014), ASTM C859-13a, ASTM C859-13, ASTM C776-06(2011), ASTM C859-10b, ASTM C859-10a, ASTM C859-10, ASTM C859-09, ASTM C888-03(2008). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM C1502-16 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: C1502 − 16
Standard Test Method for
Determination of Total Chlorine and Fluorine in Uranium
Dioxide and Gadolinium Oxide
This standard is issued under the fixed designation C1502; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 3. Terminology
1.1 This test method covers the determination of chlorine 3.1 Definitions—Except as otherwise defined herein, defini-
and fluorine in nuclear-grade uranium dioxide (UO ) powder tions of terms are given in Terminology C859.
and pellets, nuclear grade gadolinium oxide (Gd O ) powder
2 3
3.2 Definitions of Terms Specific to This Standard:
and gadolinium oxide-uranium oxide (Gd O -UO ) powder
2 3 2
3.2.1 accelerator—a chemical compound or a flux that will
and pellets.
decrease the reaction time or prohydrolysis time.
1.2 Witha2gramUO samplesizethedetectionlimitofthe
4. Summary of Test Method
method is 4 µg/g for chlorine and 2 µg/g for fluorine. The
maximum concentration determined with a 2 gram sample is
4.1 The halogens are separated from the test materials by
500 µg/g for both chlorine and fluorine. The sample size used
pyrohydrolysis in a quartz reaction tube with a stream of wet
in this test method can vary from 1 to 10 grams resulting in a
oxygenorairatatemperatureof900to1000°C (1-4).Chloride
corresponding change in the detection limits and range.
and fluoride are volatilized simultaneously as acids, absorbed
inanabsorptionsolutionaschlorideandfluorideandmeasured
1.3 The values stated in SI units are to be regarded as
with ion selective electrodes (4-6).
standard. No other units of measurement are included in this
standard.
5. Significance and Use
1.4 This standard does not purport to address all of the
5.1 The method is designed to show whether or not the
safety concerns, if any, associated with its use. It is the
tested materials meet the specifications as given in either
responsibility of the user of this standard to establish appro-
Specification C753, C776, C888 or C922.
priate safety and health practices and determine the applica-
bility of regulatory limitations prior to use.
6. Interferences
2. Referenced Documents 6.1 The absorption solution controls the pH of the measured
solutiontoavoidhydroxideioninterferenceortheformationof
2.1 ASTM Standards:
hydrogen complexes with fluoride.
C753 Specification for Nuclear-Grade, Sinterable Uranium
Dioxide Powder
6.2 Bromide, iodide, cyanide and sulfide, if present in the
C776 Specification for Sintered Uranium Dioxide Pellets condensate, interfere in the measurement of chloride with
C859 Terminology Relating to Nuclear Materials
ion-selective electrodes, but have very little effect upon the
C888 Specification for Nuclear-Grade Gadolinium Oxide measurement of fluoride with ion-selective electrodes.
(Gd O ) Powder
2 3
6.3 As the ionic activity of the chloride and fluoride ions is
C922 Specification for Sintered Gadolinium Oxide-Uranium
temperature dependent, the standard solutions and sample
Dioxide Pellets
solutions should be measured at the same temperature.
D1193 Specification for Reagent Water
7. Apparatus
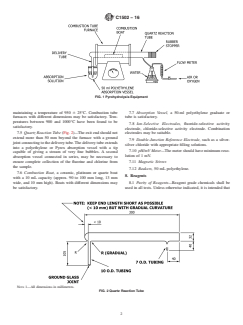
This test method is under the jurisdiction ofASTM Committee C26 on Nuclear 7.1 Pyrohydrolysis Equipment, the assembly of suitable
Fuel Cycle and is the direct responsibility of Subcommittee C26.05 on Methods of
equipment is shown in Fig. 1.
Test.
Current edition approved Jan. 15, 2016. Published February 2016. Originally 7.2 Gas Flow Regulator and Flowmeter.
approved in 2001. Last previous edition approved in 2009 as C1502 – 09. DOI:
7.3 Hot Plate, used to warm the water saturating the sparge
10.1520/C1502-16.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or gas to 50 to 80°C.
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
7.4 Combustion Tube Furnace, having a bore of about 32
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. mm with a length of about 300 mm and the capability of
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C1502 − 16
FIG. 1 Pyrohydrolysis Equipment
maintaining a temperature of 950 6 25°C. Combustion tube 7.7 Absorption Vessel, a 50-ml polyethylene graduate or
furnaces with different dimensions may be satisfactory. Tem- tube is satisfactory.
peratures between 900 and 1000°C have been found to be
7.8 Ion-Selective Electrodes, fluoride-selective activity
satisfactory.
electrode, chloride-selective activity electrode. Combination
7.5 Quartz Reaction Tube (Fig. 2)—The exit end should not electrodes may be suitable.
extend more than 50 mm beyond the furnace with a ground
7.9 Double-Junction Reference Electrode, such as a silver-
jointconnectingtothedeliverytube.Thedeliverytubeextends
silver chloride with appropriate filling solutions.
into a polyethylene or Pyrex absorption vessel with a tip
7.10 pH/mV Meter—The meter should have minimum reso-
capable of giving a stream of very fine bubbles. A second
lution of 1 mV.
absorption vessel connected in series, may be necessary to
ensure complete collection of the fluorine and chlorine from 7.11 Magnetic Stirrer.
the sample.
7.12 Beakers, 50 mL polyethylene.
7.6 Combustion Boat, a ceramic, platinum or quartz boat
8. Reagents
with a 10 mL capacity (approx. 90 to 100 mm long, 13 mm
wide, and 10 mm high). Boats with different dimensions may
8.1 Purity of Reagents—Reagent grade chemicals shall be
be satisfactory. used in all tests. Unless otherwise indicated, it is intended that
NOTE 1—All dimensions in millimetres.
FIG. 2 Quartz Reaction Tube
C1502 − 16
all reagents conform to the specifications of the Committee on 9.3 Run a pyrohydrolysis blank using a halogen-free ura-
Analytical Reagents of theAmerican Chemical Society, where nium oxide or gadolinium oxide according to the procedure in
such specifications are available. Other grades may be used, 9.5.
provided it is first ascertained that the reagent is of sufficiently 9.3.1 Alternatively an empty combustion boat can be used
high purity to permit its use without lessening the accuracy of
for the pyrohydrolysis blank.
the determination.
9.3.2 A blank run should be made each day and after any
sample that contains abnormally high levels of chlorine or
8.2 Accelerator—Two accelerators have been investigated
fluorine.
for this system, halogen free U O and a flux of sodium
3 8
tungstate and tungsten trioxide (1, 2). Halogen free U O
3 8 9.4 Run samples, controls, duplicates and spikes in accor-
requires no special preparation before use but will require a
dance with the user’s quality assurance control plan and
longer pyrohydrolysis period. The flux of sodium tungstate
requirements.
(Na WO ) with tungsten trioxide (WO ) may reduce the
2 4 3
9.5 Sample Pyrohydrolysis:
pyrohydrolysis period by half but it requires the following
9.5.1 Pellets should be crushed prior to analysis.
special preparation. Dehydrate 165 g of Na WO in a large
2 4
9.5.2 Weigh 1 to 10 g of sample and spread in the combus-
platinum dish. Transfer the dried material to a mortar, add 116
tion boat. If an accelerator is desired, mix4gofU O
3 8
gof WO , and grind the mixture to ensure good mixing.
accelerator or8gofthe tungstate flux with the sample before
Transfer the mixture into a platinum dish and heat with a
spreading in the boat. A flux to sample ratio of 1 has been
burner for 2 h. Cool the melt, transfer the flux to a mortar and
found to work satisfactorily. Other ratios may be applicable as
grind to a coarse powder. Store the flux in an airtight bottle.
determined by the analyst.
Mix about8gofflux with each portion of sample to be
9.5.3 Place15mLofabsorptionsolutioninthepolyethylene
pyrohydrolyzed.
absorption vessel and submerge the delivery tip in the solution.
8.3 Absorption Solution (0.1 M)—Dissolve 10 g, potassium
9.5.4 Remove the stopper from the entrance of the quartz
acetate (KC H O ) in water, add 5 mL of acetic acid
2 3 2
reaction tube and insert the boat into the hot area of the
(CH CO H, sp gr 1.05), and dilute to 1 L. Other absorption
3 2
furnace. Quickly stopper the quartz reaction tube.
solutions may be satisfactory. It will be necessary to validate
9.5.5 Check the gas flow and adjust to 1
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: C1502 − 09 C1502 − 16
Standard Test Method for
Determination of Total Chlorine and Fluorine in Uranium
Dioxide and Gadolinium Oxide
This standard is issued under the fixed designation C1502; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method covers the determination of chlorine and fluorine in nuclear-grade uranium dioxide (UO ) powder and
pellets, nuclear grade gadolinium oxide (Gd O ) powder and gadolinium oxide-uranium oxide (Gd O -UO ) powder and pellets.
2 3 2 3 2
1.2 With a 2 gram UO sample size the detection limit of the method is 4 μg/g for chlorine and 2 μg/g for fluorine. The maximum
concentration determined with a 2 gram sample is 500 μg/g for both chlorine and fluorine. The sample size used in this test method
can vary from 1 to 10 grams resulting in a corresponding change in the detection limits and range.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
2. Referenced Documents
2.1 ASTM Standards:
C753 Specification for Nuclear-Grade, Sinterable Uranium Dioxide Powder
C776 Specification for Sintered Uranium Dioxide Pellets
C859 Terminology Relating to Nuclear Materials
C888 Specification for Nuclear-Grade Gadolinium Oxide (Gd O ) Powder
2 3
C922 Specification for Sintered Gadolinium Oxide-Uranium Dioxide Pellets
D1193 Specification for Reagent Water
3. Terminology
3.1 Definitions—Except as otherwise defined herein, definitions of terms are given in Terminology C859.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 accelerator—a chemical compound or a flux that will decrease the reaction time or prohydrolysis time.
4. Summary of Test Method
4.1 The halogens are separated from the test materials by pyrohydrolysis in a quartz reaction tube with a stream of wet oxygen
or air at a temperature of 900 to 1000°C.1000°C (1-4)). Chloride and fluoride are volatilized simultaneously as acids, absorbed in
a bufferan absorption solution as chloride and fluoride and measured with ion selective electrodes (4-6).
5. Significance and Use
5.1 The method is designed to show whether or not the tested materials meet the specifications as given in either Specification
C753, C776, C888 or C922.
6. Interferences
6.1 The buffer absorption solution controls the pH of the measured solution to avoid hydroxide ion interference or the formation
of hydrogen complexes with fluoride.
This test method is under the jurisdiction of ASTM Committee C26 on Nuclear Fuel Cycle and is the direct responsibility of Subcommittee C26.05 on Methods of Test.
Current edition approved June 1, 2009Jan. 15, 2016. Published July 2009February 2016. Originally approved in 2001. Last previous edition approved in 20012009 as
C1502 – 01.C1502 – 09. DOI: 10.1520/C1502-09.10.1520/C1502-16.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C1502 − 16
6.2 Bromide, iodide, cyanide and sulfide, if present in the condensate, interfere in the measurement of chloride with
ion-selective electrodes, but have very little effect upon the measurement of fluoride with ion-selective electrodes.
6.3 As the ionic activity of the chloride and fluoride ions is temperature dependent, the standard solutions and sample solutions
should be measured at the same temperature.
7. Apparatus
7.1 Pyrohydrolysis Equipment, the assembly of suitable equipment is shown in Fig. 1.
7.2 Gas Flow Regulator and Flowmeter.
7.3 Hot Plate, used to warm the water saturating the sparge gas to 50–80°C.50 to 80°C.
7.4 Combustion Tube Furnace, having a bore of about 32 mm with a length of about 300 mm and the capability of maintaining
a temperature of 950 6 25°C. Combustion tube furnaces with different dimensions may be satisfactory. Temperatures between 900
and 1000°C have been found to be satisfactory.
7.5 Quartz Reaction Tube (Fig. 2)—The exit end should not extend more than 50 mm beyond the furnace with a ground joint
connecting to the delivery tube. The delivery tube extends into a polyethylene or Pyrex absorption vessel with a tip capable of
giving a stream of very fine bubbles. A second absorption vessel connected in series, may be necessary to ensure complete
collection of the fluorine and chlorine from the sample.
7.6 Combustion Boat, a ceramic, platinum or quartz boat with a 10 mL capacity (approx. 90–100 90 to 100 mm long, 13 mm
wide, and 10 mm high). Boats with different dimensions may be satisfactory.
7.7 Absorption Vessel, a 50-ml polyethylene graduate or tube is satisfactory.
7.8 Ion-Selective Electrodes, fluoride-selective activity electrode,, chloride-selective activity electrodeelectrode. Combination
electrodes may be suitable.
7.9 Double-Junction Reference Electrode, ,such as a silver-silver chloride with appropriate filling solutions.
7.10 pH/mV Meter—The meter should have minimum resolution of 1 mV.
7.11 Magnetic Stirrer.
7.12 Beakers, 50 mL polyethylene.
8. Reagents
8.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where such
specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high purity
to permit its use without lessening the accuracy of the determination.
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by
the American Chemical Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
FIG. 1 Pyrohydrolysis Equipment
C1502 − 16
NOTE 1—All dimensions in millimetres.
FIG. 2 Quartz Reaction Tube
8.2 Accelerator—Two accelerators have been investigated for this system, halogen free U O and a flux of sodium tungstate and
3 8
tungsten trioxide.trioxide (1, 2)). Halogen free U O requires no special preparation before use but will require a longer
3 8
pyrohydrolysis period. The flux of sodium tungstate (Na WO ) with tungsten trioxide (WO ) may reduce the pyrohydrolysis period
2 4 3
by half but it requires the following special preparation. Dehydrate 165 g of Na WO in a large platinum dish. Transfer the dried
2 4
material to a mortar, add 116 g of WO , and grind the mixture to ensure good mixing. Transfer the mixture into a platinum dish
and heat with a burner for 2 h. Cool the melt, transfer the flux to a mortar and grind to a coarse powder. Store the flux in an airtight
bottle. Mix about 8 g of flux with each portion of sample to be pyrohydrolyzed.
8.3 BufferAbsorption Solution (0.1 M)—Dissolve 10 g, potassium acetate (KC H O ) in water, add 5 mL of acetic acid
2 3 2
(CH CO H, sp gr 1.05), and dilute to 1 L. Other buffers absorption solutions may be satisfactory. It will be necessary to validate
3 2
the buffers absorption solutions and operating conditions with spike recovery determinations.
8.4 Chloride, Standard Solution (100 μg Cl/mL)—Dissolve 0.165 g of dry sodium chloride (NaCl) in water and dilute to 1 L.
Commercially prepared standard solutions may be used.
8.5 Fluoride, Standard Solution (50 μg F/mL)—Dissolve 0.111 g of dried sodium fluoride (NaF) in water and dilute to 1 L. Store
the solution in a polyethylene bottle. Commercially prepared standard solutions may be used.
8.6 Compressed Oxygen or Air.
8.7 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean reagent water conforming to
Specification D1193, Type I.
9. Procedure
9.1 Adjust the pyrohydrolysis system to operating condition as follows:
9.1.1 Heat the furnace to 950 6 25°C. (See 6.47.4).
9.1.2 Fill the water reservoir and heat to 50 to 80°C.
9.1.3 Adjust the gas flow to 1 to 2 L/min.
9.1.3.1 The furnace temperatu
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...