ASTM F3207-17
(Guide)Standard Guide for in vivo Evaluation of Rabbit Lumbar Intertransverse Process Spinal Fusion Model
Standard Guide for <emph type="bdit">in vivo</emph> Evaluation of Rabbit Lumbar Intertransverse Process Spinal Fusion Model
SIGNIFICANCE AND USE
4.1 This guide covers animal implantation methods and analysis of bone void fillers to determine whether a material or substance leads to lumbar intertransverse process spinal fusion, as defined by its ability to cause bone to form in vivo.
SCOPE
1.1 Historically, the single-level rabbit posterolateral, or intertransverse, lumbar spine fusion model was developed and reported on by Dr. Scott Boden, et. al. (Emory Spine Center for Orthopedics) and the model has been proposed as a non-clinical model which may be used to replicate clinically-relevant fusion rates for iliac crest autograft in the posterolateral spine (1, 2).2 This model is used routinely in submissions to regulatory bodies for the purpose of evaluating the potential efficacy of bone void filler materials as compared to other materials or iliac crest autograft to effect spinal posterolateral fusion. The use of this standard’s recommendations as part of a regulatory submission does not provide any guarantee of regulatory clearance and should be considered as a part of the data provided for regulatory submission.
1.2 This guide covers general guidelines to evaluate the effectiveness of products intended to cause and/or promote bone formation in the lumbar intertransverse process spinal fusion model in vivo. This guide is applicable to products that may be composed of one or more of the following components: natural biomaterials (such as demineralized bone), and synthetic biomaterials (such as calcium sulfate, glycerol, and reverse phase polymeric compounds) that act as additives, fillers, and/or excipients (radioprotective agents, preservatives, and/or handling agents). It should not be assumed that products evaluated favorably using this guidance will form bone when used in a clinical setting. The primary purpose of this guide is to facilitate the equitable comparison of bone void fillers and/or autograft extender products in vivo. The purpose of this guide is not to exclude other established methods.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with the use of bone void fillers. It is the responsibility of the user of this standard to establish appropriate safety and health practices involved in the development of said products in accordance with applicable regulatory guidance documents and in implementing this guide to evaluate the bone-forming/promoting capabilities of the product.
1.5 This standard does not purport to address the requirements under 21 CFR Part 58 concerning Good Laboratory Practices or international standard counterpart OECD Principles of Good Laboratory Practice (GLP). It is the responsibility of the sponsor of the study to understand the requirements for conduct of animal studies whereby the data may be used to support premarket applications, including requirements for personnel, protocol content, record retention and animal husbandry.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-May-2017
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.44 - Assessment for TEMPs
Relations
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2018
- Effective Date
- 01-Aug-2013
- Effective Date
- 01-Aug-2012
- Effective Date
- 01-Aug-2012
- Effective Date
- 01-Aug-2011
- Effective Date
- 01-Aug-2009
- Effective Date
- 15-May-2009
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-May-2008
- Effective Date
- 01-Oct-2007
- Effective Date
- 10-Oct-2002
Overview
ASTM F3207-17 provides a standardized guide for the in vivo evaluation of bone void fillers and bone formation-promoting materials using the rabbit lumbar intertransverse process spinal fusion model. Developed by ASTM International, this standard is widely referenced in the preclinical assessment of medical devices and biomaterials aimed at achieving spinal fusion. The model, established by Dr. Scott Boden and collaborators, has become a critical non-clinical method for assessing the efficacy of both autografts and synthetic or natural bone graft substitutes before clinical application.
The guide outlines procedures for animal implantation, surgical techniques, recommended materials, sample sizing, and various fusion assessment methods. It is commonly used in regulatory submissions to support new bone void filler materials, comparing their performance against established benchmarks like iliac crest autograft.
Key Topics
- Spinal Fusion Evaluation: Details the rabbit lumbar intertransverse process spinal fusion model for in vivo testing of bone-forming materials.
- Material Eligibility: Covers both natural biomaterials (e.g., demineralized bone) and synthetic biomaterials (e.g., calcium sulfate, glycerol, polymers) used as bone graft fillers or extenders.
- Surgical Protocols: Describes recommended aseptic surgical techniques, appropriate animal selection (skeletally mature New Zealand White rabbits), and post-operative care.
- Fusion Assessment Methods:
- Radiographic analysis (plain film and micro-CT imaging)
- Manual palpation for biomechanical evaluation
- Histological examination of new bone formation
- Biomechanical testing for mechanical strength and stiffness
- Sample Size Guidance: Provides considerations for determining statistically robust group sizes based on the primary outcome measures.
- Regulatory Context: Designed to support, but not replace, broader regulatory frameworks (e.g., 21 CFR Part 58 Good Laboratory Practice, OECD GLP).
Applications
The ASTM F3207-17 standard is practically valuable across several key segments of biomedical research and medical device development:
- Preclinical Testing: Enables researchers and manufacturers to assess bone void filler effectiveness in a reproducible and well-characterized animal model prior to human clinical trials.
- Spinal Device Development: Supports the development and comparative evaluation of new spinal fusion materials, including synthetic grafts and autograft extenders.
- Regulatory Submissions: Facilitates the inclusion of standardized animal model data in dossiers for regulatory clearances for bone graft materials and related medical devices.
- Comparative Effectiveness Studies: Provides a framework for head-to-head evaluation of different bone graft materials to determine relative fusion performance in vivo.
- Academic Research: Standardizes methodologies for academic investigations into bone healing, biomaterials testing, and spinal fusion mechanisms.
Related Standards
- ASTM F2529: Guide for in vivo Evaluation of Osteoinductive Potential for Materials Containing Demineralized Bone (DBM)
- ASTM F2884: Guide for Pre-clinical in vivo Evaluation of Spinal Fusion
- ISO 10993-6: Biological evaluation of medical devices - Part 6: Tests for local effects after implantation
- 21 CFR Part 58: Good Laboratory Practice for Nonclinical Laboratory Studies
- ASTM E122: Practice for Calculating Sample Size to Estimate the Average for a Characteristic of a Lot or Process
- ASTM E1488: Guide for Statistical Procedures to Use in Developing and Applying Test Methods
Conclusion
ASTM F3207-17 is a key international standard for preclinical in vivo evaluation of bone graft substitutes and bone formation-promoting materials in spinal fusion. Adherence to this guide helps ensure test consistency, reliable comparison, and relevance to clinical outcomes, thus supporting innovation and safety in spinal device development and regulatory approval processes.
Buy Documents
ASTM F3207-17 - Standard Guide for <emph type="bdit">in vivo</emph> Evaluation of Rabbit Lumbar Intertransverse Process Spinal Fusion Model
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F3207-17 is a guide published by ASTM International. Its full title is "Standard Guide for <emph type="bdit">in vivo</emph> Evaluation of Rabbit Lumbar Intertransverse Process Spinal Fusion Model". This standard covers: SIGNIFICANCE AND USE 4.1 This guide covers animal implantation methods and analysis of bone void fillers to determine whether a material or substance leads to lumbar intertransverse process spinal fusion, as defined by its ability to cause bone to form in vivo. SCOPE 1.1 Historically, the single-level rabbit posterolateral, or intertransverse, lumbar spine fusion model was developed and reported on by Dr. Scott Boden, et. al. (Emory Spine Center for Orthopedics) and the model has been proposed as a non-clinical model which may be used to replicate clinically-relevant fusion rates for iliac crest autograft in the posterolateral spine (1, 2).2 This model is used routinely in submissions to regulatory bodies for the purpose of evaluating the potential efficacy of bone void filler materials as compared to other materials or iliac crest autograft to effect spinal posterolateral fusion. The use of this standard’s recommendations as part of a regulatory submission does not provide any guarantee of regulatory clearance and should be considered as a part of the data provided for regulatory submission. 1.2 This guide covers general guidelines to evaluate the effectiveness of products intended to cause and/or promote bone formation in the lumbar intertransverse process spinal fusion model in vivo. This guide is applicable to products that may be composed of one or more of the following components: natural biomaterials (such as demineralized bone), and synthetic biomaterials (such as calcium sulfate, glycerol, and reverse phase polymeric compounds) that act as additives, fillers, and/or excipients (radioprotective agents, preservatives, and/or handling agents). It should not be assumed that products evaluated favorably using this guidance will form bone when used in a clinical setting. The primary purpose of this guide is to facilitate the equitable comparison of bone void fillers and/or autograft extender products in vivo. The purpose of this guide is not to exclude other established methods. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with the use of bone void fillers. It is the responsibility of the user of this standard to establish appropriate safety and health practices involved in the development of said products in accordance with applicable regulatory guidance documents and in implementing this guide to evaluate the bone-forming/promoting capabilities of the product. 1.5 This standard does not purport to address the requirements under 21 CFR Part 58 concerning Good Laboratory Practices or international standard counterpart OECD Principles of Good Laboratory Practice (GLP). It is the responsibility of the sponsor of the study to understand the requirements for conduct of animal studies whereby the data may be used to support premarket applications, including requirements for personnel, protocol content, record retention and animal husbandry. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This guide covers animal implantation methods and analysis of bone void fillers to determine whether a material or substance leads to lumbar intertransverse process spinal fusion, as defined by its ability to cause bone to form in vivo. SCOPE 1.1 Historically, the single-level rabbit posterolateral, or intertransverse, lumbar spine fusion model was developed and reported on by Dr. Scott Boden, et. al. (Emory Spine Center for Orthopedics) and the model has been proposed as a non-clinical model which may be used to replicate clinically-relevant fusion rates for iliac crest autograft in the posterolateral spine (1, 2).2 This model is used routinely in submissions to regulatory bodies for the purpose of evaluating the potential efficacy of bone void filler materials as compared to other materials or iliac crest autograft to effect spinal posterolateral fusion. The use of this standard’s recommendations as part of a regulatory submission does not provide any guarantee of regulatory clearance and should be considered as a part of the data provided for regulatory submission. 1.2 This guide covers general guidelines to evaluate the effectiveness of products intended to cause and/or promote bone formation in the lumbar intertransverse process spinal fusion model in vivo. This guide is applicable to products that may be composed of one or more of the following components: natural biomaterials (such as demineralized bone), and synthetic biomaterials (such as calcium sulfate, glycerol, and reverse phase polymeric compounds) that act as additives, fillers, and/or excipients (radioprotective agents, preservatives, and/or handling agents). It should not be assumed that products evaluated favorably using this guidance will form bone when used in a clinical setting. The primary purpose of this guide is to facilitate the equitable comparison of bone void fillers and/or autograft extender products in vivo. The purpose of this guide is not to exclude other established methods. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with the use of bone void fillers. It is the responsibility of the user of this standard to establish appropriate safety and health practices involved in the development of said products in accordance with applicable regulatory guidance documents and in implementing this guide to evaluate the bone-forming/promoting capabilities of the product. 1.5 This standard does not purport to address the requirements under 21 CFR Part 58 concerning Good Laboratory Practices or international standard counterpart OECD Principles of Good Laboratory Practice (GLP). It is the responsibility of the sponsor of the study to understand the requirements for conduct of animal studies whereby the data may be used to support premarket applications, including requirements for personnel, protocol content, record retention and animal husbandry. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F3207-17 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics; 11.100.99 - Other standards related to laboratory medicine. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3207-17 has the following relationships with other standards: It is inter standard links to ASTM E1402-13(2023), ASTM E1488-23, ASTM E1402-13(2018), ASTM E1402-13, ASTM E1488-12, ASTM E1488-12e1, ASTM E122-09e1, ASTM E122-09, ASTM E1488-09, ASTM E1402-08e1, ASTM E1488-08a, ASTM E1402-08, ASTM E1488-08, ASTM E122-07, ASTM E1488-02. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F3207-17 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F3207 − 17
Standard Guide for
in vivo Evaluation of Rabbit Lumbar Intertransverse Process
Spinal Fusion Model
This standard is issued under the fixed designation F3207; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope fillers. It is the responsibility of the user of this standard to
establish appropriate safety and health practices involved in
1.1 Historically, the single-level rabbit posterolateral, or
the development of said products in accordance with appli-
intertransverse, lumbar spine fusion model was developed and
cable regulatory guidance documents and in implementing this
reportedonbyDr.ScottBoden,et.al.(EmorySpineCenterfor
guide to evaluate the bone-forming/promoting capabilities of
Orthopedics) and the model has been proposed as a non-
the product.
clinical model which may be used to replicate clinically-
1.5 This standard does not purport to address the require-
relevant fusion rates for iliac crest autograft in the posterolat-
2 ments under 21 CFR Part 58 concerning Good Laboratory
eral spine (1, 2). This model is used routinely in submissions
Practices or international standard counterpart OECD Prin-
to regulatory bodies for the purpose of evaluating the potential
ciples of Good Laboratory Practice (GLP). It is the responsi-
efficacy of bone void filler materials as compared to other
bility of the sponsor of the study to understand the require-
materials or iliac crest autograft to effect spinal posterolateral
ments for conduct of animal studies whereby the data may be
fusion. The use of this standard’s recommendations as part of
used to support premarket applications, including require-
a regulatory submission does not provide any guarantee of
ments for personnel, protocol content, record retention and
regulatory clearance and should be considered as a part of the
animal husbandry.
data provided for regulatory submission.
1.6 This international standard was developed in accor-
1.2 This guide covers general guidelines to evaluate the
dance with internationally recognized principles on standard-
effectiveness of products intended to cause and/or promote
ization established in the Decision on Principles for the
bone formation in the lumbar intertransverse process spinal
Development of International Standards, Guides and Recom-
fusion model in vivo. This guide is applicable to products that
mendations issued by the World Trade Organization Technical
maybecomposedofoneormoreofthefollowingcomponents:
Barriers to Trade (TBT) Committee.
natural biomaterials (such as demineralized bone), and syn-
2. Referenced Documents
thetic biomaterials (such as calcium sulfate, glycerol, and
reverse phase polymeric compounds) that act as additives,
2.1 ASTM Standards:
fillers, and/or excipients (radioprotective agents, preservatives,
E122PracticeforCalculatingSampleSizetoEstimate,With
and/orhandlingagents).Itshouldnotbeassumedthatproducts
Specified Precision, the Average for a Characteristic of a
evaluated favorably using this guidance will form bone when
Lot or Process
used in a clinical setting. The primary purpose of this guide is
E1402Guide for Sampling Design
tofacilitatetheequitablecomparisonofbonevoidfillersand/or
E1488GuideforStatisticalProcedurestoUseinDeveloping
autograft extender products in vivo. The purpose of this guide
and Applying Test Methods
is not to exclude other established methods.
F2529Guideforin vivoEvaluationofOsteoinductivePoten-
tial for Materials Containing Demineralized Bone (DBM)
1.3 The values stated in SI units are to be regarded as
F2884Guide for Pre-clinical in vivo Evaluation of Spinal
standard. No other units of measurement are included in this
Fusion
standard.
2.2 Federal Documents:
1.4 This standard does not purport to address all of the
21 CFR 58Good Laboratory Practice for Nonclinical Labo-
safety concerns, if any, associated with the use of bone void
ratory Studies
This guide is under the jurisdiction ofASTM Committee F04 on Medical and
Surgical Materials and Devices and is the direct responsibility of Subcommittee For referenced ASTM standards, visit the ASTM website, www.astm.org, or
F04.44 on Assessment for TEMPs. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Current edition approved June 1, 2017. Published July 2017. DOI: 10.1520/ Standards volume information, refer to the standard’s Document Summary page on
F3207-17 the ASTM website.
2 4
Theboldfacenumbersinparenthesesrefertothelistofreferencesattheendof Available from U.S. Food and Drug Administration (FDA), 10903 New
this standard. Hampshire Ave., Silver Spring, MD 20993, http://www.fda.gov.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3207 − 17
2.3 AAMI/ISO Documents: 5. Animal Models
ISO 10993-6 Third edition 2016-12-01Biological evalua-
5.1 General Note—Appropriate positive or comparative
tion of medical devices — Part 6: Tests for local effects
controlsmaybeused.Forexample,comparativecontrolscould
after implantation
be similar devices, and positive controls could be autograft
from the animal.
3. Terminology
5.2 Skeletally mature New Zealand white rabbits—
3.1 Definitions:
(typically>7monthsand3.5-4.5kg).Proximaltibialanddistal
3.1.1 biomechanical fusion, n—the increased strength
femoral physes should be closed and verified via plain radio-
and/orstiffnessandreducedROMofaspinalunitascompared
graphs. Radiographic and histologic closure of the distal
to that measured before surgical intervention.
femoral growth plates occur at an average age of 21 and 22
3.1.2 biomechanical properties, n—as used in this
weeks, respectively. The proximal tibial physes close radio-
document, evaluation of the operative functional spinal unit
graphically and histologically at an average age of 26 and 28
multidirectional range of motion (ROM: Lateral bending,
weeks, respectively. A lateral radiograph is a more reliable
Flexion – Extension and Axial Rotation) properties under
method for assessing physeal closure in the rabbit, and radio-
non-destructive conditions, tensile stiffness and ultimate load.
graphic confirmation of tibial physeal closure should be
obtained prior to using rabbits that are younger than approxi-
3.1.3 fusion, n—a multifactorial outcome which can be
mately 7 months of age. Radiographic confirmation of physeal
characterized in terms of the radiographic, biomechanical and
closure is probably not necessary in rabbits 8 months or older
histological results of the intended spinal arthrodesis proce-
but should be provided for the sake of completeness. Some
dure.
minor variation in age of tibial growth plate closure may be
3.1.4 histological evidence of fusion, n—based on light
expected with different strains of New Zealand White rabbits.
microscopyofnewlyformedandremodeledbonespanningthe
Weight is not a reliable indicator of skeletal maturity in the
intertransverse region, with contiguous osseous connectivity
New Zealand White rabbit. All rabbits used should be of the
observed between the adjacent transverse processes. Assess-
same sex. (3, 4)
ment rationale must be justified.
5.3 Implant Mass/Volume—In general, implant mass (~1.6-
3.1.5 manual palpation, n—a method for evaluating spinal
2.2 grams; useful only for autograft)/volume (~2.5-3.0 cc) per
fusionstatusbyestimatingthestiffnessoftheoperativemotion
side is used. It is recommended that the experimental group
segment and adjacent superior motion segment by the appli-
contain the same total implant volume as any comparative
cation of multidirectional loads in lateral bending and flexion-
groupssotheresultsarecomparableandthepotentialeffectsof
extension using the hands.
the implant on intertransverse process spinal fusion can be
3.1.6 micro-computed tomographic (micro-CT) fusion,
determined.
n—tomographic fusion is based on interpretation of the
5.4 Sample Size:
micro-CT images, with fusion success based on the three-
5.4.1 Sample sizes should be justified in the study protocol
dimensional appearance of contiguous bone from transverse
and,ifpossible,shouldprovidestatisticalpowerappropriateto
process to transverse process (i.e. bridging bone).
the endpoint using appropriate statistical methods to justify as
3.1.7 non-union, n—a multifactorial outcome which can be
required. Interim time points may be used as appropriate and
characterized in terms of the radiographic, biomechanical and
justifications should be provided. Should statistical numbers
histological results indicating a lack of trabecular or cortical
not be practical or possible, empirical testing in the literature
bone spanning the intertransverse region, without contiguous
has shown an n=6-8 to be a target sample size minimum.
osseous connectivity observed between the adjacent transverse
5.4.2 Sample size should be determined with reference to
processes.
theprimaryoutcomeofthestudy,whichistypicallythefusion
3.1.8 radiographic fusion, n—status of radiographic fusion
rate at 8 or 12 weeks. Additionally, it may be necessary to
is based on interpretation of the posteroanterior (P/A) plain
consider the sampling requirements of other analyses in the
filmx-rayimages,withfusionsuccessbasedontheappearance
study; in particular, quantitative endpoints such as morphom-
of contiguous bone from transverse process to transverse
etry.
process (i.e. bridging bone).
NOTE 1—The sample size recommendations refer to the number of
samples expected to be available for analysis.Attrition, or loss of animals
4. Significance and Use
duetosurgicalcomplications,iscommonintherabbitspinalfusionmodel
(especially with autograft harvesting). It may be necessary to plan for
4.1 This guide covers animal implantation methods and
additional animals to replace those lost to attrition. Make sure you report
analysisofbonevoidfillerstodeterminewhetheramaterialor
all animals treated, any unexpected or early deaths, etc.
substanceleadstolumbarintertransverseprocessspinalfusion,
as defined by its ability to cause bone to form in vivo. 5.5 End Points—Each implant group should have an imme-
diate post-operative assessment and end points should be
justified by the resorption profile of the materials; there should
be at least 2 time points less than the maximum assessment
Available from Association for the Advancement of Medical Instrumentation
time (an early and mid-phase) in order to assess any irregu-
(AAMI), 4301 N. Fairfax Dr., Suite 301, Arlington, VA 22203-1633, http://
www.aami.org. larities (unexpected or excessive inflammation, etc.) at the
F3207 − 17
implant and peri-implant site (recommended time periods tionpriortofirstincision.Startfromthecenterandworktothe
representativeintheliteratureare4,8,and12weeks)orlonger edge of the surgical area. Wipe off the solution with a clean,
periods may be warranted and should be justified. sterilegauzepad.Thespinalleveltobefused,mostcommonly
L4–L5 or L5–L6, is then identified by palpation.Aline drawn
6. Recommended Surgical Protocol Methodologies
from the most cranial aspect of one iliac crest to the other, the
intercrestal line, will generally pass between the L6 and L7
6.1 Rabbit Lumbar Intertransverse Process Spinal Fusion
spinous processes (Fig. 1). A second method to verify the
Recommended Surgical Technique:
correct operative level is based on the anatomy of the lum-
6.1.1 Aseptic technique should be employed during the
bosacral spinous processes. Specifically, there is often a much
surgical implantation procedures.
wider interspinous distance at L6–L7 than there is at L5–L6,
6.1.2 Animals should be singly housed in standard cages
L7–S1orbetweenthesacralprocesses(Fig.1),althoughthisis
and fed with rabbit food and water.
not always the case.
NOTE 2—Handling of the animals during the first 14 days post-op
6.1.9.2 Using both techniques of localization, the L4–L5 or
should be avoided unless medically required.
L5–L6 level can be correctly identified in the vast majority of
6.1.3 Pre-operative analgesics: 0.05 mg/ kg buprenorphine
animals.Errorscanoccur,however,becauseofthepresenceof
administered subcutaneously and the application of a fentanyl
osseous anomalies of the lumbosacral vertebrae. A preopera-
patch (25 µg/hr) to the inner ear pinna, or other analgesic
tivedorsoventralradiographisadvisable.Inthepresenceofan
approved by the IACUC. A 25 µg/hr fentanyl patch is an
abnormality,whichaltersthetypicalnumberoflumbarmotion
effective analgesic with duration of up to 72 hours, but may
segments,theanimalshouldbeexcludedorthespinalleveljust
require up to 12 hours after application until blood levels are
cranial to the intercrestal line can be used.
sufficient to provide pain relief. Patches may be placed the
6.1.9.3 It is acceptable to perform the surgery at either
evening prior to surgery or animals dosed with an analgesic
L4–L5 or L5–L6, vertebral levels, although the choice of level
such as butorphanol prior to surgery and several hours after
shouldbeconsistentwithinthestudy.Variabilityinthenumber
surgery to ensure analgesisc coverage while fentanyl blood
of lumbar vertebrae is common in certain strains of NZW
levels rise.
rabbits-somerabbitsexhibiting6lumbarvertebraewhileothers
6.1.4 Anesthetics: Induction and maintenance: 34 mg/ kg
have7.Insuchcases,pre-operativeradiographyisadvisableto
ketamine and 5 mg/ kg xylazine administered intramuscularly.
positivelyidentifythetargetoperativesite.Performingsurgery
Isoflurane should be administered via laryngeal mask within a
at L5–L6 in such populations will result in the operative space
range of 2%-3%, but increasing and decreasing the percentage
adjacent to the lumbosacral space in some rabbits and at one
administered should be based on the individual animal re-
spaceproximalinothercases.Itisunknownifthebiomechani-
sponse. Ophthalmic ointment should be applied to the eyes
calforcesacrosstheinter-lumbarjointsareallequalorifthere
following pre-anesthesia and prior to surgery.
are differences between the joint adjacent to the lumbosacral
6.1.5 Identify each animal with a unique identifier (ear tag,
joint and more proximal joints. Selecting L4–L5 as the
tattoo, etc.). Record the individual animal identification num-
operative site may minimize this potential problem, as L4–L5
bers along with the body weights.
is separated from the lumbosacral joint by at least one motion
6.1.6 Sedate the animal with an IACUC approved medica-
segment.FusionmassesatL4–L5maybeeasiertoharvestthan
tion and maintain general anesthesia with Isoflurane or other
afusionmassatL5–L6incaseswherethereareonly6lumbar
anesthetic approved by the IACUC. The depth of anesthesia
vertebrae.Finally,inmanycaseswherethereareonly6lumbar
should be sufficient to prevent muscular movement. This can
vertebrae, the 6th transverse process may be quite narrow than
be checked by pinching the toe (between the digits) of the
the transverse process of L4 or L5. There may be no signifi-
animal’s hind limbs. If there is a reflex reaction, the animal is
cance to this observation.
not sufficiently anesthetized to continue with the implantation.
Atechnicianshallmonitortheanimal’svitals/parameterswhile
6.1.9.4 SurgicalApproach—Arepresentativedescriptionas-
under anesthesia and record every 15 minutes. suming an L5–L6 fusion site is described hereafter. A dorsal
6.1.7 Place the anesthetized animal in a sternal or ventral midline skin incision measuring approximately 6 cm in length
recimbant position on a clean flat surface in a procedure room is centered over the L5–L6 level.Afull-thickness flap of skin
and shave the dorsum of the animal from the mid thoracic andsubcutaneoustissueisdevelopedandretractedtooneside.
region well below the iliac crests with clippers. Scrub the Approximately 2 cm lateral to the midline at the L5–L6 level,
clipped area with surgical scrub (chlorhexidine scrub or povi- a 4-6 cm longitudinal incision is made through the lumbar
done scrub). Start from the center and work, in a circular fascia. Through this fascial incision, the iliocostalis muscle is
fashion, to the edge of the surgical area. Wipe off the surgical divided exposing the underlying longissimus muscle (Fig. 2).
scrub with 70 % isopropyl alcohol (repeat entire scrub proce- (1)To reach the transverse processes, blunt dissection is
dure at least 3 times). The surgeon will complete final performed along the lateral border of the longissimus muscle.
preparation for aseptic surgery. Exposure of the posterolateral fusion site is accomplished by
6.1.8 Transfer the anesthetized animal to the surgical suite. elevating the iliocostalis muscle in a lateral direction off the
6.1.9 Lumbar Posterolateral Intertransverse spinal fusion is transverse processes and intertransverse ligament. Dorsome-
detailed as follows: dial retraction of the longissimus muscle is required to expose
6.1.9.1 Final sterile prep of the surgical site is completed in the medial aspect of the transverse processes and the pars
the operating room with 2% chlorhexidine or povidone solu- interarticularis. Care should be taken to avoid inadvertent
F3207 − 17
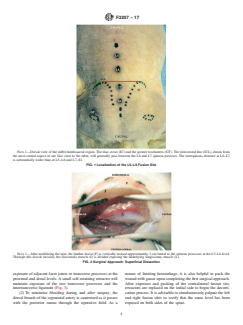
NOTE 1—Dorsal view of the rabbit lumbosacral region. The iliac crests (IC) and the greater trochanters (GT). The intercrestal line (ICL), drawn from
the most cranial aspect of one iliac crest to the other, will generally pass between the L6 and L7 spinous proceses. The interspinous distance at L6–L7
is substantially wider than at L5–L6 and L7–S1.
FIG. 1 Localization of the L5–L6 Fusion Site
NOTE 1—After mobilizing the skin, the lumbar fascia (F) is vertically incised approximately 2 cm lateral to the spinous processes at the L5–L6 level.
Through this fascial incision, the iliocostalis muscle (I) is divided exposing the underlying longissimus muscle (L).
FIG. 2 Surgical Approach: Superficial Dissection
exposure of adjacent facet joints or transverse processes at the means of limiting hemorrhage, it is also helpful to pack the
proximal and distal levels.Asmall self-retaining retractor will woundwithgauzeuponcompletingthefirstsurgicalapproach.
maintain exposure of the two transverse processes and the After exposure and packing of the contralateral fusion site,
intertransverse ligament (Fig. 3). retractors are replaced on the initial side to begin the decorti-
(2)To minimize bleeding during and after surgery, the cationprocess.Itisadvisabletosimultaneouslypalpatetheleft
dorsal branch of the segmental artery is cauterized as it passes and right fusion sites to verify that the same level has been
with the posterior ramus through the operative field. As a exposed on both sides of the spine.
F3207 − 17
NOTE 1—To reach the transverse processes, blunt dissection is performed along the lateral border of the longissi- mus muscle. Exposure of the
posterolateralfusionsiteisaccomplishedbyelevatingtheiliocostailismuscle(I)offthetransverseprocesses(TP)andintertransverseligament(ITL)and
then retracted ventrolaterally. Dor- somedial retraction of the longissimus muscle (L) is required in order to expose the pars interarticularis (P).
FIG. 3 Surgical Approach: Deep Dissection Demonstration to Show the Anatomy
(3)Decortication of the transverse processes is performed medial half to one-third of the transverse processes where
with a motorized burr until punctate bleeding observed.Trans-
decortication was done (Fig. 6).
verse process decortication should be performed as indicated
6.1.9.8 Close the fascial incisions with 3-0 absorbable
by Fig. 4. The extent of decortication has been shown to be a
suture and the skin edges are approximated using absorbable
determining factor in fusion rate, so care should be taken to
3-0 or 4-0 suture, with or without 35W staples.
ensure that decortication does not extend on to the vertebral
6.2 Recovery—Post-operative Care and Analgesics:
body, as this may result in higher than expected fusion rates.
(4)The fifth lumbar root is vulnerable to injury as it exits
6.2.1 Warm blankets and heated mats should be used both
the L5–L6 intervertebral foramen immediately dorsal to the
intra-operatively and post operatively to keep the animal’s
plane of the intertransverse ligament and transverse processes.
body temperature within normal range. For analgesics, 0.05
Thelumbarplexusisalsovulnerabletoinjuryasitscomponent
mg/ kg buprenorphine should be administered subcutaneously
nerves pass just ventral to the intertransverse ligament making
approximately 6 hours after the first dose. The second dose of
it essential to preserve the integrity of this ligament during the
buprenorphine should give the animals an adequate plane of
exposure and decortication process.
analgesia until the fentanyl patch reaches therapeutic levels.
6.1.9.5 Harvest of Iliac Crest Bone—Arthrodesis using
Fentanyl patches should be replaced approximately every 72
autogenous bone from the ilium is often implemented as a
hours until the animal is no longer deemed painful. Pain levels
control group in spinal fusion research using the NZW rabbit
can be monitored based on how well the animals are eating,
model. Working through the same dorsal skin incision, the
posture and ease of movement within the cage. When these 3
cranial and lateral surface of the iliac crest is exposed in a
observations are deemed normal, the animal can then be
subperiosteal plane (Fig. 5, top). This central part of the iliac
considered pain free. If not all of those items are normal, then
wing contains the greatest amount of cancellous bone and can
consideration needs to be given for additional analgesics.
be localized by palpation of the medial iliac spine (Fig. 5,
6.2.2 During the first hour after surgery, pulse and respira-
bottom). The recommended quantity of graft, 2.5 to 3.0 cc per
tory rate are monitored; supplemental fluids are administered
side of the spine, generally requires harvesting a significant
intravenously or subcutaneously as needed.Animals should be
proportion of both ilia. During graft harvest, it is critical to be
monitored until ambulatory, handled carefully, and then re-
gentle when elevating the muscles off the inner cortex when
turned to their cage.
takingthetricorticalilaccrestgraft.Dissectioninthisareacan
traumatize the neurovascular structures that pass through the
6.3 Post-operative Care:
sciaticnotchleadingtoserioushemorrhageand/orsciaticnerve
6.3.1 The general condition of the rabbit should be moni-
palsy. Some amount of palsy (~10%) is an expected conse-
toredtwiceeachdayforthefirst3daysaftersurgery,followed
quence of harvesting the recommended 2.5 to 3.0 cc of graft.
by once a day for the remainder of the study. If used, skin
6.1.9.6 Morselize the corticocancellous autograft bone with
staples are removed 2 weeks after the operation.
arongeurinto<5mmirregularpieces.Makesuretoremoveall
6.3.2 Post-operative anorexia in rabbits may be a serious
soft tissues from the morselized iliac crest bone.
complication and can result in death within 4-6 days. To that
6.1.9.7 Decortication and grafting material should be con-
end, rabbits should be encouraged to eat after surgery. Rabbits
fined to the medial one half of the two transverse processes
may be supplemented with fresh fruits or vegetables (apples,
(i.e., half of the transverse process that is close to the vertebral
carrots, timothy hay cubes) during the acute post-operative
body).As shown in Fig. 4, the graft material should be placed
period if dietary intake of their normal ration is reduced. A
ontopoftheredzonesandfilledinbetweenthetwotransverse
particularly effective supplement is Critical Care by Oxbow
processes.Placeeithertheiliacboneautograftorthetestarticle
Animal Health, which is a highly palatable, high fiber supple-
between the transverse processes in the paraspinal bed, paying
particular attention to placing the graft material along the ment for herbivores that is highly effective in stimulating
F3207 − 17
NOTE 1—(Top) Schematic representation of the decortication area. (Middle)Virgin transverse processes. (Bottom) DecorticatedTransverse processes.
FIG. 4 Decortication of Transverse Processes
appetite in post-operative rabbits. Rabbits are weaned off 6.4.1 General Health—Observations can occur through
supplements as they regain their appetite for their normal close, cage-side observations. If any abnormal clinical signs
ration.
including signs of inflammation and/or infection, hind limb
6.4 Recommended Observations:
F3207 − 17
NOTE 1—(Top) Working through the same skin incision, the cranial and lateral surface of the iliac crest (IC) is exposed by elevating the gluteal
musculature (G) in a subperiosteal plane. (Bottom) The greatest amount of cancellous bone can be harvested from the central part of the iliac wing.
FIG. 5 Harvest of Iliac Crest Bone Graft
paresis, decreased food and water intake or decrease urine and 7. Experimental Endpoints: Techniques to Analyze New
fecal output are observed, inform a staff veterinarian and/or Bone Formation
designated personnel.
7.1 Animal Necropsy:
6.4.2 Early Deaths—If a rabbit should die during the study,
7.1.1 Test samples immediately when possible (i.e. not ever
performanecropsyontheanimal.Iftheanimaldiedonorafter
frozen). It is possible, but not recommended, to store samples
day 14 of the study, harvest and fix the implant sites until a
frozen. If they are frozen then all samples in the study should
decision is made about the potential utility of processing
be treated the same way. If samples have not been tested
tissues for histopathology. If the animal dies before day 14,
immediately after necropsy, storage conditions (e.g., frozen,
perform a necropsy and contact the staff veterinarian and
etc.) should be recorded in the test report. With materials that
appropriate personnel. A terminal weight should always be
do not have a long history of use, it is advisable that select
recorded.
systemic tissues and any gross lesions should also be excised
6.5 Recommended Euthanasia—At the end of the implanta- and preserved using fixation methods appropriate to the ex-
tion period, record the terminal weight of each animal. Record periment’srequirements.Thesetissuesmayincludebutarenot
any abnormalities, with respect to the animals’ health, on the limited to the following: heart, kidneys, liver, lungs, spleen,
macroscopic observations section of the animal record. Eutha- pancreas, axillary lymph nodes, mesenteric lymph nodes,
nize the animals using an IACUC-approved method consistent periaorticlymphnodes,andasampleoflocalparaspinaltissue
with AVMA Guidelines on euthanasia. sample overlying the fusion site. Following careful posterior
F3207 − 17
NOTE1—(Top)Weightmaybedeterminedfollowingremovalofanyexcessblood/fluidswithgauze.(Bottom)Volumemaybedeterminedbypacking
into an open bore syringe under light compression.
FIG. 6 Iliac Crest Autograft is Morselized to Approximately <5 mm in Size
NOTE 3—It is recommended to complete all radiographic tests prior to
dissectionoftheparaspinaltissues,thespinalcolumnfromthe
biomechanical testing.
mid-thoracic region through the pelvis should be excised en
NOTE 4—It is recommended that specimens used for destructive
bloc. The operative lumbar spine and operative site should be
biomechanical testing NOT be used for histological or histomorphometry
examined for gross evidence of infection or general inflamma-
testing as destructive biomechanical testing performed on the specimens
tion.Afull gross necropsy should also include examination of
may introduce artifacts in further tests.
external surfaces/orifices of the body, the cranial, thoracic,
abdominal cavities, and all viscera.
8. Techniques for Assessing Fusion
7.1.2 The following list suggests a possible order of proce-
durestobeperformedpost-necropsy.Thetestreportshouldlist
8.1 Radiographic Analysis:
and justify the order of the procedures done during the
8.1.1 Obtaining Radiographs of the Operated Site—Obtain
experiment. Careful attention should be paid to this order so
dorsoventral plane film radiographs of all operated segments
that experimental results are not compromised by previously
immediately post-op and at the final study time point after
performed procedures. It is recommended that the researchers
animal sacrifice. It is recommended that the radiographic
note why the results of all sample testing is valid and not
equipment set-up be consistent for each assessment. Radio-
compromised by previous testing done on the samples.
graphs should be clear with minimal artifacts and should
7.1.2.1 Systemic and reticuloendothelial tissues harvest and
enablevisualizationoftheentireoperatedsegmentandatleast
lumbopelvic spine resection
one (1) caudal and cranial non-operated level. Animal desig-
7.1.2.2 Plainfilmradiographyofthelumbarspine(seeNote
nations and orientation using standard markers should be
3)
employed.
7.1.2.3 Micro CT (see Note 3)
8.1.2 Personnel—The presence of radiographic fusion will
7.1.2.4 Manual palpation
be scored by three (3) independent observers.These personnel
7.1.2.5 Biomechanical testing (see Note 4)
7.1.2.6 Histology and histomorphometry (see Note 4) mustbetrainedinthetechniqueandbeblindedtothetreatment
F3207 − 17
FIG. 7 Approximate Graft Placement Over the Paraspinal Bed and Adjacent Transverse Processes
radiography may include fusion mass size and/or maturity (presence of
group. Scoring is aided by knowledge of the post-op radio-
more organized trabecular structures and a neocortex) and resorption of
graphicappearanceoftheoperatedsiteandbonegraftmaterial.
radiopaque bone graft materials. However, alternative methods (e.g.
When possible, the post-op film should be made available
micro-CT or histomorphometry) may quantitatively evaluate these char-
during scoring.
acteristics with sensitivity beyond plain film assessment.
8.1.3 Radiographic Scoring:
NOTE 6—Assessing radiographic fusion of radiopaque bone graft
8.1.3.1 Scoring of radiographs for fusion should be con-
materials can increase false-positive fusion rate reporting. If the observer
ducted at pre-defined analysis time points. Each side (le
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...