ASTM F2260-18
(Test Method)Standard Test Method for Determining Degree of Deacetylation in Chitosan Salts by Proton Nuclear Magnetic Resonance (1H NMR) Spectroscopy
Standard Test Method for Determining Degree of Deacetylation in Chitosan Salts by Proton Nuclear Magnetic Resonance (<sup>1</sup>H NMR) Spectroscopy
SIGNIFICANCE AND USE
4.1 The degree of deacetylation of chitosan salts is an important characterization parameter since the charge density of the chitosan molecule is responsible for potential biological and functional effects.
4.2 The degree of deacetylation (% DDA) of water-soluble chitosan salts can be determined by 1H nuclear magnetic resonance spectroscopy (1H NMR). Several workers have reported on the NMR determination of chemical composition and sequential arrangement of monomer units in chitin and chitosan. The test method described is primarily based on the work of Vårum et al. (1991),5 which represents the first publication on routine determination of chemical composition in chitosans by solution state 1H NMR spectroscopy. This test method is applicable for determining the % DDA of chitosan chloride and chitosan glutamate salts. It is a simple, rapid, and suitable method for routine use. Quantitative 1H NMR spectroscopy reports directly on the relative concentration of chemically distinct protons in the sample, consequently, no assumptions, calibration curves or calculations other than determination of relative signal intensity ratios are necessary.
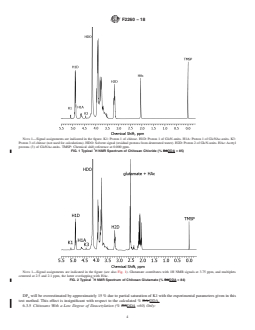
4.3 In order to obtain well-resolved NMR spectra, depolymerization of chitosans to a number average degree of polymerization (DPn) of ~15 to 30 is required. This reduces the viscosity and increases the mobility of the molecules. Although there are several options for depolymerization of chitosans, the most convenient procedure is that of nitrous acid degradation in deuterated water. The reaction is selective, stoichiometric with respect to GlcN, rapid, and easily controlled (Allan & Peyron, 1995).6 The reaction selectively cleaves after a GlcN-residue, transforming it into 2,5-anhydro-D-mannose (chitose), consequently, depletion of GlcN after depolymerization is expected. On the other hand, the chitose unit displays characteristic 1H NMR signals the intensity of which may be estimated and utilized in the calculation of % DDA,...
SCOPE
1.1 This test method covers the determination of the degree of deacetylation in chitosan and chitosan salts intended for use in biomedical and pharmaceutical applications as well as in Tissue Engineered Medical Products (TEMPs) by high-resolution proton NMR (1H NMR). A guide for the characterization of chitosan salts has been published as Guide F2103.
1.2 The test method is applicable for determining the degree of deacetylation (% DDA) of chitosan chloride and chitosan glutamate salts and is valid for % DDA values from 50 up to and including 99. It is simple, rapid, and suitable for routine use. Knowledge of the degree of deacetylation is important for an understanding of the functionality of chitosan salts in TEMP formulations and applications. This test method will assist end users in choosing the correct chitosan for their particular application. Chitosan salts may have utility in drug delivery applications, as scaffold or matrix material, and in cell and tissue encapsulation applications.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-May-2018
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.42 - Biomaterials and Biomolecules for TEMPs
Relations
- Effective Date
- 01-Jun-2018
- Effective Date
- 01-Jun-2018
- Effective Date
- 15-Mar-2011
- Effective Date
- 01-Mar-2011
- Effective Date
- 01-Nov-2008
- Effective Date
- 01-Feb-2007
- Effective Date
- 01-Feb-2007
- Effective Date
- 01-Feb-2007
- Effective Date
- 10-Apr-2002
- Effective Date
- 10-Apr-2002
- Effective Date
- 10-May-2001
- Effective Date
- 01-Jun-2018
Overview
ASTM F2260-18 - Standard Test Method for Determining Degree of Deacetylation in Chitosan Salts by Proton Nuclear Magnetic Resonance (1H NMR) Spectroscopy - defines a quantitative, reproducible test method to measure the degree of deacetylation (% DDA) in chitosan chloride and chitosan glutamate salts using high-resolution proton NMR. Knowing the % DDA in chitosan salts is critical for biomedical, pharmaceutical, and tissue-engineered medical products (TEMPs), as the deacetylation level influences charge density, solubility, reactivity, and biological functionality of the polymer.
This standard provides a simple, rapid, and routine approach to characterizing chitosan salts with reliable precision, supporting robust material selection and quality control for researchers, manufacturers, and regulatory bodies in the biomaterials industry.
Key Topics
- Degree of Deacetylation (% DDA): This method quantifies the fraction or percentage of glucosamine units (deacetylated monomers) within chitosan, an essential factor affecting its physical and biological properties.
- Applicable Materials: The standard targets chitosan chloride and chitosan glutamate, both water-soluble chitosan salts widely used in biomedical and pharmaceutical formulations.
- Proton NMR Methodology: Utilizes high-resolution 1H NMR spectroscopy to directly measure the relative intensities of proton signals associated with acetylated and deacetylated units-bypassing the need for external calibration curves or reference samples.
- Sample Preparation: Depolymerization of chitosans (to DPn 15–30) via selective nitrous acid degradation in deuterated water to obtain well-resolved NMR spectra.
- Precision and Range: Validates % DDA results for values from 50 to 99 with a standard deviation typically less than ±1%. Non-applicable for samples with extremely low DDA due to solubility limitations.
- Scope of Use: Supports characterization, quality control, and selection of chitosan salts for use in advanced medical and tissue engineering applications.
Applications
The standardized determination of the degree of deacetylation in chitosan salts is highly relevant in:
- Biomedical Devices and Pharmaceuticals:
- Ensures chitosan salts meet functional requirements for drug delivery systems, wound healing products, and tissue scaffolds.
- Allows tailored selection of chitosan based on precise DDA for biocompatibility, drug-release rates, and mechanical characteristics.
- Tissue Engineered Medical Products (TEMPs):
- Underpins material design and consistency for scaffolds and encapsulation matrices in regenerative medicine.
- Facilitates regulatory compliance regarding material specification and traceability.
- Routine Quality Control:
- Provides a repeatable, operator-independent method for everyday batch release and comparative studies.
- Research and Development:
- Supports the development of new chitosan-based biomaterials and blends by definitively characterizing and comparing source materials.
Related Standards
- ASTM F2103: Guide for Characterization and Testing of Chitosan Salts as Starting Materials Intended for Use in Biomedical and Tissue-Engineered Medical Product Applications.
- USP <761>: Nuclear Magnetic Resonance - methodology and general considerations for pharmaceutical analysis.
- European Pharmacopoeia Monograph 2008:1774: Specifications for chitosan chloride.
- ASTM F386: Practice for presentation of NMR data and standards for thickness in resilient materials.
Practical Value
Adopting ASTM F2260-18 supports the high-precision determination of chitosan deacetylation-critical for manufacturers, researchers, and quality managers focusing on biomedical-grade chitosan. Rapid, reproducible results underpin quality assurance, regulatory submissions, and effective material selection for therapeutic products. The use of proton NMR ensures direct measurement, robustness to impurities, and reduces subjective interpretation or reliance on historical calibration, strengthening confidence in product specifications and facilitating global market access.
Buy Documents
ASTM F2260-18 - Standard Test Method for Determining Degree of Deacetylation in Chitosan Salts by Proton Nuclear Magnetic Resonance (<sup>1</sup>H NMR) Spectroscopy
REDLINE ASTM F2260-18 - Standard Test Method for Determining Degree of Deacetylation in Chitosan Salts by Proton Nuclear Magnetic Resonance (<sup>1</sup>H NMR) Spectroscopy
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2260-18 is a standard published by ASTM International. Its full title is "Standard Test Method for Determining Degree of Deacetylation in Chitosan Salts by Proton Nuclear Magnetic Resonance (<sup>1</sup>H NMR) Spectroscopy". This standard covers: SIGNIFICANCE AND USE 4.1 The degree of deacetylation of chitosan salts is an important characterization parameter since the charge density of the chitosan molecule is responsible for potential biological and functional effects. 4.2 The degree of deacetylation (% DDA) of water-soluble chitosan salts can be determined by 1H nuclear magnetic resonance spectroscopy (1H NMR). Several workers have reported on the NMR determination of chemical composition and sequential arrangement of monomer units in chitin and chitosan. The test method described is primarily based on the work of Vårum et al. (1991),5 which represents the first publication on routine determination of chemical composition in chitosans by solution state 1H NMR spectroscopy. This test method is applicable for determining the % DDA of chitosan chloride and chitosan glutamate salts. It is a simple, rapid, and suitable method for routine use. Quantitative 1H NMR spectroscopy reports directly on the relative concentration of chemically distinct protons in the sample, consequently, no assumptions, calibration curves or calculations other than determination of relative signal intensity ratios are necessary. 4.3 In order to obtain well-resolved NMR spectra, depolymerization of chitosans to a number average degree of polymerization (DPn) of ~15 to 30 is required. This reduces the viscosity and increases the mobility of the molecules. Although there are several options for depolymerization of chitosans, the most convenient procedure is that of nitrous acid degradation in deuterated water. The reaction is selective, stoichiometric with respect to GlcN, rapid, and easily controlled (Allan & Peyron, 1995).6 The reaction selectively cleaves after a GlcN-residue, transforming it into 2,5-anhydro-D-mannose (chitose), consequently, depletion of GlcN after depolymerization is expected. On the other hand, the chitose unit displays characteristic 1H NMR signals the intensity of which may be estimated and utilized in the calculation of % DDA,... SCOPE 1.1 This test method covers the determination of the degree of deacetylation in chitosan and chitosan salts intended for use in biomedical and pharmaceutical applications as well as in Tissue Engineered Medical Products (TEMPs) by high-resolution proton NMR (1H NMR). A guide for the characterization of chitosan salts has been published as Guide F2103. 1.2 The test method is applicable for determining the degree of deacetylation (% DDA) of chitosan chloride and chitosan glutamate salts and is valid for % DDA values from 50 up to and including 99. It is simple, rapid, and suitable for routine use. Knowledge of the degree of deacetylation is important for an understanding of the functionality of chitosan salts in TEMP formulations and applications. This test method will assist end users in choosing the correct chitosan for their particular application. Chitosan salts may have utility in drug delivery applications, as scaffold or matrix material, and in cell and tissue encapsulation applications. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 The degree of deacetylation of chitosan salts is an important characterization parameter since the charge density of the chitosan molecule is responsible for potential biological and functional effects. 4.2 The degree of deacetylation (% DDA) of water-soluble chitosan salts can be determined by 1H nuclear magnetic resonance spectroscopy (1H NMR). Several workers have reported on the NMR determination of chemical composition and sequential arrangement of monomer units in chitin and chitosan. The test method described is primarily based on the work of Vårum et al. (1991),5 which represents the first publication on routine determination of chemical composition in chitosans by solution state 1H NMR spectroscopy. This test method is applicable for determining the % DDA of chitosan chloride and chitosan glutamate salts. It is a simple, rapid, and suitable method for routine use. Quantitative 1H NMR spectroscopy reports directly on the relative concentration of chemically distinct protons in the sample, consequently, no assumptions, calibration curves or calculations other than determination of relative signal intensity ratios are necessary. 4.3 In order to obtain well-resolved NMR spectra, depolymerization of chitosans to a number average degree of polymerization (DPn) of ~15 to 30 is required. This reduces the viscosity and increases the mobility of the molecules. Although there are several options for depolymerization of chitosans, the most convenient procedure is that of nitrous acid degradation in deuterated water. The reaction is selective, stoichiometric with respect to GlcN, rapid, and easily controlled (Allan & Peyron, 1995).6 The reaction selectively cleaves after a GlcN-residue, transforming it into 2,5-anhydro-D-mannose (chitose), consequently, depletion of GlcN after depolymerization is expected. On the other hand, the chitose unit displays characteristic 1H NMR signals the intensity of which may be estimated and utilized in the calculation of % DDA,... SCOPE 1.1 This test method covers the determination of the degree of deacetylation in chitosan and chitosan salts intended for use in biomedical and pharmaceutical applications as well as in Tissue Engineered Medical Products (TEMPs) by high-resolution proton NMR (1H NMR). A guide for the characterization of chitosan salts has been published as Guide F2103. 1.2 The test method is applicable for determining the degree of deacetylation (% DDA) of chitosan chloride and chitosan glutamate salts and is valid for % DDA values from 50 up to and including 99. It is simple, rapid, and suitable for routine use. Knowledge of the degree of deacetylation is important for an understanding of the functionality of chitosan salts in TEMP formulations and applications. This test method will assist end users in choosing the correct chitosan for their particular application. Chitosan salts may have utility in drug delivery applications, as scaffold or matrix material, and in cell and tissue encapsulation applications. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2260-18 is classified under the following ICS (International Classification for Standards) categories: 11.100.99 - Other standards related to laboratory medicine. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2260-18 has the following relationships with other standards: It is inter standard links to ASTM F2260-03(2012)e1, ASTM F2103-18, ASTM F386-11, ASTM F2103-11, ASTM F386-02(2008), ASTM F2103-01(2007), ASTM F2103-01(2007)e2, ASTM F2103-01(2007)e1, ASTM F386-02, ASTM F386-93, ASTM F2103-01, ASTM F2027-16. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2260-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2260 − 18
Standard Test Method for
Determining Degree of Deacetylation in Chitosan Salts by
Proton Nuclear Magnetic Resonance ( H NMR)
Spectroscopy
This standard is issued under the fixed designation F2260; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
2.1 ASTM Standards:
1.1 This test method covers the determination of the degree
F386 Test Method for Thickness of Resilient Flooring Ma-
of deacetylation in chitosan and chitosan salts intended for use
terials Having Flat Surfaces
in biomedical and pharmaceutical applications as well as in
F2103 Guide for Characterization and Testing of Chitosan
Tissue Engineered Medical Products (TEMPs) by high-
Salts as Starting Materials Intended for Use in Biomedical
resolution proton NMR ( H NMR). A guide for the character-
and Tissue-Engineered Medical Product Applications
ization of chitosan salts has been published as Guide F2103.
2.2 United States Pharmacopeia Document:
1.2 Thetestmethodisapplicablefordeterminingthedegree
USP 35-NF30 <761> Nuclear Magnetic Resonance
of deacetylation (% DDA) of chitosan chloride and chitosan
2.3 European Pharmacopoeia Document:
glutamate salts and is valid for % DDA values from 50 up to
European Pharmacopoeia Monograph 2008:1774 Chitosan
and including 99. It is simple, rapid, and suitable for routine
Chloride
use. Knowledge of the degree of deacetylation is important for
anunderstandingofthefunctionalityofchitosansaltsinTEMP
3. Terminology
formulations and applications. This test method will assist end
users in choosing the correct chitosan for their particular
3.1 Definitions:
application. Chitosan salts may have utility in drug delivery
3.1.1 chitosan, n—a linear polysaccharide consisting of
applications, as scaffold or matrix material, and in cell and
β(1→4) linked 2-acetamido-2-deoxy-D-glucopyranose (Glc-
tissue encapsulation applications.
NAc) and 2-amino-2-deoxy-D-glucopyranose (GlcN). Chito-
san is a polysaccharide derived by N-deacetylation of chitin.
1.3 The values stated in SI units are to be regarded as
3.1.2 degradation, n—change in the chemical structure,
standard. No other units of measurement are included in this
physical properties, or appearance of a material. Degradation
standard.
ofpolysaccharidesoccursviacleavageoftheglycosidicbonds.
1.4 This standard does not purport to address all of the
It is important to note that degradation is not synonymous with
safety concerns, if any, associated with its use. It is the
decomposition. Degradation is often used as a synonym for
responsibility of the user of this standard to establish appro-
depolymerization when referring to polymers.
priate safety, health, and environmental practices and deter-
3.1.3 degree of deacetylation, n—the fraction or percentage
mine the applicability of regulatory limitations prior to use.
of glucosamine units (GlcN: deacetylated monomers) in a
1.5 This international standard was developed in accor-
chitosan polymer molecule.
dance with internationally recognized principles on standard-
3.1.4 depolymerization, n—reduction in the length of a
ization established in the Decision on Principles for the
polymer chain to form shorter polymeric units.
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on
This test method is under the jurisdiction ofASTM Committee F04 on Medical the ASTM website.
and Surgical Materials and Devices and is the direct responsibility of Subcommittee Available from U.S. Pharmacopeia (USP), 12601 Twinbrook Pkwy., Rockville,
F04.42 on Biomaterials and Biomolecules for TEMPs. MD 20852-1790, http://www.usp.org.
Current edition approved June 1, 2018. Published August 2018. Originally Available from European Directorate for the Quality of Medicines (EDQM),
ɛ1
approved in 2003. Last previous edition approved in 2012 as F2260 – 03 (2012) . Publications and Services, European Pharmacopoeia, BP 907, F-67029 Strasbourg,
DOI: 10.1520/F2260-18. France.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2260 − 18
3.1.5 water soluble chitosan salt, n—an ionic compound below), artifactual deacetylation of the sample does not occur
between chitosan molecule (cation) and a negatively charged during the short equilibration and analysis time.
anion (e.g. glutamate, acetate, lactate, chloride) that is soluble
4.5 Ageneral description of NMR can be found in <761> of
in water.
the USP 35-NF30.
4. Significance and Use
5. Materials
4.1 The degree of deacetylation of chitosan salts is an
5.1 Chemicals:
important characterization parameter since the charge density
5.1.1 Chitosan chloride or chitosan glutamate sample.
of the chitosan molecule is responsible for potential biological
and functional effects.
5.1.2 D O (99.9 %).
5.1.3 DCl (deuterium chloride), 0.1 M and1MinD O.
4.2 The degree of deacetylation (% DDA) of water-soluble
chitosan salts can be determined by H nuclear magnetic
5.1.4 NaOD (sodium deuteroxide), 0.1 M and1MinD O.
resonance spectroscopy ( H NMR). Several workers have
5.1.5 NaNO .
reported on the NMR determination of chemical composition
5.1.6 0.15 M TMSP (sodium 3-trimethylsilylpropionate-2,
and sequential arrangement of monomer units in chitin and
2’,3,3’-d)inD O.
4 2
chitosan. The test method described is primarily based on the
work of Vårum et al. (1991), which represents the first 5.2 Instruments:
publication on routine determination of chemical composition
5.2.1 Analytical balance (0.1 mg).
in chitosans by solution state H NMR spectroscopy. This test
5.2.2 Laboratory shaking device.
method is applicable for determining the % DDA of chitosan
5.2.3 pH meter or pH paper.
chloride and chitosan glutamate salts. It is a simple, rapid, and
5.2.4 5 mm NMR tubes.
suitable method for routine use. Quantitative H NMR spec-
5.2.5 NMR spectrometer (300 MHz field strength or higher
troscopy reports directly on the relative concentration of
is recommended although analysis at 100 MHz is possible),
chemically distinct protons in the sample, consequently, no
with variable temperature option, capable of maintaining 90 6
assumptions, calibration curves or calculations other than
1°C sample temperature during analysis, Analog-digital con-
determination of relative signal intensity ratios are necessary.
version (ADC) with a minimum of 16 bits is recommended.
4.3 In order to obtain well-resolved NMR spectra, depo-
lymerization of chitosans to a number average degree of
6. Procedure
polymerization(DP )of~15to30isrequired.Thisreducesthe
n
6.1 Sample Preparation:
viscosityandincreasesthemobilityofthemolecules.Although
there are several options for depolymerization of chitosans, the
6.1.1 Dissolve 33 mg chitosan chloride or 47 mg chitosan
most convenient procedure is that of nitrous acid degradation
glutamate in 3.3 mL D O by gentle shaking until completely
in deuterated water. The reaction is selective, stoichiometric
dissolved.
with respect to GlcN, rapid, and easily controlled (Allan &
6.1.2 Add 250 µL of 1 M DCl and shake. Check that the
Peyron, 1995). The reaction selectively cleaves after a GlcN-
sample pH* is <2.
residue, transforming it into 2,5-anhydro-D-mannose (chitose),
6.1.3 Add 100 µLfreshly made NaNO solution (10 mg/mL
consequently, depletion of GlcN after depolymerization is
in D O).
expected. On the other hand, the chitose unit displays charac-
6.1.4 Store the sample at room temperature in the dark for 4
teristic H NMR signals the intensity of which may be
h.
estimated and utilized in the calculation of % DDA, eliminat-
6.1.5 Use 0.1 M or 1 M NaOD to adjust the sample to pH*
ing the need for correction factors. Using the intensity of the
3.8 to 4.2.
chitose signals, the number average degree of polymerization
6.1.6 Transfer 0.7 mL of the sample solution to an NMR
can easily be calculated as a control of the depolymerization.
tube.
4.4 Samples are equilibrated and analyzed at a temperature
6.1.7 Add 5 µL of 0.15 M TMSP for chemical shift
of 90 6 1°C. Elevated sample temperature contributes to
referencing.
reducing sample viscosity and repositions the proton signal of
residualwatertoanareaoutsidethatofinterest.Whilesamples
NOTE 1—For a sample in 100 % D O, the pH reading on a pH meter is
are not stored at 90°C but only analyzed at this elevated
0.4 units lower than the true pD, due to an isotope effect on the glass
temperature, the NMR tubes should be sealed with a stopper to electrode. The meter reading in such solvents is normally reported
uncorrected and design
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
´1
Designation: F2260 − 03 (Reapproved 2012) F2260 − 18
Standard Test Method for
Determining Degree of Deacetylation in Chitosan Salts by
Proton Nuclear Magnetic Resonance ( H NMR)
Spectroscopy
This standard is issued under the fixed designation F2260; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Editorial changes were made to subsections 2.2, 2.3, and 4.5 in November 2012.
1. Scope
1.1 This test method covers the determination of the degree of deacetylation in chitosan and chitosan salts intended for use in
biomedical and pharmaceutical applications as well as in Tissue Engineered Medical Products (TEMPs) by high-resolution proton
NMR ( H NMR). A guide for the characterization of chitosan salts has been published as Guide F2103.
1.2 The test method is applicable for determining the degree of deacetylation (% DA)DDA) of chitosan chloride and chitosan
glutamate salts and is valid for % DADDA values from 50 up to and including 99. It is simple, rapid, and suitable for routine use.
Knowledge of the degree of deacetylation is important for an understanding of the functionality of chitosan salts in TEMP
formulations and applications. This test method will assist end users in choosing the correct chitosan for their particular application.
Chitosan salts may have utility in drug delivery applications, as a scaffold or matrix material, and in cell and tissue encapsulation
applications.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
F386 Test Method for Thickness of Resilient Flooring Materials Having Flat Surfaces
F2103 Guide for Characterization and Testing of Chitosan Salts as Starting Materials Intended for Use in Biomedical and
Tissue-Engineered Medical Product Applications
2.2 United States Pharmacopeia Document:
USP 35-NF30 <761> Nuclear Magnetic Resonance
2.3 European Pharmacopoeia Document:
European Pharmacopoeia Monograph 2008:1774 Chitosan Chloride
3. Terminology
3.1 Definitions:
This test method is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.42 on Biomaterials and Biomolecules for TEMPs.
Current edition approved Oct. 1, 2012June 1, 2018. Published November 2012August 2018. Originally approved in 2003. Last previous edition approved in 20082012
ɛ1
as F2260 – 03 (2008).(2012) . DOI: 10.1520/F2260-03R12E01.10.1520/F2260-18.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from U.S. Pharmacopeia (USP), 12601 Twinbrook Pkwy., Rockville, MD 20852-1790, http://www.usp.org.
Available from European Directorate for the Quality of Medicines (EDQM), Publications and Services, European Pharmacopoeia, BP 907, F-67029 Strasbourg, France.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2260 − 18
3.1.1 chitosan, n—a linear polysaccharide consisting of β(1→4) linked 2-acetamido-2-deoxy-D-glucopyranose (GlcNAc) and
2-amino-2-deoxy-D-glucopyranose (GlcN). Chitosan is a polysaccharide derived by N-deacetylation of chitin.
3.1.2 degradation, n—change in the chemical structure, physical properties, or appearance of a material. Degradation of
polysaccharides occurs via cleavage of the glycosidic bonds. It is important to note that degradation is not synonymous with
decomposition. Degradation is often used as a synonym for depolymerization when referring to polymers.
3.1.3 degree of deacetylation, n—the fraction or percentage of glucosamine units (GlcN: deacetylated monomers) in a chitosan
polymer molecule.
3.1.4 depolymerization, n—reduction in the length of a polymer chain to form shorter polymeric units.
3.1.5 water soluble chitosan salt, n—an ionic compound between chitosan molecule (cation) and a negatively charged anion
(e.g. glutamate, acetate, lactate, chloride) that is soluble in water.
4. Significance and Use
4.1 The degree of deacetylation of chitosan salts is an important characterization parameter since the charge density of the
chitosan molecule is responsible for potential biological and functional effects.
4.2 The degree of deacetylation (% DA)DDA) of water-soluble chitosan salts can be determined by H nuclear magnetic
resonance spectroscopy ( H NMR). Several workers have reported on the NMR determination of chemical composition and
sequential arrangement of monomer units in chitin and chitosan. The test method described is primarily based on the work of
Vårum et al. (1991), which represents the first publication on routine determination of chemical composition in chitosans by
solution state H NMR spectroscopy. This test method is applicable for determining the % DADDA of chitosan chloride and
chitosan glutamate salts. It is a simple, rapid, and suitable method for routine use. Quantitative H NMR spectroscopy reports
directly on the relative concentration of chemically distinct protons in the sample, consequently, no assumptions, calibration curves
or calculations other than determination of relative signal intensity ratios are necessary.
4.3 In order to obtain well-resolved NMR spectra, depolymerization of chitosans to a number average degree of polymerization
(DP ) of ~15 to 30 is required. This reduces the viscosity and increases the mobility of the molecules. Although there are several
n
options for depolymerization of chitosans, the most convenient procedure is that of nitrous acid degradation in deuterated water.
The reaction is selective, stoichiometric with respect to GlcN, rapid, and easily controlled (Allan & Peyron, 1995). The reaction
selectively cleaves after a GlcN-residue, transforming it into 2,5-anhydro-D-mannose (chitose), consequently, depletion of GlcN
after depolymerization is expected. On the other hand, the chitose unit displays characteristic H NMR signals the intensity of
which may be estimated and utilized in the calculation of % DA,DDA, eliminating the need for correction factors. Using the
intensity of the chitose signals, the number average degree of polymerization can easily be calculated as a control of the
depolymerization.
4.4 Samples are equilibrated and analyzed at a temperature of 90 6 1°C. Elevated sample temperature contributes to reducing
sample viscosity and repositions the proton signal of residual water to an area outside that of interest. While samples are not stored
at 90°C but only analyzed at this elevated temperature, the NMR tubes should be sealed with a stopper to avoid any evaporation.
At a sample pH* of 3.8-4.3 (see 6.1.5 below), artifactual deacetylation of the sample does not occur during the short equilibration
and analysis time.
4.5 A general description of NMR can be found in <761> of the USP 35-NF30.
5. Materials
5.1 Chemicals:
5.1.1 Chitosan chloride or chitosan glutamate sample.
5.1.2 D O (99.9 %).
5.1.3 DCl (deuterium chloride), 0.1 M and 1 M in D O.
5.1.4 NaOD (sodium deuteroxide), 0.1 M and 1 M in D O.
5.1.5 NaNO .
5.1.6 0.15 M TMSP (sodium 3-trimethylsilylpropionate-2,2’,3,3’-d ) in D O.
4 2
5.2 Instruments:
5.2.1 Analytical balance (0.1 mg).
5.2.2 Laboratory shaking device.
5.2.3 pH meter or pH paper.
5.2.4 5 mm NMR tubes.
Vårum, K. M., Anthonsen, M. W., Grasdalen, H., and Smidsrød, O., “Determination of the Degree of N-acetylation and the Distribution of N-acetyl Groups in Partially
N-deacetylated Chitins (Chitosans) by High-Field N.M.R. Spectroscopy,” Carbohydr. Res., Vol 211, 1991, pp. 17–23.
Allan, G. G. and Peyron, M., “Molecular Weight Manipulation of Chitosan 1: Kinetics of Depolymerization by Nitrous Acid,” Carbohydr. Res., Vol 277, 1995, pp.
257-272.
F2260 − 18
5.2.5 NMR spectrometer (300 MHz field strength or higher is recommended although analysis at 100 MHz is possible), with
variable temperature option, capable of maintaining 90 6 1°C sample temperature during analysis, Analog-digital conversion
(ADC) with a minimum of 16 bitbits is recommended.
6. Procedure
6.1 Sample Preparation:
6.1.1 Dissolve 33 mg chitosan chloride or 47 m
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...