ASTM F2345-21
(Test Method)Standard Test Methods for Determination of Cyclic Fatigue Strength of Ceramic Modular Femoral Heads
Standard Test Methods for Determination of Cyclic Fatigue Strength of Ceramic Modular Femoral Heads
SIGNIFICANCE AND USE
4.1 These test methods can be used to determine the effects of head and cone materials, design variables, manufacturing, and other conditions on the cyclic load-carrying ability of modular femoral heads mounted on the cones of femoral stem prostheses.
4.2 The loading of modular femoral heads in vivo will, in general, differ from the loading defined in these methods. The results obtained here cannot be used to directly predict in-vivo performance. However, these methods are designed to allow for comparisons between the fatigue performance of different ceramic modular femoral head designs, when tested under similar conditions.
4.3 These test methods may use actual femoral prostheses or neck-cone models of simplified geometry with the same geometrical and material characteristics as in the implants. In either case, the matching metallic cone region of the test specimen selected shall be of the same material, tolerances, and finish as the final femoral stem prosthesis.
4.4 In the fatigue test methods, it is recognized that actual loading in vivo is quite varied, and that no one set of experimental conditions can encompass all possible variations. Thus, the test methods included here represent a simplified model for the purposes of comparisons between designs and materials. These test methods are intended to be performed in physiological solution.
4.5 The test data may yield valuable information about the relative strengths of different head and cone designs.
SCOPE
1.1 These test methods cover the evaluation of the cyclic fatigue strength of ceramic modular femoral heads, mounted on a cone as used on the femoral stem of the total hip arthroplasty.
1.2 These test methods were primarily developed for evaluation of ceramic (Specification F603, ISO 6474-1, ISO 6474-2, ISO 13356) head designs on metal cones but may have application to other materials.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2021
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.22 - Arthroplasty
Relations
- Effective Date
- 01-Aug-2020
- Effective Date
- 01-Oct-2016
- Effective Date
- 01-Jun-2014
- Refers
ASTM F603-12 - Standard Specification for High-Purity Dense Aluminum Oxide for Medical Application - Effective Date
- 01-Jan-2012
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Nov-2009
- Effective Date
- 01-Apr-2009
- Effective Date
- 01-Dec-2008
- Effective Date
- 01-Jan-2007
- Effective Date
- 10-Aug-2003
- Effective Date
- 10-Nov-2002
- Effective Date
- 10-Apr-2001
- Effective Date
- 10-Apr-2001
- Effective Date
- 01-Jan-2000
- Effective Date
- 01-Jan-2000
Overview
ASTM F2345-21 establishes standardized test methods for measuring the static and cyclic fatigue strength of ceramic modular femoral heads mounted on metallic cones, as used in total hip arthroplasty systems. Developed by ASTM International, this standard is widely referenced by manufacturers, testing laboratories, and regulatory agencies in the orthopaedic industry. It specifically evaluates the mechanical integrity and comparative fatigue performance of ceramic femoral head designs, allowing the assessment of how design, materials, and manufacturing processes affect cyclic load-carrying ability. While these laboratory tests do not replicate all physiological loading conditions, they provide critical insights for improving implant safety and durability.
Key Topics
- Static and Cyclic Fatigue Testing: The standard details procedures to test femoral heads for both single-event (static) and repeated (cyclic) loading scenarios, simulating the demanding forces experienced in clinical use.
- Test Specimens: Evaluation can be performed with actual hip prostheses or simplified test models matched in geometry and material characteristics, ensuring relevance to real-world applications.
- Assembly and Loading Procedures: Consistent protocols for mounting the ceramic head to the cone, including specified installation forces and rates, ensure repeatability and comparability of test outcomes.
- Test Environment: Fatigue tests are performed in physiologic solution at body temperature to mimic in vivo conditions, although actual physiological loading can differ from laboratory test parameters.
- Reporting Requirements: Extensive documentation is required, including test conditions, sample identification, and test results such as maximum load, failure mode, cycles to failure, and comprehensive specimen descriptions.
Applications
ASTM F2345-21 is crucial in the following contexts:
- Design Verification: Manufacturers use these test methods during product development to verify the fatigue strength of new ceramic femoral head designs and materials, aiding in product safety and regulatory submissions.
- Quality Assurance: Routine batch testing ensures that manufactured implants meet fatigue strength requirements, supporting consistent product quality and patient safety.
- Regulatory Compliance: The standard is often referenced for premarket notification submissions (such as 510(k) filings with the FDA) for ceramic hip systems.
- Comparative Assessment: Provides a reliable framework for comparing the relative performance of different modular femoral head designs under standardized conditions, helping clinicians and device developers select the most suitable implant options.
Related Standards
Professionals using ASTM F2345-21 may also reference:
- ASTM F603: Specification for High-Purity Dense Aluminum Oxide for Medical Application.
- ISO 6474-1 & ISO 6474-2: International standards for ceramic materials used in surgical implants, including alumina and composite ceramics.
- ISO 13356: Implants for surgery - Ceramic materials based on yttria-stabilized tetragonal zirconia (Y-TZP).
- ISO 7206-10: Determination of resistance to static load for modular femoral heads.
- ASTM E4: Practices for the force verification of testing machines, ensuring the validity of mechanical test results.
- FDA Guidance: For premarket notifications relevant to ceramic ball hip systems.
Practical Value
By providing a robust, standardized approach to fatigue testing, ASTM F2345-21 enables medical device manufacturers, laboratories, and regulators to evaluate and compare the mechanical reliability of ceramic modular femoral heads. This directly contributes to safer, more durable orthopedic implants, supports innovation in implant design, and helps to meet strict regulatory requirements for total hip replacement components.
Buy Documents
ASTM F2345-21 - Standard Test Methods for Determination of Cyclic Fatigue Strength of Ceramic Modular Femoral Heads
REDLINE ASTM F2345-21 - Standard Test Methods for Determination of Cyclic Fatigue Strength of Ceramic Modular Femoral Heads
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2345-21 is a standard published by ASTM International. Its full title is "Standard Test Methods for Determination of Cyclic Fatigue Strength of Ceramic Modular Femoral Heads". This standard covers: SIGNIFICANCE AND USE 4.1 These test methods can be used to determine the effects of head and cone materials, design variables, manufacturing, and other conditions on the cyclic load-carrying ability of modular femoral heads mounted on the cones of femoral stem prostheses. 4.2 The loading of modular femoral heads in vivo will, in general, differ from the loading defined in these methods. The results obtained here cannot be used to directly predict in-vivo performance. However, these methods are designed to allow for comparisons between the fatigue performance of different ceramic modular femoral head designs, when tested under similar conditions. 4.3 These test methods may use actual femoral prostheses or neck-cone models of simplified geometry with the same geometrical and material characteristics as in the implants. In either case, the matching metallic cone region of the test specimen selected shall be of the same material, tolerances, and finish as the final femoral stem prosthesis. 4.4 In the fatigue test methods, it is recognized that actual loading in vivo is quite varied, and that no one set of experimental conditions can encompass all possible variations. Thus, the test methods included here represent a simplified model for the purposes of comparisons between designs and materials. These test methods are intended to be performed in physiological solution. 4.5 The test data may yield valuable information about the relative strengths of different head and cone designs. SCOPE 1.1 These test methods cover the evaluation of the cyclic fatigue strength of ceramic modular femoral heads, mounted on a cone as used on the femoral stem of the total hip arthroplasty. 1.2 These test methods were primarily developed for evaluation of ceramic (Specification F603, ISO 6474-1, ISO 6474-2, ISO 13356) head designs on metal cones but may have application to other materials. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 These test methods can be used to determine the effects of head and cone materials, design variables, manufacturing, and other conditions on the cyclic load-carrying ability of modular femoral heads mounted on the cones of femoral stem prostheses. 4.2 The loading of modular femoral heads in vivo will, in general, differ from the loading defined in these methods. The results obtained here cannot be used to directly predict in-vivo performance. However, these methods are designed to allow for comparisons between the fatigue performance of different ceramic modular femoral head designs, when tested under similar conditions. 4.3 These test methods may use actual femoral prostheses or neck-cone models of simplified geometry with the same geometrical and material characteristics as in the implants. In either case, the matching metallic cone region of the test specimen selected shall be of the same material, tolerances, and finish as the final femoral stem prosthesis. 4.4 In the fatigue test methods, it is recognized that actual loading in vivo is quite varied, and that no one set of experimental conditions can encompass all possible variations. Thus, the test methods included here represent a simplified model for the purposes of comparisons between designs and materials. These test methods are intended to be performed in physiological solution. 4.5 The test data may yield valuable information about the relative strengths of different head and cone designs. SCOPE 1.1 These test methods cover the evaluation of the cyclic fatigue strength of ceramic modular femoral heads, mounted on a cone as used on the femoral stem of the total hip arthroplasty. 1.2 These test methods were primarily developed for evaluation of ceramic (Specification F603, ISO 6474-1, ISO 6474-2, ISO 13356) head designs on metal cones but may have application to other materials. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2345-21 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2345-21 has the following relationships with other standards: It is inter standard links to ASTM F603-12(2020), ASTM F603-12(2016), ASTM E4-14, ASTM F603-12, ASTM E4-10, ASTM E4-09a, ASTM E4-09, ASTM E4-08, ASTM E4-07, ASTM E4-03, ASTM E4-02, ASTM E4-99, ASTM E4-01, ASTM F603-00, ASTM F603-83(1995). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2345-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2345 − 21
Standard Test Methods for
Determination of Cyclic Fatigue Strength of Ceramic
Modular Femoral Heads
This standard is issued under the fixed designation F2345; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2.2 Other Documents:
DIN 4768 Determination of Surface Roughness R,R , and
a z
1.1 These test methods cover the evaluation of the cyclic
R with Electric Stylus Instruments; Basic Data
max
fatigue strength of ceramic modular femoral heads, mounted
ISO 6474-1 Implants for surgery—Ceramic materials—Part
on a cone as used on the femoral stem of the total hip 4
1: Ceramic materials based on high purity alumina
arthroplasty.
ISO 6474-2 Implants for surgery—Ceramic materials—Part
2: Composite materials based on a high-purity alumina
1.2 These test methods were primarily developed for evalu-
matrix with zirconia reinforcement
ation of ceramic (Specification F603, ISO 6474-1, ISO 6474-2,
ISO 7206-10 Implants for surgery—Partial and total hip-
ISO 13356) head designs on metal cones but may have
joint prostheses—Part 10: Determination of resistance to
application to other materials.
static load of modular femoral head
1.3 The values stated in SI units are to be regarded as
ISO 13356 Implants for surgery—Ceramic materials based
standard. No other units of measurement are included in this
on yttria-stabilized tetragonal zirconia (Y-TZP)
standard.
3. Terminology
1.4 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the 3.1 Definitions of Terms Specific to This Standard:
3.1.1 bore—conical blind hole in the surface of the modular
responsibility of the user of this standard to establish appro-
femoral head.
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
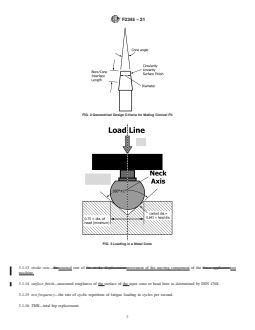
3.1.2 bore angle—included angle of the conical surface of
the bore (Fig. 1).
1.5 This international standard was developed in accor-
dance with internationally recognized principles on standard-
3.1.3 circularity—deviations of taper cross section from a
ization established in the Decision on Principles for the
perfect circle.
Development of International Standards, Guides and Recom-
3.1.4 cone—metal truncated right-circular cone (male com-
mendations issued by the World Trade Organization Technical
ponent) used to engage with a mating conical bore (female
Barriers to Trade (TBT) Committee.
component) of the modular femoral head.
3.1.5 cone angle—included angle of the conical surface of
2. Referenced Documents
the cone (Fig. 2).
2.1 ASTM Standards:
3.1.6 femoral neck-axis—centerline or axis of symmetry of
E4 Practices for Force Verification of Testing Machines
the femoral cone.
F603 Specification for High-Purity Dense Aluminum Oxide
3.1.7 head size—nominal spherical diameter of the head
for Medical Application
(generally standardized, but not limited to 22, 26, 28, 32, 36,
40, and 44 mm for total hips.)
3.1.8 installation load—the force, applied at 0° from the
These test methods are under the jurisdiction of ASTM Committee F04 on
femoral neck-axis and used to connect the head and neck
Medical and Surgical Materials and Devices and are the direct responsibility of
components prior to testing.
Subcommittee F04.22 on Arthroplasty.
Current edition approved May 1, 2021. Published June 2021. Originally
approved in 2003. Last previous edition approved in 2013 as F2345 – 03 (2013).
DOI: 10.1520/F2345-21.
2 3
For referenced ASTM standards, visit the ASTM website, www.astm.org, or Available from Beuth Verlag GmbH (DIN—DIN Deutsches Institut fur
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Normung e.V.), Burggrafenstrasse 6, 10787, Berlin, Germany.
Standards volume information, refer to the standard’s Document Summary page on Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
the ASTM website. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2345 − 21
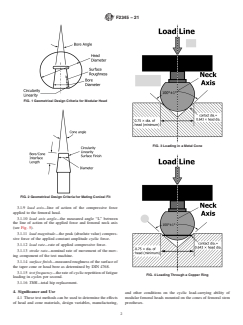
FIG. 1 Geometrical Design Criteria for Modular Head
FIG. 3 Loading in a Metal Cone
FIG. 2 Geometrical Design Criteria for Mating Conical Fit
3.1.9 load axis—line of action of the compressive force
applied to the femoral head.
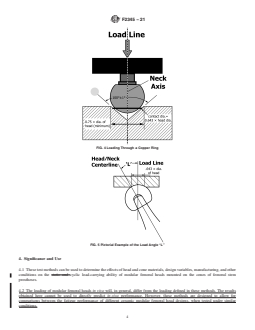
3.1.10 load axis angle—the measured angle “L” between
the line of action of the applied force and femoral neck axis
(see Fig. 5).
3.1.11 load magnitude—the peak (absolute value) compres-
sive force of the applied constant amplitude cyclic force.
3.1.12 load rate—rate of applied compressive force.
3.1.13 stroke rate—nominal rate of movement of the mov-
ing component of the test machine.
3.1.14 surface finish—measured roughness of the surface of
the taper cone or head bore as determined by DIN 4768.
3.1.15 test frequency—therateofcyclicrepetitionoffatigue
FIG. 4 Loading Through a Copper Ring
loading in cycles per second.
3.1.16 THR—total hip replacement.
4. Significance and Use
and other conditions on the cyclic load-carrying ability of
4.1 These test methods can be used to determine the effects modular femoral heads mounted on the cones of femoral stem
of head and cone materials, design variables, manufacturing, prostheses.
F2345 − 21
capacity (up to 50 kN). The test equipment should meet the
requirements outlined in Practices E4.
6.2 The varying force, as determined by suitable dynamic
verification, should be maintained at all times to be within
62 % of the largest compressive force being used for the
duration of the test.
7. Procedure
7.1 Sample Characterization:
7.1.1 Femoral heads and cones shall be characterized by the
manufacturer and shall fulfill the manufacturer’s requirements
of actual implants, according to 6.1 and 6.3 of ISO 7206-10.
7.2 Sample Assembly:
7.2.1 Following normal laboratory cleaning procedures to
remove any debris or other surface contaminants, the head and
cone are assembled on a suitable test machine. A suggested
procedure for cleaning and drying of the specimens is given in
FIG. 5 Pictorial Example of the Load Angle “L”
Appendix X1. Any cleaning procedures used should be con-
sistent with typical manufacturing practices.
7.2.2 Follow the assembly procedure according to 7.2 of
4.2 The loading of modular femoral heads in vivo will, in
ISO 7206-10.
general, differ from the loading defined in these methods. The
results obtained here cannot be used to directly predict in-vivo 7.3 General Test Requirements:
performance. However, these methods are designed to allow
7.3.1 The tests are performed at 37 °C in physiological
for comparisons between the fatigue performance of different
solution (for example, Ringer’s solution). The tests can be
ceramic modular femoral head designs, when tested under
performed at room temperature, if it is shown that such
similar conditions.
temperature change does not influence the test results signifi-
cantly. Several weeks of soaking components made from
4.3 Thesetestmethodsmayuseactualfemoralprosthesesor
inorganic materials like metal alloys or ceramics in physiologi-
neck-cone models of simplified geometry with the same
cal solution is not necessary.
geometrical and material characteristics as in the implants. In
7.3.2 New test cones and femoral heads shall be used for
either case, the matching metallic cone region of the test
each test. Note that it is imperative that components that
specimenselectedshallbeofthesamematerial,tolerances,and
survive the test should not be used for clinical purposes after
finish as the final femoral stem prosthesis.
testing.
4.4 In the fatigue test methods, it is recognized that actual
7.3.3 The load axis angle “L” shall be maintained within
loading in vivo is quite varied, and that no one set of
61° for all test samples.
experimental conditions can encompass all possible variations.
Thus, the test methods included here represent a simplified
NOTE 1—Precautions should be taken to protect the test operator from
injury by fragments should the specimen shatter when under load or when
model for the purposes of comparisons between designs and
disassembling or when storing the specimen after removal of the force
materials. These test methods are intended to be performed in
from unfractured specimens.
physiological solution.
7.4 On-Axis Fatigue Test Method:
4.5 The test data may yield valuable information about the
7.4.1 The maximum test frequency shall not exceed 30 Hz.
relative strengths of different head and cone designs.
7.4.2 The load axis angle “L” is 0 6 1°.
5. Apparatus 7.4.3 Number of Test Specimens—Aminimum of five speci-
mens is recommended for a test group.
5.1 The loading fixtures should be capable of sustaining
7.4.4 The femoral head may be loaded through a hardened
static forces up to the anticipated fracture level. The fatigue
metal 100 6 1° cone (Fig. 3) or alternatively, the contact ring
testsshouldusefixtureswithfatigueloadcapacityupto50kN.
may be protected by means of a copper ring (Fig. 4)in
5.2 The fixtures shall be constructed so that the line of force
accordance with ISO 7206-10.
application passes through the center of the femoral head.
7.4.5 The conical metal loading fixture may be damaged if
5.3 Due to the high forces anticipated in this type of cyclic, the test fractures the sample. It should be examined after each
potentially destructive test, appropriate shielding of the modu-
test fracture and be discarded if damaged. If a copper ring is
lar ball test site is recommended. usedforthecontactsurface,anewringshouldbeusedforeach
test specimen.
6. Equipment Characteristics
7.4.6 The fatigue force shall have a sinusoidal waveform
6.1 The fatigue tests should generally be performed on appliedfromtheforcemagnitudetoaminimumthatis10 %of
hydraulic (servo-hydraulic) load frames with adequate load the load magnitude.
F2345 − 21
7.4.7 The cyclic forces should be applied until 10 million amplitude, and a description of the loading contact for each
cycles without failure of the components or until fracture has sample. The report shall also contain information about the
occurred. time interval between the fatigue test and the post-fatigue burst
test.
7.5 Off-Axis Fatigue Test Method:
7.5.1 The maximum test frequency shall not exceed 30 Hz. 8.3 Test Results—The number of cycles completed by the
7.5.2 The load axis angle “L” is 30 6 1°.
sample and whether the sample failed. A statement justifying
7.5.3 A polymeric spherical concave component with the the number and kinds of samples should be included.
same segment diameter 0.643 times the head diameter should
8.4 Additional optional information characterizing the bore
be used (Fig. 5). The segment diameter should not change
and cone dimensions and tolerances (Figs. 1 and 2) would be
during the test.
desirable to better interpret the test results. This information
7.5.4 The fatigue force shall have a sinusoidal waveform
may include, but is not limited to the following: cone type,
appliedfromtheforcemagnitudetoaminimumthatis10 %of
head bore angle, head bore major/minor diameters, bore
the load magnitude.
surface roughness (R,R per DIN 4768), cone angle, cone
a z
7.6 Post-Fatigue Burst Test Method: diameter, cone surface roughness (R,R per DIN 4768),
a z
7.6.1 Following the on-axis fatigue test or off-axis fatigue
length of mating interface between the bore and cone, and
test, respectively, a burst test according to ISO 7206-10 in air method of femoral ball sterilization.
and at ambient conditions shall be performed to determine the
9. Precision and Bias
static strength of the femoral heads remaining after the cyclic
9.1 Precision—It is not possible to have a precision state-
fatigue loading.
ment because there is not a standard implant available to all
NOTE 2—The post-fatigue burst test should be done within a reasonable
users of the test method to develop such a statement.
time interval (for example, one or two days) after the fatigue test loading
Additionally, it is not possible to specify the precision of the
has been completed.
procedure in this test method because of the wide variance in
8. Report
design of the components to be tested.
8.1 The report shall identify the manufacturer(s), head size,
9.2 Bias—No statement can be made as to the bias of this
femoral head material, the definition of failure used in the test,
test method since no acceptable reference values are available.
the cone material, and the description of the cone and taper
geometries. 10. Keywords
8.2 The report shall also describe the test method, test 10.1 bore; ceramic; cone; fatigue; modular head; static;
frequency, the peak force, the load axis angle “L,” load strength
APPENDIXES
(Nonmandatory Information)
X1. RATIONALE
X1.1 Modular
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2345 − 03 (Reapproved 2013) F2345 − 21
Standard Test Methods for
Determination of Static and Cyclic Fatigue Strength of
Ceramic Modular Femoral Heads
This standard is issued under the fixed designation F2345; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 These test methods cover the evaluation of the static and cyclic fatigue strength of ceramic modular femoral heads, mounted
on a cone as used on the femoral stem of the total hip arthroplasty.
1.2 These test methods were primarily developed for evaluation of ceramic (Specifications(Specification F603 and, ISO F1873)
6474-1, ISO 6474-2, ISO 13356) head designs on metal cones but may have application to other materials.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E4 Practices for Force Verification of Testing Machines
F603 Specification for High-Purity Dense Aluminum Oxide for Medical Application
F1873 Specification for High-Purity Dense Yttria Tetragonal Zirconium Oxide Polycrystal (Y-TZP) for Surgical Implant
Applications (Withdrawn 2007)
F1875 Practice for Fretting Corrosion Testing of Modular Implant Interfaces: Hip Femoral Head-Bore and Cone Taper Interface
2.2 Other Documents:
DIN 4768 Determination of Surface Roughness R , R , and R with Electric Stylus Instruments; Basic Data
a z max
ISO 6474-1 Implants for surgery—Ceramic materials—Part 1: Ceramic materials based on high purity alumina
FDA Guidance DocumentISO 6474-2 for the Preparation of Premarket Notifications for Ceramic Ball Hip Systems
ThisThese test method ismethods are under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and isare the direct responsibility
of Subcommittee F04.22 on Arthroplasty.
Current edition approved March 15, 2013May 1, 2021. Published April 2013June 2021. Originally approved in 2003. Last previous edition approved in 20082013 as
F2345 – 03 (2008).(2013). DOI: 10.1520/F2345-03R13.10.1520/F2345-21.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from Beuth Verlag GmbH (DIN—DIN Deutsches Institut fur Normung e.V.), Burggrafenstrasse 6, 10787, Berlin, Germany.
Available from Food and Drug Administration (FDA), 5600 Fishers Ln., Rockville, MD 20857.American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor,
New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2345 − 21
(draftImplants for surgery—Ceramic materials—Part 2: Composite materials based on a high-purity alumina matrix Jan. 10,
1995)with zirconia reinforcement
ISO 7206-10 Implants for surgery—Partial and total hip-joint prostheses—Part 10: Determination of resistance to static load of
modular femoral head
ISO 13356 Implants for surgery—Ceramic materials based on yttria-stabilized tetragonal zirconia (Y-TZP)
3. Terminology
3.1 Definitions:Definitions of Terms Specific to This Standard:
3.1.1 bore—conical blind hole in the surface of the modular femoral head.
3.1.2 bore angle—included angle of the conical surface of the bore (Fig. 1).
3.1.3 circularity—deviations of taper cross section from a perfect circle.
3.1.4 cone—the proximal end of the femoral component fabricated as a truncated right cone and metal truncated right-circular
cone (male component) used to engage with a mating conical bore (female component) of the modular femoral head.
3.1.5 cone angle—included angle of the conical surface of the cone (Fig. 12).
3.1.6 femoral neck-axis—centerline or axis of symmetry of the femoral cone.
3.1.7 head size—nominal spherical diameter of the head (generally standardized, but not limited to 22, 26, 28, 32, 36, 40, and 3644
mm for total hips.)
3.1.8 installation load—the force, applied at 0° from the femoral neck axis,neck-axis and used to settleconnect the head on the
coneand neck components prior to testing.
3.1.9 load axis—line of action of the compressive force applied to the femoral head.
3.1.10 load axis angle—the measured angle “L” between the line of action of the applied force and femoral neck axis (see Fig.
5).
3.1.11 load magnitude—the peak (absolute value) compressive force of the applied constant amplitude cyclic force.
3.1.12 load rate—rate of applied compressive force.
FIG. 1 Geometrical Design Criteria for Modular BallHead
F2345 − 21
FIG. 2 Geometrical Design Criteria for Mating Conical Fit
FIG. 3 Loading in a Metal Cone
3.1.13 stroke rate—thenominal rate of the stroke displacementmovement of the moving component of the force applicator.test
machine.
3.1.14 surface finish—measured roughness of the surface of the taper cone or head bore as determined by DIN 4768.
3.1.15 test frequency—the rate of cyclic repetition of fatigue loading in cycles per second.
3.1.16 THR—total hip replacement.
F2345 − 21
FIG. 4 Loading Through a Copper Ring
FIG. 5 Pictorial Example of the Load Angle “L”
4. Significance and Use
4.1 These test methods can be used to determine the effects of head and cone materials, design variables, manufacturing, and other
conditions on the static and cyclic load-carrying ability of modular femoral heads mounted on the cones of femoral stem
prostheses.
4.2 The loading of modular femoral heads in vivo will, in general, differ from the loading defined in these methods. The results
obtained here cannot be used to directly predict in-vivo performance. However, these methods are designed to allow for
comparisons between the fatigue performance of different ceramic modular femoral head designs, when tested under similar
conditions.
F2345 − 21
4.3 These test methods may use actual femoral prostheses or neck-cone models of simplified geometry with the same geometrical
and material characteristics as in the implants. In either case, the matching metallic cone region of the test specimen selected shall
be of the same material, tolerances, and finishingfinish as the final femoral stem prosthesis.
4.3 The static test data may yield valuable information about the relative strengths and merits of different head and cone designs
for particular applications. Due to the high forces anticipated for this type of destructive test (>40 kN), the boundary conditions
and load levels far exceed possible in vivo loading parameters and therefore may not necessarily be applicable as a quantitative
indicator of expected in vivo device performance.
4.4 In the fatigue test methods, it is recognized that actual loading in vivo is quite varied, and that no one set of experimental
conditions can encompass all possible variations. Thus, the test methods included here represent a simplified model for the
purposes of comparisons between designs and materials. These test methods are intended to be performed in air.physiological
solution.
4.5 The test data may yield valuable information about the relative strengths of different head and cone designs.
5. Apparatus
5.1 The loading fixtures should be capable of sustaining static forces up to the anticipated fracture level. The static loading fixtures
may require load capacity up to 200 kN in some circumstances. The fatigue tests should use fixtures with fatigue load capacity
up to 50 kN.
5.2 The fixtures shall be constructed so that the line of force application passes through the center of the femoral head.
5.3 Due to the high forces anticipated in this type of cyclic, potentially destructive test, appropriate shielding of the modular ball
test site is recommended.
6. Equipment Characteristics
6.1 Generally, the static tests should be performed on either mechanical (power screws) or hydraulic (servo-hydraulic) load frames
with adequate load capacity (up to 200 kN). The fatigue tests should generally be performed on hydraulic (servo-hydraulic) load
frames with adequate load capacity (up to 50 kN). The test equipment should meet the requirements outlined in Practices E4.
6.2 The varying force, as determined by suitable dynamic verification, should be maintained at all times to be within 62 % of the
largest compressive force being used for the duration of the test.
7. Procedure
7.1 Sample Characterization:
7.1.1 Femoral heads and cones shall be characterized by the manufacturer and shall fulfill the manufacturer’s requirements of
actual implants, according to 6.1 and 6.3 of ISO 7206-10.
7.2 Sample Assembly:
7.2.1 Following normal laboratory cleaning procedures to remove any debris or other surface contaminants, the head and cone are
assembled on a suitable test machine. A suggested procedure for cleaning and drying of the specimens is given in Appendix X1.
Any cleaning procedures used should be consistent with typical manufacturing practices.
7.1.2 The stem taper cones are mounted at 0° load angle (L = 0°). An assembly force of 2 kN is used to mount the femoral ball
and achieve a standard head/cone reference position prior to all tests.
7.2.2 Pre-assembly of the head on the taper should be conducted under stroke or load control at a rate that will consistently
produce the required 2 kN assembly load with less than 50 N of overshoot. One of the following loading conditions for assembly
is suggested:Follow the assembly procedure according to 7.2 of ISO 7206-10.
F2345 − 21
7.1.3.1 A loading rate of 500 N/s 6 100.
7.1.3.2 A stroke rate of 0.04 mm/s.
7.3 General Test Requirements:
7.3.1 The tests are performed at room temperature in air.37 °C in physiological solution (for example, Ringer’s solution). The tests
can be performed at room temperature, if it is shown that such temperature change does not influence the test results significantly.
Several weeks of soaking components made from inorganic materials like metal alloys or ceramics in physiological solution is not
necessary.
7.3.2 New test cones and femoral heads shall be used for each test. Note that it is imperative that components that survive the test
should not be used for clinical purposes after testing.
7.3.3 The load axis angle “L” shall be maintained within 61° for all test samples.
NOTE 1—Precautions should be taken to protect the test operator from injury by fragments should the specimen shatter when under load or when
disassembling or when storing the specimen after removal of the force from unfractured specimens.
7.3 Static On Axis Test Method:
7.3.1 The load axis angle “L” is 0°.
7.3.2 Number of Test Specimens—A minimum of five specimens is recommended for a test group.
7.3.3 The femoral head may be loaded through a hardened (minimum 150 HB) metal 100 6 1° cone with a minimum surface
diameter of 0.75 times the head diameter (Fig. 3) or alternatively, the contact surface may be protected by means of a copper ring
(Fig. 4). A suggested minimum thickness for the copper ring is 1.25 mm and it should extend about 2.25 mm on either side of the
contact diameter. The diameter of contact for the applied force should be approximately 0.643 times the head size.
7.3.4 The conical metal loading fixture may be damaged if the test fractures the sample. It shall be examined after each test fracture
and be discarded if damaged. If a copper ring is used for the contact, a new ring shall be used for each test.
7.3.5 Use of one of the following loading conditions are recommended:
7.3.5.1 Position control with a stroke rate of 0.04 mm/s (0.0015 in./s) or,
7.3.5.2 Load control with a loading rate of 1 kN/s (224.8 lb/s) or less.
7.4 On Axis On-Axis Fatigue Test Method:
7.4.1 The maximum test frequency shall not exceed 30 Hz.
7.4.2 The load axis angle “L” is 0°.0 6 1°.
7.4.3 Number of Test Specimens—A minimum of five specimens is recommended for a test group.
7.4.4 The femoral head may be loaded through a hardened metal 100 6 1° cone (Fig. 3) or alternatively, the contact ring may be
protected by means of a copper ring (Fig. 4). A suggested minimum thickness for the copper ring is 1.25 mm and it should extend
about 2.25 mm on either side of the contact diameter. The diameter of contact for the applied force should be the head diameter
multiplied by the cosine of 50° or 0.643 times the head diameter.) in accordance with ISO 7206-10.
7.4.5 The conical metal loading fixture may be damaged if the test fractures the sample. It should be examined after each test
fracture and be discarded if damaged. If a copper ring is used for the contact surface, a new ring should be used for each test.test
specimen.
F2345 − 21
7.4.6 The fatigue force shall have a sinusoidal waveform applied from the force magnitude to a minimum that is 10 % of the load
magnitude.
7.4.7 The cyclic forces should be applied until 10 million cycles without failure of the components or until fracture has occurred.
7.5 Off Axis Off-Axis Fatigue Test Method:
7.5.1 The maximum test frequency shall not exceed 30 Hz.
7.5.2 The load axis angle “L” is 30°.30 6 1°.
7.5.3 A polymeric spherical concave component with the same segment diameter as suggested in0.643 times the 7.4.3head
diameter should be used (Fig. 5). The segment diameter should not change during the test.
7.5.4 The fatigue force shall have a sinusoidal waveform applied from the force magnitude to a minimum that is 10 % of the load
magnitude.
7.6 Post-Fatigue Burst Test Method:
7.6.1 Following the on-axis fatigue test or off-axis fatigue test, respectively, a burst test according to ISO 7206-10 in air and at
ambient conditions shall be performed to determine the static strength of the femoral heads remaining after the cyclic fatigue
loading.
NOTE 2—The post-fatigue burst test should be done within a reasonable time interval (for example, one or two days) after the fatigue test loading has
been completed.
8. Report
8.1 The minimum required report shall identify the manufacturer(s), head size, femoral head material, the definition of failure used
in the test, the cone material, and the description of the cone and taper geometries.
8.2 The report shall also describe the test equipment and all test parameters.method, test frequency, the peak force, the load axis
angle “L,” load amplitude, and a description of the loading contact for each sample. The report shall also contain information about
the time interval between the fatigue test and the post-fatigue burst test.
8.2.1 For the static test, the control mode, the loading rate, and a description of the loading contact.
8.2.2 For the fatigue tests, the test frequency, the peak force, the load axis angle “L,” load amplitude, and a description of the
loading contact for each sample.
8.3 Test Results: Results—
8.3.1 For the static test, the maximum failure force for each sample is required. Reporting of the mean failure force, standard
deviation, and range is also recommended.The number of cycles completed by the sample and whether the sample failed. A
statement justifying the number and kinds of samples should be included.
8.3.2 For the fatigue test methods, the number of cycles completed by the sample and whether the sample failed. A statement
justifying the number and kinds of samples should be included (FDA Guidance Document).
8.4 Additional optional information characterizing the bore and cone dimensions and tolerances (Figs. 1 and 2) would be desirable
to better interpret the test results. This information may include, but is not limited to the following: cone type, head bore angle,
head bore major/minor diameters, bore surface roughness (R , R per DIN 4768), cone angle, cone diameter, cone surface
a z
roughness (R , R per DIN 4768), length of mating interface between the bore and cone, and method of femoral ball sterilization.
a z
F2345 − 21
9. Precision and Bias
9.1 Precision—For a destructive test, wherein replicate measurements cannot be made on a single test sample, disagreement
between replicate measurements on different samples includ
...





Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...