ASTM E3351-22
(Test Method)Standard Test Method for Detection of Nitric Oxide Production In Vitro
Standard Test Method for Detection of Nitric Oxide Production <emph type="bdit">In Vitro</emph >
SIGNIFICANCE AND USE
5.1 This test method is designed to evaluate nanomaterial capacity to induce nitric oxide production by macrophages.
5.2 Activated macrophages generate large quantities of NO. NO generated from activated macrophages is a cytostatic/cytotoxic agent (3-6).
5.3 The production of NO in excessive amounts leads to the generation of peroxynitrite by its spontaneous reaction with superoxide. Peroxynitrite causes tissue injury through its capability to damage lipids, proteins, and DNA (2).
5.4 NO is a proinflammatory mediator and it is an important marker for activation of inflammation (5, 6).
5.5 Testing the capacity of a nanomaterial to induce NO production in vitro helps in predicting the nanomaterial’s biocompatibility through anticipating and understanding the potential problems that might be encountered during its in vivo administration.
SCOPE
1.1 This test method delivers a protocol for a quantitative measure of nitrite (NO2–), a stable end-product of nitric oxide (NO), in cell culture medium due to exposure to nanomaterial(s).
1.2 NO has a critical role in several pathological conditions in addition to its role in many physiological processes.
1.3 This test method uses murine macrophage cell line RAW 264.7 as an in vitro model.
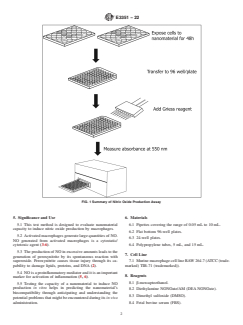
1.4 The nitrite is measured in the cell culture medium by a colorimetric analysis using Griess reagent as shown in Fig. 1.
FIG. 1 Summary of Nitric Oxide Production Assay
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Jun-2022
- Technical Committee
- E56 - Nanotechnology
- Drafting Committee
- E56.08 - Nano-Enabled Medical Products
Relations
- Effective Date
- 01-Oct-2018
- Effective Date
- 01-Oct-2016
- Effective Date
- 01-Apr-2012
- Effective Date
- 01-Jun-2010
- Refers
ASTM F1903-10 - Standard Practice for Testing For Biological Responses to Particles <i>In Vitro</i> - Effective Date
- 01-Jun-2010
- Effective Date
- 01-Apr-2009
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Nov-2005
- Effective Date
- 01-Nov-2005
- Refers
ASTM F1903-98(2003) - Standard Practice for Testing for Biological Responses to Particles In Vitro - Effective Date
- 01-Nov-2003
- Effective Date
- 10-Apr-2003
- Effective Date
- 10-Jun-1998
- Effective Date
- 10-Apr-1998
Overview
ASTM E3351-22: Standard Test Method for Detection of Nitric Oxide Production In Vitro establishes a protocol for quantitatively measuring nitric oxide (NO) production by macrophages in response to nanomaterial exposure, using nitrite (NO₂⁻) levels as an indicator. The test is based on the colorimetric analysis of nitrite in cell culture media via the Griess reagent. This standardized method is a critical tool for assessing the immunological impact and biocompatibility of nanomaterials, with implications for biomedical research and product safety evaluation.
Nitric oxide is a key proinflammatory mediator generated by activated macrophages. Its overproduction can lead to cytotoxic effects, tissue injury, and is implicated in both normal physiological and pathological processes. By evaluating NO production in vitro, this standard aids researchers and manufacturers in predicting how nanomaterials might interact with biological systems during in vivo applications.
Key Topics
- Nitric Oxide Measurement: Quantifies NO by detecting nitrite, a stable end-product, after nanomaterial exposure.
- Murine Macrophage Model: Utilizes RAW 264.7 cell lines as an established in vitro model for immune response studies.
- Colorimetric Griess Assay: Employs the Griess reagent for reliable detection and measurement of nitrite in cell culture medium.
- Controls and Calibration: Incorporates strict calibration standards, quality controls, and acceptance criteria for reproducibility and data accuracy.
- Nanomaterial Interference: Addresses protocols for identifying and mitigating nanomaterial assay interference.
- Biological Relevance: Evaluates the proinflammatory and cytotoxic potential of nanomaterials, critical for biocompatibility assessments.
- Regulatory Alignment: Developed in accordance with international standards principles, supporting global harmonization.
Applications
This standard plays a pivotal role in multiple practical contexts:
- Preclinical Safety Assessment: Provides a standardized method for screening nanomaterials used in medical devices, pharmaceuticals, and drug delivery systems, helping to ensure product biocompatibility before in vivo studies.
- Nanotoxicology Research: Supports research into the immunomodulatory effects of engineered nanoparticles, helping scientists understand potential health risks and mechanisms of action.
- Product Development: Assists manufacturers in the development and validation of nanomaterial-based products, ensuring compliance with regulatory guidelines for safety.
- Regulatory Submissions: Facilitates the generation of robust, comparable NO production data to support submissions to health authorities and regulatory bodies.
- Academic Studies: Serves as a reference protocol for academic laboratories investigating aspects of inflammation, immune response, and nanoparticle-cell interactions.
Related Standards
Several related ASTM and international standards complement or support the use of ASTM E3351-22:
- ASTM E2490: Guide for Measurement of Particle Size Distribution of Nanomaterials in Suspension by Photon Correlation Spectroscopy (PCS)
- ASTM E2834: Guide for Measurement of Particle Size Distribution of Nanomaterials in Suspension by Nanoparticle Tracking Analysis (NTA)
- ASTM F1877: Practice for Characterization of Particles
- ASTM F1903: Practice for Testing for Cellular Responses to Particles In Vitro
These standards collectively offer a comprehensive framework for characterizing nanomaterials and assessing their biological interactions, supporting the rigorous evaluation of immune response, cytotoxicity, and overall biosafety in research and commercial applications.
Keywords: ASTM E3351-22, nitric oxide detection, nanomaterial biocompatibility, in vitro assay, macrophage response, Griess reagent, nanotoxicology, immune response, biomedical research, standard test method
Buy Documents
ASTM E3351-22 - Standard Test Method for Detection of Nitric Oxide Production <emph type="bdit">In Vitro</emph >
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM E3351-22 is a standard published by ASTM International. Its full title is "Standard Test Method for Detection of Nitric Oxide Production <emph type="bdit">In Vitro</emph >". This standard covers: SIGNIFICANCE AND USE 5.1 This test method is designed to evaluate nanomaterial capacity to induce nitric oxide production by macrophages. 5.2 Activated macrophages generate large quantities of NO. NO generated from activated macrophages is a cytostatic/cytotoxic agent (3-6). 5.3 The production of NO in excessive amounts leads to the generation of peroxynitrite by its spontaneous reaction with superoxide. Peroxynitrite causes tissue injury through its capability to damage lipids, proteins, and DNA (2). 5.4 NO is a proinflammatory mediator and it is an important marker for activation of inflammation (5, 6). 5.5 Testing the capacity of a nanomaterial to induce NO production in vitro helps in predicting the nanomaterial’s biocompatibility through anticipating and understanding the potential problems that might be encountered during its in vivo administration. SCOPE 1.1 This test method delivers a protocol for a quantitative measure of nitrite (NO2–), a stable end-product of nitric oxide (NO), in cell culture medium due to exposure to nanomaterial(s). 1.2 NO has a critical role in several pathological conditions in addition to its role in many physiological processes. 1.3 This test method uses murine macrophage cell line RAW 264.7 as an in vitro model. 1.4 The nitrite is measured in the cell culture medium by a colorimetric analysis using Griess reagent as shown in Fig. 1. FIG. 1 Summary of Nitric Oxide Production Assay 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method is designed to evaluate nanomaterial capacity to induce nitric oxide production by macrophages. 5.2 Activated macrophages generate large quantities of NO. NO generated from activated macrophages is a cytostatic/cytotoxic agent (3-6). 5.3 The production of NO in excessive amounts leads to the generation of peroxynitrite by its spontaneous reaction with superoxide. Peroxynitrite causes tissue injury through its capability to damage lipids, proteins, and DNA (2). 5.4 NO is a proinflammatory mediator and it is an important marker for activation of inflammation (5, 6). 5.5 Testing the capacity of a nanomaterial to induce NO production in vitro helps in predicting the nanomaterial’s biocompatibility through anticipating and understanding the potential problems that might be encountered during its in vivo administration. SCOPE 1.1 This test method delivers a protocol for a quantitative measure of nitrite (NO2–), a stable end-product of nitric oxide (NO), in cell culture medium due to exposure to nanomaterial(s). 1.2 NO has a critical role in several pathological conditions in addition to its role in many physiological processes. 1.3 This test method uses murine macrophage cell line RAW 264.7 as an in vitro model. 1.4 The nitrite is measured in the cell culture medium by a colorimetric analysis using Griess reagent as shown in Fig. 1. FIG. 1 Summary of Nitric Oxide Production Assay 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E3351-22 is classified under the following ICS (International Classification for Standards) categories: 11.100.10 - In vitro diagnostic test systems. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E3351-22 has the following relationships with other standards: It is inter standard links to ASTM F1903-18, ASTM F1877-16, ASTM E2834-12, ASTM F1877-05(2010), ASTM F1903-10, ASTM E2490-09, ASTM E2490-08, ASTM F1877-05e1, ASTM F1877-05, ASTM F1903-98(2003), ASTM F1877-98(2003)e1, ASTM F1903-98, ASTM F1877-98. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E3351-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E3351 − 22
Standard Test Method for
Detection of Nitric Oxide Production In Vitro
This standard is issued under the fixed designation E3351; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 3. Terminology
1.1 This test method delivers a protocol for a quantitative 3.1 Definitions:
–
measure of nitrite (NO ), a stable end-product of nitric oxide 3.1.1 Cal—calibration standards
(NO), in cell culture medium due to exposure to nanomateri-
3.1.2 C —maximum serum concentration
max
al(s).
3.1.3 CV—coefficient of variation
1.2 NO has a critical role in several pathological conditions
3.1.4 DEA NONOate—diethylamine NONOate/AM
in addition to its role in many physiological processes.
3.1.5 DMSO—dimethyl sulfoxide
1.3 This test method uses murine macrophage cell line
3.1.6 FBS—fetal bovine serum
RAW 264.7 as an in vitro model.
3.1.7 Int.—intermediate
1.4 The nitrite is measured in the cell culture medium by a
colorimetric analysis using Griess reagent as shown in Fig. 1. 3.1.8 LPS—lipopolysaccharide
1.5 This standard does not purport to address all of the
3.1.9 PBS—phosphate buffered saline
safety concerns, if any, associated with its use. It is the
3.1.10 PDFT—percent difference from theoretical
responsibility of the user of this standard to establish appro-
3.1.11 RPMI—Roswell Park Memorial Institute
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use. 3.1.12 QC—quality control
1.6 This international standard was developed in accor-
3.1.13 SD—standard deviation
dance with internationally recognized principles on standard-
3.1.14 w/v—weight to volume ratio
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
4. Summary of Test Method
mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee. 4.1 This test method is used to assess the capability of
nanomaterials to induce nitric oxide production by macro-
2. Referenced Documents
phages in vitro(see Fig. 1).
2.1 ASTM Standards:
4.2 TheNOmoleculehasashorthalf-lifeandreactsquickly
E2490Guide for Measurement of Particle Size Distribution
with free oxygen, oxygen radicals, redox metals and even with
of Nanomaterials in Suspension by Photon Correlation
oxygenated hemoglobin to generate other reactive nitrogen
Spectroscopy (PCS) –
intermediates which decomposes to form nitrite (NO ) and
– 3
E2834Guide for Measurement of Particle Size Distribution
nitrate (NO ) (1). NO molecule can react with oxygenated
of Nanomaterials in Suspension by NanoparticleTracking –
hemoglobin to produce nitrate (NO ) (1, 2).
Analysis (NTA)
4.3 This test method describes a protocol for assessing and
F1877Practice for Characterization of Particles
measuring nitrite as a replacement marker and quantitative
F1903Practice for Testing for Cellular Responses to Par-
indicator of NO production.
ticles in vitro
4.4 In this test method, nitrite is measured in cell culture
medium using the Griess reagent.
This test method is under the jurisdiction of ASTM Committee E56 on
Nanotechnology and is the direct responsibility of Subcommittee E56.08 on
4.5 The upper limit of nitrite quantification is 250µM and
Nano-Enabled Medical Products.
the lower limit of quantification is 1.95µM.
Current edition approved July 1, 2022. Published July 2022. DOI: 10.1520/
E3351-22.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on Theboldfacenumbersinparenthesesrefertothelistofreferencesattheendof
the ASTM website. the standard.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E3351 − 22
FIG. 1 Summary of Nitric Oxide Production Assay
5. Significance and Use 6. Materials
5.1 This test method is designed to evaluate nanomaterial
6.1 Pipettes covering the range of 0.05mL to 10mL.
capacity to induce nitric oxide production by macrophages.
6.2 Flat bottom 96-well plates.
5.2 Activated macrophages generate large quantities of NO.
6.3 24-well plates.
NO generated from activated macrophages is a cytostatic/
6.4 Polypropylene tubes, 5mL, and 15mL.
cytotoxic agent (3-6).
5.3 TheproductionofNOinexcessiveamountsleadstothe
7. Cell Line
generation of peroxynitrite by its spontaneous reaction with
superoxide. Peroxynitrite causes tissue injury through its ca-
7.1 MurinemacrophagecelllineRAW264.7(ATCC(trade-
pability to damage lipids, proteins, and DNA (2). marked) TIB-71 (trademarked)).
5.4 NOisaproinflammatorymediatoranditisanimportant
8. Reagents
marker for activation of inflammation (5, 6).
8.1 β-mercaptoethanol.
5.5 Testing the capacity of a nanomaterial to induce NO
production in vitro helps in predicting the nanomaterial’s
8.2 Diethylamine NONOate/AM (DEA NONOate).
biocompatibility through anticipating and understanding the
8.3 Dimethyl sulfoxide (DMSO).
potentialproblemsthatmightbeencounteredduringits in vivo
administration. 8.4 Fetal bovine serum (FBS).
E3351 − 22
8.5 Lipopolysaccharides from Escherichia coli O55:B5 10.6 Griess Reagent—Consists of 2 reagents: reagentAand
(LPS). reagent B.
10.6.1 Reagent A—Dissolve sulfanilamide in 2.5% phos-
8.6 Naphthylethylenediamine dihydrochloride.
phoric acid (H PO ) to a final concentration of 1% (w/v), for
3 4
8.7 Penicillin-Streptomycin.
example, dissolve 1g of sulfanilamide in 100mL of 2.5%
H PO .
8.8 Phosphate buffered saline (PBS), pH 7.4.
3 4
10.6.2 Reagent B—Dissolvenaphthylethylenediaminedihy-
8.9 Phosphoric acid.
drochloride in 2.5% H PO to a final concentration of 0.1%
3 4
8.10 RPMI-1640 without phenol red.
(w/v), for example, dissolve 100mg of naphthylethylenedi-
amine dihydrochloride in 100mL of 2.5% H PO .
3 4
8.11 Sodium nitrite (NaNO ) standard, 0.1M stock solu-
10.6.3 Store both solutions in amber glass bottles at 4°C;
tion.
discard if discoloration occurs or solutions are not clear.
8.12 Sulfanilamide.
10.6.4 Combine equal volumes of reagents A and B just
8.13 Trypan blue solution. prior to use to form the Griess reagent.
10.6.5 Griess should be used immediately after preparation
9. Apparatus and any remaining reagent should be discarded.
10.7 Preparation of Sodium Nitrite (NaNO ) Calibration
9.1 Biohazard safety cabinet approved for level II handling
Standards:
of biological material.
10.7.1 PrepareNaNO calibrationstandardsasshowninthe
9.2 Cellcultureincubatorwith5%CO and95%humidity.
example table below. These calibration standards will be used
9.3 Centrifuge.
for building the standard curve. The standard curve will be
used to calculate the NO concentration of the test samples.
9.4 Freezer, –20°C.
Stock is reagent 8.11 (sodium nitrite (NaNO ) standard, 0.1M
9.5 Hemocytometer or cell counter.
stock solution). The stock will be diluted to prepare the
9.6 Inverted light microscope. intermediate (Int.) stock A, intermediate stock B, and calibra-
tion standard (Cal) 1–8.
9.7 Plate reader.
10.7.2 Volumes of NaNO calibration standards can be
9.8 Refrigerator, 2°C to 8°C.
adjusted based on the need.
9.9 Vortex mixer.
Nominal
Level Concentration Preparation Procedure
9.10 Water bath set at 37 °C.
(µM)
Int. A 10 000 100 µL Stock + 900 µL complete RPMI-1640 medium
Int. B 1000 100 µL Int. A + 900 µL complete RPMI-1640 medium
10. Preparation of Complete Cell Culture Medium,
Cal 1 250 200 µL Int. B + 600 µL complete RPMI-1640 medium
Lipopolysaccharide, Diethylamine NONOate/AM,
Cal 2 125 400 µL Cal 1 + 400 µL complete RPMI-1640 medium
Griess Reagent, Controls, Nitrite Calibration
Cal 3 62.5 400 µL Cal 2 + 400 µL complete RPMI-1640 medium
Cal 4 31.3 400 µL Cal 3 + 400 µL complete RPMI-1640 medium
Standards and Test Samples
Cal 5 15.6 400 µL Cal 4 + 400 µL complete RPMI-1640 medium
10.1 Preparation of Complete RPMI-1640 Medium—The Cal 6 7.81 400 µL Cal 5 + 400 µL complete RPMI-1640 medium
Cal 7 3.91 400 µL Cal 6 + 400 µL complete RPMI-1640 medium
complete RPMI-1640 medium should contain 10% FBS (heat
Cal 8 1.95 400 µL Cal 7 + 400 µL complete RPMI-1640 medium
inactivated), 2mM L-glutamine, 50 µM β-mercaptoethanol,
10.8 Preparation of Sodium Nitrite (NaNO ) Quality Con-
100U⁄mLpenicillin,and100µg⁄mLstreptomycinsulfate.The
trols:
medium should be stored at 2°C to 8°C protected from light
10.8.1 Prepare NaNO quality control as shown in the
for no longer than 1 month. Before use, warm in a water bath.
example table below. Stock is reagent 8.11 (sodium nitrite
10.2 Lipopolysaccharide 1 mg/mL (LPS, Stock)—Add1mL
(NaNO ) standard, 0.1 M stock solution). The stock will be
of sterile PBS or cell culture medium per 1 mg of LPS to the
diluted to prepare intermediate (Int.) stock A, intermediate
vial and vortex to mix.Aliquot the stock into 100 µL portions
stock B, and quality control (QC) 1–3.
and store at –20°C. Individual aliquots are thawed and used
10.8.2 Volumes of NaNO can be adjusted according to the
only once.
need.
10.3 Diethylamine NONOate/AM—Reconstitute with
Nominal
Level Concentration Preparation Procedure
DMSO, aliquot (20 µl aliquots) and freeze (–20°C). DMSO
(µM)
stock solution aliquots are stable for 2 months at –20°C.
Int. A 10 000 100 µL Stock + 900 µL complete RPMI-1640 medium
Int. B 1000 100 µL Int. A + 900 µL complete RPMI-1640 medium
10.4 Positive Control—Dilute stock LPS solution in cell
QC 1 100 100 µL Int. B + 900 µL complete RPMI-1640 medium
culture medium to final concentrations of 100ng⁄mL. Store at
QC 2 50 400 µL QC1 + 400 µL complete RPMI-1640 medium
room temperature. Discard unused portion after experiment.
QC 3 5 100 µL QC 2 + 900 µL complete RPMI-1640 medium
For DEA NONOate; dilute stock aliquots in cell culture
10.9 Specification of Nanomaterial Test Samples—This as-
medium to final concentrations of 6.25µM, 12.5µM, and
say requires a minimum of 3500 µLof nanomaterial (at 1× the
25µM. Discard unused portion after experiment.
highest final test concentration, this volume is calculated from
10.5 Negative Control—Use PBS as a negative control. the number and concentration of test samples prepared as
E3351 − 22
NOTE 1—Removal of nanomaterials from culture medium may be
specified below including the replicates) dissolved/
required prior to this step if nanomaterials can interfere with assay, for
resuspended in complete culture medium. The following shall
example, if nanomaterial reacts with either or both components of the
be considered when choosing the nanomaterial concentration:
Griess reagent or have absorbance at or close to 550 nm. If nanomaterial
(1)Dispersibility,solubility,andhomogeneityofnanomaterials
removal is not feasible, results obtained for “no cells nanomaterials”
in a biocompatible buffer, (2) pH shall be maintained in the
control may be subtracted from that obtained for “nanomaterial test
sample”tocorrectforparticlebackgroundinterference.SeeAnnexA1for
physiological range, and (3) stability of nanomaterials during
anoptionalpre-testplateprocedurethatcanbeusedtoobtaininformation
testing. Before testing, the nanomaterial shall be characterized
aboutpotentialnanomaterialinterferencebeforeperformingthefullassay.
(for example, size, size distribution, and charge) under physi-
11.7 Inaseparatetube,combineequalvolumesofreagentA
ological conditions and according to standard methods includ-
and reagent B at room temperature; this is the Griess reagent
ing those recommended in Guides E2490 and E2834 for
(for example, combine 6 ml of reagentAwith 6 ml of reagent
nanomaterials and Practices F1877 and F1903 for powders.
B).
Nanomaterials are tested at four concentrations. For example,
in the absence of data from PK studies, if the highest tested
11.8 Add 100 µL of the Griess reagent to each well of the
concentration is 1.0 mg/mL then the stock nanomaterial
“NO Test Plate”.
concentrationshouldalsobe1.0mg/mLandthethreeserial1:5
11.9 Placetheplateonashakerfor2minto3min,allowing
dilutions will be 0.2mg⁄mL, 0.04mg⁄mL, and 0.008 mg/mL.
all ingredients to mix.
Six 0.5 mL samples will be used for each test concentration
(fourfortheassaytestsamplesandtwoforacell-freecontrol). 11.10 Measure absorbance at 550 nm using a 96-well plate
However, when the nanomaterial’s plasma concentration is reader machine.
known from in vivo PK studies, the highest concentration
tested in vitro is chosen as 10×, 30×, or 100× of that C 12. Calculations
max
(depending on the solubility of the nanomaterial).
12.1 Note that superimposing the two culture plates maps
10.10 Preparation of Nanomaterial Test Samples—Prepare
ontheNOplatemapinAppendixX1(Fig.X1.1andFig.X1.2)
a nanomaterial stock solution in complete cell culture medium
yields four replicate wells for each test sample, positive
ataconcentrationof1mg/ml.Preparethreeserial1:5dilutions
control, negative control and blank. There are three replicates
using this stock to obtain nanomaterial test samples of 0.2
for each calibration standard and quality control.
mg/mL, 0.04mg⁄mL, and 0.008 mg/mL.
12.2 Subtractthemeanabsorbancevaluesoftheblankwells
10.11 Cell Maintenance—Grow RAW 264.7 cells in com-
from all the absorbance values of all other wells.
plete RPMI
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...