ASTM D4273-23
(Test Method)Standard Test Method for Polyurethane Raw Materials: Determination of Primary Hydroxyl Content of Polyether Polyols
Standard Test Method for Polyurethane Raw Materials: Determination of Primary Hydroxyl Content of Polyether Polyols
SIGNIFICANCE AND USE
5.1 The primary hydroxyl content provides information about the relative reactivities of polyols in the urethane forming reaction with isocyanates.
SCOPE
1.1 Carbon-13 Nuclear Magnetic Resonance Spectroscopy (13C NMR) measures the primary hydroxyl content of ethylene oxide (EO)-propylene oxide (PO) polyether polyols used in preparing flexible polyurethane foams. This method is best suited for polyether polyols with primary hydroxyl contents of 10 to 90 %.
1.2 The values stated in SI units are to be regarded as standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
Note 1: There is no known ISO equivalent to this standard.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 28-Feb-2023
- Technical Committee
- D20 - Plastics
- Drafting Committee
- D20.22 - Cellular Materials - Plastics and Elastomers
Relations
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Apr-2022
- Effective Date
- 01-Jan-2020
- Effective Date
- 01-Aug-2019
- Effective Date
- 15-Apr-2019
- Effective Date
- 01-Feb-2019
- Effective Date
- 01-Dec-2018
- Effective Date
- 01-Nov-2018
- Effective Date
- 01-Oct-2017
- Refers
ASTM E2935-17 - Standard Practice for Conducting Equivalence Testing in Laboratory Applications - Effective Date
- 01-Oct-2017
- Effective Date
- 01-Oct-2017
- Effective Date
- 15-Aug-2017
- Refers
ASTM E2935-16 - Standard Practice for Conducting Equivalence Testing in Laboratory Applications - Effective Date
- 15-Nov-2016
- Refers
ASTM E2935-15 - Standard Practice for Conducting Equivalence Testing in Laboratory Applications - Effective Date
- 01-Oct-2015
Overview
ASTM D4273-23: Standard Test Method for Polyurethane Raw Materials: Determination of Primary Hydroxyl Content of Polyether Polyols outlines a precise method for measuring the primary hydroxyl content in polyether polyols used for flexible polyurethane foam production. Determining the primary hydroxyl content is critical for understanding polyol reactivity during urethane formation with isocyanates, which directly impacts the properties and performance of the final polyurethane products.
This test method uses Carbon-13 Nuclear Magnetic Resonance Spectroscopy (13C NMR) to quantify primary hydroxyl content in polyether polyols derived from ethylene oxide (EO) and propylene oxide (PO). The standard is applicable to polyether polyols with primary hydroxyl contents between 10 and 90%, and is developed in accordance with internationally recognized standardization principles.
Key Topics
- 13C NMR Spectroscopy: The method relies on high-resolution 13C NMR to distinguish between primary and secondary hydroxyl carbons within polyols. By integrating the resolved peaks in specific ppm regions, it is possible to accurately determine the relative proportion of primary hydroxyl groups.
- Sample Preparation: Solutions of polyol samples are prepared in deuterated solvents with a tetramethylsilane (TMS) internal standard, enabling precise spectral analysis.
- Instrumentation Requirements: A Fourier-Transform NMR spectrometer with carbon-13 capability and a minimum signal-to-noise ratio is essential for accurate measurements.
- Result Calculation: The percentage of primary hydroxyl is calculated as the ratio of the area corresponding to primary hydroxyl peaks over the total hydroxyl area (primary plus secondary), providing results directly relevant for process optimization.
- Significance: The primary hydroxyl content influences the reactivity of polyols with isocyanates, crucial for controlling polyurethane foam properties such as flexibility, durability, and processing stability.
- Safety Considerations: The standard requires users to follow all relevant safety practices related to NMR equipment and sample handling, especially with regard to strong magnetic fields and the use of deuterated solvents.
Applications
ASTM D4273-23 is widely applicable in the following contexts:
- Quality Control in Polyurethane Manufacturing: Polyol producers and polyurethane foam manufacturers use this standard to evaluate the reactivity and quality of raw materials, ensuring optimal foam performance and consistency.
- Formulation Development: Chemists and R&D teams employ the method to tailor polyol blends that yield desirable physical properties in the final polyurethane products, ranging from soft foams for furniture to formative flexible foams for automotive and appliance applications.
- Interlaboratory Comparisons: The standard promotes consistency across manufacturing locations and laboratories by providing a uniform method for hydroxyl content determination, supporting reliable supply chains and product development cycles.
- Research and Material Certification: Academic and industrial researchers adopt ASTM D4273-23 for materials characterization, formulation certification, and publication of consistent, reproducible data.
Related Standards
For comprehensive assessment and compatibility, ASTM D4273-23 references and aligns with the following key ASTM standards:
- ASTM D883 - Terminology Relating to Plastics: Provides definitions for key terms.
- ASTM E456 - Terminology Related to Quality and Statistics: Standardizes terminology regarding precision and bias.
- ASTM E691 - Practice for Conducting Interlaboratory Studies to Determine Precision of Test Methods.
- ASTM E2935 - Practice for Evaluating Equivalence of Two Testing Processes.
- ASTM E2977 - Practice for Measuring and Reporting Performance of FT-NMR Spectrometers.
There is currently no ISO equivalent to ASTM D4273-23, positioning it as a unique guideline for global polyurethane and polyether polyol industries.
Keywords: ASTM D4273-23, polyurethane raw materials, polyether polyols, primary hydroxyl content, 13C NMR, flexible polyurethane foam, quality control, polyol reactivity, standard test method, NMR spectroscopy.
Buy Documents
ASTM D4273-23 - Standard Test Method for Polyurethane Raw Materials: Determination of Primary Hydroxyl Content of Polyether Polyols
REDLINE ASTM D4273-23 - Standard Test Method for Polyurethane Raw Materials: Determination of Primary Hydroxyl Content of Polyether Polyols
Get Certified
Connect with accredited certification bodies for this standard

Smithers Quality Assessments
US management systems and product certification.

DIN CERTCO
DIN Group product certification.
Sponsored listings
Frequently Asked Questions
ASTM D4273-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Polyurethane Raw Materials: Determination of Primary Hydroxyl Content of Polyether Polyols". This standard covers: SIGNIFICANCE AND USE 5.1 The primary hydroxyl content provides information about the relative reactivities of polyols in the urethane forming reaction with isocyanates. SCOPE 1.1 Carbon-13 Nuclear Magnetic Resonance Spectroscopy (13C NMR) measures the primary hydroxyl content of ethylene oxide (EO)-propylene oxide (PO) polyether polyols used in preparing flexible polyurethane foams. This method is best suited for polyether polyols with primary hydroxyl contents of 10 to 90 %. 1.2 The values stated in SI units are to be regarded as standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Note 1: There is no known ISO equivalent to this standard. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 The primary hydroxyl content provides information about the relative reactivities of polyols in the urethane forming reaction with isocyanates. SCOPE 1.1 Carbon-13 Nuclear Magnetic Resonance Spectroscopy (13C NMR) measures the primary hydroxyl content of ethylene oxide (EO)-propylene oxide (PO) polyether polyols used in preparing flexible polyurethane foams. This method is best suited for polyether polyols with primary hydroxyl contents of 10 to 90 %. 1.2 The values stated in SI units are to be regarded as standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Note 1: There is no known ISO equivalent to this standard. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D4273-23 is classified under the following ICS (International Classification for Standards) categories: 83.080.20 - Thermoplastic materials. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D4273-23 has the following relationships with other standards: It is inter standard links to ASTM D883-24, ASTM D883-23, ASTM E456-13a(2022)e1, ASTM D883-20, ASTM D883-19c, ASTM D883-19a, ASTM D883-19, ASTM D883-18a, ASTM D883-18, ASTM E456-13A(2017)e1, ASTM E2935-17, ASTM E456-13A(2017)e3, ASTM D883-17, ASTM E2935-16, ASTM E2935-15. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D4273-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D4273 − 23
Standard Test Method for
Polyurethane Raw Materials: Determination of Primary
Hydroxyl Content of Polyether Polyols
This standard is issued under the fixed designation D4273; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* Fourier-Transform Nuclear Magnetic Resonance (FT-
NMR) Spectrometers for Liquid Samples
1.1 Carbon-13 Nuclear Magnetic Resonance Spectroscopy
( C NMR) measures the primary hydroxyl content of ethylene
3. Terminology
oxide (EO)-propylene oxide (PO) polyether polyols used in
3.1 Definitions—Terms used in this standard are defined in
preparing flexible polyurethane foams. This method is best
accordance with Terminology D883 and Practice E2977, unless
suited for polyether polyols with primary hydroxyl contents of
otherwise specified. For terms relating to precision and bias
10 to 90 %.
and associated issues, the terms used in this standard are
1.2 The values stated in SI units are to be regarded as
defined in accordance with Terminology E456.
standard.
1.3 This standard does not purport to address all of the
4. Summary of Test Method
safety concerns, if any, associated with its use. It is the
4.1 Peaks of the primary and secondary hydroxyl carbons of
responsibility of the user of this standard to establish appro-
polyether polyols used in flexible polyurethane foams are
priate safety, health, and environmental practices and deter- 13
well-resolved in high-resolution C NMR spectra. The pri-
mine the applicability of regulatory limitations prior to use.
mary hydroxyl content is determined from the ratio of the
primary hydroxyl area to the total hydroxyl (primary and
NOTE 1—There is no known ISO equivalent to this standard.
secondary) area.
1.4 This international standard was developed in accor-
dance with internationally recognized principles on standard-
5. Significance and Use
ization established in the Decision on Principles for the
5.1 The primary hydroxyl content provides information
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical about the relative reactivities of polyols in the urethane
Barriers to Trade (TBT) Committee. forming reaction with isocyanates.
6. Interferences
2. Referenced Documents
6.1 Primary hydroxyl PO methylene carbons (where the
2.1 ASTM Standards:
methylene carbon is next to the hydroxyl group and the
D883 Terminology Relating to Plastics
methine carbon is next to the ether oxygen) are integrated with
E456 Terminology Relating to Quality and Statistics
the secondary hydroxyl carbons and are therefore not included
E691 Practice for Conducting an Interlaboratory Study to
in the primary hydroxyl content as measured by this method.
Determine the Precision of a Test Method
Any additional non-polyether polyol compounds in the sample
E2935 Practice for Evaluating Equivalence of Two Testing
that produce NMR peaks in the 68-60 ppm region may
Processes
interfere with the measurement.
E2977 Practice for Measuring and Reporting Performance of
7. Apparatus
This test method is under the jurisdiction of ASTM Committee D20 on Plastics
7.1 Fourier-Transform NMR (FT-NMR) Spectrometer, with
and is the direct responsibility of Subcommittee D20.22 on Cellular Materials -
carbon-13 capability and a carbon-13 resonance frequency of
Plastics and Elastomers.
50 MHz (proton resonance frequency of 200 MHz) or higher.
Current edition approved March 1, 2023. Published March 2023. Originally
The spectrometer is to have a minimum carbon-13 signal-to-
approved in 1983. Last previous edition approved in 2018 as D4273 - 18. DOI:
10.1520/D4273-23.
noise ratio of 70:1 based on the benzene carbon signal in a
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
60 % benzene-d6, 40 % p-dioxane (v/v) sample (ASTM NMR
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
standard) that has been pulsed once using a 90° pulse angle
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. under the conditions specified in Practice E2977.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4273 − 23
7.2 NMR sample tubes having outside diameters of 5 mm or 12.4 Obtain a stable deuterium lock on the solvent.
more. 13 1
12.5 Tune and match the probe for C and H channels.
7.3 NMR spinners.
12.6 Shim the sample to optimize magnetic field-
homogeneity.
8. Reagents and Materials
12.7 Acquire the NMR data.
8.1 All reagents are to be spectroscopic-grade and free of
magnetic materials.
12.8 Zero fill the data. The recommended value is 1 or 2 ×
8.1.1 Deuterated chloroform or deuterated acetone, con-
number of data points.
taining tetramethylsilane (TMS) as an internal standard.
12.9 Apply a spectral weighting function (apodization) and
Fourier Transform the Free Induction Decay (FID). The
9. Hazards
recommended apodization is an exponential window multipli-
9.1 Magnetic Fields—Follow the manufacturer’s recom-
cation and a typical line broadening value is 1/acquisition time.
mendation for the safe operation of the instrument.
12.10 Phase and baseline correct the spectrum.
9.1.1 Persons with implanted or attached medical devices
such as pacemakers and prosthetic parts must remain outside
12.11 Set the internal TMS reference to 0 ppm.
the 5-gauss perimeter.
12.12 Integrate the peaks of interest. The primary hydroxyl
9.1.2 Objects made of ferromagnetic material will be at-
peaks typically resonate in the 61.0 to 62.5 ppm region and the
tracted to the magnet and are to be kept a safe distance away,
secondary hydroxyl peaks typically resonate in the 65.5 to 67.5
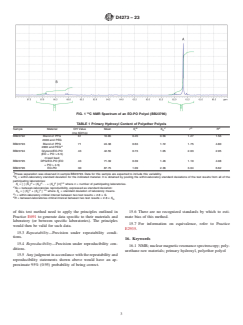
outside the 5-gauss perimeter.
ppm region. An example is shown in Fig. 1.
10. Preparation of Apparatus
NOTE 2—The use of 0.025 to 0.05 M Cr(acac) has been found to
shorten relaxation times allowing for shorter data acquisition times.
10.1 Prepare a proton decoupled carbon-13 NMR
experiment, selecting appropriate parameters to obtain quanti-
13. Calculation
tative integration of the peaks in the 68-60 ppm region.
13.1 Calculate the percent primary hydroxyl content using
10.1.1 Inverse Gated Decoupling—Decouple only during
the following equation:
acquisition.
10.1.2 Pulse Angle and Sequence Delay Time—Select a 90
Primary hydroxyl, % 5 A* (1)
degree pulse angle with a sequence delay of 5 to 10 × T of the
1 A1B
peak with the longest relaxation time in the 68-60 ppm region.
where:
It is acceptable to use a different pulse angle/sequence delay
A = area of terminal EO methylene carbons (primary hy-
combination to reduce acquisition time provided that quantita-
droxyl peaks),
tive data acquisition is not compromised.
B = area of terminal PO methine carbons (secondary hy-
10.1.3 Number of Scans—Select the appropriate number to
droxyl peaks—does not correct for primary hydroxyl
yield a minimum signal to noise of > 10:1 for the smallest peak
PO terminations).
integrated over the 68-60 ppm region (usually 1024 to 2048).
10.1.4 Sweep Width— ~ 220 ppm.
14. Report
10.1.5 Transmitter Frequency— ~100 ppm.
14.1 Report the % primary hydroxyl content to the nearest
10.1.6 Acquisition Time—1 to 2 s.
two decimal places.
11. Calibration and Standardization
15. Precision and Bias
11.1 This test method does not require standards. To evalu-
ate the test method, it is feasible to prepare standards by 15.1 Table 1 is based on a round robin conducted in 2016 in
accordance with Practice E691, involving five materials tested
blending commercially available poly(propylene oxide) and
poly(ethylene oxide) diols. Ideally, the molecular weights of by eight laboratories. For each material, all the samples were
prepared at one source, but the individual specimens were
the diols are to be 300 or more since lower molecular weight
polyols can contain structural configurations that are not prepared at the laboratories which tested them. Each test result
typical of polyether polyols used in flexible polyurethane was a single determination. Each laboratory obtained two test
foams. results for each material.
15.2 Caution—The explanation of repeatability (r) and re-
12. Procedure
producibility (R) is only intended to present a meaningful way
12.1 Prepare a solution of the polyol sample in deuterated
of considering the approximate precision of this test method.
chloroform or deuterated acetone containing TMS as an
Do not apply the data in Table 1 to accept or reject materials,
internal standard. A 30-60 % solution is recommended. (See
as these data apply only to the materials tested in the round
Note 2.)
robin and are unlikely to be rigorously representative of other
12.2 Transfer an appropriate amount of the sample solution lots, formulations, conditions, materials, or laboratories. Users
to an NMR tube.
12.3 Place the NMR tube into a spinner, adjust it to the
Supporting data are available from ASTM Headquarters. Request RR:D20-
appropriate depth and insert it into the spectrometer probe. 1270.
D4273 − 23
FIG. 1 C NMR Spectrum of an EO-PO Polyol (BB23796)
TABLE 1 Primary Hydroxyl Content of Polyether Polyols
B C D E
Sample Material OH Value Mean S S r R
r R
(mg KOH/g)
BB23792 Blend of PPG 61 10.85 0.45 0.56 1.27 1.56
2000 and PEG
BB23793 Blend of PPG 71 24.38 0.63 1.72 1.75 4.80
A
2000 and PEG
BB23794 Glycerol/EO-PO 43 42.55 0.73 1.05 2.03 2.95
(EO + PO > 6.5)
mixed feed
BB23795 DPG/EO-PO (EO 43 71.39 0.39 1.46 1.10 4.08
+ PO > 4.5)
BB23796 EO-PO 30 87.75 1.09 2.36 3.04 6.62
A
Phase separation was observed in sample BB23793. Data for this sample are expected to include this variability.
B
S = within-laboratory standard deviation for the indicated material. It is obtained by pooling the within-laboratory standard deviations of the test results from all of the
r
participating laboratories:
2 2 2 1/2
S = [ [ (S ) + (S ) { { + (S ) ]/n] where n = number of participating laboratories.
r 1 2 n
C
S = between-laboratories reproducibility, expressed as standard deviation:
R
2 2 1/2
S = [ (S ) + (S ) ] where S = standard deviation of laboratory means.
R r L L
D
r = within-laboratory critical interval between two test results = 2.8 × S
r
E
R = between-laboratories critical interval between two test results = 2.8 × S
R
of this test method need to apply the principles outlined in 15.6 There are no recognized standards by which to esti-
Practice E691 to generate data specific to their materials and mate bias of this method.
laboratory (or between specific laboratories). The principles
15.7 For information on equivalence, refer to Practice
would then be valid for such data.
E2935.
15.3 Repeatability—Precision under repeatability condi-
tions.
16. Keywords
15.4 Reproducibility—Precision under reproducibility con-
16.1 NMR; nuclear magnetic resonance spectroscopy; poly-
ditions.
urethane raw materials; primary hydroxyl, polyether polyol
15.5 Any judgment in accordance with the repeatability and
reproducibility statements shown above would have an ap-
proximate 95% (0.95) probability of being correct.
D4273 − 23
APPENDIX
(Nonmandatory Information)
X1. FLUORINE-19 NUCLEAR MAGNETIC RESONANCE SPECTROSCOPY METHOD FOR DETERMINATION OF PRIMARY
HYDROXYL CONTENT OF POLYE
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D4273 − 18 D4273 − 23
Standard Test Method for
Polyurethane Raw Materials: Determination of Primary
Hydroxyl Content of Polyether Polyols
This standard is issued under the fixed designation D4273; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 Carbon-13 Nuclear Magnetic Resonance Spectroscopy ( C NMR) measures the primary hydroxyl content of ethylene oxide
(EO)-propylene oxide (PO) polyether polyols used in preparing flexible polyurethane foams. This method is best suited for
polyether polyols with primary hydroxyl contents of 10 to 90 %.
1.2 The values stated in SI units are to be regarded as standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
NOTE 1—There is no known ISO equivalent to this standard.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D883 Terminology Relating to Plastics
E456 Terminology Relating to Quality and Statistics
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E2935 Practice for Evaluating Equivalence of Two Testing Processes
E2977 Practice for Measuring and Reporting Performance of Fourier-Transform Nuclear Magnetic Resonance (FT-NMR)
Spectrometers for Liquid Samples
3. Terminology
3.1 Definitions—For definitions of terms that appear in this method, refer to Terms used in this standard are defined in accordance
with Terminology D883 and Practice E2977, unless otherwise specified. For terms relating to precision and bias and associated
issues, the terms used in this standard are defined in accordance with Terminology E456.
This test method is under the jurisdiction of ASTM Committee D20 on Plastics and is the direct responsibility of Subcommittee D20.22 on Cellular Materials - Plastics
and Elastomers.
Current edition approved April 1, 2018March 1, 2023. Published April 2018March 2023. Originally approved in 1983. Last previous edition approved in 20112018 as
D4273 - 11.D4273 - 18. DOI: 10.1520/D4273-18.10.1520/D4273-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4273 − 23
4. Summary of Test Method
4.1 Peaks of the primary and secondary hydroxyl carbons of polyether polyols used in flexible polyurethane foams are
well-resolved in high-resolution C NMR spectra. The primary hydroxyl content is determined from the ratio of the primary
hydroxyl area to the total hydroxyl (primary and secondary) area.
5. Significance and Use
5.1 The primary hydroxyl content provides information about the relative reactivities of polyols.polyols in the urethane forming
reaction with isocyanates.
6. Interferences
6.1 Primary hydroxyl PO methylene carbons (where the methylene carbon is next to the hydroxyl group and the methine carbon
is next to the ether oxygen) are integrated with the secondary hydroxyl carbons and are therefore not included in the primary
hydroxyl content as measured by this method. Any additional non-polyether polyol compounds in the sample that produce NMR
peaks in the 68-60 ppm region may interfere with the measurement.
7. Apparatus
7.1 Fourier-Transform NMR (FT-NMR) Spectrometer, with carbon-13 capability and a carbon-13 resonance frequency of 50 MHz
(proton resonance frequency of 200 MHz) or higher. The spectrometer is to have a minimum carbon-13 signal-to-noise ratio of
70:1 based on the benzene carbon signal in a 60 % benzene-d6, 40 % p-dioxane (v/v) sample (ASTM NMR standard) that has been
pulsed once using a 90° pulse angle under the conditions specified in Practice E2977.
7.2 NMR sample tubes having outside diameters of 5 mm or more.
7.3 NMR spinners.
8. Reagents and Materials
8.1 All reagents are to be spectroscopic-grade and free of magnetic materials.
8.1.1 Deuterated chloroform or deuterated acetone, containing tetramethylsilane (TMS) as an internal standard.
9. Hazards
9.1 Magnetic Fields—Follow the manufacturer’s recommendation for the safe operation of the instrument.
9.1.1 Persons with implanted or attached medical devices such as pacemakers and prosthetic parts must remain outside the 5-gauss
perimeter.
9.1.2 Objects made of ferromagnetic material will be attracted to the magnet and are to be kept a safe distance away.away, outside
the 5-gauss perimeter.
10. Preparation of Apparatus
10.1 Prepare a proton decoupled carbon-13 NMR experiment, selecting appropriate parameters to obtain quantitative integration
of the peaks in the 68-60 ppm region.
10.1.1 Inverse Gated Decoupling—Decouple only during acquisition.
10.1.2 Pulse Angle and Sequence Delay Time—Select a 90 degree pulse angle with a sequence delay of 5 to 10 × T of the peak
with the longest relaxation time in the 68-60 ppm region. It is acceptable to use a different pulse angle/sequence delay combination
to reduce acquisition time provided that quantitative data acquisition is not compromised.
10.1.3 Number of Scans—Select the appropriate number to yield a minimum signal to noise of > 10:1 for the smallest peak
integrated over the 68-60 ppm region (usually 1024 to 2048).
D4273 − 23
10.1.4 Sweep Width—~ ~ 220 ppm.
10.1.5 Transmitter Frequency—~100 ~100 ppm.
10.1.6 Acquisition Time—1 to 2 s.
11. Calibration and Standardization
11.1 This test method does not require standards. To evaluate the test method, standards can be prepared it is feasible to prepare
standards by blending commercially available poly(propylene oxide) and poly(ethylene oxide) diols. The Ideally, the molecular
weights of the diols should ideallyare to be 300 or more since lower molecular weight polyols can contain structural configurations
that are not typical of polyether polyols used in flexible polyurethane foams.
12. Procedure
12.1 Prepare a solution of the polyol sample in deuterated chloroform or deuterated acetone containing TMS as an internal
standard. A 30-60 % solution is recommended. (See Note 2.)
12.2 Transfer an appropriate amount of the sample solution to an NMR tube.
12.3 Place the NMR tube into a spinner, adjust it to the appropriate depth and insert it into the spectrometer probe.
12.4 Obtain a stable deuterium lock on the solvent.
13 1
12.5 Tune and match the probe.probe for C and H channels.
12.6 Shim the sample to optimize magnetic field-homogeneity.
12.7 Acquire the NMR data.
12.8 Zero fill the data. The recommended value is 1 or 2 × number of data points.
12.9 Apply a spectral weighting function (apodization) and Fourier Transform the Free Induction Decay (FID). The recommended
apodization is an exponential window multiplication and a typical line broadening value is 1/acquisition time.
12.10 Phase and baseline correct the spectrum.
12.11 Set the internal TMS reference to 0 ppm.
12.12 Expand and integrate Integrate the peaks of interest. The primary hydroxyl peaks typically resonate in the 61.0 to 62.5 ppm
region and the secondary hydroxyl peaks typically resonate in the 65.5 to 67.5 ppm region. An example is shown in Fig. 1.
NOTE 2—The use of 0.025 to 0.05 M Cr(acac) has been found to shorten relaxation times allowing for shorter data acquisition times.
13. Calculation
13.1 Calculate the percent primary hydroxyl content using the following equation:
Primary hydroxyl,%5 A* (1)
A1B
where:
A = area of terminal EO methylene carbons (primary hydroxyl peaks),
D4273 − 23
FIG. 1 C NMR Spectrum of an EO-PO Polyol (BB23796)
B = area of terminal PO methine carbons (secondary hydroxyl peaks—does not correct for primary hydroxyl PO terminations).
14. Report
14.1 Report the % primary hydroxyl content to the nearest two decimal places.
15. Precision and Bias
15.1 Table 1 is based on a round robin conducted in 2016 in accordance with Practice E691, involving five materials tested by
eight laboratories. For each material, all the samples were prepared at one source, but the individual specimens were prepared at
the laboratories which tested them. Each test result was a single determination. Each laboratory obtained two test results for each
material.
15.2 Caution—The explanation of repeatability (r) and reproducibility (R) is only intended to present a meaningful way of
considering the approximate precision of this test method. Do not apply the data in Table 1 to accept or reject materials, as these
data apply only to the materials tested in the round robin and are unlikely to be rigorously representative of other lots, formulations,
conditions, materials, or laboratories. Users of this test method need to apply the principles outlined in Practice E691 to generate
data specific to their materials and laboratory (or between specific laboratories). The principles would then be valid for such data.
15.3 Repeatability—Precision under repeatability conditions.
15.4 Reproducibility—Precision under reproducibility conditions.
15.5 Any judgment in accordance with the repeatability and reproducibility statements shown above would have an approximate
95% (0.95) probability of being correct.
15.6 There are no recognized standards by which to estimate bias of this method.
15.7 For information on equivalence, refer to Practice E2935.
16. Keywords
16.1 NMR; nuclear magnetic resonance spectroscopy; polyurethane raw materials; primary hydroxyl, polyether polyol
Supporting data are available from ASTM Headquarters. Request RR:D20-1270.
D4273 − 23
TABLE 1 Primary Hydroxyl Content of Polyether Polyols
B C D E
Sample Material OH Value Mean S S r R
r R
(mg KOH/g)
BB23792 Blend of PPG 61 10.85 0.45 0.56 1.27 1.56
2000 and PEG
BB23793 Blend of PPG 71 24.38 0.63 1.72 1.75 4.80
A
2000 and PEG
BB23794 Glycerol/EO-PO 43 42.55 0.73 1.05 2.03 2.95
(EO + PO > 6.5)
mixed feed
BB23795 DPG/EO-PO (EO 43 71.39 0.39 1.46 1.10 4.08
+ PO > 4.5)
BB23796 EO-PO 30 87.75 1.09 2.36 3.04 6.62
A
Phase separation was observed in sample BB23793. Data for this sample are expected to include this variability.
B
S = within-laboratory standard deviation for the indicated material. It is obtained by pooling the within-laboratory standard deviations of the test results from all of the
r
participating laboratories:
2 2 2 1/2
S = [ [ (S ) 2 + (S ) { { + (S ) ]/n] where n = number of participating laboratories.
r 1 2 n
C
S = between-laboratories reproducibility, expressed as standard deviation:
R
2 2 1/2
S = [ (S ) + (S ) ] where S = standard deviation of laboratory means.
R r L L
D
r = within-laboratory critical interval between two test results = 2.8 × S
r
E
R = between-laboratories critical interval between two test results = 2.8 × S
R
APPENDIX
(Nonmandatory Information)
X1. FLUORINE-19 NUCLEAR MAGNETIC RESONANCE SPECTROSCOPY METHOD FOR DETERMINATION OF PRIMARY
HYDROXYL CONTENT OF POLYETHER POLYOLS
X1.1 Scope
X1.1.1 Fluorine-19 Nuclear Magnetic Resonance Spectroscopy (fluorine-19 NMR), measures the primary hydroxyl content in
ethylene oxide-propylene oxide polyethers used in flexible urethane foams. It is suitable for polyethers with hydroxyl numbers of
24 to 300 and primary hydroxyl percentages of 2 to
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...