ASTM F2528-06(2014)
(Test Method)Standard Test Methods for Enteral Feeding Devices with a Retention Balloon
Standard Test Methods for Enteral Feeding Devices with a Retention Balloon
ABSTRACT
These test methods cover the establishment of performance requirements for the utilization of a single-use, enteral feeding device with a retention balloon, used by medical professionals for providing a means of nutrition and/or administration of medication to patients by means of natural orifice (nasal, oral, transluminal) and or a surgically created stoma. The product is manufactured in various sizes and materials such as silicone, urethane, and various polymers (as well as combinations of these) and is provided nonsterile for sterilization and sterile for single use only. The following test methods are: Flow rate through feeding lumen test method which covers the determination of flow rates through the drainage lumen of the enteral feeding device with retention balloon, balloon burst volume test method which covers the determination of balloon integrity of enteral feeding devices with retention balloon, balloon volume maintenance test method which is applicable enteral feeding devices with retention balloon to test the integrity of the inflation system to maintain balloon volume, balloon concentricity test method which is applicable enteral feeding devices with retention balloon to test the concentricy of the balloon, balloon size and shaft size test method which evaluates the retention balloon shaft size, balloon integrity test method which evaluates the integrtity of the retention balloon of the enteral feeding device, and balloon integrity in simulated gastric fluid test method which assesses the ability of the retention balloon to withstand gastric acidity levels without rupture, therefore, maintaining its functional purpose of retention.
SCOPE
1.1 These test methods cover the establishment of performance requirements for the utilization of a single-use, enteral feeding device with a retention balloon, used by medical professionals for providing a means of nutrition and/or administration of medication to patients by means of natural orifice (nasal, oral, transluminal) and or a surgically created stoma. The product is manufactured in various sizes and materials such as silicone, urethane, and various polymers (as well as combinations of these) and is provided nonsterile for sterilization and sterile for single use only. Rationale for these test methods can be found in Appendix X1.
1.2 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and to determine the applicability of regulatory limitations prior to use.
General Information
Buy Standard
Standards Content (Sample)
NOTICE: This standard has either been superseded and replaced by a new version or withdrawn.
Contact ASTM International (www.astm.org) for the latest information
Designation: F2528 − 06 (Reapproved 2014)
Standard Test Methods for
Enteral Feeding Devices with a Retention Balloon
This standard is issued under the fixed designation F2528; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 3.1.3 enteral feeding device with retention balloon, n—a
two-way medical device intended to provide a means of
1.1 These test methods cover the establishment of perfor-
nutrition or administration of medication, or both, to patients
mance requirements for the utilization of a single-use, enteral
by means of natural orifice (nasal, oral, transluminal) or a
feeding device with a retention balloon, used by medical
surgically created stoma, or both, consisting of a drainage
professionals for providing a means of nutrition and/or admin-
lumen and inflation lumen (see Fig. 1). Common balloon
istration of medication to patients by means of natural orifice
3 3 3
inflation sizes are 5 cm , 15 cm , and 20 cm .
(nasal, oral, transluminal) and or a surgically created stoma.
3.1.4 French size (Fr), n—a scale used for denoting the size
The product is manufactured in various sizes and materials
of catheters and other tubular instruments. The French size
such as silicone, urethane, and various polymers (as well as
value is three times the outer diameter of the tube as measured
combinations of these) and is provided nonsterile for steriliza-
in millimetres. For example, a diameter of 18 Fr indicates a
tion and sterile for single use only. Rationale for these test
diameter of 6 mm.
methods can be found in Appendix X1.
3.1.5 inflation volume, n—volume of liquid used to inflate
1.2 This standard does not purport to address all of the
the retention balloon of the enteral feeding device for proposed
safety concerns, if any, associated with its use. It is the
testing in this standard.
responsibility of the user of this standard to establish appro-
priate safety and health practices and to determine the
3.1.6 rated volume, n—stated volume of inflation of the
applicability of regulatory limitations prior to use.
retention balloon of the enteral feeding device in the manufac-
turers labeling and instructions for use.
2. Referenced Documents
3.1.7 simulated gastric fluid, n—a solution consisting of
2.1 ASTM Standards:
hydrochloric acid, salt and pepsin with a pH of approximately
F623 Performance Specification for Foley Catheter
1.2, per USP standard recipe.
2.2 Other Standard:
3.1.8 sterility, n—the state of being free from viable micro-
Simulated Gastric Fluid, USP Official Compendia of Stan-
organisms.
dards
4. Specimen Preparation
3. Terminology
4.1 All test specimens for test methods listed below shall
consist of the manufacturers’ new, finished, untested, unsteril-
3.1 Definitions:
ized product. At the minimum, statistically valid samples of the
3.1.1 balloon integrity (resistance to rupture), n—volume of
smallest and the largest diameter of enteral feeding devices
liquid that corresponds with balloon failure, or bursting.
shall be tested.
3.1.2 distal, n—refers to the balloon end of the enteral
feeding device
5. Test Methods
PROCEDURE A: FLOW RATE THROUGH FEEDING
LUMEN
These test methods are under the jurisdiction of ASTM Committee F04 on
Medical and Surgical Materials and Devices and are the direct responsibility of
5.1 Scope—This test method covers the determination of
Subcommittee F04.35 on GI Applications.
flow rates through the drainage lumen of the enteral feeding
Current edition approved June 1, 2014. Published June 2014. Originally
approved in 2006. Last previous edition approved in 2006 as F2528 – 06. DOI:
device with retention balloon.
10.1520/F2528-06R14.
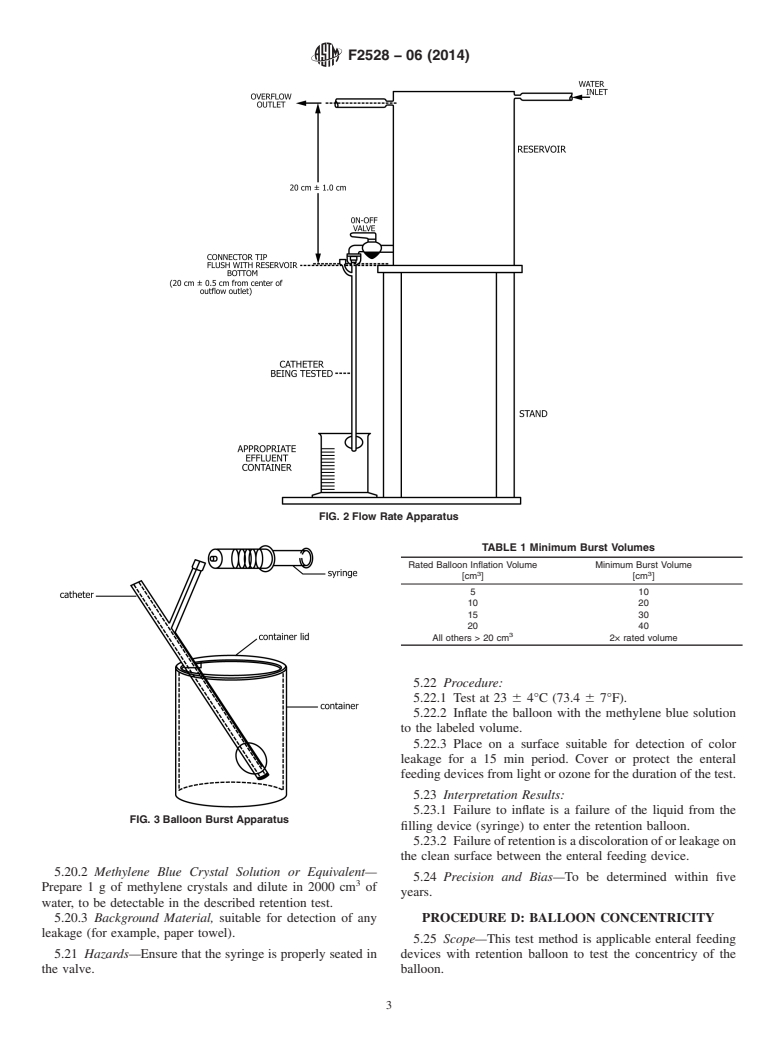
5.2 Summary of Test Method—The apparatus is set up as
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
shown in Fig. 2. The flow rate is adjusted through the water
Standards volume information, refer to the standard’s Document Summary page on
inlet to a rate sufficient to maintain flow through the overflow
the ASTM website.
3 outlet while each enteral feeding device is tested. A head
USP Official Compendia of Standards, available from U.S. Pharmacopeia
(USP), 12601 Twinbrook Pkwy., Rockville, MD 20852. pressure of 20 6 1.0 cm of water (196 6 10 kPa) above the
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2528 − 06 (2014)
with water. The balloon is then inflated with water until
rupture, which enables the volume at which the balloon bursts
to be observed.
5.11 Significance and Use—The balloon burst volume is
measured to quantify the resistance of rupture of the enteral
feeding device with retention balloon member.
5.12 Apparatus—The testing apparatus is set up as shown in
FIG. 1 Enteral Feeding Device with Retention Balloon
Fig. 3.
5.12.1 System Reservoir.
5.12.2 Syringe.
tank bottom shall be maintained throughout the test to approxi-
5.12.3 Water.
mate actual physiological conditions. The overflow outlet
should not be covered by water.
5.13 Hazards—Water should be emptied from system res-
ervoir through purge valve when fill marked is reached.
5.3 Significance and Use—The flow rate is measured in
reverse flow for ease in testing, since differences in the flow
5.14 Procedure:
rate as a result of flow direction are theoretically insignificant.
5.14.1 Test at 23 6 4°C (73.4 6 7°F).
5.4 Apparatus:
5.14.2 Insert uninflated enteral feeding device into test
5.4.1 Water Reservoir, capable of maintaining 20 6 1.0 cm
orifice in system reservoir per Fig. 3.
(7.9 6 0.4 in.) of water (196 6 10 kPa) above the tip of the
5.14.3 Close orifice so that it is positioned proximal to the
enteral feeding device connection throughout the test as shown
enteral feeding device with retention balloon member. The
in Fig. 2. (See Performance Specification F623.)
device is not to be immersed in water within the reservoir per
5.4.2 Graduated Cylinder, calibrated for suitable measure-
Fig. 3.
ment of the effluent.
5.14.4 Fill syringe with amount of water greater than that
5.4.3 Syringe, with appropriate tip for inflation of enteral
listed in Table 1 for the desired French size. Attach tip of
feeding device balloon.
syringe to enteral feeding device inflation valve.
5.5 Hazards: 5.14.5 Inflate retention balloon at 1 cm /sec with water until
5.5.1 Overflow should not be covered. Head pressure must
balloon bursts. Record amount of water injected into balloon at
be kept constant; water should always be exiting through the time of burst.
overflow outlet.
5.15 Interpretation of Results—Burst volumes for enteral
5.5.2 Establish equilibrium before testing.
feeding devices tested must meet or exceed those listed in
5.5.3 Flow rates through all fittings must exceed that of the
Table 1.
enteral feeding device being tested.
5.16 Precision and Bias—To be determined within five
5.6 Procedure:
years.
5.6.1 Test at 23 6 4°C (73.4 6 7°F).
5.6.2 Inflate the retention balloon of the test specimen with
PROCEDURE C: BALLOON VOLUME
water to labeled volume.
MAINTENANCE
5.6.3 Connect the enteral feeding device to enteral feeding
5.17 Scope—This test method is applicable enteral feeding
device connector and open the stopcock. The tip of the enteral
devices with retention balloon to test the integrity of the
feeding device connection at the junction of enteral feeding
inflation system to maintain balloon volume.
device on-off valve should be level with the bottom of the tank
61 cm and it should deliver fluid at 20 6 1 cm (196 6 10 kPa)
5.18 Summary of Test Method—The balloon retention de-
head pressure at that junction.
vice of the enteral feeding device is inflated with a test liquid.
5.6.4 Establish flow equilibrium before taking test measure-
This test liquid contains a colorant which enables a leak of this
ments.
fluid to be observed. If no leak is observed, the integrity of the
5.6.5 Record the amount of fluid through the device feeding
inflation system is upheld, therefore maintaining the balloon
lumen in 30 seconds.
volume.
5.7 Interpretation of Results—Flow rates for enteral feeding
5.19 Significance and Use—This test method establishes a
devices tested must meet or exceed 9 cm /min.
standard test method for determining the functional integrity of
the inflation system of the enteral feeding device with retention
5.8 Precision and Bias—To be determined within five years.
balloon enteral feeding device by observing the consistancy of
PROCEDURE B: BALLOON BURST VOLUME
volume of the balloon after it is filled with test liquid.
Additionally, since it is the function of the inflated balloon to
5.9 Scope—This test method covers the determination of
retain the feeding device in position, the ballon must inflate,
balloon integrity of enteral feeding devices with retention
retain inflation volume, and release that volume when required.
balloon.
5.20 Apparatus:
5.10 Summary of Test Method—The enteral feeding device
with retention balloon is submerged in a small container filled 5.20.1 Syringe.
F2528 − 06 (2014)
FIG. 2 Flow Rate Apparatus
TABLE 1 Minimum Burst Volumes
Rated Balloon Inflation Volume Minimum Burst Volume
3 3
[cm ] [cm ]
5 10
10 20
15 30
20 40
All others > 20 cm 2× rated volume
5.22 Procedure:
5.22.1 Test at 23 6 4°C (73.4 6 7°F).
5.22.2 Inflate the balloon with the methylene blue solution
to the labeled volume.
5.22.3 Place on a surface suitable for detection of color
leakage for a 15 min period. Cover or protect the enteral
feeding devices from light or ozone for the duration of the test.
5.23 Interpretation Results:
5.23.1 Failure to inflate is a failure of the liquid from the
FIG. 3 Balloon Burst Apparatus
filling device (syringe) to enter the retention balloon.
5.23.2 Failure of retention is a discoloration of or leakage on
the clean surface between the enteral feeding device.
5.20.2 Methylene Blue Crystal Solution or Equivalent—
5.24 Precision and Bias—To be determined within five
Prepare 1 g of methylene crystals and dilute in 2000 cm of
years.
water, to be detectable in the described retention test.
5.20.3 Background Material, suitable for detection of any PROCEDURE D: BALLOON CONCENTRICITY
leakage (for example, paper towel).
5.25 Scope—This test method is applicable enteral feeding
5.21 Hazards—Ensure that the syringe is properly seated in devices with retention balloon to test the concentricy of the
the valve. balloon.
F2528 − 06 (2014)
TABLE 2 Concentricity Ratios
Rated Balloon Inflation Volume Maximum Concentricity
[cm ] Ratio
5 2:1
10 2:1
15 2:1
20 2:1
All others > 20 cm 2:1
5.37 Hazards:
FIG. 4 Concentricity Test Apparatus
5.37.1 No lubrication or undue force shall be applied to the
enteral feeding device.
5.26 Summary of Test Method—The retention balloon of the 5.37.2 The edges of each hole should be smooth to avoid
enteral feeding device is inflated with water, and with the use
interference to the passage of the test enteral feeding device.
of a gauge, evaluted for concentricty.
5.38 Procedure:
5.27 Significance and Use—This test is designed to quantify
5.38.1 Test at 23 6 4°C (73.4 6 7°F).
balloon concentricity and the overall shape geometry of the 5.38.2 Per Fig. 5, without lubrication, push the proximal end
balloon. It is the purpose of the balloon to retain the feeding
of the uninflated enteral feeding device through the various
device in position during use, therefore, the balloon must be of holes of the French size gauge, advancing it to the uninflated
a functional uniformity that will not allow the enteral feeding
balloon.
device to move from its desired position. 5.38.3 Uninflated balloon should fit in appropriate French
size gauge hole snugly without undue insertion force. Label
5.28 Apparatus—The testing apparatus is set up as shown in
each test unit and the measured French size. Remove device
Fig. 4.
3 3 3 from gauge.
5.28.1 Syringes—1 cm , 5 cm , and 60 cm .
5.28.2 Water.
5.39 Interpretation of Results—The balloon section may
wrinkle but shall not tear or distort, and the enteral feeding
5.29 Hazards—Not applicable.
device shaft or tip may offer resistance but if distortion or
5.30 Procedure:
stretching occurs it is considered a failure.
5.30.1 Test at 23 6 4°C (73.4 6 7°F).
5.40 Precision and Bias—To be determined within five
5.30.2 Fill syringe with volume of water equal to balloon
years.
rating.
5.30.3 Attach syringe to enteral feeding device inflation
PROCEDURE F: BALLOON INTEGRITY
valve and inflate with water.
5.41 Scope—This test method is to evaluate the integrtity of
5.30.4 Per Fig. 4, use snap gauge and measure the two sides
the retention balloon of the enteral feeding device.
of the balloon that visually appear to have the least symmetry.
Measurement should be taken 180° from each other.
5.42 Summary of Test Method—The retention balloons are
5.30.5 Divide larger measurement by smaller measurement
inflated with water and submerged in water at 37.8 6 3°C (100
and quotient equals Concentricity Ratio. Tabulate all results.
6 5°F) for seven days. The retention balloons are evaluated to
determine if they hold their integrity and do not rupture.
5.31 Interpretation of Results—Balloon concentricity ratio
must not exceed those established in Table 2.
5.43 Significance and Use—This test method is designed to
subject the retention balloons to the inflation volume they
5.32 Precision and Bias—To be determined within five
would be subjected to during use in the field, in order to
years.
determine the integrity of the balloon. It is the purpose of the
PROCEDURE E: BALLOON SIZE AND SHAFT SIZE
balloon to retain the feeding device in position during use,
therefore, the performance of the retention balloon must be
5.33 Scope—This test method is to evaluate the retention
maintained and the balloon must not rupture.
balloon shaft size.
5.44 Apparatus—The testing apparatus is as shown in Fig.
5.34 Summary of Test Method—Using a French size gauge,
6.
the distal balloon tip is measured to determine the size of the
5.44.1 Corrosion Resistant Tanks—The tanks should con-
retention balloon over the shaft.
tain no exposed iron, copper, or brass elements and have a
5.35 Significance and Use—The overall outside diameter of
removable mixing elements. The tanks should also have
the enteral feeding device during passage should conform to
sufficient covers/lids in order to prevent evaporation of solu-
the requi
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2528 − 06 F2528 − 06 (Reapproved 2014)
Standard Test Methods for
Enteral Feeding Devices with a Retention Balloon
This standard is issued under the fixed designation F2528; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 These test methods cover the establishment of performance requirements for the utilization of a single-use, enteral feeding
device with a retention balloon, used by medical professionals for providing a means of nutrition and/or administration of
medication to patients by means of natural orifice (nasal, oral, transluminal) and or a surgically created stoma. The product is
manufactured in various sizes and materials such as silicone, urethane, and various polymers (as well as combinations of these)
and is provided nonsterile for sterilization and sterile for single use only. Rationale for these test methods can be found in Appendix
X1.
1.2 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and to determine the applicability of regulatory
limitations prior to use.
2. Referenced Documents
2.1 ASTM Standards:
F623 Performance Specification for Foley Catheter
2.2 Other Standard:
Simulated Gastric Fluid, USP Official Compendia of Standards
3. Terminology
3.1 Definitions:
3.1.1 balloon integrity (resistance to rupture)—rupture), n—volume of liquid that corresponds with balloon failure, or bursting.
3.1.2 distal—distal, n—refers to the balloon end of the enteral feeding device
3.1.3 enteral feeding device with retention balloon—balloon, n—a two-way medical device intended to provide a means of
nutrition or administration of medication, or both, to patients by means of natural orifice (nasal, oral, transluminal) or a surgically
created stoma, or both, consisting of a drainage lumen and inflation lumen (see Fig. 1). Common balloon inflation sizes are 5 cm ,
3 3
15 cm , and 20 cm .
These test methods are under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and isare the direct responsibility of Subcommittee
F04.35 on GI Applications.
Current edition approved June 1, 2006June 1, 2014. Published June 2006June 2014. Originally approved in 2006. Last previous edition approved in 2006 as F2528 – 06.
DOI: 10.1520/F2528-06.10.1520/F2528-06R14.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
USP Official Compendia of Standards, available from U.S. Pharmacopeia (USP), 12601 Twinbrook Pkwy., Rockville, MD 20852.
FIG. 1 Enteral Feeding Device with Retention Balloon
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2528 − 06 (2014)
FIG. 2 Flow Rate Apparatus
3.1.4 French size (Fr)—(Fr), n—a scale used for denoting the size of catheters and other tubular instruments. The French size
value is three times the outer diameter of the tube as measured in millimetres. For example, a diameter of 18 Fr indicates a diameter
of 6 mm.
3.1.5 inflation volume—volume, n—volume of liquid used to inflate the retention balloon of the enteral feeding device for
proposed testing in this standard.
3.1.6 rated volume—volume, n—stated volume of inflation of the retention balloon of the enteral feeding device in the
manufacturers labeling and instructions for use.
3.1.7 simulated gastric fluid—fluid, n—a solution consisting of hydrochloric acid, salt and pepsin with a pH of approximately
1.2, per USP standard recipe.
3.1.8 sterility—sterility, n—the state of being free from viable micro-organisms.
4. Specimen Preparation
4.1 All test specimens for test methods listed below shall consist of the manufacturers’ new, finished, untested, unsterilized
product. At the minimum, statistically valid samples of the smallest and the largest diameter of enteral feeding devices shall be
tested.
5. Test Methods
PROCEDURE A: FLOW RATE THROUGH FEEDING LUMEN
5.1 Scope—This test method covers the determination of flow rates through the drainage lumen of the enteral feeding device
with retention balloon.
5.2 Summary of Test Method—The apparatus is set up as shown in Fig. 2. The flow rate is adjusted through the water inlet to
a rate sufficient to maintain flow through the overflow outlet while each enteral feeding device is tested. A head pressure of 20 6
1.0 cm of water (196 6 10 kPa) above the tank bottom shall be maintained throughout the test to approximate actual physiological
conditions. The overflow outlet should not be covered by water.
F2528 − 06 (2014)
FIG. 3 Balloon Burst Apparatus
5.3 Significance and Use—The flow rate is measured in reverse flow for ease in testing, since differences in the flow rate as a
result of flow direction are theoretically insignificant.
5.4 Apparatus:
5.4.1 Water Reservoir, capable of maintaining 20 6 1.0 cm (7.9 6 0.4 in.) of water (196 6 10 kPa) above the tip of the enteral
feeding device connection throughout the test as shown in Fig. 2. (See Performance Specification F623.)
5.4.2 Graduated Cylinder, calibrated for suitable measurement of the effluent.
5.4.3 Syringe, with appropriate tip for inflation of enteral feeding device balloon.
5.5 Hazards:
5.5.1 Overflow should not be covered. Head pressure must be kept constant; water should always be exiting through the
overflow outlet.
5.5.2 Establish equilibrium before testing.
5.5.3 Flow rates through all fittings must exceed that of the enteral feeding device being tested.
5.6 Procedure:
5.6.1 Test at 23 6 4°C (73.4 6 7°F).
5.6.2 Inflate the retention balloon of the test specimen with water to labeled volume.
5.6.3 Connect the enteral feeding device to enteral feeding device connector and open the stopcock. The tip of the enteral
feeding device connection at the junction of enteral feeding device on-off valve should be level with the bottom of the tank 61
cm and it should deliver fluid at 20 6 1 cm (196 6 10 kPa) head pressure at that junction.
5.6.4 Establish flow equilibrium before taking test measurements.
5.6.5 Record the amount of fluid through the device feeding lumen in 30 seconds.
5.7 Interpretation of Results—Flow rates for enteral feeding devices tested must meet or exceed 9 cm /min.
5.8 Precision and Bias—To be determined within five years.
PROCEDURE B: BALLOON BURST VOLUME
5.9 Scope—This test method covers the determination of balloon integrity of enteral feeding devices with retention balloon.
5.10 Summary of Test Method—The enteral feeding device with retention balloon is submerged in a small container filled with
water. The balloon is then inflated with water until rupture, which enables the volume at which the balloon bursts to be observed.
5.11 Significance and Use—The balloon burst volume is measured to quantify the resistance of rupture of the enteral feeding
device with retention balloon member.
5.12 Apparatus—The testing apparatus is set up as shown in Fig. 3.
5.12.1 System Reservoir.
5.12.2 Syringe.
5.12.3 Water.
5.13 Hazards—Water should be emptied from system reservoir through purge valve when fill marked is reached.
F2528 − 06 (2014)
TABLE 1 Minimum Burst Volumes
Rated Balloon Inflation Volume Minimum Burst Volume
3 3
[cm ] [cm ]
5 10
10 20
15 30
20 40
All others > 20 cm 2× rated volume
5.14 Procedure:
5.14.1 Test at 23 6 4°C (73.4 6 7°F).
5.14.2 Insert uninflated enteral feeding device into test orifice in system reservoir per Fig. 3.
5.14.3 Close orifice so that it is positioned proximal to the enteral feeding device with retention balloon member. The device
is not to be immersed in water within the reservoir per Fig. 3.
5.14.4 Fill syringe with amount of water greater than that listed in Table 1 for the desired French size. Attach tip of syringe to
enteral feeding device inflation valve.
5.14.5 Inflate retention balloon at 1 cm /sec with water until balloon bursts. Record amount of water injected into balloon at
time of burst.
5.15 Interpretation of Results—Burst volumes for enteral feeding devices tested must meet or exceed those listed in Table 1.
5.16 Precision and Bias—To be determined within five years.
PROCEDURE C: BALLOON VOLUME MAINTENANCE
5.17 Scope—This test method is applicable enteral feeding devices with retention balloon to test the integrity of the inflation
system to maintain balloon volume.
5.18 Summary of Test Method—The balloon retention device of the enteral feeding device is inflated with a test liquid. This test
liquid contains a colorant which enables a leak of this fluid to be observed. If no leak is observed, the integrity of the inflation
system is upheld, therefore maintaining the balloon volume.
5.19 Significance and Use—This test method establishes a standard test method for determining the functional integrity of the
inflation system of the enteral feeding device with retention balloon enteral feeding device by observing the consistancy of volume
of the balloon after it is filled with test liquid. Additionally, since it is the function of the inflated balloon to retain the feeding device
in position, the ballon must inflate, retain inflation volume, and release that volume when required.
5.20 Apparatus:
5.20.1 Syringe.
5.20.2 Methylene Blue Crystal Solution or Equivalent—Prepare 1 g of methylene crystals and dilute in 2000 cm of water, to
be detectable in the described retention test.
5.20.3 Background Material, suitable for detection of any leakage (for example, paper towel).
5.21 Hazards—Ensure that the syringe is properly seated in the valve.
5.22 Procedure:
5.22.1 Test at 23 6 4°C (73.4 6 7°F).
5.22.2 Inflate the balloon with the methylene blue solution to the labeled volume.
5.22.3 Place on a surface suitable for detection of color leakage for a 15 min period. Cover or protect the enteral feeding devices
from light or ozone for the duration of the test.
5.23 Interpretation Results:
5.23.1 Failure to inflate is a failure of the liquid from the filling device (syringe) to enter the retention balloon.
5.23.2 Failure of retention is a discoloration of or leakage on the clean surface between the enteral feeding device.
5.24 Precision and Bias—To be determined within five years.
PROCEDURE D: BALLOON CONCENTRICITY
5.25 Scope—This test method is applicable enteral feeding devices with retention balloon to test the concentricy of the balloon.
5.26 Summary of Test Method—The retention balloon of the enteral feeding device is inflated with water, and with the use of
a gauge, evaluted for concentricty.
5.27 Significance and Use—This test is designed to quantify balloon concentricity and the overall shape geometry of the
balloon. It is the purpose of the balloon to retain the feeding device in position during use, therefore, the balloon must be of a
functional uniformity that will not allow the enteral feeding device to move from its desired position.
F2528 − 06 (2014)
FIG. 4 Concentricity Test Apparatus
5.28 Apparatus—The testing apparatus is set up as shown in Fig. 4.
3 3 3
5.28.1 Syringes—1 cm , 5 cm , and 60 cm .
5.28.2 Water.
5.29 Hazards—Not applicable.
5.30 Procedure:
5.30.1 Test at 23 6 4°C (73.4 6 7°F).
5.30.2 Fill syringe with volume of water equal to balloon rating.
5.30.3 Attach syringe to enteral feeding device inflation valve and inflate with water.
5.30.4 Per Fig. 4, use snap gauge and measure the two sides of the balloon that visually appear to have the least symmetry.
Measurement should be taken 180° from each other.
5.30.5 Divide larger measurement by smaller measurement and quotient equals Concentricity Ratio. Tabulate all results.
5.31 Interpretation of Results—Balloon concentricity ratio must not exceed those established in Table 2.
5.32 Precision and Bias—To be determined within five years.
PROCEDURE E: BALLOON SIZE AND SHAFT SIZE
5.33 Scope—This test method is to evaluate the retention balloon shaft size.
5.34 Summary of Test Method—Using a French size gauge, the distal balloon tip is measured to determine the size of the
retention balloon over the shaft.
5.35 Significance and Use—The overall outside diameter of the enteral feeding device during passage should conform to the
required clinical orifice size.
5.36 Apparatus—The testing apparatus is as shown in Fig. 5.
5.36.1 French Size Calibration Gauge, tolerance of 60.13 mm (60.005 in.).
5.36.2 Metric Scale Rule.
5.37 Hazards:
5.37.1 No lubrication or undue force shall be applied to the enteral feeding device.
5.37.2 The edges of each hole should be smooth to avoid interference to the passage of the test enteral feeding device.
5.38 Procedure:
5.38.1 Test at 23 6 4°C (73.4 6 7°F).
5.38.2 Per Fig. 5, without lubrication, push the proximal end of the uninflated enteral feeding device through the various holes
of the French size gauge, advancing it to the uninflated balloon.
5.38.3 Uninflated balloon should fit in appropriate French size gauge hole snugly without undue insertion force. Label each test
unit and the measured French size. Remove device from gauge.
5.39 Interpretation of Results—The balloon section may wrinkle but shall not tear or distort, and the enteral feeding device shaft
or tip may offer resistance but if distortion or stretching occurs it is considered a failure.
5.40 Precision and Bias—To be determined within five years.
PROCEDURE F: BALLOON INTEGRITY
5.41 Scope—This test method is to evaluate the integrtity of the retention balloon of the enteral feeding device.
5.42 Summary of Test Method—The retention balloons are inflated with water and submerged in water at 37.8 6 3°C (100 6
5°F) for seven days. The retention balloons are evaluated to determine if they hold their integrity and do not rupture.
F2528 − 06 (2014)
TABLE 2 Concentricity Ratios
Rated Balloon Inflation Volume Maximum Concentricity
[cm ] Ratio
5 2:1
10 2:1
15 2:1
20 2:1
All others > 20 cm 2:1
5.43 Significance and Use—This test method is designed to subject th
...









Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.