ASTM F921-10(2018)
(Terminology)Standard Terminology Relating to Hemostatic Forceps
Standard Terminology Relating to Hemostatic Forceps
SCOPE
1.1 This terminology covers basic terms and considerations for the components of hemostatic forceps. Instruments in this terminology are limited to those fabricated from stainless steel and for general surgical procedures. See Fig. 1 and Fig. 2.
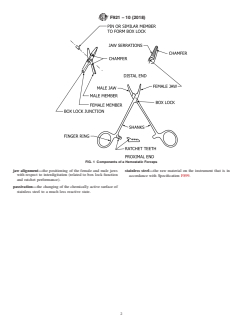
FIG. 1 Components of a Hemostatic Forceps
FIG. 2 Typical Types of Serrations and Teeth for Hemostatic Forceps
1.2 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Sep-2018
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.33 - Medical/Surgical Instruments
Relations
- Effective Date
- 01-Oct-2018
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Feb-2020
- Effective Date
- 15-Sep-2019
- Effective Date
- 01-Dec-2012
- Effective Date
- 01-Nov-2012
- Effective Date
- 01-Jun-2012
- Effective Date
- 01-Mar-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Feb-2009
- Effective Date
- 01-Feb-2009
- Effective Date
- 01-Feb-2007
- Effective Date
- 10-Apr-2002
- Effective Date
- 10-Apr-2002
- Effective Date
- 01-Oct-2018
Overview
ASTM F921-10(2018): Standard Terminology Relating to Hemostatic Forceps defines essential terms and considerations for components of hemostatic forceps made from stainless steel and intended for general surgical procedures. Developed by ASTM Committee F04 on Medical and Surgical Materials and Devices, this internationally recognized standard ensures consistent use of terminology to support safe, effective design, manufacturing, and use of hemostatic forceps across the medical device industry.
The standard aligns with the World Trade Organization (WTO) guidelines for international standards development, promoting global consistency and trade transparency. Its focus is on reusable hemostatic forceps, covering common features, key component terms, and performance characteristics relevant to device manufacturers, regulatory authorities, and healthcare professionals.
Key Topics
Essential Terminology Defined by ASTM F921-10(2018):
- Hemostatic Forceps: Surgical instruments used primarily for compressing blood vessels and grasping tissues during procedures.
- Materials: Limited to those fabricated from stainless steel, as specified in ASTM F899.
- Core Component Terms:
- Jaws: Parts with serrated surfaces for vessel compression.
- Serrations/Teeth: The gripping or clamping surfaces on the jaws or ratchets.
- Box Lock: The pivot junction enabling movement and secure closure.
- Finger Rings: Loop-shaped grips for surgeon control.
- Ratchets: Inclined teeth at the handle locking mechanism.
- Shank: The section providing length and leverage.
- Proximal End/Distal End: Respectively, the end closest to or furthest from the surgeon during use.
- Male/Female Member: Components interlocking at the box lock for forceps operation.
- Physical Properties:
- Hardness: Resistance to indentation.
- Elasticity: Capacity to withstand stress without permanent deformation.
- Finish: Surface appearance, including bright/mirror, satin, matte, or black finishes.
- Passivation: Chemical treatment to reduce reactivity of stainless steel.
- Corrosion: Rust formation and resistance.
Performance and Design Considerations:
- Importance of jaw alignment and interdigitation for function.
- Box lock construction as the standard, while not excluding alternative innovations.
Applications
Practical Value and Use Cases
- Manufacturers: Facilitates design, production, and quality assurance of surgical hemostatic forceps with standardized terminology, ensuring clear communication and product comparability.
- Regulatory Bodies: Supports the evaluation and approval of hemostatic forceps by providing a common language and reference point for compliance assessment.
- Healthcare Providers: Ensures precise identification, selection, and usage of surgical instruments with known performance and quality attributes.
- Global Trade: Promotes international harmonization and easier cross-border acceptance of medical devices that conform to standardized terminology.
Applicable only to reusable hemostatic forceps designed for general surgical use; does not cover disposable versions or forceps made from materials other than stainless steel, although definitions may be relevant to those areas.
Related Standards
- ASTM F899: Specification for Wrought Stainless Steels for Surgical Instruments - Defines material requirements applicable to forceps.
- ISO 7151: Instruments for Surgery - Hemostatic Forceps - General Requirements - Provides complementary international guidelines for manufacturing and performance.
- Other ASTM F04 Standards: Guidance on medical and surgical materials and devices, supporting broader compliance for surgical instruments.
ASTM F921-10(2018) remains a critical reference for standardizing terminology related to hemostatic forceps, promoting safety, reliability, and interoperability within the medical industry. For further information or purchasing, visit the ASTM website.
Buy Documents
ASTM F921-10(2018) - Standard Terminology Relating to Hemostatic Forceps
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F921-10(2018) is a standard published by ASTM International. Its full title is "Standard Terminology Relating to Hemostatic Forceps". This standard covers: SCOPE 1.1 This terminology covers basic terms and considerations for the components of hemostatic forceps. Instruments in this terminology are limited to those fabricated from stainless steel and for general surgical procedures. See Fig. 1 and Fig. 2. FIG. 1 Components of a Hemostatic Forceps FIG. 2 Typical Types of Serrations and Teeth for Hemostatic Forceps 1.2 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SCOPE 1.1 This terminology covers basic terms and considerations for the components of hemostatic forceps. Instruments in this terminology are limited to those fabricated from stainless steel and for general surgical procedures. See Fig. 1 and Fig. 2. FIG. 1 Components of a Hemostatic Forceps FIG. 2 Typical Types of Serrations and Teeth for Hemostatic Forceps 1.2 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F921-10(2018) is classified under the following ICS (International Classification for Standards) categories: 01.040.11 - Health care technology (Vocabularies); 11.040.30 - Surgical instruments and materials. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F921-10(2018) has the following relationships with other standards: It is inter standard links to ASTM F921-10(2011), ASTM F899-23, ASTM F899-20, ASTM F899-19, ASTM F899-12b, ASTM F899-12a, ASTM F899-12, ASTM F899-11, ASTM F899-10, ASTM F899-09e1, ASTM F899-09, ASTM F899-07, ASTM F899-95, ASTM F899-02, ASTM F1613-95(2023). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F921-10(2018) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F921 − 10 (Reapproved 2018)
Standard Terminology Relating to
Hemostatic Forceps
ThisstandardisissuedunderthefixeddesignationF921;thenumberimmediatelyfollowingthedesignationindicatestheyearoforiginal
adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.Asuperscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope finger rings—the feature of both the female and the male
members that forms the gripping surface for the surgeon
1.1 This terminology covers basic terms and considerations
(commonly classified as the ring-handled feature in ISO
for the components of hemostatic forceps. Instruments in this
7151).
terminology are limited to those fabricated from stainless steel
and for general surgical procedures. See Fig. 1 and Fig. 2.
hemostatic forceps—an instrument, available in various sizes
and configurations, used in surgical procedures for the
1.2 This international standard was developed in accor-
compression of blood vessels and the grasping of tissue.
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the
jaws—parts that contain serrations to interrupt the flow of
Development of International Standards, Guides and Recom-
blood through any vessel.
mendations issued by the World Trade Organization Technical
male member—the component that is inserted through the
Barriers to Trade (TBT) Committee.
femalememberandsecuredtothefemalememberatthebox
lock junction.
2. Referenced Documents
proximal end—that portion of the instrument that is closest to
2.1 ASTM Standards:
F899 Specification for Wrought Stainless Steels for Surgical the surgeon when in use.
Instruments
ratchets—the portion of both the female and male members at
2.2 ISO Standard:
the proximal end possessing inclined teeth and that form the
ISO 7151 Instruments for Surgery—Hemostatic Forceps—
locking mechanism.
General Requirements
serrations or teeth—the gripping or clamping surfaces of the
jaws or ratchets.
3. Terminology
shank—the part of either the female or the male member that
Definitions of Hemostatic Forceps
yields configuration, length, and leverage.
box lock—thejunctionwherethefemalememberandthemale
Definitions of Physical Properties of Hemostatic Forceps
member are secured, forming the pivoting feature.
chamfer—the broken edge of the jaw serrations and the
distal end—the working end, comprised of two jaws, that is
external edges of the box lock surfaces.
furthest from the surgeon when in use.
corrosion—the formation of rust.
female member—the component that accommodates and
encloses the male member at the box lock junction.
elasticity—the capacity of the instrument to undergo induced
stress without permanent distortion or breakage of any
component.
ThisterminologyisunderthejurisdictionofASTMCommitteeF04onMedical
finish, n—final surface visual appearance classified as follows:
and Surgical Materials and Devices and is the direct responsibility of Subcommittee
bright or mirror finish, n—highly reflective surface.
F04.33 on Medical/Surgical Instruments.
Current edition approved Oct. 1, 2018. Published November 2018. Originally
satin, matte, or black finish, n—reduced reflective surface
approved in 1985. Last previous edition approved in 2011 as F921 – 10(2011). DOI:
(as compared to bright
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...