ASTM F2577-22
(Guide)Standard Guide for Compositional Evaluation of Declarable Substances and Substances of Concern for Materials in Products
Standard Guide for Compositional Evaluation of Declarable Substances and Substances of Concern for Materials in Products
SIGNIFICANCE AND USE
4.1 Regulations, standards, and market-defined requirements for chemical constituent (“substances of interest”) compliance and conformance have become increasing numerous and complex. Specific laws and standards may pertain to certain product and industry segments, for example, electrical/electronic instruments and components, pharmaceuticals, medical devices, consumer products, agrichemicals, and so forth. Others may be broader and relate specifically to assessment and management of specific chemical compounds or classes across multiple product sectors and sources, for example Ref (2). In addition, such requirements may be issued by national authorities, international standards setting groups, or, in the case of market-defined requirements, even by customer advocacy organizations or customers through supply contracts. The resulting global landscape of requirements and market access expectations is complex, and compliance/conformance presents numerous challenges for manufacturers.
Note 1: For example, IEC 62474:2012 provides some standardized definitions for reporting thresholds and declaration statements for electrotechnical industry. In another example, ISO 14021:2016 addresses self-declared environmental claims.
4.2 Declarable substances may be found on various lists and forms, including those listed in Refs (2-10) and on supply agreements and questionnaires. This guide is not intended to be exhaustive nor cover all declarable substances. Nor does this guide address specific declaration and labeling requirements within these regulation and standards nor address product safety and compliance requirements as dictated by law for specific products, industries, or market regions.
4.3 In addition to new laws and standards, more chemical substances are continually added to a variety of screening lists for review of potential hazards, identification and quantification of possible health or environmental hazards, or both, and consideration of control measures. To ...
SCOPE
1.1 This guide uses case studies to illustrate the decision process to assess materials and products for declarable substances when evaluating conformance to relevant requirements. This may be accomplished by applying existing knowledge to determine the need for further action (for example, testing).
1.2 This guide assists in utilization and interpretation of various forms of information gathered to enable compliance or conformance or both to regulations, standards, supply agreements, or customer enquiries related to identified declarable substances, including the evidence required to issue declarations for the absence or presence of a declarable substance. Several examples are referenced. The target declarable substances will be specific to the product and product classification, the regulatory jurisdiction, customer/supplier requirements, and other relevant considerations. Consideration of which regulations and standards apply to a given product is beyond the scope of this guide.
1.3 The framework covered in this guide attempts to harmonize language used in the absence of objective data or specific regulatory requirements or both. This guide draws on a variety of existing documentation, which will be cited and referenced, as well as basic scientific principles for communication of chemical hazard and risk, and may be used as an approach for assessing composition of products and their components as part of product risk assessment.
1.4 This guide is applicable to a variety of materials, including polymeric and elastomeric materials, which are used in regulated industries and products.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international st...
General Information
- Status
- Published
- Publication Date
- 30-Sep-2022
- Technical Committee
- F40 - Declarable Substances in Materials
- Drafting Committee
- F40.02 - Management Practices and Guides
Relations
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Jan-2020
- Effective Date
- 01-Dec-2019
- Effective Date
- 01-Aug-2019
- Effective Date
- 15-Apr-2019
- Effective Date
- 01-Feb-2019
- Effective Date
- 01-Feb-2019
- Effective Date
- 01-Dec-2018
- Effective Date
- 01-Nov-2018
- Effective Date
- 15-Aug-2017
- Effective Date
- 01-Aug-2017
- Effective Date
- 01-Jul-2016
- Effective Date
- 01-Jul-2016
- Effective Date
- 01-Aug-2015
Overview
ASTM F2577-22 is a standardized guide developed by ASTM International for the compositional evaluation of declarable substances and substances of concern in materials used in products. With increasing complexity in global regulations and customer-specific requirements for chemical constituents, this guide provides a structured approach to assess material compositions and enables manufacturers to achieve compliance with relevant regulations, standards, supply agreements, and market demands.
The guide applies to a wide range of industry sectors, including electrotechnical, medical devices, pharmaceuticals, consumer products, and agrichemicals. It establishes methodologies for assessing the presence and concentration of substances of concern, offering practical case studies and flowcharts to illustrate the decision-making process.
Key Topics
- Declarable Substances: Identification and evaluation of substances that must be reported or restricted according to statutes, contractual requirements, or industry standards.
- Compliance Processes: Systematic steps for assessing conformance with various chemical reporting and restriction requirements, such as determining applicable regulations, the basis for conformance (per product or material), and documentation needs.
- A Priori and A Posteriori Knowledge: Utilization of scientific deduction (a priori) and empirical data (a posteriori) to assess if substances of concern are present above defined thresholds, supplemented by appropriate test methods as necessary.
- Material-Specific Considerations: Guidance is given for a variety of materials, including polymers, elastomers, and metals, recognizing distinct challenges in analyzing different product types and formulations.
- Interpretation and Communication: Harmonization of terminology for material declarations, fostering clearer supplier-customer communication (e.g., terms like “not intentionally added” or “below de minimis levels”).
- Risk Assessment: Approaches to determine when further testing is required versus when supplier declarations or process knowledge suffice.
Applications
- Product Compliance Documentation: Supports development of robust documentation to demonstrate conformance to global regulations such as RoHS, REACH, and WEEE.
- Supplier Management and Declarations: Provides manufacturers, importers, and assemblers with frameworks to assess, validate, and communicate the presence or absence of declarable substances through the supply chain.
- Quality Assurance in Regulated Industries: Useful for producers of electrical and electronic instruments, medical devices, consumer products, and more, when managing exposure to restricted substances.
- Process Optimization: Enables efficient allocation of resources by distinguishing circumstances where a priori knowledge is adequate versus when laboratory testing or analytical verification is necessary.
- Customer and Regulatory Inquiries: Assists businesses in responding to customer-specific environmental, safety, and regulatory requirements regarding chemical compositions.
Related Standards
Users of ASTM F2577-22 often reference other key standards to ensure comprehensive compliance and effective material declarations, including:
- ASTM F2576: Terminology Relating to Declarable Substances in Materials
- ASTM F2931: Guide for Analytical Testing of Substances of Very High Concern in Materials and Products
- IEC 62474: Material Declaration for Products of and for the Electrotechnical Industry
- ISO 14021: Environmental Labels and Declarations – Self-Declared Environmental Claims
- ISO 9001: Quality Management Systems – Requirements
- ISO 13485: Medical Devices – Quality Management Systems – Requirements for Regulatory Purposes
- EU RoHS Directive (2011/65/EU)
- IEC 62321-8: Determination of Certain Substances in Electrotechnical Products
Keywords: ASTM F2577-22, declarable substances, substances of concern, material compliance, chemical composition, product conformity, restricted substances, supplier declarations, regulatory compliance, RoHS, REACH, ISO standards, product testing, material declaration, risk assessment.
Buy Documents
ASTM F2577-22 - Standard Guide for Compositional Evaluation of Declarable Substances and Substances of Concern for Materials in Products
REDLINE ASTM F2577-22 - Standard Guide for Compositional Evaluation of Declarable Substances and Substances of Concern for Materials in Products
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

Bureau Veritas
Bureau Veritas is a world leader in laboratory testing, inspection and certification services.

DNV
DNV is an independent assurance and risk management provider.
Sponsored listings
Frequently Asked Questions
ASTM F2577-22 is a guide published by ASTM International. Its full title is "Standard Guide for Compositional Evaluation of Declarable Substances and Substances of Concern for Materials in Products". This standard covers: SIGNIFICANCE AND USE 4.1 Regulations, standards, and market-defined requirements for chemical constituent (“substances of interest”) compliance and conformance have become increasing numerous and complex. Specific laws and standards may pertain to certain product and industry segments, for example, electrical/electronic instruments and components, pharmaceuticals, medical devices, consumer products, agrichemicals, and so forth. Others may be broader and relate specifically to assessment and management of specific chemical compounds or classes across multiple product sectors and sources, for example Ref (2). In addition, such requirements may be issued by national authorities, international standards setting groups, or, in the case of market-defined requirements, even by customer advocacy organizations or customers through supply contracts. The resulting global landscape of requirements and market access expectations is complex, and compliance/conformance presents numerous challenges for manufacturers. Note 1: For example, IEC 62474:2012 provides some standardized definitions for reporting thresholds and declaration statements for electrotechnical industry. In another example, ISO 14021:2016 addresses self-declared environmental claims. 4.2 Declarable substances may be found on various lists and forms, including those listed in Refs (2-10) and on supply agreements and questionnaires. This guide is not intended to be exhaustive nor cover all declarable substances. Nor does this guide address specific declaration and labeling requirements within these regulation and standards nor address product safety and compliance requirements as dictated by law for specific products, industries, or market regions. 4.3 In addition to new laws and standards, more chemical substances are continually added to a variety of screening lists for review of potential hazards, identification and quantification of possible health or environmental hazards, or both, and consideration of control measures. To ... SCOPE 1.1 This guide uses case studies to illustrate the decision process to assess materials and products for declarable substances when evaluating conformance to relevant requirements. This may be accomplished by applying existing knowledge to determine the need for further action (for example, testing). 1.2 This guide assists in utilization and interpretation of various forms of information gathered to enable compliance or conformance or both to regulations, standards, supply agreements, or customer enquiries related to identified declarable substances, including the evidence required to issue declarations for the absence or presence of a declarable substance. Several examples are referenced. The target declarable substances will be specific to the product and product classification, the regulatory jurisdiction, customer/supplier requirements, and other relevant considerations. Consideration of which regulations and standards apply to a given product is beyond the scope of this guide. 1.3 The framework covered in this guide attempts to harmonize language used in the absence of objective data or specific regulatory requirements or both. This guide draws on a variety of existing documentation, which will be cited and referenced, as well as basic scientific principles for communication of chemical hazard and risk, and may be used as an approach for assessing composition of products and their components as part of product risk assessment. 1.4 This guide is applicable to a variety of materials, including polymeric and elastomeric materials, which are used in regulated industries and products. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international st...
SIGNIFICANCE AND USE 4.1 Regulations, standards, and market-defined requirements for chemical constituent (“substances of interest”) compliance and conformance have become increasing numerous and complex. Specific laws and standards may pertain to certain product and industry segments, for example, electrical/electronic instruments and components, pharmaceuticals, medical devices, consumer products, agrichemicals, and so forth. Others may be broader and relate specifically to assessment and management of specific chemical compounds or classes across multiple product sectors and sources, for example Ref (2). In addition, such requirements may be issued by national authorities, international standards setting groups, or, in the case of market-defined requirements, even by customer advocacy organizations or customers through supply contracts. The resulting global landscape of requirements and market access expectations is complex, and compliance/conformance presents numerous challenges for manufacturers. Note 1: For example, IEC 62474:2012 provides some standardized definitions for reporting thresholds and declaration statements for electrotechnical industry. In another example, ISO 14021:2016 addresses self-declared environmental claims. 4.2 Declarable substances may be found on various lists and forms, including those listed in Refs (2-10) and on supply agreements and questionnaires. This guide is not intended to be exhaustive nor cover all declarable substances. Nor does this guide address specific declaration and labeling requirements within these regulation and standards nor address product safety and compliance requirements as dictated by law for specific products, industries, or market regions. 4.3 In addition to new laws and standards, more chemical substances are continually added to a variety of screening lists for review of potential hazards, identification and quantification of possible health or environmental hazards, or both, and consideration of control measures. To ... SCOPE 1.1 This guide uses case studies to illustrate the decision process to assess materials and products for declarable substances when evaluating conformance to relevant requirements. This may be accomplished by applying existing knowledge to determine the need for further action (for example, testing). 1.2 This guide assists in utilization and interpretation of various forms of information gathered to enable compliance or conformance or both to regulations, standards, supply agreements, or customer enquiries related to identified declarable substances, including the evidence required to issue declarations for the absence or presence of a declarable substance. Several examples are referenced. The target declarable substances will be specific to the product and product classification, the regulatory jurisdiction, customer/supplier requirements, and other relevant considerations. Consideration of which regulations and standards apply to a given product is beyond the scope of this guide. 1.3 The framework covered in this guide attempts to harmonize language used in the absence of objective data or specific regulatory requirements or both. This guide draws on a variety of existing documentation, which will be cited and referenced, as well as basic scientific principles for communication of chemical hazard and risk, and may be used as an approach for assessing composition of products and their components as part of product risk assessment. 1.4 This guide is applicable to a variety of materials, including polymeric and elastomeric materials, which are used in regulated industries and products. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international st...
ASTM F2577-22 is classified under the following ICS (International Classification for Standards) categories: 03.100.10 - Purchasing. Procurement. Logistics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2577-22 has the following relationships with other standards: It is inter standard links to ASTM D883-24, ASTM D883-23, ASTM D883-20, ASTM F2931-19a, ASTM D883-19c, ASTM D883-19a, ASTM D883-19, ASTM F2931-19, ASTM D883-18a, ASTM D883-18, ASTM D883-17, ASTM F2931-17, ASTM F2931-16, ASTM F2931-16e1, ASTM F2576-15a. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2577-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2577 − 22
Standard Guide for
Compositional Evaluation of Declarable Substances and
Substances of Concern for Materials in Products
This standard is issued under the fixed designation F2577; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 1.6 This international standard was developed in accor-
dance with internationally recognized principles on standard-
1.1 This guide uses case studies to illustrate the decision
ization established in the Decision on Principles for the
process to assess materials and products for declarable sub-
Development of International Standards, Guides and Recom-
stances when evaluating conformance to relevant require-
mendations issued by the World Trade Organization Technical
ments. This may be accomplished by applying existing knowl-
Barriers to Trade (TBT) Committee.
edge to determine the need for further action (for example,
testing).
2. Referenced Documents
1.2 This guide assists in utilization and interpretation of
2.1 ASTM Standards:
various forms of information gathered to enable compliance or
D883 Terminology Relating to Plastics
conformance or both to regulations, standards, supply
F2576 Terminology Relating to Declarable Substances in
agreements, or customer enquiries related to identified declar-
Materials
able substances, including the evidence required to issue
F2931 Guide for Analytical Testing of Substances of Very
declarations for the absence or presence of a declarable
High Concern in Materials and Products
substance. Several examples are referenced. The target declar-
2.2 IEC Standards:
able substances will be specific to the product and product
IEC 62321-8:2017 Determination of certain substances in
classification, the regulatory jurisdiction, customer/supplier
electrotechnical products Part 8: Determination of specific
requirements, and other relevant considerations. Consideration
phthalates in polymer materials by mass spectrometry
of which regulations and standards apply to a given product is
IEC 62474:2012 Material declaration for products of and for
beyond the scope of this guide.
the electrotechnical industry
1.3 The framework covered in this guide attempts to har-
2.3 ISO Standards:
monize language used in the absence of objective data or
ISO 9001:2015 Quality management systems – Require-
specific regulatory requirements or both. This guide draws on
ments
a variety of existing documentation, which will be cited and
ISO 13485:2016 Medical devices – Quality management
referenced, as well as basic scientific principles for communi-
systems – Requirements for regulatory purposes
cation of chemical hazard and risk, and may be used as an
ISO 14021:2016 Environmental Labels and Declarations:
approach for assessing composition of products and their
Self-Declared Environmental Claims
components as part of product risk assessment.
ISO 14971:2019 Medical devices – Application of risk
1.4 This guide is applicable to a variety of materials,
management to medical devices
including polymeric and elastomeric materials, which are used
in regulated industries and products. 3. Terminology
1.5 This standard does not purport to address all of the
3.1 Definitions: Terms and definitions related to declarable
safety concerns, if any, associated with its use. It is the
substances in materials may be found in Terminology F2576.
responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on
This guide is under the jurisdiction of ASTM Committee F40 on Declarable the ASTM website.
Substances in Materials and is the direct responsibility of Subcommittee F40.02 on Available from the International Electrotechnical Commission, 3, rue de
Management Practices and Guides. Varembé, 1st floor, P.O. Box 131, CH - 1211 Geneva 20, Switzerland, www.iec.ch.
Current edition approved Oct. 1, 2022. Published November 2022. Originally Available from International Organization for Standardization (ISO), ISO
approved in 2006. Last previous edition approved in 2014 as F2577–14. DOI: Central Secretariat, Chemin de Blandonnet 8, CP 401, 1214 Vernier, Geneva,
10.1520/F2577-22. Switzerland, https://www.iso.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2577 − 22
3.1.1 Terms and definitions in the guide not found in of the resultant information, specifically where clear objective/
Terminology F2576 may be found in a common dictionary or numericdataarenotavailableorobtainable,andintheabsence
other reference documents such as Terminology D883 or the of directly applicable regulatory direction. This guide is
ASTM Dictionary of Engineering Science & Technology (Ref intended to harmonize language used to interpret and commu-
(1)). nicate results of these formulation-based or direct-analysis
based assessments. Examples include “not expected to
4. Significance and Use
contain,” “not intentionally added,” “not used in formulation,”
“below de minimis levels,” “below analytical detection limits,”
4.1 Regulations, standards, and market-defined require-
“not toxicologically significant,” “not material to safety,” “free
ments for chemical constituent (“substances of interest”) com-
of,” and so forth.
pliance and conformance have become increasing numerous
and complex. Specific laws and standards may pertain to 4.5 A priori knowledge is based on logical deduction and
certain product and industry segments, for example, electrical/
scientific principles, so actual testing of a material may not be
electronic instruments and components, pharmaceuticals, required to assess conformance to requirements. For example,
medical devices, consumer products, agrichemicals, and so
it is possible to deduce that organic substances will not survive
forth. Others may be broader and relate specifically to assess- the temperatures required to produce wrought steel, so there is
ment and management of specific chemical compounds or
no need to test for organic substances in wrought steel nor is it
classes across multiple product sectors and sources, for ex- possible to develop test methods and reference materials for
ampleRef (2).Inaddition,suchrequirementsmaybeissuedby
determination of organic substances within wrought steel.
national authorities, international standards setting groups, or,
4.6 A posteriori knowledge is based on observation, expe-
in the case of market-defined requirements, even by customer
rience and known facts. If a priori knowledge cannot rule out
advocacy organizations or customers through supply contracts.
the possibility that a substance is present within (or on) a
The resulting global landscape of requirements and market
material, a test method may be required to verify or generate
access expectations is complex, and compliance/conformance
information on the concentration of that substance within (or
presents numerous challenges for manufacturers.
on) the material.
NOTE 1—For example, IEC 62474:2012 provides some standardized
4.7 Statements provided by a manufacturer about declarable
definitions for reporting thresholds and declaration statements for electro-
substances in a product may contain either or both a priori and
technical industry. In another example, ISO 14021:2016 addresses self-
a posteriori information. The recipient of such statements may
declared environmental claims.
be downstream manufacturers who incorporate multiple mate-
4.2 Declarablesubstancesmaybefoundonvariouslistsand
rials and components into other products with their own
forms, including those listed in Refs (2-10) and on supply
subsequent documentation on declarable substances. Each
agreementsandquestionnaires.Thisguideisnotintendedtobe
producer is responsible for its own products’ substance
exhaustive nor cover all declarable substances. Nor does this
declarations, including any potential inaccuracies that may be
guide address specific declaration and labeling requirements
provided by suppliers. Thus, each manufacturer should take
within these regulation and standards nor address product
into account its suppliers’ risk profiles when choosing to rely
safety and compliance requirements as dictated by law for
on supplier declarations.
specific products, industries, or market regions.
4.8 Test methods can be used to verify and provide infor-
4.3 In addition to new laws and standards, more chemical
mation related to substances within materials. At the same
substances are continually added to a variety of screening lists
time, misinformation can be generated or inappropriate con-
for review of potential hazards, identification and quantifica-
clusions drawn when test methods are misapplied. This guide
tion of possible health or environmental hazards, or both, and
is intended to provide recommendations on the application of
consideration of control measures. To comply with these
test methods.
requirements, significant efforts are being directed to detailed
4.9 Test methods may be applied by producers or by interim
data gathering throughout manufacturer supply chains, docu-
or end users of materials. However, it is not necessary or cost
mentation of presence or absence of such declarable
effective to test materials at each stage of production. The
substances, and continual “update” maintenance of the resul-
decision to apply test methods and the frequency of testing
tant information. Standardized processes have been proposed
shouldbebasedonriskperceivedbytheuserorcanbeamatter
to assess the potential for a material to contain a possible
of agreement.
substance of interest/concern.
4.10 Assessment of different types and classes of materials
4.4 Because of requirements being placed on concentrations
each have their individual complexities and nuances given the
of declarable substances within (or on) materials, assessing
advancements in both materials and analytical evaluation
conformance of products has become a complex, time-
methods.
consuming, and expensive task.This guide is intended to assist
4.11 For some regulations and directives, a clear de minimis
the user in developing a protocol for product assessment. This
threshold has been defined for a substance or group of
guideisalsodirectedtowardinterpretationandcommunication
substances. In the absence of a prescribed de minimis, 0.1 %
(w/w) is a suitable threshold.
4.11.1 The de minimis level may be described as an indi-
The boldface numbers in parentheses refer to a list of references at the end of
this standard. vidual limit, or as an aggregated limit, whose details will be
F2577 − 22
dependent on a specific regulation. In no instance shall an material is considered non-conforming or the substances shall
aggregated de minimis exceed the sum of the prescribed de be declared. It is important to determine the basis to which the
minimis for the collective group of substances. limits apply to assess conformance properly. For example, the
limitsmayapplytoanentireproductortheymayapplytoeach
4.12 This guide includes a general process and case studies
material within the product.
in order to provide guidance and to distinguish when a priori
NOTE 2—Limits of “zero” for substances or statements such as
and a posteriori knowledge should be applied. Flow charts as
“products shall not contain” certain substances are indeterminate. Prod-
a guide for assessment of materials and products are provided
ucts or materials cannot be assessed to such limits without accounting for
in Appendix X1. and identifying every atom constituting the product or material being
assessed. See also 4.11.
5. General Process of Assessment of Materials and
5.4 If appropriate, apply a priori knowledge of the material
Products for Conformance with Requirements
and its manufacture to assess the probability whether each
5.1 The process of assessing materials and products for
declarable substance may be present. Lists of declarable
conformance with requirements may be broken down into the
substances in requirements may have been set with little or no
following steps:
regard to material types to be assessed. Through application of
5.1.1 Determine if the material or product is covered under
a priori knowledge of specific materials it may be possible to
the scope of the requirements, taking into account any exemp-
eliminate the need to gather empirical data for certain sub-
tions from the scope.
stances listed in the requirements.
5.1.2 Establish the basis of conformance to which limits for NOTE3—Aprioriknowledgerequiresjustificationbaseduponscientific
principles and logical deduction. For example, the statement that it is not
substances given in the requirements apply.
necessary to determine the hexavalent chromium concentration within
5.1.2.1 List each material to be assessed and assess each on
wrought metals is based upon knowledge of metallurgy and atomic
an individual basis.
structure.
5.1.3 If appropriate, apply a priori knowledge of the mate-
5.5 As necessary, obtain a posteriori knowledge of sub-
rial and its manufacture to assess the probability of whether
stance concentrations based upon observations or empirical
each declarable substance may be present.
data from the material. Data on substance concentrations
5.1.4 As necessary, obtain a posteriori knowledge of sub-
within or on materials may be obtained from suppliers in the
stance concentrations based upon observations or empirical
form of material declarations or specifications; certificates of
data from the material.
analysis; or a laboratory report of a lot, batch, or heat of
5.1.5 Assess the material or product for conformance to
material. While it is not necessary to confirm every certificate
requirements based upon a priori and a posteriori information.
obtained from suppliers via repetition of testing, it is good
5.1.5.1 If the basis of conformance to requirements is each
practice to confirm material data periodically.
material within a product, assess each material separately for
5.6 Assess the material or product for conformance to
declarable substances content.
requirements based upon a priori and a posteriori information.
5.1.5.2 If the basis of conformance to requirements is the
Theconcentrationsofanydeclarablesubstancesinmaterialsor
entireproduct,assessthecumulativecontributionofdeclarable
products are compared to the limits set by the requirements to
substances in materials for the entire product.
determine conformance or non-conformance. While a poste-
5.1.6 Document the assessment process for each material or
riori information in the form of empirical data may be directly
product.
compared to limit values, a priori information may indicate
5.2 Determine if the material or product is covered under
that it is not appropriate to assess conformity of materials for
the scope of the requirements, taking into account any exemp-
certain substances.
tions from the scope. Requirements may be regulations or
NOTE 4—A priori knowledge that a substance cannot be present in a
stipulations by clients, that is, they may be statutory or by
material precludes the possibility of generating empirical data for that
agreement. The first step in any assessment is to determine
substance within that material. It is appropriate to state in such cases that
conformity assessment is not applicable.
what is covered by requirements or the scope of the require-
ments. In some cases, exemptions to the scope are allowed, so
5.7 Document the assessment process for each material or
it is important to establish whether a particular material or
product. Documentation in the form of data and illustration of
product falls under the scope and whether the product or
the assessment process is necessary to back up statements of
material is exempt from that scope.
compliance for materials and products.
5.3 Establish the basis of conformance to which limits for
6. Keywords
substances given in the requirements apply. Requirements in
theformofsubstancerestrictionsordeclarationlimitstypically 6.1 compliance; conformity assessment; declarable sub-
list concentrations of substances above which the product or stances; de minimis; material declaration; product testing
F2577 − 22
APPENDIXES
(Nonmandatory Information)
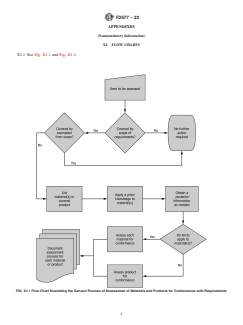
X1. FLOW CHARTS
X1.1 See Fig. X1.1 and Fig. X1.2.
FIG. X1.1 Flow Chart Illustrating the General Process of Assessment of Materials and Products for Conformance with Requirements
F2577 − 22
FIG. X1.2 Flow Chart from the United Kingdom Department of Transportation and Industry (DTI) for Use as a Guide to Help Clarify the
Compliance Process with Regard to RoHS Directive Requirements (DTI ROHS Regulations, Government Guidance Notes (4). Reproduced
by permission from the United Kingdom Department of Transportation and Industry (DTI).)
F2577 − 22
X2. CASE STUDIES
X2.1 Table X2.1 contains a directory of the case studies. mium compounds have been used as pigments in such mate-
rials. Lead may be found in some free-machining metal alloys
X2.2 A finished metal part is to be evaluated for confor-
at levels above the RoHS MCV. Cadmium can be found in
mance to the requirements of European Union Restriction of
some metals either as a contaminant or intentionally added
the Use of Certain Hazardous Substances in Electrical and
depending on the alloy t
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2577 − 14 F2577 − 22
Standard Guide for
Assessment of Materials and Products for Declarable
SubstancesCompositional Evaluation of Declarable
Substances and Substances of Concern for Materials in
Products
This standard is issued under the fixed designation F2577; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This guide uses case studies to illustrate the decision process to assess materials and products for declarable substances when
evaluating conformance to relevant requirements. This may be accomplished by applying existing knowledge to determine the
need for further action (for example, testing).
1.2 This guide assists in utilization and interpretation of various forms of information gathered to enable compliance or
conformance or both to regulations, standards, supply agreements, or customer enquiries related to identified declarable substances,
including the evidence required to issue declarations for the absence or presence of a declarable substance. Several examples are
referenced. The target declarable substances will be specific to the product and product classification, the regulatory jurisdiction,
customer/supplier requirements, and other relevant considerations. Consideration of which regulations and standards apply to a
given product is beyond the scope of this guide.
1.3 This guide is limited to the referenced European Union directives. Other regions, countries, states or local municipalities may
adopt these or similar regulations.The framework covered in this guide attempts to harmonize language used in the absence of
objective data or specific regulatory requirements or both. This guide draws on a variety of existing documentation, which will be
cited and referenced, as well as basic scientific principles for communication of chemical hazard and risk, and may be used as an
approach for assessing composition of products and their components as part of product risk assessment.
1.4 This guide is applicable to a variety of materials, including polymeric and elastomeric materials, which are used in regulated
industries and products.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
This guide is under the jurisdiction of ASTM Committee F40 on Declarable Substances in Materials and is the direct responsibility of Subcommittee F40.02 on
Management Practices and Guides.
Current edition approved March 1, 2014Oct. 1, 2022. Published April 2014November 2022. Originally approved in 2006. Last previous edition approved in 20062014
as F2577–06.–14. DOI: 10.1520/F2577-14.10.1520/F2577-22.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2577 − 22
2. Referenced Documents
2.1 ASTM Standards:
D883 Terminology Relating to Plastics
F2576 Terminology Relating to Declarable Substances in Materials
F2931 Guide for Analytical Testing of Substances of Very High Concern in Materials and Products
2.2 Other Documents: IEC Standards:
DTI ROHS Regulations Government Guidance Notes, November 2005, SI 2005 No. 2748
European Commission Decision 2005/618/ECIEC 62321-8:2017 Commission Decision of 18 August 2005 amending Directive
2002/95/EC of the European Parliament and of the Council for the purpose of establishing the maximum concentration values
for certain hazardous substances in electrical and electronic equipmentDetermination of certain substances in electrotechnical
products Part 8: Determination of specific phthalates in polymer materials by mass spectrometry
European Union Directive 2011/65/EU (recast of 2002/95/EC) on the Restriction of the Use of Certain Hazardous Substances
in Electrical and Electronic Equipment
European Union Directive 2012/19/EUIEC 62474:2012 on Waste Electrical and Electronic EquipmentMaterial declaration for
products of and for the electrotechnical industry
2.3 ISO Standards:
ISO 9001:2015 Quality management systems – Requirements
ISO 13485:2016 Medical devices – Quality management systems – Requirements for regulatory purposes
ISO 14021:2016 Environmental Labels and Declarations: Self-Declared Environmental Claims
ISO 14971:2019 Medical devices – Application of risk management to medical devices
3. Terminology
3.1 Terms and definitions related to declarable substances in materials may be found in Terminology F2576.
3.1 Definitions: Terms and definitions related to declarable substances in materials may be found in Terminology F2576.
3.1.1 Terms and definitions in the guide not found in Terminology F2576 may be found in a common dictionary or other reference
documents such as Terminology D883 or the ASTM Dictionary of Engineering Science & Technology (Ref (1)).
3.2 Terms and definitions in the guide not found in Terminology F2576 may be found in a common dictionary or other reference
documents such as the ASTM Dictionary of Engineering Science & Technology.
4. Significance and Use
4.1 Regulations, standards, and market-defined requirements for chemical constituent (“substances of interest”) compliance and
conformance have become increasing numerous and complex. Specific laws and standards may pertain to certain product and
industry segments, for example, electrical/electronic instruments and components, pharmaceuticals, medical devices, consumer
products, agrichemicals, and so forth. Others may be broader and relate specifically to assessment and management of specific
chemical compounds or classes across multiple product sectors and sources, for example Ref (2). In addition, such requirements
may be issued by national authorities, international standards setting groups, or, in the case of market-defined requirements, even
by customer advocacy organizations or customers through supply contracts. The resulting global landscape of requirements and
market access expectations is complex, and compliance/conformance presents numerous challenges for manufacturers.
NOTE 1—For example, IEC 62474:2012 provides some standardized definitions for reporting thresholds and declaration statements for electrotechnical
industry. In another example, ISO 14021:2016 addresses self-declared environmental claims.
4.2 Declarable substances may be found on various lists and forms, including those listed in Refs (2-10) and on supply agreements
and questionnaires. This guide is not intended to be exhaustive nor cover all declarable substances. Nor does this guide address
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Official Journal of the European Union, L174/88 08.06.2011. The boldface numbers in parentheses refer to a list of references at the end of this standard.
Official Journal of the European Union, L 197/1 04.07.2012.
DTI RoHS Regulations, Government Guidance Notes, November 2005, SI 2005 No. 2748,” p. 23. Reproduced by permission from the United Kingdom Department of
Transportation and Industry (DTI).Available from the International Electrotechnical Commission, 3, rue de Varembé, 1st floor, P.O. Box 131, CH - 1211 Geneva 20,
Switzerland, www.iec.ch.
Official Journal of the European Union, L 214/65, 19.8.2005; notified under document number C(2005) 3143. Available from International Organization for
Standardization (ISO), ISO Central Secretariat, Chemin de Blandonnet 8, CP 401, 1214 Vernier, Geneva, Switzerland, https://www.iso.org.
F2577 − 22
specific declaration and labeling requirements within these regulation and standards nor address product safety and compliance
requirements as dictated by law for specific products, industries, or market regions.
4.3 In addition to new laws and standards, more chemical substances are continually added to a variety of screening lists for
review of potential hazards, identification and quantification of possible health or environmental hazards, or both, and
consideration of control measures. To comply with these requirements, significant efforts are being directed to detailed data
gathering throughout manufacturer supply chains, documentation of presence or absence of such declarable substances, and
continual “update” maintenance of the resultant information. Standardized processes have been proposed to assess the potential
for a material to contain a possible substance of interest/concern.
4.4 Due toBecause of requirements being placed on concentrations of declarable substances within (or on) materials, assessing
conformance of products has become a complex, time-consumingtime-consuming, and expensive task. This guide is intended to
assist the user in developing a protocol for product assessment. This guide is also directed toward interpretation and
communication of the resultant information, specifically where clear objective/numeric data are not available or obtainable, and
in the absence of directly applicable regulatory direction. This guide is intended to harmonize language used to interpret and
communicate results of these formulation-based or direct-analysis based assessments. Examples include “not expected to contain,”
“not intentionally added,” “not used in formulation,” “below de minimis levels,” “below analytical detection limits,” “not
toxicologically significant,” “not material to safety,” “free of,” and so forth.
4.5 A priori knowledge is based on logical deduction and scientific principles, so actual testing of a material may not be required
in order to assess conformance to requirements. For example, it is possible to deduce that organic substances will not survive the
temperatures required to produce wrought steel, so there is no need to test for organic substances in wrought steel nor is it possible
to develop test methods and reference materials for determination of organic substances within wrought steel.
4.6 A posteriorposteriorii knowledge is based on observation, experience and known facts. If a priori knowledge cannot rule out
the possibility that a substance is present within (or on) a material, a test method may be required to verify or generate information
on the concentration of that substance within (or on) the material.
4.7 Statements provided by a manufacturer about declarable substances in a product may contain either or both a priori and a
posteriori information. The recipient of such statements may be downstream manufacturers who incorporate multiple materials and
components into other products with their own subsequent documentation on declarable substances. Each producer is responsible
for its own products’ substance declarations, including any potential inaccuracies that may be provided by suppliers. Thus, each
manufacturer should take into account its suppliers’ risk profiles when choosing to rely on supplier declarations.
4.8 Test methods can be used as a means to verify and provide information related to substances within materials. At the same
time, misinformation can be generated or inappropriate conclusions drawn when test methods are misapplied. This guide is
intended to provide recommendations on the application of test methods.
4.9 Test methods may be applied by producers or by interim or end users of materials. However, it is not necessary or cost effective
to test materials at each stage of production. The decision to apply test methods and the frequency of testing should be based on
risk perceived by the user or can be a matter of agreement.
4.10 Assessment of different types and classes of materials each have their individual complexities and nuances given the
advancements in both materials and analytical evaluation methods.
4.11 Assessment of plastics For some regulations and directives, a clear de minimisfor presence of declarable substances is more
complex than assessment of metals and threshold has been defined for a substance or group of substances. In the absence of a
prescribed alloys, sincede minimis, the possible ingredients are comparatively much more numerous in plastic manufacture. 0.1
% (w/w) is a suitable threshold.
4.11.1 The de minimis level may be described as an individual limit, or as an aggregated limit, whose details will be dependent
on a specific regulation. In no instance shall an aggregated de minimis exceed the sum of the prescribed de minimis for the
collective group of substances.
F2577 − 22
4.12 This guide includes a general process and case studies in order to provide guidance and to distinguish wherewhen a priori
and a posteriori knowledge should be applied. Flow charts as a guide for assessment of materials and products are provided in
Appendix X1.
5. General Process of Assessment of Materials and Products for Conformance with Requirements
5.1 The process of assessing materials and products for conformance with requirements may be broken down into the following
steps:
5.1.1 Determine if the material or product is covered under the scope of the requirements, taking into account any exemptions from
the scope.
5.1.2 Establish the basis of conformance to which limits for substances given in the requirements apply.
5.1.2.1 List each material to be assessed and assess each on an individual basis.
5.1.3 If appropriate, apply a priori knowledge of the material and its manufacture to assess the probability of whether each
declarable substance may be present.
5.1.4 As necessary, obtain a posteriori knowledge of substance concentrations based upon observations or empirical data from the
material.
5.1.5 Assess the material or product for conformance to requirements based upon a priori and a posteriori information.
5.1.5.1 If the basis of conformance to requirements is each material within a product, assess each material separately for declarable
substances content.
5.1.5.2 If the basis of conformance to requirements is the entire product, assess the cumulative contribution of declarable
substances in materials for the entire productproduct.
5.1.6 Document the assessment process for each material or product.
5.2 Determine if the material or product is covered under the scope of the requirements, taking into account any exemptions from
the scope. Requirements may be regulations or stipulations by clients, that is, they may be statutory or by agreement. The first step
in any assessment is to determine what is covered by requirements,requirements or the scope of the requirements. In some cases,
exemptions to the scope are allowed, so it is important to establish whether a particular material or product falls under the scope
and whether the product or material is exempt from that scope.
5.3 Establish the basis of conformance to which limits for substances given in the requirements apply. Requirements in the form
of substance restrictions or declaration limits typically list concentrations of substances above which the product or material is
considered non-conforming or the substances mustshall be declared. It is important to determine the basis to which the limits apply
in order to properly assess conformance. conformance properly. For example, the limits may apply to an entire product or they may
apply to each material within the productproduct.
NOTE 2—Limits of “zero” for substances or statements such as “products shall not contain” certain substances are indeterminate. Products or materials
cannot be assessed to such limits without accounting for and identifying every atom constituting the product or material being assessed. See also 4.11.
5.4 If appropriate, apply a priori knowledge of the material and its manufacture to assess the probability whether each declarable
substance may be present. Lists of declarable substances in requirements may have been set with little or no regard to material
types to be assessed. Through application of a priori knowledge of specific materials it may be possible to eliminate the need to
gather empirical data for certain substances listed in the requirements.
NOTE 3—A priori knowledge requires justification based upon scientific principles and logical deduction. For example, the statement that it is not
necessary to determine the hexavalent chromium concentration within wrought metals is based upon knowledge of metallurgy and atomic structure.
5.5 As necessary, obtain a posteriori knowledge of substance concentrations based upon observations or empirical data from the
material. Data on substance concentrations within or on materials may be obtained from suppliers in the form of Certificates of
F2577 − 22
Analysismaterial declarations or specifications; certificates of analysis; or a laboratory report of a lot, batch, or heat of material.
While it is not necessary to confirm each and every certificate obtained from suppliers via repetition of testing, it is good practice
to periodically confirm material data.data periodically.
5.6 Assess the material or product for conformance to requirements based upon a priori and a posteriori information. information.
The concentrations of any declarable substances in materials or products are compared to the limits set by the requirements in order
to determine conformance or non-conformance. While a posteriori information in the form of empirical data may be directly
compared to limit values, a priori information may indicate that it is not appropriate to assess conformity of materials for certain
substances.
NOTE 4—A priori knowledge that a substance cannot be present in a material precludes the possibility of generating empirical data for that substance
within that material. It is appropriate to state in such cases that conformity assessment is not applicable.
5.7 Document the assessment process for each material or product. Documentation in the form of data and illustration of the
assessment process is necessary to back up statements of compliance for materials and productsproducts.
6. Case Studies
6.1 A finished aluminum part is to be evaluated for conformance to the requirements of European Union Directive 2002/95/EC
on the Restriction of the Use of Certain Hazardous Substances in Electrical and Electronic Equipment (RoHS). It was established
that the part would be incorporated into a finished electrical product to be sold on the European market after the effective date of
the RoHS Directive and the finished product fits the description of electrical products to be regulated per the RoHS Directive and
European Union Directive 2011/65/EU (recast of 2002/95/EC) on Waste Electrical and Electronic Equipment (WEEE).
6.1.1 The chemical requirements of the RoHS Directive are given in European Commission Decision 2005/618/EC as follows:
the Maximum Concentration Value (MCV) for lead, mercury, hexavalent chromium, polybrominated biphenyls (PBB) and
polybrominated diphenyl ethers (PBDE) is 0.1% by weight per homogeneous material and the MCV for cadmium is 0.01% by
weight per homogeneous material. Per the Annex of the RoHS Directive, aluminum is exempt from the restriction on lead and is
allowed to contain up to 0.4% by weight lead. No other exemption was applicable to the aluminum part.
6.1.2 A priori knowledge of aluminum and its method of manufacture were used to evaluate the part. Aluminum is not flammable
under normal conditions, so there is a low probability that flame retardants such as PBB or PBDE were part of the formulation.
Furthermore, the temperature of a typical aluminum smelter is maintained at 920°C – 980°C at which organic substances such as
PBB or PBDE are decomposed and become volatile gases, so organic substances will not remain in the finished aluminum metal.
While metal alloys may contain the element chromium, the chromium within the metal is in the metallic state with an atomic
valence of zero. With regard to metals, hexavalent chromium is not found within the metal but may be found on the exterior
surfaces in the form of a chromate conversion coating. Hexavalent chromium may also be found in coatings such as paint and
plastic since hexavalent chromium compounds have been employed as pigments in such materials. Lead may be found in some
free machining aluminum alloys at levels above the RoHS MCV. Cadmium can be found in aluminum as a contaminant, but is
added intentionally in only two alloys (2021 and 4013) which are not commonly produced at this time. Mercury is volatile and
is not likely to be found in aluminum or its alloys, though there are some formulations used as sacrificial anodes containing
mercury at 0.03 to 0.06% Small concentrations of mercury in aluminum will cause the aluminum to be rapidly oxidized and
converted into aluminum oxide, so a piece of solid aluminum is unlikely to contain more than 0.1% mercury.
6.1.3 Application of a priori knowledge of aluminum and its manufacture results in a preliminary assessment concerning the
presence of certain substances. The preliminary assessment of the aluminum part to the requirements of RoHS points to which
substances should be measured and which should not: there is a good probability that cadmium or lead may be present within the
aluminum part; there is a low probability that mercury may be present and there is a possibility that hexavalent chromium may
be found on the surface of the part. Conversely, there is low probability that PBB or PBDE are present in the aluminum.
Completing the evaluation of the aluminum part to RoHS requirements requires a posteriori knowledge, or knowledge that is based
on measurement. At some point in the life of the aluminum metal, testing would be required to quantify the concentrations of lead,
cadmium, mercury and hexavalent chromium associated with the aluminum. Information on the lead and cadmium content, as well
as total chromium, may be available in the form of a Certificate of Analysis generated by the producer or by an independent
laboratory. If such a document is not available or the available information requires verification, testing of the part is required to
evaluate conformance to requirements. Aluminum Association specifications require that aluminum producers maintain a
surveillance program for mercury so that production is monitored on a periodic (quarterly within North America) basis to ensure
that mercury has not been introduced into any of the production processes. The test method(s) applied to the aluminum part should
be designed specifically for analysis of the substances on or within aluminum or aluminum alloys; in other words, both the
F2577 − 22
substance sought and the material to be analyzed should be included in the scope of the test method. If a standard method does
not exist, it is incumbent upon the laboratory to demonstrate that the method employed is valid for analysis of the substance in
(or on) the specific material tested.
6.1.4 Validation of a method can be accomplished through analysis of a reference material, preferably a certified reference material
with traceability to SI units, using the same method of analysis as is used on the unknown sample. The reference material used
for validation of the method shall be of the same or similar composition of the unknown sample; for example, the reference
material used to validate the analysis of aluminum or its alloys for a substance shall be covered by the scope of the test method
for aluminum and its alloys and shall also contain a known concentration of the substance to be measured.
NOTE 4—A reference material to be used for validation of a test method should not also be used for calibration of the instrument.
NOTE 5—The instrument used for analysis of the sample is not as important as demonstration that the instrument is capable of analyzing substances in
specific materials; such capability may be demonstrated through validation of the test method.
6.2 A red piece of polyvinyl chloride (PVC) is to be evaluated for conformance to the requirements of the RoHS Directive. It was
established that the PVC sample represented material which would be incorporated into a finished electronic product to be sold
on the European market after the effective date of the RoHS Directive. The finished product fits the description of electronic
products to be regulated per the RoHS and WEEE Directives.
6.2.1 After review of the directive, it was determined no exemptions were applicable to the red PVC sample.
6.2.2 Application of a priori knowledge of PVC and its manufacture results in a preliminary risk assessment concerning the
presence of certain substances. The sample was colored
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...