ASTM D5827-22
(Test Method)Standard Test Method for Analysis of Engine Coolant for Chloride and Other Anions by Ion Chromatography
Standard Test Method for Analysis of Engine Coolant for Chloride and Other Anions by Ion Chromatography
SIGNIFICANCE AND USE
4.1 This test method provides for the qualitative and quantitative determination of common anions in engine coolant in the milligrams per litre to low percent range and requires only a few millilitres or microlitres of sample per test, with results available in less than 30 min. Acceptable levels of chloride and other anions vary with manufacturer's blending specifications and applicable ASTM minimum or maximum specifications.
SCOPE
1.1 This test method covers the chemical analysis of engine coolant for chloride ion by high-performance ion chromatography (HPIC). Several other common anions found in engine coolant can be determined in one chromatographic analysis by this test method.
1.2 This test method is applicable to both new and used engine coolant.
1.3 Coelution of other ions may cause interferences for any of the listed anions. In the case of unfamiliar formulations, identification verification should be performed by either or both fortification and dilution of the sample matrix with the anions of interest.
1.4 Analysis can be performed directly by this test method without pretreatment, other than dilution, as required by the linear ranges of the equipment. Table 1 indicates several applicable anions and approximate detection limits.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to its use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-May-2022
- Technical Committee
- D15 - Engine Coolants and Related Fluids
- Drafting Committee
- D15.04 - Chemical Properties
Relations
- Effective Date
- 01-Oct-2019
- Effective Date
- 01-May-2014
- Effective Date
- 01-Apr-2014
- Effective Date
- 01-May-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Oct-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Apr-2008
- Refers
ASTM E177-06b - Standard Practice for Use of the Terms Precision and Bias in ASTM Test Methods - Effective Date
- 15-Nov-2006
- Refers
ASTM E177-06a - Standard Practice for Use of the Terms Precision and Bias in ASTM Test Methods - Effective Date
- 01-Nov-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 01-Nov-2005
- Effective Date
- 01-Nov-2004
Overview
ASTM D5827-22: Standard Test Method for Analysis of Engine Coolant for Chloride and Other Anions by Ion Chromatography provides an internationally recognized procedure for determining the qualitative and quantitative presence of chloride and other common anions in engine coolant. Developed by ASTM International, this test method enables laboratories and manufacturers to evaluate both new and used engine coolants quickly and efficiently, facilitating quality assurance, regulatory compliance, and optimized coolant formulation. The method is precise, requiring only small sample volumes and delivering results in less than 30 minutes.
Keywords: ASTM D5827-22, engine coolant analysis, chloride detection, anions, ion chromatography, laboratory testing, coolant quality.
Key Topics
- Test Scope: The method applies to both new and used engine coolants and assesses chloride as well as other common anions (such as nitrite, bromide, nitrate, phosphate, sulfate, and oxalate).
- Analytical Technique: Utilizes high-performance ion chromatography (HPIC) to enable reliable, simultaneous multi-analyte detection in a single run.
- Detection Limits: The technique offers detection from milligrams per liter to low percent ranges, supporting stringent product and regulatory standards.
- Interference and Verification: Includes guidance for mitigating analytical interferences-such as coeluting ions or matrix effects-through sample dilution, matrix spiking, and identification verification.
- Efficiency: Requires minimal sample preparation beyond dilution; standard SI units apply throughout for universal measurement reference.
- Precision and Quality Control: Incorporates duplicate analysis, matrix spikes, calibration standards, and routine checks to maintain method accuracy and reproducibility.
- Safety and Compliance: Stresses user responsibility for safety and environmental practices and recognizes WTO standardization principles in development.
Applications
- Quality Assurance in Manufacturing: Engine coolant manufacturers use ASTM D5827-22 to verify that factory blends meet formulation specifications for anion content, including chloride, which is critical for corrosion prevention.
- Used Coolant Evaluation and Recycling: Laboratories analyze used engine coolant samples to assess ongoing performance, contaminant ingress, or suitability for recycling and re-use.
- Regulatory Compliance: The method supports compliance with industry guidelines and customer requirements by providing objective, standardized anion concentration data.
- Formulation Research and Development: Researchers deploy the test method to evaluate new coolant chemistries, optimize performance, or troubleshoot formulation issues related to anion contamination or depletion.
- Laboratory Accreditation and Routine Testing: Accrediting bodies and service laboratories reference ASTM D5827-22 as a reliable protocol for routine coolant analysis, supporting cross-laboratory data comparability and quality.
Related Standards
- ASTM D1193 - Specification for Reagent Water: Defines water purity requirements for sample and solution preparation in laboratory testing.
- ASTM D1176 - Practice for Sampling and Preparing Aqueous Solutions of Engine Coolants or Antirusts for Testing: Provides sampling guidance to ensure representative, contamination-free coolant specimen collection.
- ASTM E691 - Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method: Supports the determination of precision and reproducibility for test methods.
- ASTM E177 - Practice for Use of the Terms Precision and Bias in ASTM Test Methods: Clarifies definitions and reporting of analytical method accuracy.
By adhering to ASTM D5827-22, organizations in the automotive, chemical, and laboratory testing sectors ensure high standards for engine coolant quality, safety, and performance. The method’s rapid, reliable measurement of chloride and other anions supports proactive maintenance, regulatory adherence, and innovation in coolant technology.
Buy Documents
ASTM D5827-22 - Standard Test Method for Analysis of Engine Coolant for Chloride and Other Anions by Ion Chromatography
REDLINE ASTM D5827-22 - Standard Test Method for Analysis of Engine Coolant for Chloride and Other Anions by Ion Chromatography
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D5827-22 is a standard published by ASTM International. Its full title is "Standard Test Method for Analysis of Engine Coolant for Chloride and Other Anions by Ion Chromatography". This standard covers: SIGNIFICANCE AND USE 4.1 This test method provides for the qualitative and quantitative determination of common anions in engine coolant in the milligrams per litre to low percent range and requires only a few millilitres or microlitres of sample per test, with results available in less than 30 min. Acceptable levels of chloride and other anions vary with manufacturer's blending specifications and applicable ASTM minimum or maximum specifications. SCOPE 1.1 This test method covers the chemical analysis of engine coolant for chloride ion by high-performance ion chromatography (HPIC). Several other common anions found in engine coolant can be determined in one chromatographic analysis by this test method. 1.2 This test method is applicable to both new and used engine coolant. 1.3 Coelution of other ions may cause interferences for any of the listed anions. In the case of unfamiliar formulations, identification verification should be performed by either or both fortification and dilution of the sample matrix with the anions of interest. 1.4 Analysis can be performed directly by this test method without pretreatment, other than dilution, as required by the linear ranges of the equipment. Table 1 indicates several applicable anions and approximate detection limits. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to its use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This test method provides for the qualitative and quantitative determination of common anions in engine coolant in the milligrams per litre to low percent range and requires only a few millilitres or microlitres of sample per test, with results available in less than 30 min. Acceptable levels of chloride and other anions vary with manufacturer's blending specifications and applicable ASTM minimum or maximum specifications. SCOPE 1.1 This test method covers the chemical analysis of engine coolant for chloride ion by high-performance ion chromatography (HPIC). Several other common anions found in engine coolant can be determined in one chromatographic analysis by this test method. 1.2 This test method is applicable to both new and used engine coolant. 1.3 Coelution of other ions may cause interferences for any of the listed anions. In the case of unfamiliar formulations, identification verification should be performed by either or both fortification and dilution of the sample matrix with the anions of interest. 1.4 Analysis can be performed directly by this test method without pretreatment, other than dilution, as required by the linear ranges of the equipment. Table 1 indicates several applicable anions and approximate detection limits. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to its use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D5827-22 is classified under the following ICS (International Classification for Standards) categories: 71.100.45 - Refrigerants and antifreezes. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D5827-22 has the following relationships with other standards: It is inter standard links to ASTM D1176-14(2019), ASTM E177-14, ASTM D1176-14, ASTM E691-13, ASTM E177-13, ASTM E691-11, ASTM E177-10, ASTM E691-08, ASTM E177-08, ASTM D1176-98(2008), ASTM E177-06b, ASTM E177-06a, ASTM D1193-06, ASTM E691-05, ASTM E177-04. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D5827-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D5827 − 22
Standard Test Method for
Analysis of Engine Coolant for Chloride and Other Anions
by Ion Chromatography
This standard is issued under the fixed designation D5827; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 2. Referenced Documents
2.1 ASTM Standards:
1.1 This test method covers the chemical analysis of engine
D1193Specification for Reagent Water
coolant for chloride ion by high-performance ion chromatog-
D1176Practice for Sampling and Preparing Aqueous Solu-
raphy (HPIC). Several other common anions found in engine
tionsofEngineCoolantsorAntirustsforTestingPurposes
coolant can be determined in one chromatographic analysis by
E691Practice for Conducting an Interlaboratory Study to
this test method.
Determine the Precision of a Test Method
1.2 This test method is applicable to both new and used E177Practice for Use of the Terms Precision and Bias in
engine coolant. ASTM Test Methods
1.3 Coelution of other ions may cause interferences for any
3. Summary of Test Method
of the listed anions. In the case of unfamiliar formulations,
3.1 A small volume of working sample is prepared by
identification verification should be performed by either or
dilution of the sample with the method eluant. This diluted
both fortification and dilution of the sample matrix with the
sample is filtered and pumped through two ion exchange
anions of interest.
columnsandasuppressorandintoaconductivitydetector.Ions
are separated based on their affinity for exchange sites of the
1.4 Analysis can be performed directly by this test method
resin with respect to the resin’s affinity for the eluant. The
without pretreatment, other than dilution, as required by the
suppressor increases the sensitivity of the method by both
linear ranges of the equipment. Table 1 indicates several
increasing the conductivity of the analytes and decreasing the
applicable anions and approximate detection limits.
conductivity of the eluant. The suppressor converts the eluant
1.5 The values stated in SI units are to be regarded as and the analytes to the corresponding hydrogen form acids.
standard. No other units of measurement are included in this Anions are quantitated by integration of their response com-
pared with an external calibration curve and are reported as
standard.
milligrams per litre (mg/L).
1.6 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
4. Significance and Use
responsibility of the user of this standard to establish appro-
4.1 This test method provides for the qualitative and quan-
priate safety, health, and environmental practices and deter-
titative determination of common anions in engine coolant in
mine the applicability of regulatory limitations prior to its use.
the milligrams per litre to low percent range and requires only
1.7 This international standard was developed in accor-
a few millilitres or microlitres of sample per test, with results
dance with internationally recognized principles on standard-
availableinlessthan30min.Acceptablelevelsofchlorideand
ization established in the Decision on Principles for the
other anions vary with manufacturer’s blending specifications
Development of International Standards, Guides and Recom-
and applicable ASTM minimum or maximum specifications.
mendations issued by the World Trade Organization Technical
5. Interferences
Barriers to Trade (TBT) Committee.
5.1 Interferences can be caused by substances with similar
retention times, especially if they are in high concentration
This test method is under the jurisdiction ofASTM Committee D15 on Engine
Coolants and Related Fluids and is the direct responsibility of Subcommittee
D15.04 on Chemical Properties. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved June 1, 2022. Published June 2022. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1995. Last previous edition approved in 2015 as D5827–09(2015). Standards volume information, refer to the standard’s Document Summary page on
DOI: 10.1520/D5827-22. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5827 − 22
TABLE 1 Analytes and Minimum Detection Limits
6.8 Drying Oven, controlled at 105°C, 150°C, and 600°C
A
Analyte Detection Limit, mg/L 6 5°C.
−
Chloride (Cl)2.0
6.9 Desiccator.
−
Nitrite (NO)5.0
Bromide (Br) 4.0
− 7. Reagents
Nitrate (NO)7.1
2−
o-Phosphate (HPO ) 20.0
7.1 Purity of Reagents—Reagent grade or higher purity
2−
Sulfate (SO ) 8.0
2−
chemicals shall be used for the preparation of all samples,
Oxalate (C O ) 12.0
2 4
standards, eluants, and regenerator solutions. Unless otherwise
A
Determined using 100 µL sample volume. Sample diluted 99 + 1 (v/v) with
chromatographic eluant 30 µS ⁄cm full scale, suppressed conductivity detection. indicated, it is intended that all reagents conform to the
Dionex AS4ASC column with AG4ASC guard columns. Other systems will require
specifications of the Committee onAnalytical Reagents of the
MDL determinations using chosen dilution factors, eluants, columns, and detector.
American Chemical Society, where such specification are
available. Other grades may be used, provided it is first
ascertained that the reagent is of sufficiently high purity to
compared to those of the analyte of interest. Sample dilution
permit its use without lessening the accuracy of the determi-
will be used to minimize or solve most interference problems.
nation.
5.2 A water dip (solvent system peak) can cause interfer-
7.2 Purity of Water—Unless otherwise indicated, references
ence with some integrators.This is eliminated by dilution with
to water shall be understood to mean reagent water as defined
the eluant if the sample dilution factor is 49+1 (v⁄v) or
by Type II of Specification D1193. It is recommended that all
greater. Below this dilution, it is best to add a spike of eluant
waterbefilteredthrougha0.2µmfilter.Foreluantpreparation,
concentrate to the sample such that the sample is not diluted
degas the water by sparging with helium or vacuum degassing
significantly and the resulting test solution matches the eluant
and sonication.
used in the system. One method is the addition of 100 µL of
100X eluant concentrate to 10.0 mL of sample or standard.
7.3 Eluant Buffer Stock Solution—Sodium bicarbonate
(NaHCO ) 1.5 mM and sodium carbonate (Na CO ) 1.2 mM.
5.3 Method interferences can be caused by the contamina-
3 2 3
Dissolve 2.5203g 6 0.0005 g of NaHCO and 2.5438g 6
tion of glassware, eluant, reagents, etc. Great care must be
0.0005 g of Na CO in reagent water in a 1000mL Type A
taken to ensure that contamination, especially by chloride, is
2 3
volumetric flask and dilute to 1 L. Dilute 100.0mL of this
kept at the lowest possible levels.
stock solution to 2000 mL in a 2L Type A volumetric flask
5.4 Pre-rinsing of the sample preparation containers with
with degassed reagent water. The pH of the stock solution is
deionized water is mandatory.
10.1 to 10.3 (based on pK calculation). The eluant solution
a
5.5 The use of latex gloves is highly recommended to
usedmaybedifferentifothersystemoranalyticalcolumnsare
prevent contamination.
used.
6. Apparatus 7.4 Stock Bromide Solution—Dry approximately2gof
sodium bromide (NaBr) for 6 h at 150°C and cool in a
6.1 Analytical Balance, capable of weighing accurately to
desiccator. Weigh and dissolve 1.2877 g of the dried salt in
0.0001 g.
reagent water and dilute to 1 L (1.00 mL=1.00 mg bromide).
6.2 Ion Chromatograph—Analytical system with all re-
7.5 Stock Chloride Solution—Dry approximately2gof
quired accessories including syringes, columns, suppressor,
sodium chloride (NaCl) for1hat600°C and cool in a
gasses, and detector. Column life and performance are en-
desiccator. Weigh and dissolve 1.6485 g and dilute to 1 Lwith
hanced by the use of a two-eluant channel gradient pump, if
−
reagent water (1.00 mL=1.00 mg Cl ).
available.
7.6 Stock Formate Solution—Dry approximately2gof
6.3 Guard Column, for protection of the analytical column
sodium formate (NaHCO ) at 105°C for 6 h and cool in a
from strongly retained constituents. Better separations are
desiccator. Weigh and dissolve 1.4775 g of the salt in reagent
obtained with additional plates.
water and dilute to 1 L (1.00 mL=1.00 mg formic acid).
6.4 Anion Separator Column, capable of producing analyte
7.7 Stock Glycolic Acid Solution—Weigh and dissolve
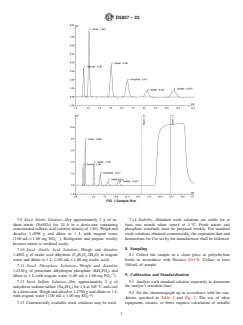
separation equivalent to or better than that shown in Fig. 1.
1.0000 g of the solid acid in reagent water and dilute to 1 L
6.5 Anion Suppressor Device—Micro membrane suppressor
(1.00 mL=1.00 mg glycolate).
or equivalent.Acation exchange column in the hydrogen form
7.8 Stock Nitrate Solution—Dry approximately2gofso-
has been used successfully, but it will periodically need to be
dium nitrate (NaNO ) for 24 h at 105°C and cool in a
regenerated as required, being indicated by a high background
desiccator. Weigh and dissolve 1.3707 g and dilute to 1 Lwith
conductivity and low analyte response.
−
reagent water (1.00 mL=1.00 mg NO ).
6.6 Conductivity Detector, low volume (<2 µL) and flow,
temperature compensated, capable of at least 0 to 1000 µS/cm
Reagent Chemicals, American Chemical Society Specifications, American
on a linear scale.
Chemical Society, Washington, DC. For suggestions on the testing of reagents not
listed by the American Chemical Society, see Analar Standards for Laboratory
6.7 Integrator or Chromatography Data System Software,
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
capable of obtaining approximately the same detection limits
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
as are listed in Table 1. MD.
D5827 − 22
FIG. 1 Sample Run
7.9 Stock Nitrite Solution—Dry approximately2gofso- 7.14 Stability—Standard stock solutions are stable for at
dium nitrite (NaNO ) for 24 h in a desiccator containing least one month when stored at 4°C. Fresh nitrite and
concentratedsulfuricacid(relativedensityof1.84).Weighand phosphate standards must be prepared weekly. For standard
dissolve 1.4998 g and dilute to 1 L with reagent water stock solutions obtained commercially, the expiration date and
−
(1.00mL=1.00 mg NO ). Refrigerate and prepare weekly Instructions for Use set by the manufacturer shall be followed.
because nitrite is oxidized easily.
8. Sampling
7.10 Stock Oxalic Acid Solu
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D5827 − 09 (Reapproved 2015) D5827 − 22
Standard Test Method for
Analysis of Engine Coolant for Chloride and Other Anions
by Ion Chromatography
This standard is issued under the fixed designation D5827; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the chemical analysis of engine coolant for chloride ion by high-performance ion chromatography
(HPIC). Several other common anions found in engine coolant can be determined in one chromatographic analysis by this test
method.
1.2 This test method is applicable to both new and used engine coolant.
1.3 Coelution of other ions may cause interferences for any of the listed anions. In the case of unfamiliar formulations,
identification verification should be performed by either or both fortification and dilution of the sample matrix with the anions of
interest.
1.4 Analysis can be performed directly by this test method without pretreatment, other than dilution, as required by the linear
ranges of the equipment. Table 1 indicates several applicable anions and approximate detection limits.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to its use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
D1176 Practice for Sampling and Preparing Aqueous Solutions of Engine Coolants or Antirusts for Testing Purposes
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E177 Practice for Use of the Terms Precision and Bias in ASTM Test Methods
This test method is under the jurisdiction of ASTM Committee D15 on Engine Coolants and Related Fluids and is the direct responsibility of Subcommittee D15.04 on
Chemical Properties.
Current edition approved May 1, 2015June 1, 2022. Published June 2015June 2022. Originally approved in 1995. Last previous edition approved in 20092015 as
ε1
D5827D5827–09(2015).-09 . DOI: 10.1520/D5827-09R15.10.1520/D5827-22.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’sstandard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5827 − 22
TABLE 1 Analytes and Minimum Detection Limits
A
Analyte Detection Limit, mg/L
−
Chloride (Cl ) 2.0
−
Nitrite (NO ) 5.0
Bromide (Br) 4.0
−
Nitrate (NO ) 7.1
2−
o-Phosphate (HPO ) 20.0
2−
Sulfate (SO ) 8.0
2−
Oxalate (C O ) 12.0
2 4
A
Determined using 100-μL100 μL sample volume. Sample diluted 99 + 1 (v/v)
with chromatographic eluant 30-μS/cm30 μS ⁄cm full scale, suppressed conduc-
tivity detection. Dionex AS4ASC column with AG4ASC guard columns. Other
systems will require MDL determinations using chosen dilution factors, eluants,
columns, and detector.
3. Summary of Test Method
3.1 A small volume of working sample is prepared by dilution of the sample with the method eluant. This diluted sample is filtered
and pumped through two ion exchange columns and a suppressor and into a conductivity detector. Ions are separated based on their
affinity for exchange sites of the resin with respect to the resin’sresin’s affinity for the eluant. The suppressor increases the
sensitivity of the method by both increasing the conductivity of the analytes and decreasing the conductivity of the eluant. The
suppressor converts the eluant and the analytes to the corresponding hydrogen form acids. Anions are quantitated by integration
of their response compared with an external calibration curve and are reported as milligrams per litre (mg/L).
4. Significance and Use
4.1 This test method provides for the qualitative and quantitative determination of common anions in engine coolant in the
milligrams per litre to low percent range and requires only a few millilitres or microlitres of sample per test, with results available
in less than 30 min. Acceptable levels of chloride and other anions vary with manufacturer’s blending specifications and applicable
ASTM minimum or maximum specifications.
5. Interferences
5.1 Interferences can be caused by substances with similar retention times, especially if they are in high concentration compared
to those of the analyte of interest. Sample dilution will be used to minimize or solve most interference problems.
5.2 A water dip (solvent system peak) can cause interference with some integrators. This is eliminated by dilution with the eluant
if the sample dilution factor is 49 + 1 (v/v)(v ⁄v) or greater. Below this dilution, it is best to add a spike of eluant concentrate to
the sample such that the sample is not diluted significantly and the resulting test solution matches the eluant used in the system.
One method is the addition of 100 μL of 100X eluant concentrate to 10.0 mL of sample or standard.
5.3 Method interferences can be caused by the contamination of glassware, eluant, reagents, etc. Great care must be taken to
ensure that contamination, especially by chloride, is kept at the lowest possible levels.
5.4 Pre-rinsing of the sample preparation containers with deionized water is mandatory.
5.5 The use of latex gloves is highly recommended to prevent contamination.
6. Apparatus
6.1 Analytical Balance, capable of weighing accurately to 0.0001 g.
6.2 Ion Chromatograph—Analytical system with all required accessories including syringes, columns, suppressor, gasses, and
detector. Column life and performance are enhanced by the use of a two-eluant channel gradient pump, if available.
6.3 Guard Column, for protection of the analytical column from strongly retained constituents. Better separations are obtained
with additional plates.
D5827 − 22
6.4 Anion Separator Column, capable of producing analyte separation equivalent to or better than that shown in Fig. 1.
6.5 Anion Suppressor Device—Micro membrane suppressor or equivalent. A cation exchange column in the hydrogen form has
been used successfully, but it will periodically need to be regenerated as required, being indicated by a high background
conductivity and low analyte response.
6.6 Conductivity Detector, low volume (<2 μL) and flow, temperature compensated, capable of at least 0 to 1000 μS/cm on a linear
scale.
6.7 Integrator or Chromatography Data System Software, capable of obtaining approximately the same detection limits as are
listed in Table 1.
6.8 Drying Oven, controlled at 105, 150,105 °C, 150 °C, and 600600 °C 6 5°C.5 °C.
6.9 Desiccator.
FIG. 1 Sample Run—Chloride Peak at 1.7 minRun
D5827 − 22
7. Reagents
7.1 Purity of Reagents—Reagent grade or higher purity chemicals shall be used for the preparation of all samples, standards,
eluants, and regenerator solutions. Unless otherwise indicated, it is intended that all reagents conform to the specifications of the
Committee on Analytical Reagents of the American Chemical Society, where such specification are available. Other grades may
be used, provided it is first ascertained that the reagent is of sufficiently high purity to permit its use without lessening the accuracy
of the determination.
7.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean reagent water as defined by Type
II of Specification D1193. It is recommended that all water be filtered through a 0.2-μm0.2 μm filter. For eluant preparation, degas
the water by sparging with helium or vacuum degassing and sonication.
7.3 Eluant Buffer Stock Solution—Sodium bicarbonate (NaHCO ) 1.5 mM and sodium carbonate (Na CO ) 1.2 mM. Dissolve
3 2 3
2.52032.5203 g 6 0.0005 g of NaHCO and 2.54382.5438 g 6 0.0005 g of Na CO in reagent water in a 1000-mL1000 mL Type
3 2 3
A volumetric flask and dilute to 1 L. Dilute 100.0 mL 100.0 mL of this stock solution to 2000 mL in a 2-L2 L Type A volumetric
flask with degassed reagent water. The pH of the stock solution is 10.1 to 10.3 (based on pK calculation). The eluant solution used
a
may be different if other system or analytical columns are used.
7.4 Stock Bromide Solution—Dry approximately 2 g of sodium bromide (NaBr) for 6 h at 150°C150 °C and cool in a desiccator.
Weigh and dissolve 1.2877 g of the dried salt in reagent water and dilute to 1 L (1.00 mL = 1.00 mg bromide).
7.5 Stock Chloride Solution—Dry approximately 2 g of sodium chloride (NaCl) for 1 h at 600°C600 °C and cool in a desiccator.
−
Weigh and dissolve 1.6485 g and dilute to 1 L with reagent water (1.00 mL = 1.00 mg Cl ).
7.6 Stock Formate Solution—Dry approximately 2 g of sodium formate (NaHCO ) at 105°C105 °C for 6 h and cool in a desiccator.
Weigh and dissolve 1.4775 g of the salt in reagent water and dilute to 1 L (1.00 mL = 1.00 mg formic acid).
7.7 Stock Glycolic Acid Solution—Weigh and dissolve 1.0000 g of the solid acid in reagent water and dilute to 1 L (1.00 mL = 1.00
mg glycolate).
7.8 Stock Nitrate Solution—Dry approximately 2 g of sodium nitrate (NaNO ) for 24 h at 105°C105 °C and cool in a desiccator.
−
Weigh and dissolve 1.3707 g and dilute to 1 L with reagent water (1.00 mL = 1.00 mg NO ).
7.9 Stock Nitrite Solution—Dry approximately 2 g of sodium nitrite (NaNO ) for 24 h in a desiccator containing concentrated
sulfuric acid (relative density of 1.84). Weigh and dissolve 1.4998 g and dilute to 1 L with reagent water (1.00 mL = 1.00
−
(1.00 mL = 1.00 mg NO ). Refrigerate and prepare weekly because nitrite is oxidized easily.
7.10 Stock Oxalic Acid Solution—Weigh and dissolve 1.4002 g of oxalic acid dihydrate (C H O ·2H O) in reagent water and
2 2 4 2
dilute to 1 L (1.00 mL = 1.00 mg oxalic acid).
7.11 Stock Phosphate Solution—Weigh and dissolve 1.4330 g 1.4330 g of potassium dihydrogen phosphate (KH PO ) and dilute
2 4
−3
to 1 L with reagent water (1.00 mL = 1.00 mg PO ).
7.12 Stock Sulfate Solution—Dry approximately 2 g of anhydrous sodium sulfate (Na SO ) for 1 h at 105°C105 °C and cool in
2 4
−2
a desiccator. Weigh and dissolve 1.4790 g and dilute to 1 L with reagent water (1.00 mL = 1.00 mg SO ).
7.13 Suppressor Solution for Membrane Suppressor—0.025Commercially Navailable Hstock SO . Carefully add 13.7 mL of
2 4
reagent sulfuric acid (relative density of 1.84) to approximately 500 mL reagent water in a 1-L volumetric flask. Dilute to 1000
mL with reagent water. Dilute 100 mL of this concentrate to 2000 mL with reagent water for the final working suppressor solution.
solutions may be used.
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by
the American Chemical Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...