ASTM E1750-23
(Guide)Standard Guide for Use of Water Triple Point Cells
Standard Guide for Use of Water Triple Point Cells
SIGNIFICANCE AND USE
4.1 This guide describes a procedure for placing a water triple-point cell in service and for using it as a reference temperature in thermometer calibration.
4.2 The reference temperature attained is that of a fundamental state of pure water, the equilibrium between coexisting solid, liquid, and vapor phases.
4.3 The cell is subject to qualification but not to calibration. The cell may be qualified as capable of representing the fundamental state (see 4.2) by comparison with a bank of similar qualified cells of known history, and it may be so qualified and the qualification documented by its manufacturer.
4.4 The temperature to be attributed to a qualified water triple-point cell is exactly 273.16 K on the ITS-90, unless corrected for isotopic composition (refer to Appendix X3).
4.5 Continued accuracy of a qualified cell depends upon sustained physical integrity. This may be verified by techniques described in Section 6.
4.6 The commercially available triple point of water cells described in this standard are capable of achieving an expanded uncertainty (k=2) of between ±0.1 mK and ±0.05 mK, depending upon the method of preparation. Specified measurement procedures shall be followed to achieve these levels of uncertainty.
4.7 Commercially-available triple point of water cells of unknown isotopic composition should be capable of achieving an expanded uncertainty (k=2) of no greater than 0.25 mK, depending upon the actual isotopic composition (3). These types of cells are acceptable for use at this larger value of uncertainty.
SCOPE
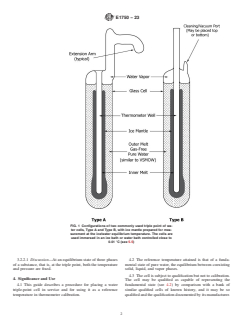
1.1 This guide covers the nature of two commercial water triple-point cells (types A and B, see Fig. 1) and provides a method for preparing the cell to realize the water triple-point and calibrate thermometers. The qualifications concerning preparation and the types of glass used for a cell are discussed. Tests for assuring the integrity of a qualified cell and of cells yet to be qualified are given. Precautions for handling the cell to avoid breakage are also described.
FIG. 1 Configurations of two commonly used triple point of water cells, Type A and Type B, with ice mantle prepared for measurement at the ice/water equilibrium temperature. The cells are used immersed in an ice bath or water bath controlled close to 0.01 °C (see 5.5)
1.2 The effect of hydrostatic pressure on the temperature of a water triple-point cell is discussed.
1.3 Procedures for adjusting the observed SPRT resistance readings for the effects of self-heating and hydrostatic pressure are described in Appendix X1 and Appendix X2.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Oct-2023
- Technical Committee
- E20 - Temperature Measurement
- Drafting Committee
- E20.07 - Fundamentals in Thermometry
Relations
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Referred By
ASTM E644-11(2019) - Standard Test Methods for Testing Industrial Resistance Thermometers - Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
Overview
ASTM E1750-23: Standard Guide for Use of Water Triple Point Cells provides comprehensive guidance on the use of water triple-point cells as a reference temperature for the calibration of thermometers. The water triple point is a thermometric fixed point fundamental to defining the Kelvin Thermodynamic Temperature Scale (KTTS) and the International Temperature Scale of 1990 (ITS-90). This guide covers procedures for preparing and qualifying different types of commercially available water triple-point cells (Type A and B), maintaining their integrity, and employing them in precise temperature measurements.
Key Topics
- Nature and Configuration of Triple-Point Cells: The standard discusses the features of Type A and Type B water triple-point cells, which comprise high-purity sealed glass vessels filled with pure, air-free water.
- Realization of the Triple Point: Guidance on preparing the cell to establish the water triple-point by forming a uniform ice mantle, ensuring equilibrium between water’s solid, liquid, and vapor phases.
- Qualification Process: Details on qualifying a triple-point cell by comparison with a bank of similar cells of known history, as opposed to calibration.
- Reference Temperature: A qualified water triple-point cell is assigned the temperature 273.16 K on ITS-90, unless corrected for isotopic composition.
- Expanded Uncertainty: Specified procedures enable the realization of the triple-point temperature with expanded uncertainties ranging from ±0.05 mK to ±0.25 mK, depending on cell preparation and water isotopic composition.
- Integrity and Handling: Emphasizes maintaining the physical integrity of the cell, including tests for detecting air or soluble impurities and precautions to avoid breakage during handling.
- Measurement Procedures: Covers procedures for using Standard Platinum Resistance Thermometers (SPRTs) with the triple-point cell, including corrections for self-heating and hydrostatic head effects.
Applications
ASTM E1750-23 is crucial for laboratories, calibration service providers, and metrology institutes that require precise and reliable temperature calibration methods. Its primary applications include:
- Calibration of Thermometers: Used for calibrating high-precision thermometers, particularly standard platinum resistance thermometers (SPRTs), by providing a stable, intrinsic temperature reference.
- Temperature Scale Realization: Supports national and international metrology institutes in realizing and disseminating the ITS-90 temperature scale.
- Quality Assurance: Provides procedures to verify and maintain the integrity of reference cells, critical for sustained accuracy in temperature measurement and instrument calibration.
- Research and Development: Essential for scientific research that demands tight control and verification of temperature standards and ensuring traceability to the kelvin unit.
Related Standards
Several related ASTM and international standards provide complementary guidelines and terminology:
- ASTM E344 - Terminology Relating to Thermometry and Hydrometry: Provides key definitions used in temperature measurement.
- ASTM E1594 - Guide for Expression of Temperature: Offers guidance on expressing and reporting temperature measurements.
- ITS-90 (International Temperature Scale of 1990): The definitive scale for accurate temperature measurement, with the water triple point as a foundational reference point.
- ISO and BIPM Documentation: International references outlining the principles for temperature scale realization and the isotopic composition of reference water.
By adhering to ASTM E1750-23, organizations ensure the highest level of precision in temperature measurements, facilitate comparability across laboratories worldwide, and maintain compliance with internationally recognized measurement standards. This guide is an indispensable reference for anyone involved in precise thermometry and calibration activities.
Buy Documents
ASTM E1750-23 - Standard Guide for Use of Water Triple Point Cells
REDLINE ASTM E1750-23 - Standard Guide for Use of Water Triple Point Cells
Get Certified
Connect with accredited certification bodies for this standard

BSMI (Bureau of Standards, Metrology and Inspection)
Taiwan's standards and inspection authority.
Sponsored listings
Frequently Asked Questions
ASTM E1750-23 is a guide published by ASTM International. Its full title is "Standard Guide for Use of Water Triple Point Cells". This standard covers: SIGNIFICANCE AND USE 4.1 This guide describes a procedure for placing a water triple-point cell in service and for using it as a reference temperature in thermometer calibration. 4.2 The reference temperature attained is that of a fundamental state of pure water, the equilibrium between coexisting solid, liquid, and vapor phases. 4.3 The cell is subject to qualification but not to calibration. The cell may be qualified as capable of representing the fundamental state (see 4.2) by comparison with a bank of similar qualified cells of known history, and it may be so qualified and the qualification documented by its manufacturer. 4.4 The temperature to be attributed to a qualified water triple-point cell is exactly 273.16 K on the ITS-90, unless corrected for isotopic composition (refer to Appendix X3). 4.5 Continued accuracy of a qualified cell depends upon sustained physical integrity. This may be verified by techniques described in Section 6. 4.6 The commercially available triple point of water cells described in this standard are capable of achieving an expanded uncertainty (k=2) of between ±0.1 mK and ±0.05 mK, depending upon the method of preparation. Specified measurement procedures shall be followed to achieve these levels of uncertainty. 4.7 Commercially-available triple point of water cells of unknown isotopic composition should be capable of achieving an expanded uncertainty (k=2) of no greater than 0.25 mK, depending upon the actual isotopic composition (3). These types of cells are acceptable for use at this larger value of uncertainty. SCOPE 1.1 This guide covers the nature of two commercial water triple-point cells (types A and B, see Fig. 1) and provides a method for preparing the cell to realize the water triple-point and calibrate thermometers. The qualifications concerning preparation and the types of glass used for a cell are discussed. Tests for assuring the integrity of a qualified cell and of cells yet to be qualified are given. Precautions for handling the cell to avoid breakage are also described. FIG. 1 Configurations of two commonly used triple point of water cells, Type A and Type B, with ice mantle prepared for measurement at the ice/water equilibrium temperature. The cells are used immersed in an ice bath or water bath controlled close to 0.01 °C (see 5.5) 1.2 The effect of hydrostatic pressure on the temperature of a water triple-point cell is discussed. 1.3 Procedures for adjusting the observed SPRT resistance readings for the effects of self-heating and hydrostatic pressure are described in Appendix X1 and Appendix X2. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This guide describes a procedure for placing a water triple-point cell in service and for using it as a reference temperature in thermometer calibration. 4.2 The reference temperature attained is that of a fundamental state of pure water, the equilibrium between coexisting solid, liquid, and vapor phases. 4.3 The cell is subject to qualification but not to calibration. The cell may be qualified as capable of representing the fundamental state (see 4.2) by comparison with a bank of similar qualified cells of known history, and it may be so qualified and the qualification documented by its manufacturer. 4.4 The temperature to be attributed to a qualified water triple-point cell is exactly 273.16 K on the ITS-90, unless corrected for isotopic composition (refer to Appendix X3). 4.5 Continued accuracy of a qualified cell depends upon sustained physical integrity. This may be verified by techniques described in Section 6. 4.6 The commercially available triple point of water cells described in this standard are capable of achieving an expanded uncertainty (k=2) of between ±0.1 mK and ±0.05 mK, depending upon the method of preparation. Specified measurement procedures shall be followed to achieve these levels of uncertainty. 4.7 Commercially-available triple point of water cells of unknown isotopic composition should be capable of achieving an expanded uncertainty (k=2) of no greater than 0.25 mK, depending upon the actual isotopic composition (3). These types of cells are acceptable for use at this larger value of uncertainty. SCOPE 1.1 This guide covers the nature of two commercial water triple-point cells (types A and B, see Fig. 1) and provides a method for preparing the cell to realize the water triple-point and calibrate thermometers. The qualifications concerning preparation and the types of glass used for a cell are discussed. Tests for assuring the integrity of a qualified cell and of cells yet to be qualified are given. Precautions for handling the cell to avoid breakage are also described. FIG. 1 Configurations of two commonly used triple point of water cells, Type A and Type B, with ice mantle prepared for measurement at the ice/water equilibrium temperature. The cells are used immersed in an ice bath or water bath controlled close to 0.01 °C (see 5.5) 1.2 The effect of hydrostatic pressure on the temperature of a water triple-point cell is discussed. 1.3 Procedures for adjusting the observed SPRT resistance readings for the effects of self-heating and hydrostatic pressure are described in Appendix X1 and Appendix X2. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E1750-23 is classified under the following ICS (International Classification for Standards) categories: 17.200.20 - Temperature-measuring instruments. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E1750-23 has the following relationships with other standards: It is inter standard links to ASTM E1750-10(2016), ASTM E344-23, ASTM E879-20, ASTM E2593-17(2023), ASTM D445-23, ASTM E644-11(2019), ASTM E2730-22, ASTM D8164-21, ASTM D8278-20, ASTM D2162-21, ASTM D2532-22, ASTM D6466-10(2018), ASTM E344-20, ASTM D7279-20. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E1750-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E1750 − 23
Standard Guide for
Use of Water Triple Point Cells
This standard is issued under the fixed designation E1750; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
INTRODUCTION
The triple point of water is an important thermometric fixed point as defined in the International
Temperature Scale of 1990 (ITS-90). The ITS-90 assigns a value of 273.16 K to the triple point of
water which was exactly equivalent to the thermodynamic temperature in the international System (SI)
of units at the time of the adoption of the ITS-90 (1, 2). The triple point of water is the temperature
to which the resistance ratios W(T) = R(T) ⁄R(273.16 K) of the standard platinum resistance
thermometer (SPRT) calibrations are referred to in the ITS-90.
The triple points of various materials (where three distinct phases, for example, their solid, liquid,
and vapor phases, coexist in a state of thermal equilibrium) have fixed pressures and temperatures and
are highly reproducible. Of the ITS-90 fixed points, six are triple points. The water triple point is one
of the most accurately realizable of the defining fixed points of the ITS-90; under the best of
conditions, it can be realized with an expanded uncertainty (k=2) of less than 60.00005 K. In
comparison, it is difficult to prepare and use an ice bath with an expanded uncertainty (k=2) of less
than 60.002 K (3).
1. Scope 1.5 This international standard was developed in accor-
dance with internationally recognized principles on standard-
1.1 This guide covers the nature of two commercial water
ization established in the Decision on Principles for the
triple-point cells (types A and B, see Fig. 1) and provides a
Development of International Standards, Guides and Recom-
method for preparing the cell to realize the water triple-point
mendations issued by the World Trade Organization Technical
and calibrate thermometers. The qualifications concerning
Barriers to Trade (TBT) Committee.
preparation and the types of glass used for a cell are discussed.
Tests for assuring the integrity of a qualified cell and of cells
2. Referenced Documents
yet to be qualified are given. Precautions for handling the cell
2.1 ASTM Standards:
to avoid breakage are also described.
E344 Terminology Relating to Thermometry and Hydrom-
1.2 The effect of hydrostatic pressure on the temperature of
etry
a water triple-point cell is discussed.
E1594 Guide for Expression of Temperature
1.3 Procedures for adjusting the observed SPRT resistance
3. Terminology
readings for the effects of self-heating and hydrostatic pressure
are described in Appendix X1 and Appendix X2.
3.1 Definitions—The definitions given in Terminology E344
apply to terms used in this guide.
1.4 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
3.2 Definitions of Terms Specific to This Standard:
responsibility of the user of this standard to establish appro-
3.2.1 inner melt, n—a thin continuous layer of water be-
priate safety, health, and environmental practices and deter-
tween the thermometer well and the ice mantle of a water
mine the applicability of regulatory limitations prior to use.
triple-point cell.
3.2.2 reference temperature, n—the temperature of a phase
1 equilibrium state of a pure substance at a specified pressure, for
This guide is under the jurisdiction of ASTM Committee E20 on Temperature
Measurement and is the direct responsibility of Subcommittee E20.07 on Funda- example, the assigned temperature of a fixed point.
mentals in Thermometry.
Current edition approved Nov. 1, 2023. Published December 2023. Originally
approved in 1995. Last previous edition approved in 2016 as E1750 – 10 (2016). For referenced ASTM standards, visit the ASTM website, www.astm.org, or
DOI: 10.1520/E1750-23. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
The boldface numbers in parentheses refer to the list of references at the end of Standards volume information, refer to the standard’s Document Summary page on
this standard. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1750 − 23
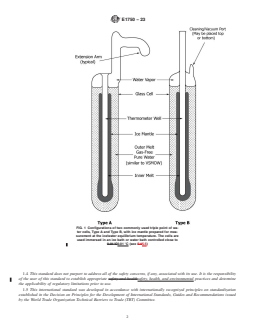
FIG. 1 Configurations of two commonly used triple point of wa-
ter cells, Type A and Type B, with ice mantle prepared for mea-
surement at the ice/water equilibrium temperature. The cells are
used immersed in an ice bath or water bath controlled close to
0.01 °C (see 5.5)
3.2.2.1 Discussion—At an equilibrium state of three phases 4.2 The reference temperature attained is that of a funda-
of a substance, that is, at the triple point, both the temperature mental state of pure water, the equilibrium between coexisting
and pressure are fixed. solid, liquid, and vapor phases.
4.3 The cell is subject to qualification but not to calibration.
4. Significance and Use
The cell may be qualified as capable of representing the
4.1 This guide describes a procedure for placing a water fundamental state (see 4.2) by comparison with a bank of
triple-point cell in service and for using it as a reference similar qualified cells of known history, and it may be so
temperature in thermometer calibration. qualified and the qualification documented by its manufacturer.
E1750 − 23
4.4 The temperature to be attributed to a qualified water 5.4 For use, a portion of the water is frozen within the cell
triple-point cell is exactly 273.16 K on the ITS-90, unless to form a mantle of ice that surrounds the well and controls its
corrected for isotopic composition (refer to Appendix X3). temperature.
4.5 Continued accuracy of a qualified cell depends upon 5.5 The temperature of the triple point of water realized in
sustained physical integrity. This may be verified by techniques a cell is independent of the environment outside the cell;
described in Section 6. however, to reduce heat transfer and keep the ice mantle from
melting quickly, it is necessary to minimize heat flow between
4.6 The commercially available triple point of water cells
the cell and its immediate environment. This may be done by
described in this standard are capable of achieving an expanded
immersing the cell in an ice bath that maintains the full length
uncertainty (k=2) of between 60.1 mK and 60.05 mK,
of the outer cell wall at or near the melting point of ice.
depending upon the method of preparation. Specified measure-
Alternatively, commercial automatic maintenance baths, built
ment procedures shall be followed to achieve these levels of
specifically for this purpose, are available. In such baths, the
uncertainty.
triple point of water equilibrium of the cell, once established,
4.7 Commercially-available triple point of water cells of
can be maintained for many months of continual use. To avoid
unknown isotopic composition should be capable of achieving
radiation heat transfer to the cell and to the thermometer, the
an expanded uncertainty (k=2) of no greater than 0.25 mK,
outer surface of the maintenance bath is made opaque to
depending upon the actual isotopic composition (3). These
radiation.
types of cells are acceptable for use at this larger value of
6. Assurance of Integrity
uncertainty.
6.1 The temperature attained within a water triple-point cell
5. Apparatus
is an intrinsic property of the solid and liquid phases of water
5.1 The essential features of type A and type B water
under its own vapor pressure. If the water triple-point condi-
triple-point cells are shown in Fig. 1. A transparent flask is
tions are satisfied, the temperature attained within the cell is
made from glass tubing that has been cleaned in such a way more reproducible than any measurements that can be made of
that all the surface contaminants have been removed from all
it.
internal surfaces without etching or otherwise damaging the
6.2 The accuracy of realization of the water triple-point
glass. The cell is made from a type and grade of glass that is
temperature with a qualified cell depends on the physical
suitable to withstand the stresses associated with normal use
integrity of the seal and of the walls of the glass cell and on
(for example, freezing expansion, thermal cycling, etc.). These
their ability to exclude environmental air and contaminants.
may include any of several grades of borosilicate glass as well
6.3 Initial and continued physical integrity is confirmed by
as quartz glass.
the following procedures:
5.2 The cell’s internal volume is filled with purified water in
6.3.1 Test for the Presence of Air:
such a way that all displaced and dissolved air or other gas
6.3.1.1 Remove all objects from the thermometer well.
content has been removed from the cell and the water. The cell
6.3.1.2 The solubility and the pressure of air at 101 325 Pa
is designed to accommodate any one of various filling and
lower the ice/water equilibrium temperature 0.01 °C below the
purification modalities. These include decanting, distillation,
triple-point temperature. Since air is more soluble in water at
and vacuum degassing via one or more ports in the glass wall
lower temperatures, the test for air shall be done at room
that are permanently sealed once the filling is complete. A
temperature. The test is less definitive when performed on a
small fraction of the cell’s internal volume is left unfilled to
chilled cell. At room temperature, with the cell initially upright
allow for the vapor-phase water to coexist with the condensed
and the well opening upward, slowly invert the cell. As the axis
phases at the free surface. A reentrant well on the axis of the
of the cell passes through horizontal and as the water within the
flask (see “Thermometer Well” in Fig. 1) receives thermom-
cell strikes the end of the cell, a sharp “glassy clink” sound
eters that equilibrate with the condensed phases at a suitable
should be heard. The distinctive sound results from the sudden
immersion depth below the free surface. A small correction is
collapse of water vapor and the “water hammer” striking the
necessary to account for this immersion depth (see Appendix
glass cell. The smaller the amount of air, the sharper the clink
X2).
sound; a large amount of air cushions the water-hammer action
5.3 For the lowest level of uncertainty, the water used as the and the sound is duller.
reference medium shall be very pure and of known isotopic 6.3.1.3 With a type A cell, continue to tilt the cell to make
composition. Often it is distilled directly into the cell. The a McLeod-gauge type test until the vapor (water saturated air)
isotopic composition of cells filled with “rain water” is bubble is entirely captured in the space provided in the handle.
expected not to vary enough to cause more than 0.05 mK The vapor bubble should be compressed to a volume no larger
difference in their triple points. Extreme variations in isotopic than about 0.03 cm (4 mm diameter). It may even vanish as it
composition, such as between ocean water and water from old is compressed by the weight of the water column. As in the tilt
polar ice, can affect the realized temperature by as much as test, the bubble test is more definitive when the cell is at room
0.25 mK (4). In cases where the isotopic composition is temperature (see 6.3.1.2). Since type B cells do not have a
unknown, or if the cell has not been qualified by comparison space to capture the vapor, the amount of air in the cell is
with a cell of known isotopic composition, the larger value of estimated by comparing the sharpness of the clink sound with
uncertainty (60.25 mK) should be assumed. that of a type A cell.
E1750 − 23
6.3.2 Test for the Presence of Water Soluble Impurities: for a period of 2 to 3 min. In repeated use of the cell, the ice
6.3.2.1 When ice is slowly formed around the thermometer mantle melts mostly at the bottom; hence, it is desirable that
well, impurities are rejected into the remaining unfrozen water. the ice mantle be thicker at the bottom. Crushed dry ice may be
Therefore, the impurity concentration of the unfrozen water prepared from a block or by expansion from a siphon-tube tank
increases as the ice mantle thickens. The ice is purer than the of liquid CO .
unfrozen water. Consequently, the inner melt (see 7.1.3) that is
7.1.7 At the interface of the well, the water is initially
formed from the ice mantle is purer than the unfrozen water
supercooled, and the well becomes abruptly coated with fine
outside of the mantle.
needles of ice frozen from the supercooled water.
6.3.2.2 Prepare a relatively thick ice mantle, according to
7.1.8 After a layer of ice forms around the bottom of the
Section 7, by maintaining the dry ice level full for about
well, fill the well with crushed dry ice up to the vapor/liquid
20 min. Make certain that the ice does not bridge to the cell
interface.
wall (see 7.1.9).
7.1.9 Replenish the dry ice as it sublimes, maintaining the
6.3.2.3 Prepare an inner melt according to 7.1.13. Using an
well filled to the liquid surface, until a continuous ice mantle as
SPRT, make measurements on the cell and determine the
thick as desired forms on the surface of the well within the
zero-power resistance according to Section 8 and Appendix
water (usually 4 mm to 8 mm thick). The mantle will appear
X1.
thicker than its actual thickness because of the lenticular shape
6.3.2.4 After 6.3.2.3, remove the SPRT. Gently invert the
of the cell and the refractive index of water. The actual
water triple-point cell and then return it to the upright position
thickness may be best estimated by viewing from the bottom of
several times to exchange the unfrozen water on the outside of
the cell while it is inverted or by immersing the cell in a large
the ice mantle with the inner melt water. (Warning—When
glass container of water. (Warning—During preparation, the
inverting the cell, do not allow the floating ice mantle to
mantle should never be allowed to grow at any place to
severely strike the bottom of the water triple-point cell.)
completely bridge the space between the well and the inner
6.3.2.5 Reinsert the pre-chilled SPRT used in 6.3.2.3 into
wall of the cell, as the expansion of the ice may break the cell.
the well. Make measurements on the cell and determine the
In particular, if bridging occurs at the surface of the water at the
zero-power resistance, according to Section 8 and Appendix
top of the cell under the vapor space, melt the ice bridge by
X1.
warming the cell locally with heat from the hand, while gently
6.3.2.6 Typically, for high quality water triple-point cells,
shaking the cell.)
the results of 6.3.2.3 and 6.3.2.5 will not differ by more than
7.1.10 When the mantle attains nearly the desired thickness
60.03 mK.
or after maintaining the dry ice level in the well at the water
6.4 Any cell that had previously been qualified by compari-
surface for about 20 min, return the cell to the ice bath with the
son with cells of known integrity (as in 4.3), that has not
entrance to the thermometer well slightly above the ice bath
thereafter been modified, and which currently passes the tests
surface and allow the dry ice to sublime completely. By
of 6.3.1 and 6.3.2, is qualified as a water triple-point cell.
allowing the dry ice to sublime completely, the bottom of the
well stays cold longer and the mantle grows thicker there.
6.5 Any cell that fails to pass the tests of 6.3.1 and 6.3.2,
7.1.11 After the thermometer well becomes free of dry ice,
even though previously qualified, is no longer qualified for use
immerse the cell deeper into the ice bath and fill the well with
as a water triple-point cell.
ice bath water.
7. Realization of the Water Triple-Point Temperature
7.1.12 Allow the cell to remain packed in ice or in the ice
bath for two days to stabilize its temperature. Because of the
7.1 The ice mantle that is required to realize the triple-point
strains in the ice mantle prepared using dry ice, a freshly
temperature of water can be prepared in a number of ways.
prepared mantle can give a temperature that is as much as
They produce essentially the same result. A common procedure
0.2 mK lower.
is as follows:
7.1.1 Empty the well of any solids or liquids. Wipe the well 7.1.13 When initially prepared, the mantle will be fixed to
clean and dry, and seal the well opening with a rubber stopper. the wall of the well. Before the cell can be used, a thin layer of
7.1.2 If the triple point of water cell has not already been ice next to the thermometer well shall be melted. To prepare
tested for the presence of air, perform the tests indicated in this “inner melt,” briefly and gently insert a metal or glass rod,
6.3.1 for presence of air. initially at room temperature, into the well to heat the well
7.1.3 To obtain an ice mantle of fairly uniform thickness slightly. The rod should have a smooth rounded end to avoid
that extends to the top, immerse the cell completely in an ice scratching or possibly breaking the cell. Upon removal of the
bath, and chill the cell to near 0 °C. rod, tilt the cell to an angle of about 45° from the vertical axis
7.1.4 Remove the cell from the bath and mount it upright on and observe for the rotation of the mantle. If the mantle is
a plastic foam cushion. Wipe the cell dry around the rubber properly detached, it will spin freely about the well. The liquid
stopper before removing the rubber stopper. water film should be thin to minimize the thermal resistance
7.1.5 Remove the rubber stopper and place about 1 cm of between the thermometer well and the ice/water interface. The
dry alcohol in the well to serve as a heat-transfer medium while liquid water film between the mantle and the well surface is
forming an ice mantle around the well within the sealed cell. essential to the proper realization of the triple-point tempera-
7.1.6 Place a small amount of crushed dry ice at the bottom ture. The freedom of the mantle should always be checked by
of the well, maintaining the height of the dry ice at about 1 cm tilting the cell prior to and after calibrating thermometers.
----------------------
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E1750 − 10 (Reapproved 2016) E1750 − 23
Standard Guide for
Use of Water Triple Point Cells
This standard is issued under the fixed designation E1750; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
INTRODUCTION
The triple point of water is an important thermometric fixed point common to the definition of two
temperature scales of science and technology, the Kelvin Thermodynamic Temperature Scale (KTTS)
and the as defined in the International Temperature Scale of 1990 (ITS-90). The ITS-90 was designed
to be as close to the KTTS as the experimental data available at the time of the adoption of the ITS-90
would permit. The temperatures (T) on the KTTS are defined by assigning the value assigns a value
of 273.16 K to the triple point of water, thus defining the thermodynamic unit of temperature, kelvin
(K), as 1/273.16 of the thermodynamic temperature of the triple point of water water which was
exactly equivalent to the thermodynamic temperature in the international System (SI) of units at the
time of the adoption of the ITS-90 (1, 2). The triple point of water, one of the fixed points used to
define the ITS-90, water is the temperature to which the resistance ratios W(T) = R(T) ⁄R(273.16 K)
of the standard platinum resistance thermometer (SPRT) calibrations are referred.referred to in the
ITS-90.
The triple points of various materials (where three distinct phases, for example, their solid, liquid,
and vapor phases, coexist in a state of thermal equilibrium) have fixed pressures and temperatures and
are highly reproducible. Of the ITS-90 fixed points, six are triple points. The water triple point is one
of the most accurately realizable of the defining fixed points of the ITS-90; under the best of
conditions, it can be realized with an expanded uncertainty (k=2) of less than 60.00005 K. In
comparison, it is difficult to prepare and use an ice bath with an expanded uncertainty (k=2) of less
than 60.002 K (3).
1. Scope
1.1 This guide covers the nature of two commercial water triple-point cells (types A and B, see Fig. 1) and provides a method for
preparing the cell to realize the water triple-point and calibrate thermometers. The qualifications concerning preparation and the
types of glass used for a cell are discussed. Tests for assuring the integrity of a qualified cell and of cells yet to be qualified are
given. Precautions for handling the cell to avoid breakage are also described.
1.2 The effect of hydrostatic pressure on the temperature of a water triple-point cell is discussed.
1.3 Procedures for adjusting the observed SPRT resistance readings for the effects of self-heating and hydrostatic pressure are
described in Appendix X1 and Appendix X2.
This guide is under the jurisdiction of ASTM Committee E20 on Temperature Measurement and is the direct responsibility of Subcommittee E20.07 on Fundamentals
in Thermometry.
Current edition approved May 1, 2016Nov. 1, 2023. Published May 2016December 2023. Originally approved in 1995. Last previous edition approved in 20102016 as
E1750 – 10.E1750 – 10 (2016). DOI: 10.1520/E1750-10R16.10.1520/E1750-23.
The boldface numbers in parentheses refer to the list of references at the end of this standard.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1750 − 23
FIG. 1 Configurations of two commonly used triple point of wa-
ter cells, Type A and Type B, with ice mantle prepared for mea-
surement at the ice/water equilibrium temperature. The cells are
used immersed in an ice bath or water bath controlled close to
0.01°C0.01 °C (see 5.45.5)
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and healthsafety, health, and environmental practices and determine
the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
E1750 − 23
2. Referenced Documents
2.1 ASTM Standards:
E344 Terminology Relating to Thermometry and Hydrometry
E1594 Guide for Expression of Temperature
3. Terminology
3.1 Definitions—The definitions given in Terminology E344 apply to terms used in this guide.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 inner melt, n—a thin continuous layer of water between the thermometer well and the ice mantle of a water triple-point cell.
3.2.2 reference temperature, n—the temperature of a phase equilibrium state of a pure substance at a specified pressure, for
example, the assigned temperature of a fixed point.
3.2.2.1 Discussion—
At an equilibrium state of three phases of a substance, that is, at the triple point, both the temperature and pressure are fixed.
4. Significance and Use
4.1 This guide describes a procedure for placing a water triple-point cell in service and for using it as a reference temperature in
thermometer calibration.
4.2 The reference temperature attained is that of a fundamental state of pure water, the equilibrium between coexisting solid,
liquid, and vapor phases.
4.3 The cell is subject to qualification but not to calibration. The cell may be qualified as capable of representing the fundamental
state (see 4.2) by comparison with a bank of similar qualified cells of known history, and it may be so qualified and the qualification
documented by its manufacturer.
4.4 The temperature to be attributed to a qualified water triple-point cell is exactly 273.16 K on the ITS-90, unless corrected for
isotopic composition (refer to Appendix X3).
4.5 Continued accuracy of a qualified cell depends upon sustained physical integrity. This may be verified by techniques described
in Section 6.
4.6 The commercially available triple point of water cells described in this standard are capable of achieving an expanded
uncertainty (k=2) of between 60.1 mK and 60.05 mK, depending upon the method of preparation. Specified measurement
procedures shall be followed to achieve these levels of uncertainty.
4.7 Commercially-available triple point of water cells of unknown isotopic composition should be capable of achieving an
expanded uncertainty (k=2) of no greater than 0.25 mK, depending upon the actual isotopic composition (3). These types of cells
are acceptable for use at this larger value of uncertainty.
5. Apparatus
5.1 The essential features of type A and type B water triple-point cells are shown in Fig. 1. A transparent glass flask free of soluble
material is filled with pure, air-free water and then is permanently sealed, air-free, at the vapor pressure of the water. A reentrant
well on the axis of the flask receives thermometers that are to be exposed to the reference temperature.flask is made from glass
tubing that has been cleaned in such a way that all the surface contaminants have been removed from all internal surfaces without
etching or otherwise damaging the glass. The cell is made from a type and grade of glass that is suitable to withstand the stresses
associated with normal use (for example, freezing expansion, thermal cycling, etc.). These may include any of several grades of
borosilicate glass as well as quartz glass.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
E1750 − 23
5.2 The cell’s internal volume is filled with purified water in such a way that all displaced and dissolved air or other gas content
has been removed from the cell and the water. The cell is designed to accommodate any one of various filling and purification
modalities. These include decanting, distillation, and vacuum degassing via one or more ports in the glass wall that are permanently
sealed once the filling is complete. A small fraction of the cell’s internal volume is left unfilled to allow for the vapor-phase water
to coexist with the condensed phases at the free surface. A reentrant well on the axis of the flask (see “Thermometer Well” in Fig.
1) receives thermometers that equilibrate with the condensed phases at a suitable immersion depth below the free surface. A small
correction is necessary to account for this immersion depth (see Appendix X2).
5.3 For the lowest level of uncertainty, the water used as the reference medium shall be very pure and of known isotopic
composition. Often it is distilled directly into the cell. The isotopic composition of cells filled with “rain water” is expected not
to vary enough to cause more than 0.05 mK difference in their triple points. Extreme variations in isotopic composition, such as
between ocean water and water from old polar ice, can affect the realized temperature by as much as 0.25 mK (4). In cases where
the isotopic composition is unknown, or if the cell has not been qualified by comparison with a cell of known isotopic composition,
the larger value of uncertainty (60.25 mK) should be assumed.
5.4 For use, a portion of the water is frozen within the cell to form a mantle of ice that surrounds the well and controls its
temperature.
5.5 The temperature of the triple point of water realized in a cell is independent of the environment outside the cell; however, to
reduce heat transfer and keep the ice mantle from melting quickly, it is necessary to minimize heat flow between the cell and its
immediate environment. This may be done by immersing the cell in an ice bath that maintains the full length of the outer cell wall
at or near the melting point of ice. Alternatively, commercial automatic maintenance baths, built specifically for this purpose, are
available. In such baths, the triple point of water equilibrium of the cell, once established, can be maintained for many months of
continual use. To avoid radiation heat transfer to the cell and to the thermometer, the outer surface of the maintenance bath is made
opaque to radiation.
6. Assurance of Integrity
6.1 The temperature attained within a water triple-point cell is an intrinsic property of the solid and liquid phases of water under
its own vapor pressure. If the water triple-point conditions are satisfied, the temperature attained within the cell is more
reproducible than any measurements that can be made of it.
6.2 The accuracy of realization of the water triple-point temperature with a qualified cell depends on the physical integrity of the
seal and of the walls of the glass cell and on their ability to exclude environmental air and contaminants.
6.3 Initial and continued physical integrity is confirmed by the following procedures:
6.3.1 Test for the Presence of Air:
6.3.1.1 Remove all objects from the thermometer well.
6.3.1.2 The solubility and the pressure of air at 101 325 Pa lower the ice/water equilibrium temperature 0.01°C0.01 °C below the
triple-point temperature. Since air is more soluble in water at lower temperatures, the test for air shall be done at room temperature.
The test is less definitive when performed on a chilled cell. At room temperature, with the cell initially upright and the well opening
upward, slowly invert the cell. As the axis of the cell passes through horizontal and as the water within the cell strikes the end of
the cell, a sharp “glassy clink” sound should be heard. The distinctive sound results from the sudden collapse of water vapor and
the “water hammer” striking the glass cell. The smaller the amount of air, the sharper the clink sound; a large amount of air
cushions the water-hammer action and the sound is duller.
6.3.1.3 With a type A cell, continue to tilt the cell to make a McLeod-gauge type test until the vapor (water saturated air) bubble
is entirely captured in the space provided in the handle. The vapor bubble should be compressed to a volume no larger than about
0.03 cm (4 mm diameter). It may even vanish as it is compressed by the weight of the water column. As in the tilt test, the bubble
test is more definitive when the cell is at room temperature (see 6.3.1.2). Since type B cells do not have a space to capture the vapor,
the amount of air in the cell is estimated by comparing the sharpness of the clink sound with that of a type A cell.
E1750 − 23
6.3.2 Test for the Presence of Water Soluble Impurities:
6.3.2.1 When ice is slowly formed around the thermometer well, impurities are rejected into the remaining unfrozen water.
Therefore, the impurity concentration of the unfrozen water increases as the ice mantle thickens. The ice is purer than the unfrozen
water. Consequently, the inner melt (see section 7.1.3) that is formed from the ice mantle is purer than the unfrozen water outside
of the mantle.
6.3.2.2 Prepare a relatively thick ice mantle, according to Section 7, by maintaining the dry ice level full for about 20 minutes.
20 min. Make certain that the ice does not bridge to the cell wall (see 7.1.9).
6.3.2.3 Prepare an inner melt according to 7.1.13. Using an SPRT, make measurements on the cell and determine the zero-power
resistance according to Section 8 and Appendix X1.
6.3.2.4 After 6.3.2.3, remove the SPRT. Gently invert the water triple-point cell and then return it to the upright position several
times to exchange the unfrozen water on the outside of the ice mantle with the inner melt water. (Warning—When inverting the
cell, do not allow the floating ice mantle to severely strike the bottom of the water triple-point cell.)
6.3.2.5 Reinsert the pre-chilled SPRT used in 6.3.2.3 into the well. Make measurements on the cell and determine the zero-power
resistance, according to Section 8 and Appendix X1.
6.3.2.6 Typically, for high quality water triple-point cells, the results of 6.3.2.3 and 6.3.2.5 will not differ by more than 60.03 mK.
6.4 Any cell that had previously been qualified by comparison with cells of known integrity (as in 4.3), that has not thereafter been
modified, and which currently passes the tests of 6.3.1 and 6.3.2, is qualified as a water triple-point cell.
6.5 Any cell that fails to pass the tests of 6.3.1 and 6.3.2, even though previously qualified, is no longer qualified for use as a water
triple-point cell.
7. Realization of the Water Triple-Point Temperature
7.1 The ice mantle that is required to realize the triple-point temperature of water can be prepared in a number of ways. They
produce essentially the same result. A common procedure is as follows:
7.1.1 Empty the well of any solids or liquids. Wipe the well clean and dry, and seal the well opening with a rubber stopper.
7.1.2 If the triple point of water cell has not already been tested for the presence of air, perform the tests indicated in 6.3.1 for
presence of air.
7.1.3 To obtain an ice mantle of fairly uniform thickness that extends to the top, immerse the cell completely in an ice bath, and
chill the cell to near 0°C.0 °C.
7.1.4 Remove the cell from the bath and mount it upright on a plastic foam cushion. Wipe the cell dry around the rubber stopper
before removing the rubber stopper.
7.1.5 Remove the rubber stopper and place about 1 cm of dry alcohol in the well to serve as a heat-transfer medium while forming
an ice mantle around the well within the sealed cell.
7.1.6 Place a small amount of crushed dry ice at the bottom of the well, maintaining the height of the dry ice at about 1 cm for
a period of 2 to 3 min. In repeated use of the cell, the ice mantle melts mostly at the bottom; hence, it is desirable that the ice mantle
be thicker at the bottom. Crushed dry ice may be prepared from a block or by expansion from a siphon-tube tank of liquid CO .
7.1.7 At the interface of the well, the water is initially supercooled, and the well becomes abruptly coated with fine needles of ice
frozen from the supercooled water.
7.1.8 After a layer of ice forms around the bottom of the well, fill the well with crushed dry ice up to the vapor/liquid interface.
7.1.9 Replenish the dry ice as it sublimes, maintaining the well filled to the liquid surface, until a continuous ice mantle as thick
E1750 − 23
as desired forms on the surface of the well within the water (usually 4 to 8 mm 4 mm to 8 mm thick). The mantle will appear
thicker than its actual thickness because of the lenticular shape of the cell and the refractive index of water. The actual thickness
may be best estimated by viewing from the bottom of the cell while it is inverted or by immersing the cell in a large glass container
of water. (Warning—During preparation, the mantle should never be allowed to grow at any place to completely bridge the space
between the well and the inner wall of the cell, as the expansion of the ice may break the cell. In particular, if bridging occurs at
the surface of the water at the top of the cell under the vapor space, melt the ice bridge by warming the cell locally with heat from
the hand, while gently shaking the cell.)
7.1.10 When the mantle attains nearly the desired thickness or after maintaining the dry ice level in the well at the water surface
for about 20 min, return the cell to the ice bath with the entrance to the thermometer well slightly above the ice bath surface and
allow the dry ice to sublime completely. By allowing the dry ice to sublime completely, the bottom of the well stays cold longer
and the mantle grows thicker there.
7.1.11 After the thermometer well becomes free of dry ice, immerse the cell deeper into the ice bath and fill the well with ice bath
water.
7.1.12 Allow the cell to remain packed in ice or in the ice bath for two days to stabilize its temperature. Because of the strains
in the ice mantle prepared using dry ice, a freshly prepared mantle can give a temperature that is as much as 0.2 mK low.0.2 mK
lower.
7.1.13 When initially prepared, the mantle will be fixed to the wall of the well. Before the cell can be used, a thin layer of ice
next to the thermometer well shall be melted. To prepare this “inner melt,” briefly and gently insert a metal or glass rod, initially
at room temperature, into the well to heat the well slightly. The rod should have a smooth rounded end to avoid scratching or
possibly breaking the cell. Upon removal of the rod, tilt the cell to an angle of about 45° fro
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...