ASTM C1458-16
(Test Method)Standard Test Method for Nondestructive Assay of Plutonium, Tritium and 241Am by Calorimetric Assay
Standard Test Method for Nondestructive Assay of Plutonium, Tritium and <sup >241</sup>Am by Calorimetric Assay
SIGNIFICANCE AND USE
5.1 This test method is considered to be the most accurate NDA technique for the assay of many physical forms of Pu. Isotopic measurements by gamma-ray spectroscopy or destructive analysis techniques are part of this test method when it is applied to the assay of Pu.
5.1.1 Calorimetry has been applied to a wide variety of Pu-bearing solids including metals, alloys, oxides, fluorides, mixed Pu-U oxides, mixed oxide fuel pins, waste, and scrap, for example, ash, ash heels, salts, crucibles, and graphite scarfings) (2, 3). This test method has been routinely used at U.S. and European facilities for Pu process measurements and nuclear material accountability since the mid 1960’s (2-9).
5.1.2 Pu-bearing materials have been measured in calorimeter containers ranging in size from about 0.025 m to about 0.63 m in diameter and from about 0.076 m to about 1.38 m in height.
5.1.3 Gamma-ray spectroscopy typically is used to determine the Pu isotopic composition and 241Am to Pu ratio (see Test Method C1030). However, isotopic information from mass spectrometry and alpha counting measurements may be used instead (see Test Method C697).
5.2 This test method is considered to be the most accurate NDA method for the measurement of tritium. For many physical forms of tritium compounds calorimetry is currently the only practical measurement technique available.
5.3 Physical standards representative of the materials being assayed are not required for the test method.
5.3.1 This test method is largely independent of the elemental distribution of the nuclear materials in the matrix.
5.3.2 The accuracy of the method can be degraded for materials with inhomogeneous isotopic composition.
5.4 The thermal power measurement is traceable to national measurement systems through electrical standards used to directly calibrate the calorimeters or to calibrate secondary 238Pu heat standards.
5.5 Heat-flow calorimetry has been used to prepare secondary standards for neutron...
SCOPE
1.1 This test method describes the nondestructive assay (NDA) of plutonium, tritium, and 241Am using heat flow calorimetry. For plutonium the typical range of applicability, depending on the isotopic composition, corresponds to ~0.1 g to ~5 g quantities while for tritium the typical range extends from ~0.001 g to ~400 g. This test method can be applied to materials in a wide range of container sizes up to 380 L. It has been used routinely to assay items whose thermal power ranges from 0.001 W to 135 W.
1.2 This test method requires knowledge of the relative abundances of the plutonium isotopes and the 241Am/Pu mass ratio to determine the total plutonium mass.
1.3 This test method provides a direct measure of tritium content.
1.4 This test method provides a measure of 241Am either as a single isotope or mixed with plutonium.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 29-Feb-2016
- Technical Committee
- C26 - Nuclear Fuel Cycle
- Drafting Committee
- C26.10 - Non Destructive Assay
Relations
- Effective Date
- 01-Mar-2016
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Jun-2016
- Effective Date
- 01-Jan-2013
- Effective Date
- 01-Nov-2010
- Effective Date
- 01-Nov-2010
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Jun-2007
- Effective Date
- 01-Jun-2007
- Effective Date
- 01-Jan-2006
- Effective Date
- 01-Feb-2004
- Effective Date
- 01-Jan-2004
- Effective Date
- 10-Jul-2003
- Effective Date
- 10-Feb-1998
Overview
ASTM C1458-16 is the international standard established by ASTM for the nondestructive assay (NDA) of plutonium (Pu), tritium, and americium-241 (^241Am) using calorimetric assay methods. The standard outlines procedures to accurately measure these isotopes in various physical and chemical forms, leveraging heat flow calorimetry as the principal technique. Calorimetric assay is recognized as the most precise NDA technique for Pu and is often the only practical methodology for tritium in many compound forms. The method has been widely adopted in nuclear facilities across the US and Europe since the 1960s for nuclear material process monitoring and accountability.
Key Topics
- Heat Flow Calorimetry: This standard specifies the use of heat flow calorimetry to assay Pu, tritium, and ^241Am within a broad range of item sizes and shapes, including metals, oxides, alloys, and waste materials.
- Isotopic Measurement Requirements: Accurate plutonium assay additionally requires the determination of isotopic composition, typically by gamma-ray spectroscopy (see ASTM C1030), but alternatively by mass spectrometry or alpha counting (see ASTM C697).
- Direct Measurement of Tritium and Americium-241: For tritium and ^241Am, calorimetry provides a direct measure, making it invaluable when other assay techniques are impractical or less accurate.
- Wide Applicability: The standard covers a variety of container sizes (up to 380 L) with tested item power ranges from 0.001 W to 135 W, and mass ranges from ~0.1 g to ~5 g for Pu, ~0.001 g to ~400 g for tritium.
- Traceability and Calibration: Calorimeter calibration relies on electrical standards traceable to national measurement systems, ensuring reliable and auditable assaying results.
- No Need for Physical Standards: Assay can be performed without physical reference standards matching the material, as the method is largely independent of elemental distribution within the item matrix. However, materials with inhomogeneous isotopic composition may reduce accuracy.
Applications
ASTM C1458-16’s standardized calorimetric assay method is essential in the following contexts:
- Nuclear Material Accountability: Used routinely in commercial and government nuclear facilities for inventory verification and safeguards, supporting regulatory compliance and nonproliferation efforts.
- Process Control in Reprocessing and Fabrication: Essential for monitoring and controlling Pu, tritium, and ^241Am in nuclear fuel cycle operations, such as mixed oxide (MOX) fuel processing, recycling, and waste management.
- Assaying Scrap and Waste: Applied to a diverse range of Pu-bearing residues and wastes-including metals, oxides, crucibles, salts, and graphite-improving waste characterization and disposal safety.
- Measurement in Variable Container Formats: Capable of handling small samples to large waste drums, increasing flexibility in nuclear operations and minimizing sample handling risks.
- Support for Nuclear Safeguards: Provides high-precision, non-destructive measurements crucial for treaty verification and facility auditing.
Related Standards
Several key standards complement or are referenced within ASTM C1458-16 for effective implementation and integration:
- ASTM C697: Test Methods for Chemical, Mass Spectrometric, and Spectrochemical Analysis of Nuclear-Grade Plutonium Dioxide Powders and Pellets.
- ASTM C1030: Test Method for Determination of Plutonium Isotopic Composition by Gamma-Ray Spectrometry.
- ASTM C1592: Guide for Nondestructive Assay Measurements.
- ASTM C1673: Terminology relevant to NDA methods.
- ANSI N15.36: Measurement Control Program for Nondestructive Assay Measurement Control and Assurance.
- ASTM C1009: Guide for Establishing and Maintaining Quality Assurance Programs for Analytical Laboratories in the Nuclear Industry.
In summary, ASTM C1458-16 provides a robust framework for nondestructive assay of nuclear materials, ensuring measurement accuracy, process integrity, and regulatory compliance in the nuclear sector. Facilities adhering to this standard benefit from enhanced safety, improved accountability, and streamlined NDA operations.
Buy Documents
ASTM C1458-16 - Standard Test Method for Nondestructive Assay of Plutonium, Tritium and <sup >241</sup>Am by Calorimetric Assay
REDLINE ASTM C1458-16 - Standard Test Method for Nondestructive Assay of Plutonium, Tritium and <sup >241</sup>Am by Calorimetric Assay
Get Certified
Connect with accredited certification bodies for this standard

DNV
DNV is an independent assurance and risk management provider.

Lloyd's Register
Lloyd's Register is a global professional services organisation specialising in engineering and technology.

DNV Energy Systems
Energy and renewable energy certification.
Sponsored listings
Frequently Asked Questions
ASTM C1458-16 is a standard published by ASTM International. Its full title is "Standard Test Method for Nondestructive Assay of Plutonium, Tritium and <sup >241</sup>Am by Calorimetric Assay". This standard covers: SIGNIFICANCE AND USE 5.1 This test method is considered to be the most accurate NDA technique for the assay of many physical forms of Pu. Isotopic measurements by gamma-ray spectroscopy or destructive analysis techniques are part of this test method when it is applied to the assay of Pu. 5.1.1 Calorimetry has been applied to a wide variety of Pu-bearing solids including metals, alloys, oxides, fluorides, mixed Pu-U oxides, mixed oxide fuel pins, waste, and scrap, for example, ash, ash heels, salts, crucibles, and graphite scarfings) (2, 3). This test method has been routinely used at U.S. and European facilities for Pu process measurements and nuclear material accountability since the mid 1960’s (2-9). 5.1.2 Pu-bearing materials have been measured in calorimeter containers ranging in size from about 0.025 m to about 0.63 m in diameter and from about 0.076 m to about 1.38 m in height. 5.1.3 Gamma-ray spectroscopy typically is used to determine the Pu isotopic composition and 241Am to Pu ratio (see Test Method C1030). However, isotopic information from mass spectrometry and alpha counting measurements may be used instead (see Test Method C697). 5.2 This test method is considered to be the most accurate NDA method for the measurement of tritium. For many physical forms of tritium compounds calorimetry is currently the only practical measurement technique available. 5.3 Physical standards representative of the materials being assayed are not required for the test method. 5.3.1 This test method is largely independent of the elemental distribution of the nuclear materials in the matrix. 5.3.2 The accuracy of the method can be degraded for materials with inhomogeneous isotopic composition. 5.4 The thermal power measurement is traceable to national measurement systems through electrical standards used to directly calibrate the calorimeters or to calibrate secondary 238Pu heat standards. 5.5 Heat-flow calorimetry has been used to prepare secondary standards for neutron... SCOPE 1.1 This test method describes the nondestructive assay (NDA) of plutonium, tritium, and 241Am using heat flow calorimetry. For plutonium the typical range of applicability, depending on the isotopic composition, corresponds to ~0.1 g to ~5 g quantities while for tritium the typical range extends from ~0.001 g to ~400 g. This test method can be applied to materials in a wide range of container sizes up to 380 L. It has been used routinely to assay items whose thermal power ranges from 0.001 W to 135 W. 1.2 This test method requires knowledge of the relative abundances of the plutonium isotopes and the 241Am/Pu mass ratio to determine the total plutonium mass. 1.3 This test method provides a direct measure of tritium content. 1.4 This test method provides a measure of 241Am either as a single isotope or mixed with plutonium. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 5.1 This test method is considered to be the most accurate NDA technique for the assay of many physical forms of Pu. Isotopic measurements by gamma-ray spectroscopy or destructive analysis techniques are part of this test method when it is applied to the assay of Pu. 5.1.1 Calorimetry has been applied to a wide variety of Pu-bearing solids including metals, alloys, oxides, fluorides, mixed Pu-U oxides, mixed oxide fuel pins, waste, and scrap, for example, ash, ash heels, salts, crucibles, and graphite scarfings) (2, 3). This test method has been routinely used at U.S. and European facilities for Pu process measurements and nuclear material accountability since the mid 1960’s (2-9). 5.1.2 Pu-bearing materials have been measured in calorimeter containers ranging in size from about 0.025 m to about 0.63 m in diameter and from about 0.076 m to about 1.38 m in height. 5.1.3 Gamma-ray spectroscopy typically is used to determine the Pu isotopic composition and 241Am to Pu ratio (see Test Method C1030). However, isotopic information from mass spectrometry and alpha counting measurements may be used instead (see Test Method C697). 5.2 This test method is considered to be the most accurate NDA method for the measurement of tritium. For many physical forms of tritium compounds calorimetry is currently the only practical measurement technique available. 5.3 Physical standards representative of the materials being assayed are not required for the test method. 5.3.1 This test method is largely independent of the elemental distribution of the nuclear materials in the matrix. 5.3.2 The accuracy of the method can be degraded for materials with inhomogeneous isotopic composition. 5.4 The thermal power measurement is traceable to national measurement systems through electrical standards used to directly calibrate the calorimeters or to calibrate secondary 238Pu heat standards. 5.5 Heat-flow calorimetry has been used to prepare secondary standards for neutron... SCOPE 1.1 This test method describes the nondestructive assay (NDA) of plutonium, tritium, and 241Am using heat flow calorimetry. For plutonium the typical range of applicability, depending on the isotopic composition, corresponds to ~0.1 g to ~5 g quantities while for tritium the typical range extends from ~0.001 g to ~400 g. This test method can be applied to materials in a wide range of container sizes up to 380 L. It has been used routinely to assay items whose thermal power ranges from 0.001 W to 135 W. 1.2 This test method requires knowledge of the relative abundances of the plutonium isotopes and the 241Am/Pu mass ratio to determine the total plutonium mass. 1.3 This test method provides a direct measure of tritium content. 1.4 This test method provides a measure of 241Am either as a single isotope or mixed with plutonium. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM C1458-16 is classified under the following ICS (International Classification for Standards) categories: 27.120.30 - Fissile materials and nuclear fuel technology. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM C1458-16 has the following relationships with other standards: It is inter standard links to ASTM C1458-09e1, ASTM C1673-10a(2018), ASTM C697-16, ASTM C1009-13, ASTM C1673-10a, ASTM C1673-10ae1, ASTM C697-10, ASTM C1673-10, ASTM C1673-07e1, ASTM C1673-07, ASTM C1009-06, ASTM C1592-04, ASTM C697-04, ASTM C1030-03, ASTM C697-98. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM C1458-16 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: C1458 − 16
Standard Test Method for
Nondestructive Assay of Plutonium, Tritium and Am by
Calorimetric Assay
This standard is issued under the fixed designation C1458; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope C1009Guide for Establishing and Maintaining a Quality
AssuranceProgramforAnalyticalLaboratoriesWithinthe
1.1 This test method describes the nondestructive assay
241 Nuclear Industry
(NDA) of plutonium, tritium, and Am using heat flow
C1030TestMethodforDeterminationofPlutoniumIsotopic
calorimetry. For plutonium the typical range of applicability,
Composition by Gamma-Ray Spectrometry
depending on the isotopic composition, corresponds to ~0.1 g
C1592Guide for Nondestructive Assay Measurements
to ~5 g quantities while for tritium the typical range extends
C1673Terminology of C26.10 NondestructiveAssay Meth-
from ~0.001 g to ~400 g. This test method can be applied to
ods
materials in a wide range of container sizes up to 380 L. It has
beenusedroutinelytoassayitemswhosethermalpowerranges 2.2 ANSI Standard:
from 0.001 W to 135 W. ANSI N15.36Measurement Control Program–Nondestruc-
tive Assay Measurement Control and Assurance
1.2 This test method requires knowledge of the relative
abundances of the plutonium isotopes and the Am/Pu mass
3. Terminology
ratio to determine the total plutonium mass.
3.1 Definitions:
1.3 This test method provides a direct measure of tritium
3.1.1 Terms shall be defined in accordance with C26.10
content.
Terminology C1673 except for the following:
1.4 This test method provides a measure of Am either as
3.1.2 active mode—a mode of calorimeter operation where
a single isotope or mixed with plutonium.
anexternalpower,appliedbymeansofaseriesofbandheaters
1.5 The values stated in SI units are to be regarded as
for instance, is used to maintain the calorimeter at constant
standard. No other units of measurement are included in this
temperature.
standard.
3.1.3 baseline, n—the calorimeter output signal with no
1.6 This standard does not purport to address all of the
heat-generating item in the calorimeter item chamber.
safety concerns, if any, associated with its use. It is the
3.1.4 base power, n—a constant thermal power applied in a
responsibility of the user of this standard to establish appro-
calorimeter through an electrical resistance heater with no
priate safety and health practices and determine the applica-
heat-generating item in the item chamber.
bility of regulatory limitations prior to use.
3.1.5 equilibrium, n—the point at which the temperature of
2. Referenced Documents
the calorimeter measurement cell and the item being measured
stabilizes.
2.1 ASTM Standards:
C697Test Methods for Chemical, Mass Spectrometric, and
3.1.6 heat distribution error, n—the bias arising from the
Spectrochemical Analysis of Nuclear-Grade Plutonium
location of the heat source within the calorimeter chamber.
Dioxide Powders and Pellets
3.1.7 passive mode, n—a mode of calorimeter operation
wheretemperaturechange(causedbytheheattobequantified)
is measured by means of temperature sensors with no external
ThistestmethodisunderthejurisdictionofASTMCommitteeC26onNuclear
Fuel Cycle and is the direct responsibility of Subcommittee C26.10 on Non
power applied except in the case of Wheatstone bridge tem-
Destructive Assay.
peraturesensorswhereelectricalcurrentisneededtoexcitethe
Current edition approved March 1, 2016. Published April 2016. Originally
ɛ1 bridge circuit.
approved in 2000. Last previous edition approved in 2009 as C1458– 09 . DOI:
10.1520/C1458-16.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
the ASTM website. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C1458 − 16
3.1.8 power compensation mode, n—a mode of calorimeter 4.4.3 The Am mass, m , in a plutonium-bearing item is
Am
operation where a constant applied thermal power is main- determined by multiplying the Pu mass by the Am/Pu mass
tained in a calorimeter measurement chamber through the use ratio. The Am/Pu mass ratio is typically determined by
of an electric resistance heater in a closed loop control system. gamma-ray spectroscopy.
Historically, Mound Laboratory used to call this “Servo Con-
5. Significance and Use
trol.”
5.1 This test method is considered to be the most accurate
3.1.9 sensitivity, n—the change in calorimeter response per
NDA technique for the assay of many physical forms of Pu.
Watt of thermal power (usually in units of µV/W) for a heat
Isotopicmeasurementsbygamma-rayspectroscopyordestruc-
flow calorimeter.
tive analysis techniques are part of this test method when it is
3.1.10 specific power, n—the rate of energy emission by
applied to the assay of Pu.
ionizing radiation per unit mass of a radionuclide, such
241 5.1.1 Calorimetry has been applied to a wide variety of
as Am or tritium.
Pu-bearing solids including metals, alloys, oxides, fluorides,
3.1.11 thermal diffusivity, n—theratioofthermalconductiv-
mixed Pu-U oxides, mixed oxide fuel pins, waste, and scrap,
ity to the heat capacity. It measures the ability of a material to
for example, ash, ash heels, salts, crucibles, and graphite
conduct thermal energy relative to its ability to store thermal
scarfings) (2, 3). This test method has been routinely used at
energy.
U.S. and European facilities for Pu process measurements and
3.1.12 thermal power, n—therateatwhichheatisgenerated
nuclear material accountability since the mid 1960’s (2-9).
in a radioactively decaying item.
5.1.2 Pu-bearing materials have been measured in calorim-
eter containers ranging in size from about 0.025 m to about
3.1.13 thermal resistance, n—ratio of the temperature dif-
0.63 m in diameter and from about 0.076 m to about 1.38 m in
ference at two different surfaces to the heat flux through the
height.
surfaces at equilibrium.
5.1.3 Gamma-ray spectroscopy typically is used to deter-
3.1.14 thermal time constant, n—an exponential decay con-
mine the Pu isotopic composition and Am to Pu ratio (see
stant describing the rate at which a temperature approaches a
Test Method C1030). However, isotopic information from
constant value. The combination of the item and its container
mass spectrometry and alpha counting measurements may be
will have numerous thermal time constants.
used instead (see Test Method C697).
4. Summary of Test Method
5.2 This test method is considered to be the most accurate
NDA method for the measurement of tritium. For many
4.1 The item is placed in the calorimeter measurement
physical forms of tritium compounds calorimetry is currently
chamber and the heat flow at equilibrium, that is, the thermal
the only practical measurement technique available.
power,fromtheitemisdeterminedbytemperaturesensorsand
associated electronic equipment.
5.3 Physical standards representative of the materials being
assayed are not required for the test method.
4.2 The thermal power emitted by a test item is directly
5.3.1 Thistestmethodislargelyindependentoftheelemen-
related to the quantity of the radioactive material in it. The
tal distribution of the nuclear materials in the matrix.
power generated by ionizing radiation absorbed in the item is
5.3.2 The accuracy of the method can be degraded for
measured by the calorimeter.
materials with inhomogeneous isotopic composition.
4.3 The total power W from a mixture of isotopes in the
item
5.4 Thethermalpowermeasurementistraceabletonational
test item is the sum of the power from each heat-producing
measurement systems through electrical standards used to
isotope:
directly calibrate the calorimeters or to calibrate second-
W 5Σm ·P (1)
item i i 238
ary Pu heat standards.
i
th
where m is the mass of the i isotope and P is the specific
i i
5.5 Heat-flow calorimetry has been used to prepare second-
power (W/g isotope) with the sum taken over all heat-
238 239 240 241
arystandardsforneutronandgamma-rayassaysystems (7-12).
producing isotopes, most usually Pu, Pu, Pu, Pu,
242 241
Pu, and Am for Pu-bearing items
5.6 Four parameters of the item and the item packaging
4.4 ThemassofPu,tritium,or Amiscalculatedfromthe affect measurement time. These four parameters are density,
measured thermal power of an item, refer to 11.3. mass, thermal conductivity, and change in temperature. The
4.4.1 When tritium is the only heat source the measured measurement well of passive calorimeters will also affect
thermal power can be directly converted into the tritium mass measurement time because it too will need to come to the new
using the specific power of tritium, P = (0.3240 6 0.00045) equilibrium temperature. Calorimeters operated in power com-
eff
(SD) W/g (1). pensation mode maintain a constant measurement well tem-
4.4.2 When Am is the only heat source, as a single perature and have no additional effect on measurement time.
isotope, the measured thermal power can be directly converted 5.6.1 Calorimeter measurement times range from 20 min-
into mass using the specific power of Am, P = (0.1142 6 utes (13) for smaller, temperature-conditioned containers up to
eff
0.00042) (SD) W/g (see Table 1). 72 h (14) for larger containers and items with long thermal-
time constants.
5.6.2 Measurement times may be reduced by using equilib-
Theboldfacenumbersinparenthesesrefertothelistofreferencesattheendof
this standard. rium prediction techniques, by temperature preconditioning of
C1458 − 16
theitemtobemeasured,byoperatingthecalorimeterusingthe 7.1.3 Temperature Sensors—Temperature sensors consist of
power compensation technique, or by optimization of the item thermistors, thermocouples, temperature sensitive resistance
container (low thermal mass and high thermal conductivity) wires, thermopiles, or Peltier modules working in Seebeck
and packaging. mode. Temperature sensors should be placed as close as
possible in a geometrical homogeneous network all around the
6. Interferences can.
7.1.4 Thermal Sink—The temperature increases due to heat
6.1 For plutonium-bearing items alpha decay heating is
flows generated by items are measured against a reference
usually the dominant heat source and any interferences are
temperature of a thermal sink. The thermal sink could be a
typically negligible. These minor effects included the points
water bath, air bath, a solid block, usually metallic block,
6.2 and 6.3.
maintained at a constant temperature or a combination of a
6.2 Interferences for calorimetry are those processes that
solid block and water bath or air bath.
would add or subtract thermal power from the power of the
7.1.5 Electrical Components—Sensitive, stable electronic
radionuclides being assayed. Some examples include phase
components are required for accurate calorimeter measure-
changes, endothermic or exothermic chemical reactions, such
ments.
as oxidation, radiolysis of liquids, and bacterial action.
7.1.5.1 High precision voltmeters or voltage measurement
6.3 Forthe P calculation,chargedparticlesareassumedto
eff
devices are required to quantify the voltage changes generated
be totally absorbed in the matrix. The contribution of high
from the temperature sensors. The resolution should be better
energy gamma-rays (for example, 1.173 MeV and 1.333 MeV
than one part per million of the voltage range.
of the Co) needs to be calculated to not underestimate their
7.1.5.2 Stable power supplies are necessary to provide
contribution which could bias the measurement results. These
constant current to Wheatstone bridge sensors and calorimeter
assessments requiring subject matter expertise are usually
heaters.
based on high-resolution gamma spectroscopy (HRGS) and
7.1.5.3 Precision resistors with certified resistances trace-
need to be considered in a case by case basis.These effects are
able to a national measurement system may be used with
usually considered negligible (15, 16).
calibrated voltmeters to accurately determine electrical power
6.4 The loss of energy by escaping neutrons following
delivered to heaters in the calorimeter chamber. If radioactive
spontaneous and induced fissions is about 1%. The loss of
heat standards are used as part of the measurement control
energy by escaping neutrons following (α, η) reactions is
program the calorimeter voltmeters need not be calibrated nor
around 0.007% per alpha particle. These effects are also
are precision resistors required.
usually considered negligible (15, 16).
7.1.5.4 For a calorimeter operated in the power compensa-
tion mode digital-to-analog controller units are used to supply
7. Apparatus
power to an internal resistance heater to maintain a constant
7.1 Calorimetersaredesignedtomeasuredifferentsizesand
temperature differential across thermal resistances.
quantities of nuclear material. Different types of heat-flow
7.1.6 Heat Standards—Thermal power standards are re-
calorimeter systems share the common attributes listed below.
quired to calibrate the calorimeter and may be used as
7.1.1 Measurement Chamber—Heat flow calorimeters typi-
measurement control standards to check the stability of calo-
cally have a cylindrical, cuboid, or hexagonal measurement
rimeter performance (19-22).
chamber from which all of the heat flow generated by
7.1.6.1 Radioactive heat standards, typically Pu heat
radioactive decay is directed through temperature sensors. It
sources,alsomaybeusedtocalibratecalorimetersoverarange
may also have a reference chamber in which a dummy can is
of thermal powers. These standards are calibrated against
placed (so-called twin cell calorimeter) (15, 17, 18).
electrical standards traceable to a national measurement sys-
7.1.1.1 Anelectricalheatermaybebuiltintothewallsorthe
tem. The certified power is typically decay corrected to the
base of the chamber to provide measured amounts of thermal
nearest day using certified decay tables.
power into the calorimeter well.
7.1.6.2 Removable electrical heaters may be used to cali-
7.1.1.2 Insulation or active heaters (or both) should sur-
brate calorimeters. For this type of standard the power gener-
round the cavity to shield the chamber from outside tempera-
ated by the heater must be measured with electrical equipment
ture variations that would influence the thermal power mea-
regularly calibrated against standards or standard methods
surement. Typically, an insulated plug or a temperature-
traceable to a national measurement system. The power sup-
controlledplugwithanindependentelectricalheaterisinserted
plied to the electrical calibration heater may be varied over the
above the item container inside the calorimeter. For some
calibration range.
calorimeter types an insulating plug is installed permanently
7.1.7 Wheatstone Bridge—When temperature sensitive re-
below the measurement chamber.
sistance wire is used as the sensor, it is arranged in a
7.1.2 Calorimeter Can—The item to be measured may be
Wheatstone bridge configuration shown in Fig. 1.
placed in a special can that is designed to be inserted and
removedeasilyfromthecalorimeter.Itwilltypicallyhaveonly 7.1.8 Data Acquisition System—Calorimeter data collection
a small air gap to provide good thermal conductivity between is performed using computer-based data acquisition systems.
the outer surface of the can and the inner surface of the Thesystemshouldbeabletoreadsignalvoltagesorresistances
measurement chamber. at a fixed time frequency and be able to calculate and report a
C1458 − 16
FIG. 1 Calorimeter Wheatstone Bridge Circuit
power value from the item using software that detects equilib- 8.1.1.1 Equilibrium may be detected by visual inspection of
rium. Graphics and numerical data indicating system power the measurement data versus time or through statistical tests
and temperatures may be displayed to aid the operator. performed on a set of the latest data points in the time series.
7.1.9 Adapters—Low mass cylindrical metal adapters may
8.1.1.2 Statistical prediction algorithms may be used during
be fabricated to accept smaller calorimeter containers in the
transient temperature conditions to predict equilibrium and
calorimeter well, and thus, provide good thermal contact
reduce measurement time. These typically consist of exponen-
betweentheoutercontainersurfaceandcalorimeterinnerwall.
tial functions that are used to fit the measurement data. The
Heat-conducting metal foil or metal gauze fill material, typi-
fitted parameters then are used to predict the final equilibrium
callyAl or Cu, or metal shot can be used in place of machined
power.
metaladapters.Smalleritemsmaybeplacedinthecalorimeter
8.1.1.3 The temperature of the item to be measured may be
container and the void space inside the container may be filled
adjusted through the use of preconditioning heaters or baths in
withmetalfillmaterialorshottoprovidegoodthermalcontact.
order to decrease the time required to reach equilibrium.
Lackoffillmaterialdoesnotprecludemeasurementoftheitem
8.2 Heat-flow calorimeters are operated typically in one of
but it will increase the time to reach the calorimetric equilib-
two modes, passive mode or power compensation mode.
rium and thus increase the total measurement time.
8.2.1 Passive Mode Operation—A plot with an example of
7.1.10 Loading Apparatus—A hoist or other assist may be
a passive mode calorimeter response to a heat source is shown
usedtoloadandunloaditems.Roboticloadingsystemsmaybe
in Fig. 2. This plot shows that after a period of time the
used to handle the items.
temperature transient caused by the insertion of the item into
the calorimeter disappears and the calorimeter and item are in
8. Heat-Flow Calorimeter Systems
thermal equilibrium after approximately7hin this example.
8.1 A heat flow calorimeter system consists of an item
8.2.1.1 The item power, W , is calculated by the follow-
item
chamber thermally insulated from a constant temperature
ing:
environment by a thermal resistance. It may also include a
reference chamber (twin calorimeter) also insulated from a W 5 BP 2 BP /S (2)
~ !
item s 0
constant temperature environment by a thermal resistance (15,
where:
17, 18). When an item is placed in the calorimeter the
S = thecalorimetersensitivity(µV/W)atthepowerlevel,
temperature difference across the thermal resistance is dis-
determined by electrical or Pu standards,
turbed and the difference changes with time until it converges
BP = the equilibrium sensor response with the item in the
s
toaconstantvalueandequilibriumisachieved.Themagnitude
calorimeter, and
oftheshiftinthemeasuredvoltage(passivemode)orsupplied
BP = the baseline sensor response with no item in the
power (power compensation mode) is used to determine the
calorimeter.
thermal power of the item in the calorimeter.
8.1.1 Thecurveofthetemperaturedifferencedisplayingthe 8.2.2 Power Compensation Mode—In this mode of
approach to the calorimetric equilibrium is a function of operation,aconstantamountofthermalpowerisappliedtothe
severalexponentialswithdifferenttimeconstantsrelatedtothe item chamber by electrical heaters. The temperature of the
specific heats and thermal conductivities of the item matrix calorimeter item chamber is held at a constant temperature
material, packaging, and the calorimeter. difference above the temperature of the thermal sink by means
C1458 − 16
FIG. 2 Approach to Equilibrium for a Calorimeter in the Passive Mode
of a controlled power unit. The constant power causes a inserted, the external power applied is decreased to precisely
constant temperature differential to be maintained across the maintain the same signal differential. When the unknown item
thermalresistanceseparatingthemeasurementchamberfroma
is placed in the calorimeter, the control power drops over time
controlled reference temperature. The temperature differential
to a lower level. The power of the unknown is the difference
is proportional to the signal, voltage or resistance, and is the
between the two control power readings at equilibrium.Aplot
temperature difference between a sensor (or sensors) located
withanexampleofthecalorimeterresponseisshowninFig.3.
adjacent to the item being measured and the other(s) located at
8.2.2.1 The item power, W , is calculated by the follow-
item
the reference temperature. A closed-loop controller monitors
ing:
the output signal, and if a radioactive heat-generating item is
FIG. 3 Approach to Equilibrium for a Calorimeter Operated in the Power Compensation Mode
C1458 − 16
W 5 W 2 W (3) 9.2.10 Bias in the determination of P will lead to a bias in
item 0 H
eff
the assay result.
where:
9.2.11 For removable electrical heat standards the heater
W = the base power with no item in the calorimeter, and
leads to the standard will serve as a heat path for heat to
W = the power supplied to the calorimeter with the item in
H
exchange between the measurement chamber and the environ-
the calorimeter.
ment; this may bias the calibration.
8.2.2.2 The measurement time for the power compensation
9.2.12 Improperly closing the calorimeter (for example, not
mode of operation is normally shorter than for the passive
completely inserting the insulating baffle) may bias the mea-
mode because the calorimeter components are at the equilib-
surement results, degrade precision, or both.
rium temperature and the controlled internal heater can supply
9.2.13 Cables or any other conductor providing a thermal
heat actively to bring the item to equilibrium.
conduction path into and out of the calorimeter may bias the
measurementresults.Variabilityinthesepathsisamoresevere
9. Hazards
hazard.
9.1 Safety Hazards:
10. Calibration Procedure
9.1.1 It is recommended that a criticality evaluation be
carried out if fissile material is to be measured.
10.1 The type of calibration procedure depends on whether
9.1.2 Precautions should be taken to minimize electrical
the calorimeter is operated in the passive or power compensa-
shock hazards.
tion mode. In the passive mode calibration consists of deter-
9.1.3 Precautions should be taken to avoid contamination
mining the calorimeter sensitivity, S, the conversion factor
with radioactive materials.
between the differential voltage or resistance output of the
9.1.4 Precautions should be taken to minimize personnel
sensor system and the thermal power of the item being
radiation exposure to ionizing radiation. measured. In the power compensation mode calibration is
9.1.5 Pinch-point and lifting hazards may be present during setting the sensor output set point voltage that corresponds to
the loading and unloading of heavy items with calorimeters. a specific base power.
Mechanical aids, such as a hoist or any other assist, should be
10.2 Calibration–Passive Mode:
used for movement of heavy items.
10.2.1 Select a series of Pu heat standards or calibrated
9.1.6 High-power items could present a burn hazard to the
electricalstandardpowersettingsthatspantheexpectedpower
operator or damages to the instrument.
range of items to be measured. A minimum of three different
9.2 Technical Hazards: standard powers must be used.
9.2.1 Room temperature variations may affect the stability
10.2.2 Initiate a baseline measurement of the voltage with a
of the reference temperature and increase the measurement
calorimeter can, if used, filled with conductive material in the
uncertainty.
calorimeter chamber. There should be no heat source in the
pre
9.2.2 Usingameasurementresultoutsideoftherangeofthe calorimeter can. Record the baseline, BP , after equilibrium
calibration is not recommended.
is reached.
9.2.3 Careshouldbetakenintheinsertionorremovalofthe
10.2.3 Remove the calorimeter can and place theappropri-
calorimetercansothatitisnotjammedinthecalorimeterwell.
ate standard in the measurement cavity as it is done for an
9.2.4 Noise in the electronics AC supply power generated unknown item.
by nearby machinery may increase the measurement uncer-
10.2.3.1 Whether using radioactive or electrical heat
tainty.
standards, the calorimeter can must be removed from the
9.2.5 The base power for power compensation mode calo- calorimeter between each measurement, baseline, or standard.
rimetersmustnotbelessthanthehighestpowerexpectedfrom If possible, this removal is necessary even when using electri-
items. calstandardstosimulateascloselyaspossiblerealcalorimeter
9.2.6 Mechanical stress on the item chamber from the operating conditions.
weight of the item may cause a bias in the final result. 10.2.4 Closethecalorimetercanandplaceitinthecalorim-
9.2.7 The calorimeter may exhibit a small heat distribution eter well. For all measurements, the calorimeter should be
errordependentoncalorimeterdesignanditemcharacteristics. properly closed before a measurement is commenced.
10.2.5 Initiate the calorimeter run. Record the voltage out-
9.2.8 The following conditions could extend measurement
time. put (BP ) after equilibrium has been achieved.
std
9.2.8.1 Large masses of material. 10.2.6 Remove the calorimeter can from the calorimeter,
9.2.8.2 Items that make poor thermal contact with their then remove the heat standard from the can and place the can
back in the calorimeter.
containers.
post
9.2.8.3 Items that contain a large amount of insulating 10.2.7 Re-measure the baseline BP after equilibrium is
material or dead air spaces caused by several layers of reached as needed.
containment.
10.2.8 Use the known power output of the heat standard,
9.2.9 Deviation between the actual approach to equilibrium W , the calorimeter sensor value, BP , and the calculated
std std
pre post
and end-point prediction algorithm can result in measurement average baseline (average of BP and BP ), BP to
0 0 0
bias. calculate the calorimeter sensitivity, S.
C1458 − 16
BP 2 BP 10.3.4 Place a calorimeter can, if used, that is filled with
std 0 av
~ !
S 5 (4)
conductive material but with no heat generating item in the
W
std
The sensitivity S may vary with the power of the standard, calorimeter.
W , over the measurement range of the calorimeter. The
std
10.3.5 Close the calorimeter and initiate the measurement
change in sensitivity with power can be described by a
run. Once equilibrium is reached take a heater power measure-
mathematical function appropriate to the specific calorimeter
ment. This is the base power W corresponding to BP .
0 sp
response:
10.3.6 Repeat the base power measurement with no item in
S 5 S 1k 3W (5)
0 std
the item chamber at least three times. This involves removing
where:
and reinserting the calorimeter can between each of the base
power measurements. The relative standard deviation of an
S = the mathematically determined sensitivity for zero
power, and individual base power measurement should typically be less
k = the slope of the varying sensitivity. than 0.1%.
As an example, the sensitivity for a nickel resistance
11. Assay Procedure
wire-wound heat flow calorimeter of the Mound Gradient
Bridge design is shown in Fig. 4.
11.1 Item Measurement—Determine the baseline voltage
10.2.9 Perform a minimum of three replicate measurements
BP orbasepower W forthecalorimeter.Thisshouldbedone
0 0
ateachpowerlevelusingsteps10.2.1–10.2.8.Thissensitivity
using step 10.2.2 for the passive mode or steps 10.3.4 and
determination at different power levels typically need only be
10.3.5 for the power compensation mode prior to the item
done once after a calorimeter is installed.
measurement.If BP or W isstablethebaselineorbasepower
0 0
10.2.10 The equation W = f(∆V) relates sensor output to
measurement need not be done prior to every item measure-
thermal power in Watts, where ∆V is the measured voltage
ment.Timebetweenzeropowermeasurementsaresetbasedon
difference and f is an algorithm function chosen to the
calorimeter stability, required accuracy, and administrative
calorimeter design and historically has the form of a quadratic
requirements.
function (24). The relative standard deviation of an individual
11.1.1 Load the item to be assayed into the calorimeter can,
base power measurement should typically be less than 0.1%.
and close the can.
10.3 Power Compensation Mode:
11.1.2 Load the can into the calorimeter measurement
10.3.1 Select an approximate base power, W, at which the
chamber.
servo system will operate.As a general rule, it should be 10 to
11.1.3 Properly close the calorimeter.
20% higher than the highest expected item power.
11.1.4 Initiate the measurement.
10.3.2 Determine the sensitivity S of the calorimeter by
11.1.5 Terminate the measurement when the thermal equi-
performing steps 10.2.2 – 10.2.8 once for one Pu or electri-
librium has been established or predicted.
calstandardpower.Anypowerwithintherangeofitempowers
is satisfactory. 11.1.6 An additional baseline (passive) or base power
10.3.3 The actual supplied heater power (W ) used to (power compensation) run may optionally be taken after the
maintain the set point bridge, BP may be slightly different itemmeasurement-usingstep10.2.2orsteps10.3.4and10.3.5.
sp
than the target value, W, taking into account the uncertainty in The average of the pre- and post-item measurement baselines
S. orbasepowersmaybeusedfo
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
´1
Designation: C1458 − 09 C1458 − 16
Standard Test Method for
Nondestructive Assay of Plutonium, Tritium and Am by
Calorimetric Assay
This standard is issued under the fixed designation C1458; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Section 6.2 was corrected editorially in March 2009.
1. Scope
1.1 This test method describes the nondestructive assay (NDA) of plutonium, tritium, and Am using heat flow calorimetry.
For plutonium the typical range of applicability applicability, depending on the isotopic composition, corresponds to ~1~0.1 g to
~2000~5 g quantities while for tritium the typical range extends from ~0.001 g to ~10~400 g. This test method can be applied to
materials in a wide range of container sizes up to 50380 L. It has been used routinely to assay items whose thermal power ranges
from 0.001 W to 135 W.
1.2 This test method requires knowledge of the relative abundances of the plutonium isotopes and the Am/Pu mass ratio to
determine the total plutonium mass.
1.3 This test method provides a direct measure of tritium content.
1.4 This test method provides a measure of Am either as a single isotope or mixed with plutonium.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
2. Referenced Documents
2.1 ASTM Standards:
C697 Test Methods for Chemical, Mass Spectrometric, and Spectrochemical Analysis of Nuclear-Grade Plutonium Dioxide
Powders and Pellets
C1009 Guide for Establishing and Maintaining a Quality Assurance Program for Analytical Laboratories Within the Nuclear
Industry
C1030 Test Method for Determination of Plutonium Isotopic Composition by Gamma-Ray Spectrometry
C1592 Guide for Nondestructive Assay Measurements
C1673 Terminology of C26.10 Nondestructive Assay Methods
2.2 ANSI Standards:Standard:
ANSI N15.22 N15.36 Plutonium—Bearing Solids–Calibration Techniques for Calorimetric AssayMeasurement Control
Program – Nondestructive Assay Measurement Control and Assurance
ANSI N15.54 Radiometric Calorimeters–Measurement Control Program
3. Terminology
3.1 Definitions:
3.1.1 Terms shall be defined in accordance with C26.10 Terminology C1673 except for the following:
This test method is under the jurisdiction of ASTM Committee C26 on Nuclear Fuel Cycle and is the direct responsibility of Subcommittee C26.10 on Non Destructive
Assay.
Current edition approved Feb. 1, 2009March 1, 2016. Published March 2009April 2016. Originally approved in 2000. Last previous edition approved in 20002009 as
ɛ1
C1458 – 00.C1458 – 09 . DOI: 10.1520/C1458-09E01.10.1520/C1458-16.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C1458 − 16
3.1.2 active mode—a mode of calorimeter operation where an external power, applied by means of a series of band heaters for
instance, is used to maintain the calorimeter at constant temperature.
3.1.3 baseline, n—the calorimeter output signal with no heat-generating item in the calorimeter item chamber.
3.1.4 basepower, base power, n—a constant thermal power applied in a calorimeter through an electrical resistance heater with
no heat-generating item in the item chamber.
3.1.5 equilibrium, n—the point at which the temperature of the calorimeter measurement cell and the item being measured stops
changing.stabilizes.
3.1.6 heat distribution error, n—the bias arising from the location of the heat source within the calorimeter chamber.
3.1.7 passive mode, n—a mode of calorimeter operation where temperature change (caused by the heat to be quantified) is
measured by means of temperature sensors with no external power is applied to the calorimeter applied except in the case of
Wheatstone bridge temperature sensors where electrical current is needed to excite the bridge circuit.
3.1.7 sensitivity, n—the change in calorimeter response per Watt of thermal power (usually in units of micro Volts per Watt) for
a heat flow calorimeter.
3.1.8 servo controlpower compensation mode, n—a mode of calorimeter operation where a constant applied thermal power is
maintained in a calorimeter measurement chamber through the use of an electric resistance heater in a closed loop control system.
Historically, Mound Laboratory used to call this “Servo Control.”
3.1.9 sensitivity, n—the change in calorimeter response per Watt of thermal power (usually in units of μV/W) for a heat flow
calorimeter.
3.1.10 specific power, n—the rate of energy emission by ionizing radiation per unit mass of a radionuclide, such as Am or
tritium.
3.1.11 thermal diffusivity, n—the ratio of thermal conductivity to the heat capacity. It measures the ability of a material to
conduct thermal energy relative to its ability to store thermal energy.
3.1.12 thermal power, n—the rate at which heat is generated in a radioactively decaying item.
3.1.13 thermal resistance, n—ratio of the temperature difference at two different surfaces to the heat flux through the surfaces
at equilibrium.
3.1.14 thermal time constant, n—an exponential decay constant describing the rate at which a temperature approaches a constant
value. An item container combination The combination of the item and its container will have numerous thermal time constants.
3.1.14 thermel, n—the THERMal ELement of the calorimeter, including the item chamber, and temperature sensor.
4. Summary of Test Method
4.1 The item is placed in the calorimeter measurement chamber and the heat flow at equilibrium, that is, the thermal power, from
the item is determined by temperature sensors and associated electronic equipment.
4.2 The thermal power emitted by a test item is directly related to the quantity of the radioactive material in it. The power
generated by ionizing radiation absorbed in the item is measured by the calorimeter.
4.3 The total power W from a mixture of isotopes in the test item is the sum of the power from each heat-producing isotope:
item
W 5Σm ·P (1)
item i i
i
th
where m is the mass of the i isotope and P is the specific power (W/g isotope) with the sum taken over all heat-producing
i i
238 239 240 241 242 241
isotopes, most usually Pu, Pu, Pu, Pu, Pu, and Am for Pu-bearing items
4.4 The mass (m) of Pu, tritium, or Am is calculated from the measured thermal power of an item (item, referW ) using to
i
11.3the following relationship:.
W
i
m5 (1)
P
eff
where:
P = the effective specific power calculated from the isotopic composition of the item (see 11.3.2 for details of the calculation
eff
of P for plutonium).
eff
4.4.1 When tritium is the only heat source the measured thermal power can be directly converted into the tritium mass using
the specific power of tritium, P = (0.3240 6 0.00045) (SD) W/g (1).
eff
4.4.2 ForWhen Am is the only heat source, as a single isotope, the measured thermal power can be directly converted into
mass using the specific power of Am, P = (0.1142 6 0.00042) (SD) W/g (see Table 1).Table 1).
eff
The boldface numbers in parentheses refer to the list of references at the end of this standard.
C1458 − 16
A
TABLE 1 Nuclear Decay Parameters for Pu Calorimetric Assay
Standard Standard
Half-Life, Specific
Isotope Deviation, Deviation, References
Years Power, W/g
Years W/g
Pu 87.74 0.04 (0.05 %) 0.56757 0.00026 (0.05 %) (2,3)
1.9288 × 0.0003 ×
Pu 24 119 16 (0.11 %) (0.02 %) (3-5)
–3 –3
10 10
7.0824 × 0.0020 ×
Pu 6564 11 (0.17 %) (0.03 %) (6-11)
–3 –3
10 10
3.412 × 0.002 ×
Pu 14.348 0.022 (0.15 %) (0.06 %) (12-16)
–3 –3
10 10
0.1159 × 0.00026 ×
Pu 376 300 900 (0.24 %) (0.22 %) (17)
–3 –3
10 10
Am 433.6 1.4 (0.32 %) 0.1142 0.00042 (0.37 %) (15,18)
A
TABLE 1 Nuclear Decay Parameters for Pu Calorimetric Assay
Specific
Half-Life,
Isotope RSD (%) Power RSD (%) References
Years
(W/g)
Pu 87.74 0.05 0.56757 0.05 (25, 26)
1.9288 ×
Pu 24 119 0.11 0.02 (26-28)
–3
7.0824 ×
Pu 6564 0.17 0.03 (29-34)
–3
3.412 ×
Pu 14.348 0.15 0.06 (35-39)
–3
0.1159 ×
Pu 376 300 0.24 0.22 (40)
–3
Am 433.6 0.32 0.1142 0.37 (38, 41)
A
Numbers in parentheses are % relative standard deviation (RSD).ANSI N15.22, the original compiled source for these parameters, is no longer an active ANSI standard.
The References are those from ANSI N15.22 and are the appropriate references for the values in Table 1.
241 241
4.4.3 ForThe Am mixed with plutonium, the Am mass, Mm , in a plutonium-bearing item is determined by
Am
M 5 R M (2)
Am Am Pu
multiplying the Pu mass by the Am/Pu mass ratio. The Am/Pu mass ratio is typically determined by gamma-ray spectroscopy.
where:
R = the mass ratio of Am to Pu, and
Am
M = the mass of Pu.
Pu
5. Significance and Use
5.1 This test method is considered to be the most accurate NDA technique for the assay of many physical forms of Pu. Isotopic
measurements by gamma-ray spectroscopy or destructive analysis techniques are part of thethis test method when it is applied to
the assay of Pu.
5.1.1 Calorimetry has been applied to a wide variety of Pu-bearing solids including metals, alloys, oxides, fluorides, mixed Pu-U
oxides, mixed oxide fuel pins, waste, and scrap, for example, ash, ash heels, salts, crucibles, and graphite scarfings) (192, 203).
This test method has been routinely used at U.S. and European facilities for Pu process measurements and nuclear material
accountability for the last 40 years since the mid 1960’s (19-2-269).
5.1.2 Pu-bearing materials have been measured in calorimeter containers ranging in size from about 0.025 m to about 0.600.63
m in diameter and from about 0.076 m to about 0.91.38 m in height.
5.1.3 Gamma-ray spectroscopy typically is used to determine the Pu-relativePu isotopic composition and Am to Pu ratio (see
Test Method C1030). Isotopic However, isotopic information from mass spectrometry and alpha counting measurements may be
used instead (see Test Method C697).
5.2 This test method is considered to be the most accurate NDA method for the measurement of tritium. For many physical
forms of tritium compounds calorimetry is currently the only practical measurement technique available.
5.3 Physical standards representative of the materials being assayed are not required for the test method.
5.3.1 This test method is largely independent of the elemental distribution of the nuclear materials in the matrix.
5.3.2 The accuracy of the method can be degraded for materials with inhomogeneous isotopic composition.
5.4 The thermal power measurement is traceable to national measurement systems through electrical standards used to directly
calibrate the calorimeters or to calibrate secondary Pu heat standards.
5.5 Heat-flow calorimetry has been used to prepare secondary standards for neutron and gamma-ray assay systems (24-7-2912).
C1458 − 16
5.6 Calorimetry measurement times are typically longer than other NDA techniques. Four parameters of the item and the item
packaging affect measurement time. These four parameters are density, mass, thermal conductivity, and change in temperature. The
measurement well of passive calorimeters will also affect measurement time because it too will need to come to the new
equilibrium temperature. Calorimeters operated in servo power compensation mode maintain a constant measurement well
temperature and have no additional effect on measurement time.
5.6.1 Calorimeter measurement times range from 20 minutes (3013) for smaller, temperature-conditioned,temperature-
conditioned containers up to 2472 h (14) for larger containers and items with long thermal-time constants.
5.6.2 Measurement times may be reduced by using equilibrium prediction techniques, by temperature preconditioning of the
item to be measured, orby operating the calorimeter using the servo-control technique.power compensation technique, or by
optimization of the item container (low thermal mass and high thermal conductivity) and packaging.
6. Interferences
6.1 For plutonium-bearing items alpha decay heating is usually the dominant heat source and any interferences are typically
negligible. These minor effects included the points 6.2 and 6.3.
6.2 Interferences for calorimetry are those processes that would add or subtract thermal power from the power of the
radionuclides being assayed. Some examples include phase changes, endothermic or exothermic chemical reactions, such as
oxidation, radiolisisradiolysis of liquids, and bacterial action.
6.3 Heat-generating radionuclidesFor the thatP are not included in the Pcalculation, charged particles are assumed to be totally
eff
absorbed in the matrix. determination will The contribution of high energy gamma-rays (for example, 1.173 MeV and 1.333 MeV
eff
of the Co) needs to be calculated to not underestimate their contribution which could bias the measurement results high. results.
These assessments requiring subject matter expertise are usually based on high-resolution gamma spectroscopy (HRGS) and need
to be considered in a case by case basis. These effects are usually considered negligible (15, 16).
6.4 The loss of energy by escaping neutrons following spontaneous and induced fissions is about 1 %. The loss of energy by
escaping neutrons following (α, η) reactions is around 0.007 % per alpha particle. These effects are also usually considered
negligible (15, 16).
7. Apparatus
7.1 Calorimeters are designed to measure different sizes and quantities of nuclear material. Different types of heat-flow
calorimeter systems share the common attributes listed below.
7.1.1 Measurement Chamber—Heat flow calorimeters typically have a cylindrical cylindrical, cuboid, or hexagonal measure-
ment chamber from which all of the heat flow generated by radioactive decay is directed through temperature sensors. It may also
have a reference chamber in which a dummy can is placed (so-called twin cell calorimeter) (15, 17, 18).
7.1.1.1 An electrical heater may be built into the walls or the base of the chamber to provide measured amounts of thermal
power into the calorimeter well.
7.1.1.2 Insulation or active heaters (or both) are used should surround the cavity to shield the chamber from outside temperature
variations that would influence the thermal power measurement. Typically, an insulated plug or a temperature-controlled plug with
an independent electrical heater is inserted above the item container inside the calorimeter. For some calorimeter types an
insulating plug is installed permanently below the measurement chamber.
7.1.2 Calorimeter Can—The item to be measured may be placed in a special can that is designed to be inserted and removed
easily from the calorimeter. It will typically have only a small air gap to provide good thermal conductivity between the outer
surface of the can and the inner surface of the measurement chamber.
7.1.3 Temperature Sensors—Temperature sensors consist of thermistors, thermocouples, temperature sensitive resistance wire,
or thermopiles.wires, thermopiles, or Peltier modules working in Seebeck mode. Temperature sensors should be placed as close
as possible in a geometrical homogeneous network all around the can.
7.1.4 Thermal Sink—The temperature increases due to heat flows generated by items are measured against a reference
temperature of a thermal sink. The thermal sink could be a water bath, air bath, or a solid, usually metal, solid block, usually
metallic block, maintained at a constant temperature.temperature or a combination of a solid block and water bath or air bath.
7.1.5 Electrical Components—Sensitive, stable electronic components are required for accurate calorimeter measurements.
7.1.5.1 High precision voltmeters or voltage measurement devices are required to measurequantify the voltage changes
generated from the temperature sensors. The resolution of the voltmeters should be better than one part per million of the voltage
range.
7.1.5.2 Stable power supplies are necessary to provide constant current to Wheatstone bridge sensors and calorimeter heaters.
7.1.5.3 Precision resistors with certified resistances traceable to a national measurement system may be used with calibrated
voltmeters to accurately determine electrical power delivered to heaters in the calorimeter chamber. If radioactive heat standards
are used as part of the measurement control program the calorimeter voltmeters need not be calibrated,calibrated nor are precision
resistors required.
7.1.5.4 For a calorimeter operated in the servo (power replacement) power compensation mode digital-to-analog controller units
are used to supply power to an internal resistance heater to maintain a constant temperature differential across thermal resistances.
C1458 − 16
7.1.6 Heat Standards—Thermal power standards are required to calibrate the calorimeter and may be used as measurement
control standards to check the stability of calorimeter performance (31-19-3422).
238 238
7.1.6.1 Radioactive heat standards, typically powered by Pu heat Pu, sources, also may be used to calibrate calorimeters
over a range of thermal powers. These standards are calibrated against electrical standards traceable to a national measurement
system. The certified power is typically decay corrected to the nearest day using certified decay tables.
7.1.6.2 Removable electrical heaters may be used to calibrate calorimeters. For this type of standard the power generated by
the heater must be measured with electrical equipment regularly calibrated against standards or standard methods traceable to a
national measurement system. The power supplied to the electrical calibration heater may be varied over the calibration range.
7.1.7 Wheatstone Bridge—When temperature sensitive resistance wire is used as the sensor, it is arranged in a Wheatstone
bridge configuration shown in Fig. 1.
7.1.8 Data Acquisition System—Calorimeter data collection is performed using computer-based data acquisition systems. The
system should be able to read signal voltages or resistances at a fixed time frequency and be able to calculate and report a power
value from the item using software that detects equilibrium. Graphics and numerical data indicating system power and
temperatures may be displayed to aid the operator.
7.1.9 Adapters—Low mass cylindrical metal adapters may be fabricated to accept smaller calorimeter containers in the
calorimeter well, and thus, provide good thermal contact between the outer container surface and calorimeter inner wall.
Heat-conducting metal foil or metal gauze fill material, typically Al or Cu, or metal shot can be used in place of machined metal
adapters. Smaller items may be placed in the calorimeter container and the void space inside the container may be filled with metal
fill material or shot to provide good thermal contact. Lack of fill material does not preclude measurement of the item.item but it
will increase the time to reach the calorimetric equilibrium and thus increase the total measurement time.
7.1.10 Loading Apparatus—A hoist or other assist may be used to load and unload items. Robotic loading systems may be used
to handle the items.
8. Heat-Flow Calorimeter Systems
8.1 Equilibrium—A heat flow calorimeter system consists of an item chamber thermally insulated from a constant temperature
environment by a thermal resistance. It may also include a reference chamber (twin calorimeter) also insulated from a constant
temperature environment by a thermal resistance (15, 17, 18). When an item is placed in the calorimeter the temperature difference
across the thermal resistance is disturbed and the difference changes with time until it converges to a constant value and equilibrium
is achieved. The magnitude of the shift in the measured voltage (passive mode) or supplied power (servo (power compensation
mode) is used to determine the thermal power of the item in the calorimeter.
8.1.1 The curve describing the approach to equilibrium of the temperature difference of the temperature difference displaying
the approach to the calorimetric equilibrium is a function of several exponentials with different time constants related to the specific
heats and thermal conductivities of the item matrix material, packaging, and the calorimeter.
8.1.1.1 Equilibrium may be detected by visual inspection of the measurement data versus time or through statistical tests
performed on a set of the latest data points in the time series.
FIG. 1 Calorimeter Wheatstone Bridge Circuit
C1458 − 16
8.1.1.2 Statistical prediction algorithms may be used earlier during transient temperature conditions to predict equilibrium and
reduce measurement time. These typically consist of exponential functions that are used to fit the measurement data. The fitted
parameters then are used to predict the final equilibrium power.
8.1.1.3 The temperature of the item to be measured may be adjusted through the use of preconditioning heaters or baths in order
to decrease the time required to reach equilibrium.
8.2 Heat-flow calorimeters are operated typically in one of two modes, passive or servo controlled (power-replacement).mode
or power compensation mode.
8.2.1 Passive Mode—Mode Operation—In this mode of calorimeter operation a Wheatstone bridge is frequently used as the heat
sensor. The only heat generated comes from the item being measured and the current required to excite the bridge inside the
thermel. A graph of the A plot with an example of a passive mode calorimeter response to a heat source is shown in Fig. 2. This
plot shows that after a period of time the temperature transient caused by the insertion of the item into the calorimeter disappears
and the calorimeter and item are in thermal equilibrium.equilibrium after approximately 7 h in this example.
8.2.1.1 The item wattage,power, W , is calculated by the following:
iitem
W 5 BP 2 BP /S (2)
~ !
i s 0
W 5 BP 2 BP /S (2)
~ !
item s 0
where:
S = the calorimeter sensitivity (microvolts/Watt) at the power level, determined by electrical or Pu standards,
S = the calorimeter sensitivity (μV/W) at the power level, determined by electrical or Pu standards,
BP = the equilibrium sensor response with the item in the calorimeter, and
s
BP = the baseline sensor response with no item in the calorimeter.
8.2.2 Servo Mode (Power Replacement Mode)—Power Compensation Mode—In this mode of operation, a constant amount of
thermal power is applied to the item chamber by electrical heaters. The temperature of the calorimeter item chamber is held at a
constant temperature difference above the temperature of the thermal sink by means of a servo-controller. controlled power unit.
The constant power causes a constant temperature differential to be maintained across the thermal resistance separating the
measurement chamber from a controlled reference temperature. The temperature differential is proportional to the signal, voltage
or resistance, and is the temperature difference between a sensor (or sensors) located adjacent to the item being measured and the
other(s) located at the reference temperature. A closed-loop controller monitors the output signal, and if a radioactive
heat-generating item is inserted, the external power applied is decreased to precisely maintain the same signal differential. When
the unknown item is placed in the calorimeter, the control power drops over time to a lower level. The power of the unknown is
the difference between the two control power readings at equilibrium. A graph plot with an example of the calorimeter response
is shown in Fig. 3.
FIG. 2 Approach to Equilibrium for a Calorimeter Operated in the Passive Mode
C1458 − 16
FIG. 3 Approach to Equilibrium for a Calorimeter Operated in the Servo (Power Replacement) Power Compensation Mode
8.2.2.1 The item wattage,power, W , is calculated by the following:
iitem
W 5 W 2 W (3)
i 0 H
W 5 W 2 W (3)
item 0 H
where:
W = the basepower with no item in the calorimeter, and
W = the base power with no item in the calorimeter, and
W = the power supplied to the calorimeter with the item in the calorimeter.
H
8.2.2.2 The measurement time for the servo power compensation mode of operation is normally shorter than for the passive
mode because the calorimeter components are at the equilibrium temperature and the servo-controlledcontrolled internal heater can
supply heat actively to bring the item to equilibrium.
9. Hazards
9.1 Safety Hazards:
9.1.1 It is recommended that a criticality evaluation be carried out if fissile material is to be measured.
9.1.2 Precautions should be taken to minimize electrical shock hazards.
9.1.3 Precautions should be taken to avoid contamination with radioactive materials.
9.1.4 Precautions should be taken to minimize personnel radiation exposure to ionizing radiation.
9.1.5 Pinch-point and lifting hazards may be present during the loading and unloading of heavy items with calorimeters.
Mechanical aids, such as a hoist, hoist or any other assist, should be used for movement of heavy items.
9.1.6 A burn hazard can exist for high-power items. Caution should be taken to avoid burns.High-power items could present
a burn hazard to the operator or damages to the instrument.
9.2 Technical Hazards:
9.2.1 Room temperature variations may affect the stability of the reference temperature and increase the measurement
uncertainty.
9.2.2 Using a measurement result outside of the range of the calibration is not recommended.
9.2.3 Care should be taken in the insertion or removal of the calorimeter can so that it is not jammed in the calorimeter well.
9.2.4 Noise in the electronics AC supply power generated by nearby machinery may increase the measurement uncertainty.
9.2.5 The base power for servo-operated power compensation mode calorimeters must not be less than the highest power
expected from items.
9.2.6 Mechanical stress on the item chamber from the weight of the item may cause a bias in the final result.
9.2.7 The calorimeter may exhibit a small heat distribution error dependent on calorimeter design and item characteristics.
9.2.8 The following conditions could extend measurement time.
9.2.8.1 Large masses of material.
9.2.8.2 Items that make poor thermal contact with their containers.
C1458 − 16
9.2.8.3 Items that contain a large amount of insulating material or dead air spaces caused by several layers of containment.
9.2.9 Errors can result from incorrect algorithms used for equilibrium detection or prediction. The algorithm used for
equilibrium prediction that is suitable for a Deviation between the actual approach to equilibrium and end-point prediction
algorithm can result Pu heat standard or electrical standard in a conductive matrix may not be suitable for items with Pu in a
matrix with poor thermal conductivity.in measurement bias.
9.2.10 Bias in the determination of P will lead to a bias in the assay result.
eff
9.2.11 The item introduced into the calorimeter should not generate enough heat such that the temperature inside the calorimeter
would exceed safe limits.
9.2.11 For removable electrical heat standards the heater leads to the standard will serve as a heat path for heat to exchange
between the measurement chamber and the environment; this may bias the calibration.
9.2.12 ImporperlyImproperly closing the calorimeter (for example, not completely inserting the insulating baffle) may bias the
measurement results, degrade precision, or both.
9.2.13 Cables or any other conductor providing a thermal conduction path into and out of the calorimeter. calorimeter may bias
the measurement results. Variability in these paths is a more severe hazard.
10. Calibration Procedure
10.1 The type of calibration procedure depends on whether the calorimeter is operated in the passive or servo power
compensation mode. In the passive mode calibration consists of determining the calorimeter sensitivity, S, the conversion factor
between the differential voltage or resistance output of the sensor system and the thermal power of the item being measured. In
the servo power compensation mode calibration is setting the sensor output setpoint set point voltage that corresponds to a specific
base power.
10.2 Calibration–Passive Mode:
10.2.1 Select a series of Pu heat standards or calibrated electrical standard power settings that span the expected power range
of items to be measured. A minimum of three different standard powers must be used.
10.2.2 Initiate a baseline measurement of the bridge potential (voltage) voltage with a calorimeter can can, if used, filled with
pre
conductive material in the calorimeter chamber. There should be no heat source in the calorimeter can. Record the baseline, BP
(, 1),after equilibrium is reached.
10.2.3 Remove the calorimeter can and place the the appropriate Pu or electrical standard in the can. Center the standard in
the can.measurement cavity as it is done for an unknown item.
10.2.3.1 Whether using radioactive or electrical heat standards, the calorimeter can must be removed from the calorimeter
between each measurement, baseline, or standard. This If possible, this removal is necessary even when using electrical standards
to simulate as closely as possible real calorimeter operating conditions.
10.2.4 Close the calorimeter can and place it in the calorimeter well. For all measurements, the calorimeter should be properly
closed before a measurement is commenced.
10.2.5 Initiate the calorimeter run. Record the bridge potential voltage output (BP ) after equilibrium has been achieved.
std
10.2.6 Remove the calorimeter can from the calorimeter, then remove the heat standard from the can and place the can back
in the calorimeter.
post
10.2.7 Repeat another baseline run. Record Re-measure the baseline BP (19) after equilibrium is reached.reached as needed.
10.2.8 Calculate the average baseline, BP (av) = [BP (1) + BP (19)]/2.
0 0 0
10.2.8 Use the known power output of the heat standard, W , the calorimeter sensor value, BP , and the calculated average
std std
pre post
baseline, baseline (average of BP and BP ), BP to calculate the calorimeter sensitivity, S.
0 0(av)0 0
BP 2 BP
std 0 av
~ !
S 5 (4)
W
std
The sensitivity S may vary with the power of the standard, W , over the measurement range of the calorimeter. The change
std
in sensitivity with power can be described by a mathematical function appropriate to the specific calorimeter response:
S 5 S 1k 3W (5)
0 std
where:
S = the mathematically determined sensitivity for zero power, and
k = the slope of the varying sensitivity.
10.2.9.1 The sensitivity S is not usually a constant, but varies slightly with the wattage of the standard, W , over the
std
measurement range of the calorimeter. For one calorimeter, with an air gap thermal resistance, a standard measurement at 1 W
yielded a sensitivity of 28 950 μV/W while for another standard measurement at 11 W, it was 28 450 μV/W, a decrease in
sensitivity of 1.7 %. The decreased sensitivity is qualitatively consistent with increased thermal conductivity of material forming
the primary thermal resistance. For example the conductivity of dry air, a common thermal gap material, increases at the rate of
0.3 % ⁄°C. The change in sensitivity with power is described by:
S 5 S 1k 3W (6)
0 std
C1458 − 16
where:
S = the mathematically determined sensitivity for zero power, and
k = the slope of the varying sensitivity.
As an example, the sensitivity for a nickel resistance wire-wound heat flow calorimeter of the Mound Gradient Bridge design
is shown in Fig. 4.
10.2.9 Perform a minimum of three replicate measurements at each power level using steps 10.2.1 – 10.2.8. This sensitivity
determination at different power levels typically need only be done once after a calorimeter is installed.
10.2.10 CombineThe Eq 5 and equation Eq 6 and rearrange for net sensor output, BPW –= BPf ,(ΔV to obtain the equation
std 0(av)
to relate ) relates sensor output to watts:thermal
BP 2 BP 5 S 3W 1k 3W (7)
~ !
std 0 av 0 std std
~ !
The fit parameterspower in Watts, Swhere Δ V is the measured voltage difference and kf are determined by a least-squares fit
to a plot of net sensor output as a function of standard watts. Since calorimeter noise is a function of the power of the item
beingis an algorithm function chosen to the calorimeter design and historically has the form of a quadratic function measured,
(24). increasing variability with increasing power, the residuals of the least-squares fit will not be normally distributed. How-
ever the parameter estimates are not affected by this abnormal distribution.The relative standard deviation of an individual
base power measurement should typically be less than 0.1 %.
10.3 Calibration–Servo Power Compensation Mode:
10.3.1 Select an approximate basepower,base power, W, at which the servo system will operate. It should be 10–20 % As a
general rule, it should be 10 to 20 % higher than the highest expected item power.
10.3.2 Determine the sensitivity S of the calorimeter by performing steps 10.2.2 – 10.2.810.2.9 once for one Pu or electrical
standard power. Any power within the range of item powers is satisfactory.
10.3.3 Calculate the setpoint bridge potential (BP ) as follows:
sp
BP 5 BP 1S 3W (8)
sp 0 av
~ !
10.3.3 Set the controller to adjust The actual supplied heater power from(W the external power source so that ) used to maintain
the set point bridge, BP may be slightly different than the target value, W, is maintained.taking into account the uncertainty in
sp
S.
10.3.4.1 The actual supplied heater power (W ) used to maintain BP may be slightly different than the target value, W, because
0 sp
of the uncertainty in S used to calculate the setpoint.
10.3.4 Place a calorimeter can can, if used, that is filled with conductive material but with no heat generating item in the
calorimeter.
10.3.5 Close the calorimeter and initiate the measurement run. Once equilibrium is reached take a heater power measurement.
This is the basepower base power W corresponding to BP .
0 sp
10.3.6 Repeat the basepower base power measurement with no item in the item chamber at least three times. This involves
removing and reinserting the calorimeter can between each of the basepower measurements at least three times and reinserting it
in the calorimeter. The base power measurements. The relative standard deviation of an individual basepower base power
measurement should typically be less than 0.1 %.
The grouping of points at 1 W are from the CALEX PuO standard; the rest of the points are from Pu heat sources (23).
FIG. 4 The Differential Sensitivity of the ARIES I Calorimeter Shows a Sensitivity of About 20 000 μV/W
C1458 − 16
11. Assay Procedure
11.1 Item Measurement—Determine the baseline bridge potential voltage BP or basepower base power W for the calorimeter.
0 0
This should be done using step 10.2.2 for the passive mode or steps 10.3.510.3.4 and 10.3.610.3.5 for the servo power
compensation mode prior to the item measurement. If BP or W is stable the baseline or basepower base power measurement need
0 0
not be done prior to every item measurement. Time between zero power measurements are set based on calorimeter stability,
required accuracy, and administrative requirements.
11.1.1 Load the item to be assayed into the calorimeter can, and close the can.
11.1.2 Load the can into the calorimeter measurement chamber.
11.1.3 Close the calorimeter. Be sure the baffle top is completely down.Properly close the calorimeter.
11.1.4 Initiate the calorimeter run.measurement.
11.1.5 When Terminate the measurement when the thermal equilibrium has been established or predicted the run is
terminated.predicted.
11.1.6 An additional baseline (passive) or basepower (servo) base power (power compensation) run may optionally be taken
after the item measurement-using step 10.2.2 or steps 10.3.510.3.4 and 10.3.610.3.5. The average of the pre- and post-item
measurement baselines or basepowers base powers may be used for BP or average base power W .
0 0
11.2 Item Thermal Power Calculation—The calculation of the item thermal power is performed differently for passive mode
compared to servo operation.power compensation mode.
11.2.1 Power Calculation–Passive Mode: Mode—
11.2.1.1 Calculate the item wattage by solving the quadratic equation for W as follows:
i
W 5 BP 2 BP / S 1k W (9)
~ ! ~ !
i i 0 0 i
using the bridge potential voltage BP for the item, the baseline BP , and the fitted parameters S and k, from step
i 0 0
10.2.11.Calculate the item power using the equation for W (24).
item
~BP 2 BP !
item 0
W (6)
item5
S
11.2.1.2 The solution to the quadratic equation when S > 0, and k < 0 is as follows (35) :
2 ½
2S 2S BP 2 BP
0 0 0 i
W 5 2 2 (10)
FS D S DG
i
23k 23k k
and when S > 0 and k > 0 then W is:
0 i
2 ½
2S 2S BP 2 BP
0 0 0 i
W 5 1 2 (11)
FS D S DG
i
23k 23k k
11.2.1.3 Eq 10 and Eq 11 are specialized rearrangements of the more familiar form of the quadratic equation:
2b6=b 2 43a 3c
x 5 (12)
23a
This solution is necessary due to the rounding errors that occur using Eq 12 when the product of a and c is much smaller than
b squared.
11.2.2 Power Calculation–Servo Calculation–Power Compensation Mode:
11.2.2.1 Calculate the item wattage,power, W as follows:
i item
W 5 W 2 W (7)
i O H
W 5 W 2 W (7)
item O H
where:
W = control power with no item in the calorimeter, and
O
W = control power with an item in the calorimeter.
H
11.2.3 If necessary, a bias correction may be made to a calorimeter measurement based on standard measurements made under
a measurement control program. It is unusual thatif a calorimeter requirerequires a bias correction. Proper operation of the
calorimeter hardware and software should be definitively verified before resorting to bias corrections.
11.2.4 Once the thermal power is determined the quantity of radioactive material in the container is calculated using the
equations in 4.311.3.1. For Pu, a separate an isotopic analysis is required to determine P .
eff
11.3 Calorimetric Assay—Plutonium:
11.3.1 The mass of plutonium in an item can be calculated from the thermal power measured by calorimetry in units of wattsW
and from the item effective specific power in units of watts per gram W/g Pu as shown in Eq 148:
W
i
m 5 (8)
P
eff
C1458 − 16
W
item
m 5 (8)
P
eff
P is the important factor required to convert the measured thermal power in Watts from the calorimeter to grams of elemen-
eff
tal plutonium. P is most often determined from an independent knowledge of the plutonium isotopic composition and Am
eff
content of the measured item. The isotopic composition information can come from destructive mass spectrometry
measurements, or nondestructively from gamma ray isotopic analysis measurements.
11.3.2 The effective specific power (W/g Pu) of the material in the item is calculated in Eq 159:
P 5 R P (9)
eff ( i i
i
P 5 R 3P (9)
eff i i
(
i
where:
238 239 240 241 242 241
R = abundance of the i-th isotope (i = Pu, Pu, Pu, Pu, Pu, and Am) of the Pu in the item expressed as a weight
i
fraction, g isotope/g-Pu, and
238 239 240 241 242 241
R = abundance of the i-th isotope (i = Pu, Pu, Pu, Pu, Pu, and Am) of the Pu in the item expressed as a weight
i
fraction, g m /m , and
i Pu
P = a physical constant, the specific power of the i-th isotope in the item in W/g.
i
Am mass is determined using the Am:Pu ratio:
M 5 R ·M (10)
Am Am Pu
where:
R = the mass ratio of Am to Pu, and
Am
M = the mass of Pu.
Pu
11.3.2.1 The isotopic fractions, R , can be determined destructively by mass spectrometry and alpha spectrometry, or
i
nondestructively by gamma-ray spectrometry. Liquid scintillation counting may be used as well as alpha spectrometry for
determination of the Am abundance by means of destructive analysis. The terms R P are the contribution of each isotope to the
i
total item power. The isotopic fractions R are not constant, but change as the constituent isotopes in the item decay or grow in.
i
11.3.2.2 The isotopic specific powers, P , for somerelevant Pu isotopes have been directly measured or can be derived from Eq
i
1611. The specific powers and half-lives for Pu isotopes and Am as listed in ANSI N15.22–1987 are listed in Table 1. The
half-lives were established after a data review of collaborative and individual experiments by the USDOE Half-Life Evaluation
239 240
Committee. The half-lives and specific powers of Pu and Pu were determined from collaborative experiments on nearly pure
241 242
isotopic samples. The specific powers of Pu and Pu were measured by individual experimenters and the specific power
238 241
of Pu and Am were determined using Eq 1611:
2119.3
P 5 Q (11)
i i
T A
½ i i
~ !
2119.3
P 5 3Q (11)
i i i
T A
1⁄2 i
where:
Q = total disintegration energy (MeV) for alpha-particle emitters, or the average energy (MeV) of beta particle emitters,
i
T = half-life (years) of isotope i, and
½(i)
A = gram atomic weight of isotope I.
i
P = specific power of the isotope i (W/g),
i
Q = total disintegration energy (MeV) for alpha-particle emitters, or the average energy (MeV) of beta particle emitters of pure
i
beta emitter,
T ⁄2 = half-life (years) of isotope i, and
i
A = molar mass of nuclide i (g/mol).
i
11.3.2.3 A recent publication (42) of a reanalysis of Pu half-life data gives a value of 14.329 6 0.015 (SD), in agreement
with the value in Table 1. While other references give slightly different values for some of the half-lives in Table 1, an analysis
of the effects of these half-life variations (43) shows very little effect on the overall sample power giving support for continuing
to recommend use of the values in Table 1.
12. Measurement Control Procedure
12.1 Measurement control procedures are similar for all calorimeter measurements.
12.1.1 Replicate measurements of Pu or electrical heat standards are performed to demonstrate that the calorimeter system,
hardware and software, is operating correctly. Other well-characterized nuclear materials may be used as heat standards.
12.1.2 Corrections to the thermal power of radioactive heat standards due to radioactive decay shouldmust be made.
C1458 − 16
12.1.3 Control charts formedmonitoring from replicate measurements of heat standards can be beneficial in providing
quantitative means for determining that the calorimeter system is operating satisfactorysatisfactorily prior to a single assay
measurement or group of assay measurements. In addition these charts can be used to demonstrate that the calorimeter was in
control during the assay runs. measurements.
12.1.3.1 Control charts monitoring may also be used to monitor baseline or basepower base power measurements. This can
provide auxiliary information in case out-of-control operating conditions are detected.
12.1.4 Calorimeter bath temperatures can be monitored continuously to flag changes that will affect calorimeter performance.
The temperatures may be evaluated using control charts or administrative limits.
12.1.5 The frequency of standards and baseline measurements are dependent on how well the calorimeter meets performance
requirements and environmental conditions. Other factors to consider are throughput/day, portable or perm
...






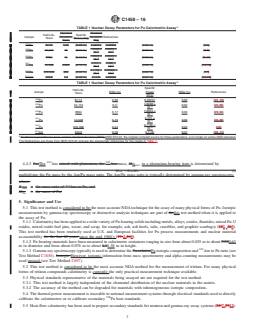

Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...