ASTM F2394-07(2022)
(Guide)Standard Guide for Measuring Securement of Balloon-Expandable Vascular Stent Mounted on Delivery System
Standard Guide for Measuring Securement of Balloon-Expandable Vascular Stent Mounted on Delivery System
SIGNIFICANCE AND USE
4.1 The securement of the endovascular stent on the balloon is a critical parameter to ensure that the stent is safely delivered to or from the treatment site.
4.2 This guide is intended for use by researchers and manufacturers for the development and selection of pre-test treatments, tests, and test endpoints to measure stent securement (displacement distances and dislodgment forces).
4.3 This guide may be used to investigate which practical combinations of in vitro tests best characterize clinical scenarios.
4.4 This guide should be used with discretion in choosing securement tests and evaluating results due to the myriad possible combinations of clinical conditions, failure modes, and stent delivery system designs.
4.5 This guide may be of use for developing a test for meeting Parts 2 and 3 of the requirements of EN 14299, Section 7.3.4.4 on Trackability.
4.6 This guide may be of use for developing a test to meet section VII-C-8 of CDRH Guidance document.
SCOPE
1.1 This guide provides guidance for the design and development of pre-test treatments, tests, and test endpoints to measure stent securement of pre-mounted, unsheathed, balloon-expandable stent delivery systems. This guide is intended to aid investigators in the design, development, and in vitro characterization of pre-mounted, unsheathed, balloon-expandable stent delivery systems.
1.2 This guide covers the laboratory determination of the shear force required to displace or dislodge a balloon-expandable endovascular stent mounted on a delivery system. The guide proposes a set of options to consider when testing stent securement. The options cover pre-test treatments, possible stent securement tests, and relevant test endpoints. An example test apparatus is given in 7.1.
1.3 This guide covers in vitro bench testing characterization only. Measured levels of securement and product design/process differentiation may be particularly influenced by selections of pre-test treatments, securement test type (for example, stent gripping method), and test endpoint. In vivo characteristics may also differ from in vitro results.
1.4 This guide does not cover all possible pre-test treatments, stent securement tests, or test endpoints. It is intended to provide a starting point from which to select and investigate securement test options.
1.5 This guide does not specify a method for mounting the stent onto the delivery system.
1.6 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system are not necessarily exact equivalents; therefore, to ensure conformance with the standard, each system shall be used independently of the other, and values from the two systems shall not be combined.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Sep-2022
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.30 - Cardiovascular Standards
Relations
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Sep-2018
- Effective Date
- 01-Oct-2017
- Effective Date
- 01-Oct-2017
- Effective Date
- 01-May-2014
- Effective Date
- 15-May-2013
- Effective Date
- 15-Apr-2013
- Effective Date
- 15-Dec-2012
- Effective Date
- 01-Nov-2012
- Effective Date
- 01-Aug-2012
- Effective Date
- 01-Aug-2012
- Effective Date
- 15-May-2009
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-May-2008
- Effective Date
- 01-Aug-2007
Overview
ASTM F2394-07(2022), officially titled the Standard Guide for Measuring Securement of Balloon-Expandable Vascular Stent Mounted on Delivery System, provides comprehensive guidance for researchers, manufacturers, and quality assurance teams on the evaluation and measurement of stent securement. Ensuring stents remain securely mounted on their delivery systems during handling and deployment is critical for patient safety and product performance in vascular interventions. This guide is particularly relevant for balloon-expandable, pre-mounted, unsheathed stent delivery systems and focuses on laboratory-based, in vitro test methods.
Key Topics
- Stent Securement Importance: Securement is vital for the safe transport, positioning, and deployment of vascular stents. Failures such as stent displacement or dislodgment can have serious clinical consequences.
- Testing Methods: The guide outlines laboratory procedures for determining the shear force needed to displace or dislodge a stent. It discusses a range of pre-test treatments and securement tests – including both guide-type and lesion-type methods – that simulate clinical conditions.
- Test Endpoints: Important measurement endpoints include displacement force and dislodgement force, both of which provide insight into how secure the stent is under simulated clinical challenges.
- Pre-Test Treatments: Treatments before testing may include simulated shipping, storage, handling, and anatomical tracking conditions (e.g., temperature, lubrication, lesion simulation).
- Clinical Scenarios: The guide addresses common failure modes encountered during procedures, such as loss of securement during navigation through tortuous arteries or withdrawal into a guide catheter.
- Flexibility and Limitations: Users are encouraged to select and adapt test methods suitable for specific product designs and clinical scenarios, due to the variability in stent and delivery system configurations.
Applications
- Product Development: Medical device developers use this guide to benchmark and refine the securement of stent delivery systems during research and development.
- Quality Control: Manufacturers rely on these test methods for routine quality assurance, ensuring each batch of stents meets safety and performance criteria.
- Regulatory Submissions: Results from securement measurements can support applications for regulatory approvals by demonstrating compliance with requirements such as those in EN 14299 on Trackability and relevant FDA guidance.
- Comparative Testing: The guide supports comparative studies between products – helping to differentiate and improve stent and delivery system designs.
- Clinical Risk Management: Insights from securement tests inform risk analysis, failure modes and effects analysis (FMEA), and the establishment of product accept/reject criteria.
Related Standards
- EN 14299: Non-Active Surgical Implants – Requirements for Cardiac and Vascular Implants, especially Sections concerning trackability.
- CDRH Guidance: FDA’s guidance documents on Non-Clinical Tests and Recommended Labeling for Intravascular Stents and Associated Delivery Systems.
- ISO 10555-1: General requirements for sterile and single-use intravascular catheters.
- ASTM E1169: Practice for Conducting Ruggedness Tests.
- ASTM E1488: Guide for Statistical Procedures to Use in Developing and Applying Test Methods.
- 21 CFR Part 820: FDA regulations for quality system, emphasizing the need for suitable and capable test methods in medical device production.
Practical Value
ASTM F2394-07(2022) plays a crucial role in the safety and efficacy of minimally invasive vascular procedures. By providing a structured, standardized approach to stent securement testing, this guide helps reduce clinical complications related to insecure stent mounting. Its adaptability ensures relevance across a wide range of product types and clinical situations, making it an essential reference for medical device manufacturers and testing laboratories.
Keywords: vascular stent securement, balloon-expandable stent, delivery system, stent displacement, stent dislodgment, medical device testing, in vitro bench testing, quality assurance, pre-test treatment, regulatory compliance, ASTM F2394
Buy Documents
ASTM F2394-07(2022) - Standard Guide for Measuring Securement of Balloon-Expandable Vascular Stent Mounted on Delivery System
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2394-07(2022) is a guide published by ASTM International. Its full title is "Standard Guide for Measuring Securement of Balloon-Expandable Vascular Stent Mounted on Delivery System". This standard covers: SIGNIFICANCE AND USE 4.1 The securement of the endovascular stent on the balloon is a critical parameter to ensure that the stent is safely delivered to or from the treatment site. 4.2 This guide is intended for use by researchers and manufacturers for the development and selection of pre-test treatments, tests, and test endpoints to measure stent securement (displacement distances and dislodgment forces). 4.3 This guide may be used to investigate which practical combinations of in vitro tests best characterize clinical scenarios. 4.4 This guide should be used with discretion in choosing securement tests and evaluating results due to the myriad possible combinations of clinical conditions, failure modes, and stent delivery system designs. 4.5 This guide may be of use for developing a test for meeting Parts 2 and 3 of the requirements of EN 14299, Section 7.3.4.4 on Trackability. 4.6 This guide may be of use for developing a test to meet section VII-C-8 of CDRH Guidance document. SCOPE 1.1 This guide provides guidance for the design and development of pre-test treatments, tests, and test endpoints to measure stent securement of pre-mounted, unsheathed, balloon-expandable stent delivery systems. This guide is intended to aid investigators in the design, development, and in vitro characterization of pre-mounted, unsheathed, balloon-expandable stent delivery systems. 1.2 This guide covers the laboratory determination of the shear force required to displace or dislodge a balloon-expandable endovascular stent mounted on a delivery system. The guide proposes a set of options to consider when testing stent securement. The options cover pre-test treatments, possible stent securement tests, and relevant test endpoints. An example test apparatus is given in 7.1. 1.3 This guide covers in vitro bench testing characterization only. Measured levels of securement and product design/process differentiation may be particularly influenced by selections of pre-test treatments, securement test type (for example, stent gripping method), and test endpoint. In vivo characteristics may also differ from in vitro results. 1.4 This guide does not cover all possible pre-test treatments, stent securement tests, or test endpoints. It is intended to provide a starting point from which to select and investigate securement test options. 1.5 This guide does not specify a method for mounting the stent onto the delivery system. 1.6 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system are not necessarily exact equivalents; therefore, to ensure conformance with the standard, each system shall be used independently of the other, and values from the two systems shall not be combined. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 The securement of the endovascular stent on the balloon is a critical parameter to ensure that the stent is safely delivered to or from the treatment site. 4.2 This guide is intended for use by researchers and manufacturers for the development and selection of pre-test treatments, tests, and test endpoints to measure stent securement (displacement distances and dislodgment forces). 4.3 This guide may be used to investigate which practical combinations of in vitro tests best characterize clinical scenarios. 4.4 This guide should be used with discretion in choosing securement tests and evaluating results due to the myriad possible combinations of clinical conditions, failure modes, and stent delivery system designs. 4.5 This guide may be of use for developing a test for meeting Parts 2 and 3 of the requirements of EN 14299, Section 7.3.4.4 on Trackability. 4.6 This guide may be of use for developing a test to meet section VII-C-8 of CDRH Guidance document. SCOPE 1.1 This guide provides guidance for the design and development of pre-test treatments, tests, and test endpoints to measure stent securement of pre-mounted, unsheathed, balloon-expandable stent delivery systems. This guide is intended to aid investigators in the design, development, and in vitro characterization of pre-mounted, unsheathed, balloon-expandable stent delivery systems. 1.2 This guide covers the laboratory determination of the shear force required to displace or dislodge a balloon-expandable endovascular stent mounted on a delivery system. The guide proposes a set of options to consider when testing stent securement. The options cover pre-test treatments, possible stent securement tests, and relevant test endpoints. An example test apparatus is given in 7.1. 1.3 This guide covers in vitro bench testing characterization only. Measured levels of securement and product design/process differentiation may be particularly influenced by selections of pre-test treatments, securement test type (for example, stent gripping method), and test endpoint. In vivo characteristics may also differ from in vitro results. 1.4 This guide does not cover all possible pre-test treatments, stent securement tests, or test endpoints. It is intended to provide a starting point from which to select and investigate securement test options. 1.5 This guide does not specify a method for mounting the stent onto the delivery system. 1.6 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system are not necessarily exact equivalents; therefore, to ensure conformance with the standard, each system shall be used independently of the other, and values from the two systems shall not be combined. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2394-07(2022) is classified under the following ICS (International Classification for Standards) categories: 11.040.20 - Transfusion, infusion and injection equipment. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2394-07(2022) has the following relationships with other standards: It is inter standard links to ASTM E1488-23, ASTM E1169-18, ASTM E1169-17, ASTM E1169-17e1, ASTM E1169-14, ASTM E1169-13a, ASTM E1169-13, ASTM E1169-12a, ASTM E1169-12, ASTM E1488-12e1, ASTM E1488-12, ASTM E1488-09, ASTM E1488-08a, ASTM E1488-08, ASTM E1169-07. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2394-07(2022) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2394 − 07 (Reapproved 2022)
Standard Guide for
Measuring Securement of Balloon-Expandable Vascular
Stent Mounted on Delivery System
This standard is issued under the fixed designation F2394; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 1.7 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
1.1 This guide provides guidance for the design and devel-
responsibility of the user of this standard to establish appro-
opment of pre-test treatments, tests, and test endpoints to
priate safety, health, and environmental practices and deter-
measure stent securement of pre-mounted, unsheathed,
mine the applicability of regulatory limitations prior to use.
balloon-expandable stent delivery systems. This guide is in-
1.8 This international standard was developed in accor-
tended to aid investigators in the design, development, and in
dance with internationally recognized principles on standard-
vitro characterization of pre-mounted, unsheathed, balloon-
ization established in the Decision on Principles for the
expandable stent delivery systems.
Development of International Standards, Guides and Recom-
1.2 This guide covers the laboratory determination of the
mendations issued by the World Trade Organization Technical
shear force required to displace or dislodge a balloon-
Barriers to Trade (TBT) Committee.
expandable endovascular stent mounted on a delivery system.
The guide proposes a set of options to consider when testing
2. Referenced Documents
stent securement. The options cover pre-test treatments, pos- 2
2.1 ASTM Standards:
sible stent securement tests, and relevant test endpoints. An
E1169 Practice for Conducting Ruggedness Tests
example test apparatus is given in 7.1.
E1488 GuideforStatisticalProcedurestoUseinDeveloping
1.3 This guide covers in vitro bench testing characterization and Applying Test Methods
only. Measured levels of securement and product design/
2.2 Other Documents:
process differentiation may be particularly influenced by selec-
ISO 10555-1 Sterile and Single-Use Intravascular
tions of pre-test treatments, securement test type (for example,
Catheters—Part 1: General Requirements
stent gripping method), and test endpoint. In vivo characteris-
Quality System Regulation, Part VII Dept. Health and Hu-
tics may also differ from in vitro results.
man Services, Food and Drug Administration, 21 CFR
Part 820 Medical Devices; Current Good Manufacturing
1.4 This guide does not cover all possible pre-test
Practice; Final Rule. Federal Register, October 7, 1996
treatments, stent securement tests, or test endpoints. It is
EN 14299 Non Active Surgical Implants—Particular Re-
intended to provide a starting point from which to select and
quirements for Cardiac and Vascular Implants—Specific
investigate securement test options.
Requirements For Arterial Stents, May 2004
1.5 This guide does not specify a method for mounting the
CDRH Guidance, Non-Clinical Tests and Recommended
stent onto the delivery system.
Labeling for Intravascular Stents andAssociated Delivery
Systems, January 13, 2005
1.6 The values stated in either SI units or inch-pound units
are to be regarded separately as standard. The values stated in
each system are not necessarily exact equivalents; therefore, to
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
ensure conformance with the standard, each system shall be
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
used independently of the other, and values from the two
Standards volume information, refer to the standard’s Document Summary page on
systems shall not be combined.
the ASTM website.
Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
4th Floor, New York, NY 10036, http://www.ansi.org.
AvailablefromU.S.GovernmentPrintingOfficeSuperintendentofDocuments,
This guide is under the jurisdiction of ASTM Committee F04 on Medical and 732 N. Capitol St., NW, Mail Stop: SDE, Washington, DC 20401, http://
Surgical Materials and Devices and is the direct responsibility of Subcommittee www.access.gpo.gov.
F04.30 on Cardiovascular Standards. Available from British Standards Institute (BSI), 389 Chiswick High Rd.,
Current edition approved Oct. 1, 2022. Published October 2022. Originally London W4 4AL, U.K., http://www.bsi-global.com.
approved in 2004. Last previous edition approved in 2017 as F2394 – 07 (2017). Available from Food and Drug Administration (FDA), 5600 Fishers Ln.,
DOI: 10.1520/F2394-07R22. Rockville, MD 20857, http://www.fda.gov/cdrh/ode/guidance/1545.pdf.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2394 − 07 (2022)
MAUDE Database makes the contact with the stent. Typical grips used to apply
force to the stent include shims (as used in Figs. X2.5-X2.8);
3. Terminology
tape which sticks to the stent but not the balloon; an iris which
can be narrowed down to allow the balloon to slip by but not
3.1 Definitions:
the stent; or nubs which contact the stent but not the balloon.
3.1.1 balloon-expandable stent, n—a stent that is expanded
at the treatment site by a balloon catheter. The stent material is
3.1.12 guide catheter, n—a tube designed to transport the
plastically deformed by the balloon expansion such that the
guide wire and the stent delivery system into the target vessel.
stent remains expanded after deflation of the balloon.
3.1.13 guide wire, n—a wire designed to aid in balloon,
3.1.2 crimp, v—tosecurethestentonthedeliverysystemby
ultrasound, atherectomy, or stent placement during endovascu-
radially compressing and plastically deforming the stent onto
lar procedures.
the balloon.
3.1.14 mandrel, n—awirethatmaybeusedasanalternative
3.1.3 delivery system, n—a system similar to a balloon
to the intended guide wire to provide support for the catheter
dilatation catheter that is used to deliver and deploy a stent at
guide wire lumen for some test procedures.
the target site and then removed.
3.1.15 non-recoverable movement, n—a displacement of the
3.1.4 displacement force, critical distance peak, n—a stent
stent relative to the balloon such that if the shearing force was
securement test endpoint characterizing the maximum force
reduced to zero, the stent would remain displaced in the
required to displace the stent with respect to the balloon a
direction of the shearing force relative to the initial placement
critical distance. This critical distance is the minimum of the
on the balloon. The force at which non-recoverable movement
following two distances. The first is the distance at which the
begins is defined as the initial displacement force (see defini-
undamaged stent could overhang the balloon body resulting in
tion above).
a clinically significant, incomplete end deployment. The sec-
3.1.16 pre-test treatment, n—a treatment of the stent deliv-
ond is the length (distance) of stent compression or buckling
ery system prior to the evaluation of securement that simulates
that could result in a clinically significant incomplete deploy-
preparatory, environmental, mechanical, or other conditions
ment of the stent against the vessel walls. (See Fig. X2.1.)
that may be encountered prior to or during clinical use of the
3.1.5 displacement force, initial, n—a stent securement test
device. Examples include subjecting the devices to elevated
endpointcharacterizingtheinitialforcerequiredtodisplacethe
shipping temperature/humidity, catheter preparation per use
stent with respect to the balloon such that the displacement is
instructions, pre-soaking, bending treatments, tracking treat-
a non-recoverable movement (see 3.1.15). (See Fig. X2.1.)
ments (tracking fixture, see definition below), and tracking
3.1.6 displacement force, initial peak, n—astentsecurement
through lesion treatments (lesion fixture, see definition below).
test endpoint characterizing the first peak in force that occurs
3.1.17 pre-test treatment tracking fixture, n—apre-testtreat-
during or after stent displacement with respect to the balloon.
mentfixtureusedtosimulateananatomicalvasculature.Useof
(See Fig. X2.1.)
the fixture with a guide catheter, a guide wire, and the
3.1.7 dislodgment force, peak, n—a stent securement test
stent-balloon catheter delivery system is intended to simulate
endpointcharacterizingthepeakormaximumforcerequiredto
the bending and frictional forces of tracking the device to the
completelydislodgethestentfromthedeliverysystemballoon.
lesion site that may be encountered in the clinical setting. See
During a test, this force will occur after or coincide with the
the engineering diagrams in Appendix X2. Note that these
initial displacement force. (See Fig. X2.1.)
engineering diagrams simulate vessels with a moderately
3.1.8 end flaring, n—a distal or proximal outward conical difficult degree of coronary tortuousity but do not include
opening of the diameter of the stent on the balloon. End flaring
simulated lesions.
is a contributing factor to the probability that the stent may
3.1.18 pre-test treatment lesion fixture, n—a pre-test treat-
become caught during withdrawal into a guide catheter while
ment fixture used to simulate an anatomical vasculature and
tracking through a lesion.
lesion. Use of the fixture with a guide catheter, a guide wire,
3.1.9 failure mode effect analysis (FMEA), n—an analytical
and the stent-balloon catheter delivery system is intended to
approach to methodically determine and address all possible
simulate the bending, frictional, and mechanical resistance
product failure modes, their associated causes, and their
forces of tracking the device across the lesion site that may be
criticality. Used to evaluate designs, prioritize testing, and
encountered in the clinical setting.
track risk reducing improvements to the product.
3.1.19 securement test, guide-type, n—a stent securement
3.1.10 gauge length, n—the initial unstressed length of
test that is similar to the clinical scenario of pulling an
catheter tubing between the proximal end of the stent to the
undeployed stent delivery system back into a guide catheter,
grips which engage the catheter tubing.
arterial sheath, or hemostasis valve. Examples include guides,
3.1.11 grips, n—a means of applying force to the stent and rings,orshimsideallydesignedtoengagethestentendorbody
balloon catheter to displace or dislodge the stent relative to the but not the catheter balloon. The shim securement test, de-
balloon. In particular, grips refer to the end of a device which scribedinSection7,usescomplementarythin,rigidplateswith
rounded “V” notches that are sized to circumferentially engage
the stent end but not the catheter balloon. See the engineering
http://www.fda.gov/cdrh/maude.html. diagrams in Appendix X2.
F2394 − 07 (2022)
3.1.20 securement test, lesion-type, n—a stent securement 6. Test Method Considerations
test that is similar to the clinical scenario of pushing or pulling
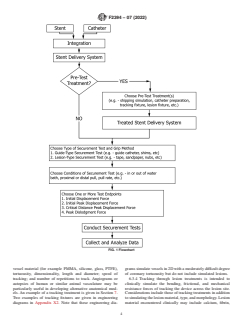
6.1 Flowchart—See Fig. 1.
an undeployed stent delivery system through or around a
6.2 Development and Evaluation of Securement Tests:
fibrous or calcified lesion. Examples include tape, nubs,
6.2.1 Securement test development and selection is ideally
protrusions, or sandpaper ideally designed to engage the stent
begun through the initial use of a battery of tests measuring a
end or body but not the catheter balloon.
variety of failure modes. These test methods may vary from a
simple intuitive tactile impression of the securement forces
4. Significance and Use
through manipulation to clinically modeled situations with
4.1 The securement of the endovascular stent on the balloon
guide catheters and stenosis models to in vivo animal studies
isacriticalparametertoensurethatthestentissafelydelivered
with representative anatomy and physician handling. From a
to or from the treatment site.
safety risk perspective, consider how securement challenges
may occur in clinical situations, what may result from loss of
4.2 This guide is intended for use by researchers and
securement, what the severity of the outcome is to the patient,
manufacturers for the development and selection of pre-test
what the frequency of these situations are, and then how to test
treatments, tests, and test endpoints to measure stent secure-
to detect these occurrences. Factors to consider in evaluating
ment (displacement distances and dislodgment forces).
securement tests include the following:
4.3 This guide may be used to investigate which practical 6.2.1.1 Review of the MAUDE database for reported prob-
combinations of in vitro tests best characterize clinical sce- lems with comparable devices.
narios.
6.2.1.2 Physician surveys for clinical relevance and prob-
lems with comparable devices.
4.4 This guide should be used with discretion in choosing
6.2.1.3 Mechanical understanding of the tests’ clinical rel-
securement tests and evaluating results due to the myriad
evance and limitations.
possible combinations of clinical conditions, failure modes,
6.2.1.4 Mechanical and statistical understanding of the test
and stent delivery system designs.
reproducibility limitations due to device variation, pre-test
4.5 This guide may be of use for developing a test for
treatments, various grips, and test conditions.
meeting Parts 2 and 3 of the requirements of EN 14299,
6.2.1.5 Ability to set accept/reject criteria by physician
Section 7.3.4.4 on Trackability.
evaluation, by historical comparisons, or by other rational
means.
4.6 This guide may be of use for developing a test to meet
6.2.2 The final securement test(s) selected must ultimately
section VII-C-8 of CDRH Guidance document.
satisfy internal manufacturer quality standards. These stan-
dards may include clinical relevance, FMEA analysis, statisti-
5. Clinical Scenarios
cal assurance of characteristics, and challenge assurance of
5.1 There are two failure modes—the stent is dislodged
characteristics.
from the catheter or the stent is displaced or deformed on the
6.2.3 The final securement test(s) must also satisfy external
catheter such that balloon inflation delivery would not produce
regulatorybodystandards.Forexample,theFDAQSR21CFR
an acceptable stent shape at the proper location. Based on
Part 820, Oct. 7, 1996 states that each test used in the process
reported clinical incidents, there are three causes for these two
ofdesignandmanufacturingoffinisheddevices“issuitablefor
types of failures:
its intended purposes and is capable of producing valid
results.” For the statistical capability evaluation, Guide E1488
5.1.1 Displacement or dislodgment of the stent while at-
is very helpful.
tempting to track through or position in tortuous bends, fibrous
or calcified lesions, or previously implanted stents, or combi-
6.3 Pre-Test Treatments:
nation thereof.
6.3.1 Pre-test treatments may be conducted prior to the
5.1.2 Displacement or dislodgment of the stent on with-
evaluation of securement to simulate preparatory,
drawal of the undeployed stent delivery system back into the
environmental, mechanical, or other conditions that may be
guide catheter, introducer sheath, or hemostasis valve. This
encountered prior to or during clinical use of the device.
failure type is usually associated with failure to cross tortuous
6.3.2 Pre-test treatments may include subjecting the devices
bends, fibrous or calcified lesions, or previously implanted
to shelf life testing, sterilization, elevated shipping
stents, or combination thereof. It is sometimes associated with
temperature/humidity, removal of the delivery system from the
less-than-ideal seating or angled placement of the guide
carriertube,andothercatheterpreparationperuseinstructions,
catheter tip in the ostium of the vessel.
pre-soaking, bending treatments, tracking treatments, and
tracking through lesion treatments.
5.1.3 Displacement or dislodgement of the stent due to
improper catheter preparation including mishandling or partial
6.3.3 Tracking treatments are intended to clinically simulate
ballooninflationduringpreparation.Thishasbeenidentifiedin thebendingandfrictionalforcesoftrackingthedevicethrough
a few cases where the loose, displaced, or dislodged stent was
the guide catheter and vasculature to the lesion site. Consider-
observed prior to use but may conceivably play a role in a ations for tracking treatments include: tracking medium (for
small percentage of cases where dislodgment occurs in pa-
example, air, water, water with lubricants, saline, blood) and
tients. temperature;guidecatheterandguidewireselection;simulated
F2394 − 07 (2022)
FIG. 1 Flowchart
vessel material (for example PMMA, silicone, glass, PTFE), gramssimulatevesselsin2Dwithamoderatelydifficultdegree
tortuousity, dimensionality, length and diameter; speed of of coronary tortuousity but do not include simulated lesions.
tracking; and number of repetitions to track. Angiograms or 6.3.4 Tracking through lesion treatments is intended to
autopsies of human or similar animal vasculature may be clinically simulate the bending, frictional, and mechanical
particularly useful in developing alternative anatomical mod- resistance forces of tracking the device across the lesion site.
els. An example of a tracking treatment is given in Section 7. Considerations include those of tracking treatments in addition
Two examples of tracking fixtures are given in engineering tosimulatingthelesionmaterial,type,andmorphology.Lesion
diagrams in Appendix X2. Note that these engineering dia- material encountered clinically may include calcium, fibrin,
F2394 − 07 (2022)
collagen, fat, cholesterol, endothelial cells, smooth muscle 6.5.2 Guide wire selection (or substituted test mandrel) may
cells, red blood cells, platelets, dead white blood cells, and particularly influence pre-test track treatments and securement
macrophages. Lesion types may be calcified, fibrous, lipidic, tests that include simulated tracking.Astiffer guide wire or test
thrombotic, or grumous. Lesion morphologies may be totally mandrel may place the stent delivery system under greater
or partially occluded, concentric or eccentric, focal or diffuse, normal and frictional forces. Or, it may span and round out
and of many possible shapes. curvature in a track model, especially flexible models, and
6.3.5 Post-Track Test Evaluation—To understand the effects reducethebendingoftheballoonandstent.Additionally,guide
of tracking on the probability that the stent delivery system wire stiffness varies along the length (for example, flexible
may encounter securement challenges, the measurement of 50 mm tip, moderate 50 mm stent/balloon support, and stiff
stent-balloon geometry changes post tracking may be a useful 500 mmshaft);guidewirematerialsmayhavestrongnonlinear
adjunct to force-measuring tests. In particular, the measure- response and recovery from bending, and each different guide
ment of end flaring may be useful. While end flaring may wire surface, typically chosen for lubricity and durability, may
increase the chance the stent will catch on a guide, previously respond to different catheter lumen surfaces. Completing
placedstents,lesions,andsoforth,endflaringdoesnotinitself ruggedness evaluations using a variety of wires may be
determine the force it takes to displace, dislodge, or deform the important to determine the optimal wire choice for the test.
stent. End flaring and other geometrical changes also provide a However, after choosing the optimal wire within the guidelines
measure of the effects of different track and lesion geometries of the Instructions for Use for your stent, maintain consistency
and materials. End flaring may be measured by measuring the and simplicity by executing the remaining testing with the
change in peak proximal and distal end heights relative to the optimal selection.
longitudinal continuation of the nominal stent diameter.
6.5.3 The gauge length chosen may influence the pull rate
transmitted to the stent. A shorter gauge length will have less
6.4 Securement Test and Grip Methods:
longitudinal material deformation and transmit a less variable
6.4.1 There are two main categories of stent securement
pull rate closer to the set pull rate. A 25 mm gauge length is a
tests: guide-types and lesion-types.
typical value.
6.4.2 A guide-type securement test simulates the clinical
6.5.4 The pull rate selected should allow meaningful dis-
scenario of pulling an undeployed stent delivery system back
crimination and measurement of the chosen test endpoint(s)
into a guide catheter, arterial sheath, or hemostasis valve.
and, as possible, model the clinical situation. Slower rates may
Examples of grip methods used to simulate guide-type secure-
help visualizing pull-off mechanics in videotaped tests. Faster
ment tests include guide catheters, rings, or shims ideally
rates may be more repeatable. In a test modeling lesion
designed to engage the stent end or body but not the catheter
dislodgment, set the crosshead extension speed (pull rate) to a
balloon.The shim securement test, described in Section 7, uses
slow rate such as at 0.5 in./min. In a test modeling withdrawal
complementary thin, rigid plates with rounded “V” notches to
into the catheter, set the crosshead extension speed to a faster
circumferentially engage the stent end but not the catheter
rate such as 10 to 30 in./min. Adjust the data sampling rate as
balloon. Engineering diagrams are included in Appendix X2.
appropriate; for example, increase the sampling rate for high
6.4.3 A lesion-type securement test simulates the clinical
pull speeds; have a high sampling rate for measuring peak
scenario of pushing or pulling an undeployed stent delivery
forces.
system through or around a fibrous or calcified lesion. Ex-
6.5.5 The direction of pull (proximally, distally, or angled)
amples of grip methods used to simulate lesion-type secure-
chosen may depend on factors such as stent and delivery
ment tests include tape, nubs, protrusions, or sandpaper ideally
system design and clinical relevance. The product design may
desi
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...