ASTM D4266-17
(Test Method)Standard Test Methods for Precoat Capacity of Powdered Ion-Exchange Resins
Standard Test Methods for Precoat Capacity of Powdered Ion-Exchange Resins
SIGNIFICANCE AND USE
4.1 The salt removal capacity of a powdered resin precoat is limited by the capacity of either the anion-exchange resin or the cation-exchange resin contained in it. Applications include condensate polishing in fossil-fueled electric generating plants, as well as condensate polishing, spent fuel pool water treatment, reactor water treatment, and low-level radioactive liquid waste treatment in nuclear-powered electric generating plants.
4.2 By determining the ion-exchange capacity profile of either a cation exchange resin or an anion-exchange resin (capacity expended per unit of time under specific conditions), it is possible to estimate runlength and remaining capacity when treating a liquid of the same makeup. Although they cannot accurately predict performance during condenser leaks, these test methods are useful for determining operating capacities as measured under the test conditions used.
4.3 These test methods may be used to monitor the performance of either powdered anion-exchange resin or powdered cation-exchange resin. The total capacity of either resin depends primarily upon the number density of ion-exchange sites within the resin. The operating capacity is a function of the total capacity, degree of conversion to the desired ionic form when received, and properties of the resin and the system that affect ion exchange kinetics.
SCOPE
1.1 These test methods cover the determination of the operating ion-exchange capacity of both powdered cation-exchange resins (hydrogen form) and powdered anion-exchange resins (hydroxide form). These test methods are intended for use in testing new powdered ion-exchange resins when used for the treatment of water. The following two test methods are included:
Sections
Test Method A—Operating Capacity, Anion-Exchange
Resin, Hydroxide Form
7 to 15
Test Method B—Operating Capacity, Cation-Exchange
Resin, Hydrogen Form
16 to 24
1.2 The values stated in SI units are to be regarded as the standard. The inch-pound units given in parentheses are for information only.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-May-2017
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.08 - Membranes and Ion Exchange Materials
Relations
- Replaces
ASTM D4266-96(2009)e1 - Standard Test Methods for Precoat Capacity of Powdered Ion-Exchange Resins - Effective Date
- 01-Jun-2017
- Effective Date
- 01-Apr-2024
- Effective Date
- 01-May-2020
- Effective Date
- 01-Aug-2017
- Effective Date
- 15-May-2016
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-May-2009
- Effective Date
- 01-Dec-2007
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 15-Feb-2006
- Effective Date
- 01-Jun-2005
- Effective Date
- 01-Apr-2005
- Refers
ASTM D1125-95(2005) - Standard Test Methods for Electrical Conductivity and Resistivity of Water - Effective Date
- 01-Apr-2005
Overview
ASTM D4266-17: Standard Test Methods for Precoat Capacity of Powdered Ion-Exchange Resins provides established methodologies for determining the operating ion-exchange capacity of both powdered cation-exchange resins (hydrogen form) and powdered anion-exchange resins (hydroxide form). Developed by ASTM International, this standard is critical for industries that employ ion-exchange resins in water treatment applications, ensuring the effective removal of dissolved salts and improving water purity.
The procedures covered in ASTM D4266-17 are designed for testing new powdered resins intended for water treatment processes. By evaluating the precoat capacity, users can better estimate runlength, monitor resin performance, and maintain optimal operation in facilities such as power plants.
Key Topics
- Ion-Exchange Resin Capacity Measurement: The standard details methods to determine the salt removal (ion-exchange) capacity of powdered resin precoats, focusing specifically on their operational effectiveness under predefined conditions.
- Test Method A: Describes the operating capacity determination for powdered anion-exchange resin in the hydroxide form.
- Test Method B: Outlines the procedure for measuring the operating capacity of powdered cation-exchange resin in the hydrogen form.
- Resin Performance Monitoring: Enables operators to assess and compare the performance of different resins in precoat filtration systems, even though direct prediction under leak or unusual conditions is limited.
- Material Definitions: Provides terminology for key parameters such as “resin dosage,” “resin floc,” and “resin ratio.”
- Applicability: Procedures address both the upper bound of potential resin performance and comparative analysis across resin batches or sources.
Applications
The test methods specified in ASTM D4266-17 are essential in multiple high-value applications, especially within the power generation sector:
- Condensate Polishing: In both fossil-fueled and nuclear-powered electric generating plants, maintaining high-purity water cycles is vital. Measuring powdered resin precoat capacity ensures efficient condensate polishing and protection of plant equipment.
- Nuclear Plant Water Treatment: Used in reactor water systems, spent fuel pool water purification, and low-level radioactive liquid waste treatment.
- Quality Assurance: Standardized testing enables utilities, water treatment companies, and resin manufacturers to monitor new batches, troubleshoot system performance, and verify supplier claims.
- Process Optimization: Operators can use results from these test methods to adjust the dosage and composition of resin precoats, maximizing service life and improving cost-efficiency.
- Regulatory Compliance: Supporting adherence to international standards and water purity requirements set by governing bodies.
Related Standards
ASTM D4266-17 refers to several other important ASTM standards related to water analysis and ion-exchange materials, including:
- ASTM D1125: Test Methods for Electrical Conductivity and Resistivity of Water
- ASTM D1129: Terminology Relating to Water
- ASTM D1193: Specification for Reagent Water
- ASTM D2687: Practices for Sampling Particulate Ion-Exchange Materials
- ASTM D4456: Test Methods for Physical and Chemical Properties of Powdered Ion Exchange Resins
- ASTM D5391: Test Method for Electrical Conductivity and Resistivity of a Flowing High Purity Water Sample
- ASTM E200: Practice for Preparation, Standardization, and Storage of Standard and Reagent Solutions for Chemical Analysis
In summary, ASTM D4266-17 is a foundational reference for professionals and organizations aiming to standardize the assessment and operational management of powdered ion-exchange resins in critical water treatment processes. By applying the methods outlined in this standard, users can achieve consistent, reliable results essential to effective plant operation and water quality management.
Buy Documents
ASTM D4266-17 - Standard Test Methods for Precoat Capacity of Powdered Ion-Exchange Resins
REDLINE ASTM D4266-17 - Standard Test Methods for Precoat Capacity of Powdered Ion-Exchange Resins
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D4266-17 is a standard published by ASTM International. Its full title is "Standard Test Methods for Precoat Capacity of Powdered Ion-Exchange Resins". This standard covers: SIGNIFICANCE AND USE 4.1 The salt removal capacity of a powdered resin precoat is limited by the capacity of either the anion-exchange resin or the cation-exchange resin contained in it. Applications include condensate polishing in fossil-fueled electric generating plants, as well as condensate polishing, spent fuel pool water treatment, reactor water treatment, and low-level radioactive liquid waste treatment in nuclear-powered electric generating plants. 4.2 By determining the ion-exchange capacity profile of either a cation exchange resin or an anion-exchange resin (capacity expended per unit of time under specific conditions), it is possible to estimate runlength and remaining capacity when treating a liquid of the same makeup. Although they cannot accurately predict performance during condenser leaks, these test methods are useful for determining operating capacities as measured under the test conditions used. 4.3 These test methods may be used to monitor the performance of either powdered anion-exchange resin or powdered cation-exchange resin. The total capacity of either resin depends primarily upon the number density of ion-exchange sites within the resin. The operating capacity is a function of the total capacity, degree of conversion to the desired ionic form when received, and properties of the resin and the system that affect ion exchange kinetics. SCOPE 1.1 These test methods cover the determination of the operating ion-exchange capacity of both powdered cation-exchange resins (hydrogen form) and powdered anion-exchange resins (hydroxide form). These test methods are intended for use in testing new powdered ion-exchange resins when used for the treatment of water. The following two test methods are included: Sections Test Method A—Operating Capacity, Anion-Exchange Resin, Hydroxide Form 7 to 15 Test Method B—Operating Capacity, Cation-Exchange Resin, Hydrogen Form 16 to 24 1.2 The values stated in SI units are to be regarded as the standard. The inch-pound units given in parentheses are for information only. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 The salt removal capacity of a powdered resin precoat is limited by the capacity of either the anion-exchange resin or the cation-exchange resin contained in it. Applications include condensate polishing in fossil-fueled electric generating plants, as well as condensate polishing, spent fuel pool water treatment, reactor water treatment, and low-level radioactive liquid waste treatment in nuclear-powered electric generating plants. 4.2 By determining the ion-exchange capacity profile of either a cation exchange resin or an anion-exchange resin (capacity expended per unit of time under specific conditions), it is possible to estimate runlength and remaining capacity when treating a liquid of the same makeup. Although they cannot accurately predict performance during condenser leaks, these test methods are useful for determining operating capacities as measured under the test conditions used. 4.3 These test methods may be used to monitor the performance of either powdered anion-exchange resin or powdered cation-exchange resin. The total capacity of either resin depends primarily upon the number density of ion-exchange sites within the resin. The operating capacity is a function of the total capacity, degree of conversion to the desired ionic form when received, and properties of the resin and the system that affect ion exchange kinetics. SCOPE 1.1 These test methods cover the determination of the operating ion-exchange capacity of both powdered cation-exchange resins (hydrogen form) and powdered anion-exchange resins (hydroxide form). These test methods are intended for use in testing new powdered ion-exchange resins when used for the treatment of water. The following two test methods are included: Sections Test Method A—Operating Capacity, Anion-Exchange Resin, Hydroxide Form 7 to 15 Test Method B—Operating Capacity, Cation-Exchange Resin, Hydrogen Form 16 to 24 1.2 The values stated in SI units are to be regarded as the standard. The inch-pound units given in parentheses are for information only. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D4266-17 is classified under the following ICS (International Classification for Standards) categories: 71.100.40 - Surface active agents. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D4266-17 has the following relationships with other standards: It is inter standard links to ASTM D4266-96(2009)e1, ASTM D2687-95(2024), ASTM D1129-13(2020)e2, ASTM D4456-17, ASTM D2687-95(2016), ASTM D1129-10, ASTM D4456-99(2009)e1, ASTM D2687-95(2007)e1, ASTM D1129-06a, ASTM D1129-06ae1, ASTM D1193-06, ASTM D1129-06, ASTM D4456-99(2005), ASTM D5391-99(2005), ASTM D1125-95(2005). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D4266-17 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D4266 − 17
Standard Test Methods for
Precoat Capacity of Powdered Ion-Exchange Resins
This standard is issued under the fixed designation D4266; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope D2687PracticesforSamplingParticulateIon-ExchangeMa-
terials
1.1 These test methods cover the determination of the
D4456Test Methods for Physical and Chemical Properties
operating ion-exchange capacity of both powdered cation-
of Powdered Ion Exchange Resins
exchange resins (hydrogen form) and powdered anion-
D5391Test Method for Electrical Conductivity and Resis-
exchange resins (hydroxide form). These test methods are
tivity of a Flowing High Purity Water Sample
intended for use in testing new powdered ion-exchange resins
E200Practice for Preparation, Standardization, and Storage
when used for the treatment of water. The following two test
of Standard and Reagent Solutions for ChemicalAnalysis
methods are included:
Sections
3. Terminology
Test Method A—Operating Capacity, Anion-Exchange 7 to 15
3.1 Definitions:
Resin, Hydroxide Form
Test Method B—Operating Capacity, Cation-Exchange 16 to 24
3.1.1 For definitions of other terms used in this standard,
Resin, Hydrogen Form
refer to Terminology D1129.
1.2 The values stated in SI units are to be regarded as the
3.2 Definitions of Terms Specific to This Standard:
standard. The inch-pound units given in parentheses are for
3.2.1 powdered ion-exchange material, n—an ion-exchange
information only.
resin that has undergone post-manufacturing size reduction to
1.3 This standard does not purport to address all of the
less than 300 µm.
safety concerns, if any, associated with its use. It is the
3.2.2 resin dosage, n—theweightofmixedresinappliedper
responsibility of the user of this standard to establish appro-
unit area of precoatable filter surface. This is expressed as dry
priate safety and health practices and determine the applica-
pounds per square foot.
bility of regulatory limitations prior to use.
3.2.3 resin floc, n—thatvoluminousaggregateformedwhen
1.4 This international standard was developed in accor-
powdered anion-exchange resin and powdered cation-
dance with internationally recognized principles on standard-
exchange resin are slurried together in an aqueous suspension.
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
3.2.4 resin ratio, n—the ratio of the weights of powdered
mendations issued by the World Trade Organization Technical
cation-exchange resin to powdered anion-exchange resin used
Barriers to Trade (TBT) Committee.
to prepare a resin slurry. If not otherwise indicated, it is
understood to be the ratio of the dry resin weights.
2. Referenced Documents
4. Significance and Use
2.1 ASTM Standards:
4.1 Thesaltremovalcapacityofapowderedresinprecoatis
D1125Test Methods for Electrical Conductivity and Resis-
limited by the capacity of either the anion-exchange resin or
tivity of Water
the cation-exchange resin contained in it.Applications include
D1129Terminology Relating to Water
condensatepolishinginfossil-fueledelectricgeneratingplants,
D1193Specification for Reagent Water
as well as condensate polishing, spent fuel pool water
treatment, reactor water treatment, and low-level radioactive
liquid waste treatment in nuclear-powered electric generating
These test methods are under the jurisdiction of ASTM Committee D19 on
plants.
Water and are the direct responsibility of Subcommittee D19.08 on Membranes and
Ion Exchange Materials.
4.2 By determining the ion-exchange capacity profile of
Current edition approved June 1, 2017. Published June 2017. Originally
ɛ1
either a cation exchange resin or an anion-exchange resin
approved in 1983. Last previous edition approved in 2009 as D4266–96 (2009) .
DOI: 10.1520/D4266-17.
(capacity expended per unit of time under specific conditions),
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
it is possible to estimate runlength and remaining capacity
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
when treating a liquid of the same makeup. Although they
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. cannot accurately predict performance during condenser leaks,
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4266 − 17
thesetestmethodsareusefulfordeterminingoperatingcapaci- 9. Apparatus
ties as measured under the test conditions used.
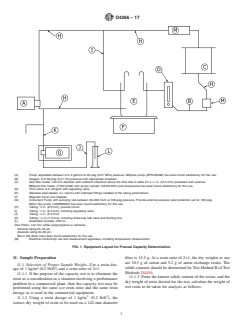
9.1 Test apparatus, as shown in Fig. 1, with the following
components:
4.3 These test methods may be used to monitor the perfor-
mance of either powdered anion-exchange resin or powdered 9.1.1 Water Pump—adjustable between 0 to 7.57 L/min (0
5 4
to 2 gal/min) at 2.76×10 Pa (40 psig) pressure.
cation-exchange resin. The total capacity of either resin de-
pendsprimarilyuponthenumberdensityofion-exchangesites 9.1.2 Pressure Gauges (two), 0 to 4.137×10 Pa (0 to 60
psig) with appropriate snubbers.
within the resin. The operating capacity is a function of the
total capacity, degree of conversion to the desired ionic form 9.1.3 Disk Filter Holder, 142-mm diameter with sufficient
clearance above the filter disk to allow for uniform application
when received, and properties of the resin and the system that
affect ion exchange kinetics. of resin precoat.
9.1.4 Filter-Disk, 142-mm diameter, with nominal retention
5. Purity of Reagents rating of 25 to 30 µm and absolute retention rating of 40 to
60µm.
5.1 Reagent grade chemicals shall be used in all tests.
9.1.5 Flow Meter, 0 to 1.89 L/min (0 to 30 gal/h) with
Unlessotherwiseindicated,itisintendedthatallreagentsshall
regulating valve.
conform to the specifications of the Committee on Analytical
9.1.6 Beaker, stainless steel, 4 L to volume with bulkhead
Reagents of the American Chemical Society, where such
fittings installed at tubing penetrations.
specifications are available.
9.1.7 Chemical Pump, with pumping rate between
−6 −5
5.2 Purity of Water—Unless otherwise indicated, references
8.33×10 and 8.33×10 L/s (30 to 300 mL/h) at 3.45×10
to water shall be understood to mean Type II reagent water,
6 Pa (500 psig) pressure. Suction tubing should be 3.2-mm
Specification D1193.
( ⁄8-in.) outside diameter stainless steel and discharge tubing
should be 1.6-mm ( ⁄16-in.) outside diameter stainless steel.
6. Sampling
9.2 Electrical Conductivity Measurement Apparatus, con-
6.1 Obtain a representative sample of the powdered ion-
formingtotherequirementsgiveninTestMethodsD1125,Test
exchange resin in accordance with Practices D2687 but sub-
Method B, and D5391.
stituting a 12.5-mm ( ⁄2-in.) inside diameter tube.
10. Reagents
TEST METHOD A—OPERATING CAPACITY,
10.1 Hydrochloric Acid Solution, Standard (0.10 N)—
ANION-EXCHANGE RESIN, HYDROXIDE FORM
Prepare and standardize as described in Practice E200.
7. Scope 10.2 Polyacrylic Acid Solution, Standard (1+99)—Pipet 1
mL of polyacrylic acid (25 weight% solids, MW<50 000)
7.1 This test method covers the determination of ion-
into a 100 mL volumetric flask and dilute to 100 mL with
exchange capacity, on a dry weight basis, of new powdered
water. Mix well. Prepare this solution fresh daily.
anion-exchange resins in the hydroxide form.
7.2 The ion-exchange capacity obtainable in commercial
installations depends not only upon the initial state of the
The sole source of supply of the Millipore pump (ZPN100400) apparatus
powdered resin, but also on how the resin floc is prepared and
knowntothecommitteeatthistimeistheMilliporeCorporation,290ConcordRd.,
applied, on the condition of the equipment on which it is to be
Billerica, MA 01821. If you are aware of alternative suppliers, please provide this
used, and the pH and general chemistry of the water system information to ASTM International Headquarters. Your comments will receive
careful consideration at a meeting of the responsible technical committee, which
being treated. Thus, this test method has comparative rather
you may attend.
than predictive value and provides an upper limit on exchange
The sole source of supply of the Millipore filter holder (YY2214230) with
capacity that may be expected.
acryliccylinder(XX4214201)andaccessoriesapparatusknowntothecommitteeat
this time is the Millipore Corporation, 290 Concord Rd., Billerica, MA 01821. If
you are aware of alternative suppliers, please provide this information to ASTM
8. Summary of Test Method
International Headquarters. Your comments will receive careful consideration at a
meeting of the responsible technical committee, which you may attend.
8.1 The powdered anion-exchange resin to be tested is
The sole source of supply of the BG or DG filter apparatus known to the
slurried with an appropriate amount of powdered cation-
committee at this time is the Pall Corporation, 30 Sea Cliff Ave., Glen Cove, NY
exchange resin in the hydrogen form, and the resulting floc is
11542. If you are aware of alternative suppliers, please provide this information to
ASTM International Headquarters. Your comments will receive careful consider-
precoatedontoafilterdisk.Thenadilutestandardizedsolution
ation at a meeting of the responsible technical committee, which you may attend.
of a strong acid is fed to the precoat while monitoring the
The sole source of supply of the Milton Roy pump (1960066002) apparatus
effluent stream conductometrically.
known to the committee at this time is Milton Roy USA, 201 Ivyland Rd., Ivyland,
PA18974. If you are aware of alternative suppliers, please provide this information
to ASTM International Headquarters. Your comments will receive careful consid-
erationatameetingoftheresponsibletechnicalcommittee, whichyoumayattend.
3 8
Reagent Chemicals, American Chemical Society Specifications, American The sole source of supply of the Accumer (1510) apparatus known to the
Chemical Society, Washington, DC. For suggestions on the testing of reagents not committee at this time is the Rohm and Haas Company, 100 Independence Mall
listed by the American Chemical Society, see Analar Standards for Laboratory West, Philadelphia, PA 19106. If you are aware of alternative suppliers, please
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia provide this information toASTM International Headquarters.Your comments will
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, receive careful consideration at a meeting of the responsible technical committee,
MD. which you may attend.
D4266 − 17
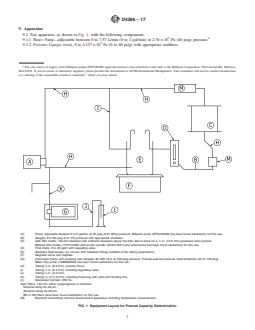
(A) Pump, adjustable between 0 to 2 gal/min at 40 psig (0.27 MPa) pressure. Millipore pump (ZPN100400) has been found satisfactory for this use.
(B) Gauges, 0 to 60 psig (0.41 Pa) pressure with appropriate snubbers.
(C)
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
´1
Designation: D4266 − 96 (Reapproved 2009) D4266 − 17
Standard Test Methods for
Precoat Capacity of Powdered Ion-Exchange Resins
This standard is issued under the fixed designation D4266; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Updated 5.2 editorially in June 2009.
1. Scope
1.1 These test methods cover the determination of the operating ion-exchange capacity of both powdered cation-exchange resins
(hydrogen form) and powdered anion-exchange resins (hydroxide form). These test methods are intended for use in testing new
powdered ion-exchange resins when used for the treatment of water. The following two test methods are included:
Sections
Test Method A—Operating Capacity, Anion-Exchange 7 to 15
Resin, Hydroxide Form
Test Method B—Operating Capacity, Cation-Exchange 16 to 24
Resin, Hydrogen Form
1.2 The values stated in SI units are to be regarded as the standard. The inch-pound units given in parentheses are for
information only.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1125 Test Methods for Electrical Conductivity and Resistivity of Water
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
D2687 Practices for Sampling Particulate Ion-Exchange Materials
D4456 Test Methods for Physical and Chemical Properties of Powdered Ion Exchange Resins
D5391 Test Method for Electrical Conductivity and Resistivity of a Flowing High Purity Water Sample
E200 Practice for Preparation, Standardization, and Storage of Standard and Reagent Solutions for Chemical Analysis
3. Terminology
3.1 Definitions:
3.1.1 For definitions of other terms used in this standard, refer to Terminology D1129.
3.2 Definitions of Terms Specific to This Standard:
3.1.1 For definitions of other terms used in these test methods, refer to Terminology D1129.
3.2.1 powdered ion-exchange material, n—an ion-exchange resin that has undergone post-manufacturing size reduction to less
than 300 μm.
These test methods are under the jurisdiction of ASTM Committee D19 on Water and are the direct responsibility of Subcommittee D19.08 on Membranes and Ion
Exchange Materials.
Current edition approved May 1, 2009June 1, 2017. Published June 2009 June 2017. Originally approved in 1983. Last previous edition approved in 20072009 as
ɛ1
D4266 – 96 (2007).(2009) . DOI: 10.1520/D4266-96R09E01.10.1520/D4266-17.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4266 − 17
3.2.2 resin dosage, n—the weight of mixed resin applied per unit area of precoatable filter surface. This is expressed as dry
pounds per square foot.
3.2.3 resin floc, n—that voluminous aggregate formed when powdered anion-exchange resin and powdered cation-exchange
resin are slurried together in an aqueous suspension.
3.2.4 resin ratio, n—the ratio of the weights of powdered cation-exchange resin to powdered anion-exchange resin used to
prepare a resin slurry. If not otherwise indicated, it is understood to be the ratio of the dry resin weights.
4. Significance and Use
4.1 The salt removal capacity of a powdered resin precoat is limited by the capacity of either the anion-exchange resin or the
cation-exchange resin contained in it. Applications include condensate polishing in fossil-fueled electric generating plants, as well
as condensate polishing, spent fuel pool water treatment, reactor water treatment, and low-level radioactive liquid waste treatment
in nuclear-powered electric generating plants.
4.2 By determining the ion-exchange capacity profile of either a cation exchange resin or an anion-exchange resin (capacity
expended per unit of time under specific conditions), it is possible to estimate runlength and remaining capacity when treating a
liquid of the same makeup. Although they cannot accurately predict performance during condenser leaks, these test methods are
useful for determining operating capacities as measured under the test conditions used.
4.3 These test methods may be used to monitor the performance of either powdered anion-exchange resin or powdered
cation-exchange resin. The total capacity of either resin depends primarily upon the number density of ion-exchange sites within
the resin. The operating capacity is a function of the total capacity, degree of conversion to the desired ionic form when received,
and properties of the resin and the system that affect ion exchange kinetics.
5. Purity of Reagents
5.1 Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all reagents shall conform
to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where such specifications are
available.
5.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean Type II reagent water,
Specification D1193.
6. Sampling
6.1 Obtain a representative sample of the powdered ion-exchange resin in accordance with Practices D2687 but substituting a
12.5-mm ( ⁄2-in.) inside diameter tube.
TEST METHOD A—OPERATING CAPACITY,
ANION-EXCHANGE RESIN, HYDROXIDE FORM
7. Scope
7.1 This test method covers the determination of ion-exchange capacity, on a dry weight basis, of new powdered
anion-exchange resins in the hydroxide form.
7.2 The ion-exchange capacity obtainable in commercial installations depends not only upon the initial state of the
powdered resin, but also on how the resin floc is prepared and applied, on the condition of the equipment on which it is to be used,
and the pH and general chemistry of the water system being treated. Thus, this test method has comparative rather than predictive
value and provides an upper limit on exchange capacity that may be expected.
8. Summary of Test Method
8.1 The powdered anion-exchange resin to be tested is slurried with an appropriate amount of powdered cation-exchange resin
in the hydrogen form, and the resulting floc is precoated onto a filter disk. Then a dilute standardized solution of a strong acid is
fed to the precoat while monitoring the effluent stream conductometrically.
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by
the American Chemical Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
D4266 − 17
9. Apparatus
9.1 Test apparatus, as shown in Fig. 1, with the following components:
5 4
9.1.1 Water Pump—adjustable between 0 to 7.57 L/min (0 to 2 gal/min) at 2.76 × 10 Pa (40 psig) pressure.
9.1.2 Pressure Gauges (two), 0 to 4.137 × 10 Pa (0 to 60 psig) with appropriate snubbers.
The sole source of supply of the Millipore pump (ZPN100400) apparatus known to the committee at this time is the Millipore Corporation, 290 Concord Rd., Billerica,
MA 01821. If you are aware of alternative suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration
at a meeting of the responsible technical committee, which you may attend.
(A) Pump, adjustable between 0 to 2 gal/min at 40 psig (0.27 MPa) pressure. Millipore pump (ZPN100400) has been found satisfactory for this use.
(B) Gauges, 0 to 60 psig (0.41 Pa) pressure with appropriate snubbers.
(C) Disk filter holder, 142-mm diameter with sufficient clearance above the filter disk to allow for a ⁄2 in. (12.5 mm) powdered resin precoat.
Millipore filter holder (YY2214230) with acrylic cylinder (XX4214201) and accessories has been found satisfactory for this use.
(D) Flow metre, 0 to 30 gal/h with regulating valve.
(E) Stainless steel beaker, 4-L volume with bulkhead fittings installed at the tubing penetrations.
(F) Magnetic stirrer and hotplate.
(G) Instrument Pump, with pumping rate between 30–300 mL/h at 100-psig pressure. Provide external pressure relief protection set for 100 psig.
Milton Roy pump (1960066002) has been found satisfactory for this use.
(H) Tubing, ⁄8 in. (9.5 mm), precoat circuit.
(I) Tubing, ⁄4 in. (6.4 mm), including regulating valve.
(J) Tubing, ⁄8 in. (3.2 mm).
(K) Tubing, ⁄16 in (1.6 mm), including three-way ball valve and flushing line.
(L) Graduated Cylinder, 250 mL.
Disk Filters, 142 mm, either polypropylene or cellulose.
Nominal rating 25–30 μm.
Absolute rating 40–60 μm.
BG or DG filters have been found satisfactory for this use.
(M) Electrical conductivity cell and measurement apparatus, including temperature measurement.
FIG. 1 Equipment Layout for Precoat Capacity Determination
D4266 − 17
9.1.3 Disk Filter Holder, 142-mm diameter with sufficient clearance above the filter disk to allow for uniform application of
resin precoat.
9.1.4 Filter-Disk, 142-mm diameter, with nominal retention rating of 25 to 30 μm and absolute retention rating of 40 to 60μm.
9.1.5 Flow Meter, 0 to 1.89 L/min (0 to 30 gal/h) with regulating valve.
9.1.6 Beaker, stainless steel, 4 L to volume with bulkhead fittings installed at tubing penetrations.
−6 −5 6
9.1.7 Chemical Pump, with pumping rate between 8.33 × 10 and 8.33 × 10 L/s (30 to 300 mL/h) at 3.45 × 10 Pa (500 psig)
1 1
pressure. Suction tubing should be 3.2-mm ( ⁄8-in.) outside diameter stainless steel and discharge tubing should be 1.6-mm ( ⁄16-in.)
outside diameter stainless steel.
9.2 Electrical Conductivity Measurement Apparatus, conforming to the requirements given in Test Methods D1125, Test
Method B.B, and D5391.
10. Reagents
10.1 Hydrochloric Acid Solution, Standard (0.10 N)—Prepare and standardize as described in Practice E200.
10.2 Polyacrylic Acid Solution, Standard (1+99)—Pipet 1 mL of polyacrylic acid (25 weight % solids, MW <50 000) into a
100 mL volumetric flask and dilute to 100 mL with water. Mix well. Prepare this solution fresh daily.
11. Sample Preparation
2 2
11.1 Selection of Proper Sample Weight—Use a resin dosage of 1 kg/m (0.2 lb/ft ) and a resin ratio of 2+1.
11.1.1 If the purpose of the capacity test is to eliminate the resin as a consideration in a situation involving a performance
problem in a commercial plant, then the capacity test ma
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...