ASTM D7597-16(2017)
(Test Method)Standard Test Method for Determination of Diisopropyl Methylphosphonate, Ethyl Hydrogen Dimethylamidophosphate, Ethyl Methylphosphonic Acid, Isopropyl Methylphosphonic Acid, Methylphosphonic Acid and Pinacolyl Methylphosphonic Acid in Water by Liquid Chromatography/Tandem Mass Spectrometry
Standard Test Method for Determination of Diisopropyl Methylphosphonate, Ethyl Hydrogen Dimethylamidophosphate, Ethyl Methylphosphonic Acid, Isopropyl Methylphosphonic Acid, Methylphosphonic Acid and Pinacolyl Methylphosphonic Acid in Water by Liquid Chromatography/Tandem Mass Spectrometry
SIGNIFICANCE AND USE
5.1 Organophosphate pesticides affect the nervous system by disrupting the enzyme that regulates acetylcholine, a neurotransmitter. They were developed during the early 19th century, but their effects on insects, which are similar to their effects on humans, were discovered in 1932. Some are poisonous and were used as chemical weapon agents. Organophosphate pesticides are usually not persistent in the environment.4,5
5.2 This test method is for the analysis of selected organophosphorous-based chemical weapon agent degradation products from Sarin (GB), Soman (GD), Tabun (GA) and VX. This test method has been investigated for use with reagent and surface water.
SCOPE
1.1 This procedure covers the determination of diisopropyl methylphosphonate (DIMP), ethyl hydrogen dimethylamidophosphate (EHDMAP), ethyl methylphosphonic acid (EMPA), isopropyl methylphosphonic acid (IMPA), methylphosphonic acid (MPA) and pinacolyl methylphosphonic acid (PMPA) (referred to collectively as organophosphonates in this test method) in surface water by direct injection using liquid chromatography (LC) and detected with tandem mass spectrometry (MS/MS) using electrospray ionization (ESI). These analytes are qualitatively and quantitatively determined by this test method. This test method adheres to single reaction monitoring (SRM) mass spectrometry.
1.2 This test method has been developed by U.S. EPA Region 5 Chicago Regional Laboratory (CRL).
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 The detection verification level (DVL) and reporting range for the organophosphonates are listed in Table 1.
TABLE 1 Detection Verification Level and Reporting Range
Analyte
ESI Mode
DVL (μg/L)
Reporting Range (μg/L)
Diisopropyl methylphosphonate
Positive
1
5–150
Ethyl hydrogen dimethylamidophosphate
Negative
0.25
5–150
Ethyl hydrogen dimethylamidophosphate
Positive
0.25
5–150
Ethyl methylphosphonic acid
Negative
5
50–1500
Ethyl methylphosphonic acid
Positive
5
50–1500
Isopropyl methylphosphonic acid
Negative
10
50–1500
Isopropyl Methylphosphonic acid
Positive
5
50–1500
Methylphosphonic acid
Negative
20
100–1500
Methylphosphonic acid
Positive
10
50–1500
Pinacolyl methylphosphonic acid
Negative
5
50–1500
Pinacolyl methylphosphonic acid
Positive
5
50–1500
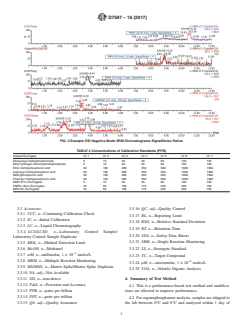
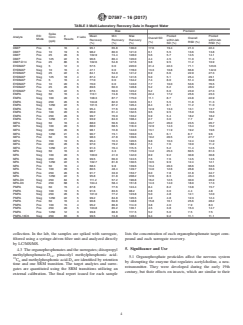
1.4.1 The DVL is required to be at a concentration at least three times below the reporting limit (RL) and have a signal/noise ratio greater than 3:1. Fig. 1 displays the signal/noise ratios at the DVLs for the organophosphonates in the ESI positive mode and Fig. 2 in the ESI negative mode.
FIG. 1 Example ESI Positive Mode SRM Chromatograms Signal/Noise Ratios
FIG. 2 Example ESI Negative Mode SRM Chromatograms Signal/Noise Ratios
1.4.2 The reporting limit is the concentration of the Level 1 calibration standard as shown in Table 2 for the organophosphonates except for MPA in the ESI negative mode which is at Level 2 due to not meeting the DVL criteria at the lower concentration level. The DVL for MPA in the ESI negative mode is at 20 μg/L, which forces a raised reporting limit. However, the multi-laboratory validation required a spike of all target analytes at Level 1 concentrations. The mean recovery for MPA in the ESI negative mode at this level was 98.7 % as shown in Table 3. If your instrument’s sensitivity can meet the requirements in this test method, MPA may have a 50 μg/L reporting limit.
TABLE 2 Concentrations of Calibration Standards (PPB)
Analyte/Surrogate
LV 1
LV 2
LV 3
LV 4
LV 5
LV 6
LV 7
Diisopropyl methylphosphonate
5
10
20
35
50
100
150
Ethyl hydrogen dimethylamidophosphate
5
10
20
35
50
100
150
Ethyl methylphosphonic acid ...
General Information
- Status
- Published
- Publication Date
- 14-Jun-2017
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.06 - Methods for Analysis for Organic Substances in Water
Relations
- Effective Date
- 15-Jun-2017
- Effective Date
- 01-Apr-2024
- Effective Date
- 01-May-2020
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Apr-2013
- Effective Date
- 15-Jun-2012
- Refers
ASTM D3856-11 - Standard Guide for Management Systems in Laboratories Engaged in Analysis of Water - Effective Date
- 15-Nov-2011
- Effective Date
- 01-May-2011
- Effective Date
- 01-Mar-2010
- Effective Date
- 15-Jan-2008
- Effective Date
- 01-May-2007
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 15-Aug-2006
Overview
ASTM D7597-16(2017) is an international standard developed by ASTM International for the determination of specific organophosphonate compounds in water. The method utilizes liquid chromatography coupled with tandem mass spectrometry (LC/MS/MS) using electrospray ionization (ESI) to identify and quantify trace levels of degradation products from organophosphorus chemical agents. These compounds include diisopropyl methylphosphonate (DIMP), ethyl hydrogen dimethylamidophosphate (EHDMAP), ethyl methylphosphonic acid (EMPA), isopropyl methylphosphonic acid (IMPA), methylphosphonic acid (MPA), and pinacolyl methylphosphonic acid (PMPA).
Designed to support environmental monitoring and water analysis efforts, this standard is applicable to both surface and reagent water and is significant for laboratories engaged in the detection of chemical warfare agent degradation products.
Key Topics
- Target Compounds: Focus on six specific organophosphonate analytes associated with the degradation of chemical warfare agents such as Sarin (GB), Soman (GD), Tabun (GA), and VX.
- Analytical Methodology: Involves direct injection of water samples followed by LC/MS/MS analysis using single reaction monitoring (SRM) to achieve both qualitative and quantitative results.
- Detection and Reporting: Establishes detection verification levels (DVLs) and reporting ranges for each analyte, ensuring sensitivity and reliability across various sample types.
- Quality Control: Places strong emphasis on laboratory quality assurance, including method blanks, laboratory control samples (LCS), matrix spikes, and surrogate recoveries to validate accuracy and precision.
- Interference Management: Addresses potential analytical interferences from sample matrices, solvents, reagents, and glassware. Recommends stringent contamination control and cleanliness procedures for accurate readings.
- Instrument Calibration: Details calibration procedures using multiple concentration levels and linear or quadratic regression to ensure instrument performance and data validity.
- Regulatory Alignment: Developed to align with internationally recognized standardization principles such as those set by the WTO TBT Committee.
Applications
- Water Quality Monitoring: Essential for environmental laboratories, regulatory agencies, and industry professionals monitoring surface and drinking water for organophosphonate contaminants resulting from chemical agent degradation.
- Public Health Protection: Supports early detection and quantification of hazardous organophosphonate compounds, thus helping mitigate risks to public health from potential water contamination.
- Defense and Security: Facilitates the surveillance of water sources for remnants of nerve agents and promotes compliance with chemical weapons demilitarization and nonproliferation efforts.
- Research and Development: Provides a validated approach for academic and governmental research laboratories focused on the environmental fate and behavior of organophosphate and organophosphonate compounds.
Related Standards
- ASTM D1129: Terminology Relating to Water
- ASTM D1193: Specification for Reagent Water
- ASTM D2777: Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
- ASTM D3694: Practices for Preparation of Sample Containers and for Preservation of Organic Constituents
- ASTM D5847: Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
- ASTM D3856: Guide for Management Systems in Laboratories Engaged in Analysis of Water
- ASTM E2554: Practice for Estimating and Monitoring the Uncertainty of Test Results of a Test Method Using Control Chart Techniques
These related standards provide essential guidance for terminology, calibration, sample preparation, quality management, and analytical precision within the scope of water analysis.
By following ASTM D7597-16(2017), laboratories can ensure standardized, reliable, and sensitive detection of organophosphonate analytes in water, supporting regulatory compliance and the protection of public and environmental health.
Buy Documents
ASTM D7597-16(2017) - Standard Test Method for Determination of Diisopropyl Methylphosphonate, Ethyl Hydrogen Dimethylamidophosphate, Ethyl Methylphosphonic Acid, Isopropyl Methylphosphonic Acid, Methylphosphonic Acid and Pinacolyl Methylphosphonic Acid in Water by Liquid Chromatography/Tandem Mass Spectrometry
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D7597-16(2017) is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Diisopropyl Methylphosphonate, Ethyl Hydrogen Dimethylamidophosphate, Ethyl Methylphosphonic Acid, Isopropyl Methylphosphonic Acid, Methylphosphonic Acid and Pinacolyl Methylphosphonic Acid in Water by Liquid Chromatography/Tandem Mass Spectrometry". This standard covers: SIGNIFICANCE AND USE 5.1 Organophosphate pesticides affect the nervous system by disrupting the enzyme that regulates acetylcholine, a neurotransmitter. They were developed during the early 19th century, but their effects on insects, which are similar to their effects on humans, were discovered in 1932. Some are poisonous and were used as chemical weapon agents. Organophosphate pesticides are usually not persistent in the environment.4,5 5.2 This test method is for the analysis of selected organophosphorous-based chemical weapon agent degradation products from Sarin (GB), Soman (GD), Tabun (GA) and VX. This test method has been investigated for use with reagent and surface water. SCOPE 1.1 This procedure covers the determination of diisopropyl methylphosphonate (DIMP), ethyl hydrogen dimethylamidophosphate (EHDMAP), ethyl methylphosphonic acid (EMPA), isopropyl methylphosphonic acid (IMPA), methylphosphonic acid (MPA) and pinacolyl methylphosphonic acid (PMPA) (referred to collectively as organophosphonates in this test method) in surface water by direct injection using liquid chromatography (LC) and detected with tandem mass spectrometry (MS/MS) using electrospray ionization (ESI). These analytes are qualitatively and quantitatively determined by this test method. This test method adheres to single reaction monitoring (SRM) mass spectrometry. 1.2 This test method has been developed by U.S. EPA Region 5 Chicago Regional Laboratory (CRL). 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 The detection verification level (DVL) and reporting range for the organophosphonates are listed in Table 1. TABLE 1 Detection Verification Level and Reporting Range Analyte ESI Mode DVL (μg/L) Reporting Range (μg/L) Diisopropyl methylphosphonate Positive 1 5–150 Ethyl hydrogen dimethylamidophosphate Negative 0.25 5–150 Ethyl hydrogen dimethylamidophosphate Positive 0.25 5–150 Ethyl methylphosphonic acid Negative 5 50–1500 Ethyl methylphosphonic acid Positive 5 50–1500 Isopropyl methylphosphonic acid Negative 10 50–1500 Isopropyl Methylphosphonic acid Positive 5 50–1500 Methylphosphonic acid Negative 20 100–1500 Methylphosphonic acid Positive 10 50–1500 Pinacolyl methylphosphonic acid Negative 5 50–1500 Pinacolyl methylphosphonic acid Positive 5 50–1500 1.4.1 The DVL is required to be at a concentration at least three times below the reporting limit (RL) and have a signal/noise ratio greater than 3:1. Fig. 1 displays the signal/noise ratios at the DVLs for the organophosphonates in the ESI positive mode and Fig. 2 in the ESI negative mode. FIG. 1 Example ESI Positive Mode SRM Chromatograms Signal/Noise Ratios FIG. 2 Example ESI Negative Mode SRM Chromatograms Signal/Noise Ratios 1.4.2 The reporting limit is the concentration of the Level 1 calibration standard as shown in Table 2 for the organophosphonates except for MPA in the ESI negative mode which is at Level 2 due to not meeting the DVL criteria at the lower concentration level. The DVL for MPA in the ESI negative mode is at 20 μg/L, which forces a raised reporting limit. However, the multi-laboratory validation required a spike of all target analytes at Level 1 concentrations. The mean recovery for MPA in the ESI negative mode at this level was 98.7 % as shown in Table 3. If your instrument’s sensitivity can meet the requirements in this test method, MPA may have a 50 μg/L reporting limit. TABLE 2 Concentrations of Calibration Standards (PPB) Analyte/Surrogate LV 1 LV 2 LV 3 LV 4 LV 5 LV 6 LV 7 Diisopropyl methylphosphonate 5 10 20 35 50 100 150 Ethyl hydrogen dimethylamidophosphate 5 10 20 35 50 100 150 Ethyl methylphosphonic acid ...
SIGNIFICANCE AND USE 5.1 Organophosphate pesticides affect the nervous system by disrupting the enzyme that regulates acetylcholine, a neurotransmitter. They were developed during the early 19th century, but their effects on insects, which are similar to their effects on humans, were discovered in 1932. Some are poisonous and were used as chemical weapon agents. Organophosphate pesticides are usually not persistent in the environment.4,5 5.2 This test method is for the analysis of selected organophosphorous-based chemical weapon agent degradation products from Sarin (GB), Soman (GD), Tabun (GA) and VX. This test method has been investigated for use with reagent and surface water. SCOPE 1.1 This procedure covers the determination of diisopropyl methylphosphonate (DIMP), ethyl hydrogen dimethylamidophosphate (EHDMAP), ethyl methylphosphonic acid (EMPA), isopropyl methylphosphonic acid (IMPA), methylphosphonic acid (MPA) and pinacolyl methylphosphonic acid (PMPA) (referred to collectively as organophosphonates in this test method) in surface water by direct injection using liquid chromatography (LC) and detected with tandem mass spectrometry (MS/MS) using electrospray ionization (ESI). These analytes are qualitatively and quantitatively determined by this test method. This test method adheres to single reaction monitoring (SRM) mass spectrometry. 1.2 This test method has been developed by U.S. EPA Region 5 Chicago Regional Laboratory (CRL). 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 The detection verification level (DVL) and reporting range for the organophosphonates are listed in Table 1. TABLE 1 Detection Verification Level and Reporting Range Analyte ESI Mode DVL (μg/L) Reporting Range (μg/L) Diisopropyl methylphosphonate Positive 1 5–150 Ethyl hydrogen dimethylamidophosphate Negative 0.25 5–150 Ethyl hydrogen dimethylamidophosphate Positive 0.25 5–150 Ethyl methylphosphonic acid Negative 5 50–1500 Ethyl methylphosphonic acid Positive 5 50–1500 Isopropyl methylphosphonic acid Negative 10 50–1500 Isopropyl Methylphosphonic acid Positive 5 50–1500 Methylphosphonic acid Negative 20 100–1500 Methylphosphonic acid Positive 10 50–1500 Pinacolyl methylphosphonic acid Negative 5 50–1500 Pinacolyl methylphosphonic acid Positive 5 50–1500 1.4.1 The DVL is required to be at a concentration at least three times below the reporting limit (RL) and have a signal/noise ratio greater than 3:1. Fig. 1 displays the signal/noise ratios at the DVLs for the organophosphonates in the ESI positive mode and Fig. 2 in the ESI negative mode. FIG. 1 Example ESI Positive Mode SRM Chromatograms Signal/Noise Ratios FIG. 2 Example ESI Negative Mode SRM Chromatograms Signal/Noise Ratios 1.4.2 The reporting limit is the concentration of the Level 1 calibration standard as shown in Table 2 for the organophosphonates except for MPA in the ESI negative mode which is at Level 2 due to not meeting the DVL criteria at the lower concentration level. The DVL for MPA in the ESI negative mode is at 20 μg/L, which forces a raised reporting limit. However, the multi-laboratory validation required a spike of all target analytes at Level 1 concentrations. The mean recovery for MPA in the ESI negative mode at this level was 98.7 % as shown in Table 3. If your instrument’s sensitivity can meet the requirements in this test method, MPA may have a 50 μg/L reporting limit. TABLE 2 Concentrations of Calibration Standards (PPB) Analyte/Surrogate LV 1 LV 2 LV 3 LV 4 LV 5 LV 6 LV 7 Diisopropyl methylphosphonate 5 10 20 35 50 100 150 Ethyl hydrogen dimethylamidophosphate 5 10 20 35 50 100 150 Ethyl methylphosphonic acid ...
ASTM D7597-16(2017) is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D7597-16(2017) has the following relationships with other standards: It is inter standard links to ASTM D7597-16, ASTM D3694-96(2024), ASTM D1129-13(2020)e2, ASTM E2554-18e1, ASTM E2554-18, ASTM E2554-13, ASTM D2777-12, ASTM D3856-11, ASTM D3694-96(2011), ASTM D1129-10, ASTM D2777-08, ASTM E2554-07, ASTM D1129-06a, ASTM D1129-06ae1, ASTM D2777-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D7597-16(2017) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:D7597 −16 (Reapproved 2017)
Standard Test Method for
Determination of Diisopropyl Methylphosphonate, Ethyl
Hydrogen Dimethylamidophosphate, Ethyl
Methylphosphonic Acid, Isopropyl Methylphosphonic Acid,
Methylphosphonic Acid and Pinacolyl Methylphosphonic
Acid in Water by Liquid Chromatography/Tandem Mass
Spectrometry
This standard is issued under the fixed designation D7597; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope concentration level. The DVL for MPA in the ESI negative
mode is at 20 µg/L, which forces a raised reporting limit.
1.1 This procedure covers the determination of diisopropyl
However,themulti-laboratoryvalidationrequiredaspikeofall
methylphosphonate (DIMP), ethyl hydrogen dimethylami-
target analytes at Level 1 concentrations. The mean recovery
dophosphate (EHDMAP), ethyl methylphosphonic acid
for MPAin the ESI negative mode at this level was 98.7% as
(EMPA), isopropyl methylphosphonic acid (IMPA), methyl-
shown in Table 3. If your instrument’s sensitivity can meet the
phosphonic acid (MPA) and pinacolyl methylphosphonic acid
requirements in this test method, MPA may have a 50 µg/L
(PMPA)(referredtocollectivelyasorganophosphonatesinthis
reporting limit.
test method) in surface water by direct injection using liquid
1.5 This standard does not purport to address all of the
chromatography (LC) and detected with tandem mass spec-
safety concerns, if any, associated with its use. It is the
trometry (MS/MS) using electrospray ionization (ESI). These
responsibility of the user of this standard to establish appro-
analytesarequalitativelyandquantitativelydeterminedbythis
priate safety, health, and environmental practices and deter-
test method. This test method adheres to single reaction
mine the applicability of regulatory limitations prior to use.
monitoring (SRM) mass spectrometry.
1.6 This international standard was developed in accor-
1.2 This test method has been developed by U.S. EPA
dance with internationally recognized principles on standard-
Region 5 Chicago Regional Laboratory (CRL).
ization established in the Decision on Principles for the
1.3 The values stated in SI units are to be regarded as
Development of International Standards, Guides and Recom-
standard. No other units of measurement are included in this
mendations issued by the World Trade Organization Technical
standard.
Barriers to Trade (TBT) Committee.
1.4 The detection verification level (DVL) and reporting
2. Referenced Documents
range for the organophosphonates are listed in Table 1.
1.4.1 The DVL is required to be at a concentration at least 2.1 ASTM Standards:
three times below the reporting limit (RL) and have a signal/ D1129Terminology Relating to Water
noise ratio greater than 3:1. Fig. 1 displays the signal/noise D1193Specification for Reagent Water
ratios at the DVLs for the organophosphonates in the ESI D2777Practice for Determination of Precision and Bias of
positive mode and Fig. 2 in the ESI negative mode. Applicable Test Methods of Committee D19 on Water
1.4.2 The reporting limit is the concentration of the Level 1 D3856Guide for Management Systems in Laboratories
calibration standard as shown in Table 2 for the organophos- Engaged in Analysis of Water
phonates except for MPAin the ESI negative mode which is at D3694Practices for Preparation of Sample Containers and
Level 2 due to not meeting the DVL criteria at the lower for Preservation of Organic Constituents
D5847Practice for Writing Quality Control Specifications
for Standard Test Methods for Water Analysis
This test method is under the jurisdiction ofASTM Committee D19 on Water
andisthedirectresponsibilityofSubcommitteeD19.06onMethodsforAnalysisfor
Organic Substances in Water. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved June 15, 2017. Published July 2017. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2009. Last previous edition approved in 2016 as D7597 – 16. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/D7597-16R17. the ASTM website.
Copyright ©ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA19428-2959. United States
D7597−16 (2017)
TABLE 1 Detection Verification Level and Reporting Range
Analyte ESI Mode DVL (µg/L) Reporting Range (µg/L)
Diisopropyl methylphosphonate Positive 1 5–150
Ethyl hydrogen dimethylamidophosphate Negative 0.25 5–150
Ethyl hydrogen dimethylamidophosphate Positive 0.25 5–150
Ethyl methylphosphonic acid Negative 5 50–1500
Ethyl methylphosphonic acid Positive 5 50–1500
Isopropyl methylphosphonic acid Negative 10 50–1500
Isopropyl Methylphosphonic acid Positive 5 50–1500
Methylphosphonic acid Negative 20 100–1500
Methylphosphonic acid Positive 10 50–1500
Pinacolyl methylphosphonic acid Negative 5 50–1500
Pinacolyl methylphosphonic acid Positive 5 50–1500
FIG. 1Example ESI Positive Mode SRM Chromatograms Signal/Noise Ratios
E2554Practice for Estimating and Monitoring the Uncer- 3.2 Definitions of Terms Specific to This Standard:
tainty of Test Results of a Test Method Using Control
3.2.1 detection verification level, DVL, n—a concentration
Chart Techniques
that has a signal/noise ratio greater than 3:1 and is at least 3
2.2 Other Documents:
times below the reporting limit (RL).
EPAPublication SW-846Test Methods for Evaluating Solid
3.2.2 independent reference material, IRM, n—amaterialof
Waste, Physical/Chemical Methods
known purity and concentration obtained either from the
3. Terminology
NationalInstituteofStandardsandTechnology(NIST)orother
reputable supplier. The IRM shall be obtained from a different
3.1 Definitions:
lot of material than is used for calibration.
3.1.1 For definitions of terms used in this standard, refer to
Terminology D1129.
3.2.3 organophosphonates, n—inthistestmethod,diisopro-
pyl methylphosphonate (DIMP), ethyl hydrogen dimethylami-
dophosphate (EHDMAP), ethyl methylphosphonic acid
(EMPA), isopropyl methylphosphonic acid (IMPA), methyl-
Available from United States Environmental Protection Agency (EPA), Ariel
phosphonic acid (MPA) and pinacolyl methylphosphonic acid
Rios Bldg., 1200 Pennsylvania Ave., NW, Washington, DC 20460, http://
www.epa.gov. (PMPA).
D7597−16 (2017)
FIG. 2Example ESI Negative Mode SRM Chromatograms Signal/Noise Ratios
TABLE 2 Concentrations of Calibration Standards (PPB)
Analyte/Surrogate LV 1 LV 2 LV 3 LV 4 LV 5 LV 6 LV 7
Diisopropyl methylphosphonate 5 10 20 35 50 100 150
Ethyl hydrogen dimethylamidophosphate 5 10 20 35 50 100 150
Ethyl methylphosphonic acid 50 100 200 350 500 1000 1500
Isopropyl methylphosphonic acid 50 100 200 350 500 1000 1500
Methylphosphonic acid 50 100 200 350 500 1000 1500
Pinacolyl methylphosphonic acid 50 100 200 350 500 1000 1500
DIMP-D14 (Surrogate) 5 10 20 35 50 100 150
PMPA-13C4 (Surrogate) 25 50 100 175 250 500 750
MPA-D3 (Surrogate) 25 50 100 175 250 500 750
3.3 Acronyms: 3.3.16 QC, adj—Quality Control
3.3.1 CCC, n—Continuing Calibration Check
3.3.17 RL, n—Reporting Limit
3.3.2 IC, n—Initial Calibration
3.3.18 RSD, n—Relative Standard Deviation
3.3.3 LC, n—Liquid Chromatography
3.3.19 RT, n—Retention Time
3.3.4 LCS/LCSD, n—Laboratory Control Sample/
3.3.20 SDS, n—Safety Data Sheets
Laboratory Control Sample Duplicate
3.3.5 MDL, n—Method Detection Limit 3.3.21 SRM, n—Single Reaction Monitoring
3.3.6 MeOH, n—Methanol
3.3.22 SS, n—Surrogate Standard
–3
3.3.7 mM, n—millimolar,1×10 moles/L
3.3.23 TC, n—Target Compound
3.3.8 MRM, n—Multiple Reaction Monitoring –6
3.3.24 µM, n—micromolar,1×10 moles/L
3.3.9 MS/MSD, n—Matrix Spike/Matrix Spike Duplicate
3.3.25 VOA, n—Volatile Organic Analysis
3.3.10 NA, adj—Not Available
3.3.11 ND, n—non-detect
4. Summary of Test Method
3.3.12 P&A, n—Precision and Accuracy
4.1 This is a performance-based test method and modifica-
3.3.13 PPB, n—parts per billion tions are allowed to improve performance.
3.3.14 PPT, n—parts per trillion
4.2 Fororganophosphonateanalysis,samplesareshippedto
3.3.15 QA, adj—Quality Assurance the lab between 0°C and 6°C and analyzed within 1 day of
D7597−16 (2017)
TABLE 3 Multi-Laboratory Recovery Data in Reagent Water
Bias Precision
Spike
ESI #
Mean Min Max Pooled Pooled
Analyte Conc. # Labs
Overall SD Overall
Mode Results
Recovery Recovery Recovery within-lab within-lab
(ppb)
(%) RSD (%)
(%) (%) (%) SD (%) RSD (%)
DIMP Pos 5 16 4 95.1 65.8 136.0 17.6 19.4 21.3 22.4
DIMP Pos 10 19 5 98.2 80.0 121.0 6.1 5.5 13.6 13.8
DIMP Pos 25 26 6 102.9 74.4 128.0 5.6 5.7 14.5 14.1
DIMP Pos 125 22 5 96.6 80.4 120.0 4.4 4.5 11.0 11.4
DIMP-D14 Pos 25 86 6 102.6 54.8 127.6 9.8 9.5 11.2 10.9
EHDMAP Neg 5 12 3 57.5 0.0 220.0 31.4 22.3 71.1 123.6
EHDMAP Neg 10 16 4 47.1 0.0 178.0 13.3 10.8 66.8 142.0
EHDMAP Neg 25 22 5 84.1 54.0 141.2 6.9 6.6 22.9 27.3
EHDMAP Neg 125 18 4 87.4 64.2 141.6 5.0 5.1 25.4 29.1
EHDMAP Pos 5 16 4 77.0 0.0 134.2 7.4 8.0 51.4 66.8
EHDMAP Pos 10 20 5 70.0 0.0 143.0 7.7 10.0 53.6 76.5
EHDMAP Pos 25 26 6 89.6 60.0 128.8 5.2 6.2 23.5 26.2
EHDMAP Pos 125 22 5 87.5 59.0 123.2 5.2 6.0 23.9 27.3
EMPA Neg 50 16 4 110.1 74.8 170.6 22.4 17.2 25.6 23.3
EMPA Neg 100 20 5 108.3 87.7 175.0 11.1 8.7 24.8 22.9
EMPA Neg 250 26 6 104.8 82.0 122.6 6.1 5.5 11.8 11.3
EMPA Neg 1250 22 5 101.5 87.2 126.4 8.4 8.1 11.2 11.0
EMPA Pos 50 16 4 95.4 77.6 122.8 12.9 13.3 13.1 13.7
EMPA Pos 100 20 5 96.0 61.4 132.5 9.5 9.5 15.9 16.6
EMPA Pos 250 26 6 99.7 70.0 133.2 5.9 5.4 18.2 18.2
EMPA Pos 1250 21 5 93.9 84.0 108.4 2.7 3.0 7.7 8.2
IMPA Neg 50 16 4 88.0 56.6 140.4 23.7 26.0 23.5 26.7
IMPA Neg 100 20 5 88.0 68.5 118.0 12.9 14.3 13.4 15.2
IMPA Neg 250 26 6 98.1 72.8 144.0 13.1 11.9 19.2 19.6
IMPA Neg 1250 21 5 90.7 73.1 103.0 5.5 6.1 8.7 9.6
IMPA Pos 50 16 4 98.3 47.8 139.6 19.2 20.5 27.2 27.7
IMPA Pos 100 19 5 95.4 72.3 120.5 9.8 10.3 12.4 13.0
IMPA Pos 250 26 6 97.0 79.2 188.4 7.4 7.6 10.9 11.2
IMPA Pos 1250 21 5 91.3 70.4 115.5 5.1 5.2 11.4 12.5
MPA Neg 50 16 4 98.7 3.3 175.0 14.2 25.3 60.5 61.3
MPA Neg 100 20 5 100.0 41.9 142.0 8.9 9.2 30.8 30.8
MPA Neg 250 26 6 99.5 66.0 124.5 7.6 7.6 14.5 14.5
MPA Neg 1250 22 5 102.7 81.8 130.5 10.5 9.9 12.4 12.1
MPA Pos 50 16 4 68.3 9.8 139.6 13.4 20.3 36.6 53.6
MPA Pos 100 20 5 80.5 48.4 149.7 14.0 12.6 26.8 33.3
MPA Pos 250 26 6 91.7 33.9 153.7 8.0 7.8 31.8 34.7
MPA Pos 1250 22 5 95.8 31.8 208.2 12.6 8.3 43.4 45.3
MPA-D3 Neg 250 84 6 111.2 57.2 190.8 16.2 12.5 30.0 26.9
MPA-D3 Pos 250 68 5 104.4 58.4 151.8 14.3 14.0 18.0 17.3
PMPA Neg 50 15 4 87.8 77.6 124.4 8.4 8.2 13.8 15.7
PMPA Neg 100 19 5 91.6 83.6 98.2 2.8 3.0 4.4 4.8
PMPA Neg 250 26 6 101.0 77.2 123.8 5.0 4.8 12.1 12.0
PMPA Neg 1250 22 5 99.2 84.8 126.5 4.9 4.8 12.3 12.4
PMPA Pos 50 16 4 90.8 60.8 148.8 15.8 16.1 25.6 28.2
PMPA Pos 100 15 4 95.2 86.8 114.0 3.8 4.0 7.9 8.3
PMPA Pos 250 20 5 103.8 85.2 136.1 4.5 3.8 15.3 14.7
PMPA Pos 1250 12 3 99.8 88.8 117.5 5.2 5.0 7.5 7.5
PMPA-13C6 Neg 250 83 6 99.5 74.8 128.3 9.4 9.2 11.1 11.1
collection. In the lab, the samples are spiked with surrogate, lists the concentration of each organophosphonate target com-
filtered using a syringe-driven filter unit and analyzed directly pound and each surrogate recovery.
by LC/MS/MS.
5. Significance and Use
4.3 Theorganophosphonatesandthesurrogates;diisopropyl
methylphosphonate-D , pinacolyl methylphosphonic acid-
5.1 Organophosphate pesticides affect the nervous system
C and methylphosphonic acid-D are identified by retention
6 3
by disrupting the enzyme that regulates acetylcholine, a neu-
time and one SRM transition. The target analytes and surro-
rotransmitter. They were developed during the early 19th
gates are quantitated using the SRM transitions utilizing an
century, but their effects on insects, which are similar to their
external calibration. The final report issued for each sample
D7597−16 (2017)
effects on humans, were discovered in 1932. Some are poison- 7.1.3 Tandem Mass Spectrometer (MS/MS) System—A
ous and were used as chemical weapon agents. Organophos- MS/MS system capable of MRM analysis. A system that is
capableofperformingattherequirementsinthisstandardmay
phate pesticides are usually not persistent in the
4,5
be used.
environment.
7.2 Filtration Device:
5.2 This test method is for the analysis of selected
7.2.1 Hypodermic Syringe—A luer-lock tip glass syringe
organophosphorous-based chemical weapon agent degradation
capable of holding syringe-driven filter unit.
products from Sarin (GB), Soman (GD), Tabun (GA) and VX.
7.2.1.1 A25-mLlocktipglasssyringesizeisrecommended
Thistestmethodhasbeeninvestigatedforusewithreagentand
since a 25-mL sample size is used in this test method.
surface water.
7.2.2 Filter Unit —A PVDF filter units were used to filter
the samples.
6. Interferences
8. Reagents and Materials
6.1 Test method interferences may be caused by contami-
8.1 Purity of Reagents—High-performance liquid chroma-
nants in solvents, reagents, glassware and other apparatus
tography (HPLC) pesticide residue analysis and spectropho-
producing discrete artifacts or elevated baselines. All of these
tometry grade chemicals shall be used in all tests. Unless
materials are demonstrated to be free from interferences by
indicated otherwise, it is intended that all reagents shall
analyzing laboratory reagent blanks under the same conditions
conform to the Committee on Analytical Reagents of the
as samples.
American Chemical Society. Other reagent grades may be
6.2 All glassware is washed in hot water with a detergent,
used provided they are first determined they are of sufficiently
rinsed in hot water followed by distilled water. The glassware
highpuritytopermittheirusewithoutaffectingtheaccuracyof
is then dried and heated in an oven at 250°C for 15 to 30
the measurements.
minutes. All glassware is subsequently cleaned with acetone,
8.2 Purity of Water—Unless otherwise indicated, references
then methanol.
towatershallbeunderstoodtomeanreagentwaterconforming
6.3 All reagents and solvents should be pesticide residue toType1ofSpecificationD1193.Itmustbedemonstratedthat
this water does not contain contaminants at concentrations
purity or higher to minimize interference problems.
sufficient to interfere with the analysis.
6.4 Matrix interferences may be caused by contaminants
8.3 Gases—Ultrapure nitrogen and argon.
that are co-extracted from the sample. The extent of matrix
interferences can vary considerably from sample source de- 8.4 Acetonitrile (CAS # 75-05-8).
pending on variations of the sample matrix.
8.5 Methanol (CAS # 67-56-1).
8.6 Acetone (CAS # 67-64-1).
7. Apparatus
8.7 Formic acid (≥95 %, CAS # 64-18-6).
7.1 LC/MS/MS System:
8.8 Diisopropyl methylphosphonate (DIMP, CAS # 1445-
7.1.1 LiquidChromatography(LC)System—AcompleteLC
6 75-6).
system is needed in order to analyze samples. This should
8.9 Ethyl hydrogen dimethylamidophosphate (EHDMAP,
include a sample injection system, a solvent pumping system
capable of mixing solvents, a sample compartment capable of CAS # 2632-86-2).
maintaining required temperature and a temperature controlled
8.10 Ethylmethylphosphonicacid(EMPA,CAS#1832-53-
columncompartment.Asystemthatiscapableofperformingat
7).
the flows, pressures, controlled temperatures, sample volumes
8.11 Isopropylmethylphosphonicacid(IMPA,CAS#1832-
and requirements of the standard may be used.
54-8).
7.1.2 Analytical Column —A reverse phase C18 particle
8.12 Methylphosphonic acid (MPA, CAS # 993-13-5).
columnwasusedtodevelopthistestmethod.Anycolumnthat
achieves adequate resolution may be used.The retention times 8.13 Pinacolyl methylphosphonic acid (PMPA, CAS # 616-
andorderofelutionmaychangedependingonthecolumnthat 52-4).
is used and need to be monitored.
AWaters Quattro (a trademark of theWaters Corporation, Milford, MA) micro
API mass spectrometer, or equivalent, was found suitable for use. The multi-
laboratory study included applied Biosystems and Waters mass spectrometers.
4 9
Additional information about organophosphate pesticides is available on the A Millex (a trademark of Merck KGAA, Darmstadt, Germany) HV Syringe
Internet at http://www.epa.gov (2009). Driven Filter Unit PVDF 0.45 µm (Millipore Corporation, Catalog # SLHV033NS)
Additional information about chemical weapon agents is available on the has been found suitable for use for this test method, any filter unit may be used that
Internet at http://www.opcw.org (2009). meets the performance of this test method may be used.
AWatersAlliance (a trademark of theWaters Corporation, Milford, MA) High Reagent Chemicals, American Chemical Society Specifications, American
Performance Liquid Chromatography (HPLC) System, or equivalent, was found Chemical Society, Washington, DC. For Suggestions on the testing of reagents not
suitable for use. The multi-laboratory study included Agilent and Waters LC listed by the American Chemical Society, see Annual Standards for Laboratory
systems. Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
AWatersAtlantis(atrademarkoftheWatersCorporation,Milford,MA)dC18, and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
150mm×2.1mm,3µmparticlesize,orequivalent,hasbeenfoundsuitableforuse. MD.
D7597−16 (2017)
8.14 Diisopropyl methylphosphonate-D (DIMP-D14, Un- 11.1.5 Autosampler Purge—Three loop volumes.
labeled CAS # 1445-75-6). 11.1.6 Specific instrument manufacturer wash/purge speci-
8.14.1 DIMP-D14 represents deuterium labeled diisopropyl fications should be followed in order to eliminate sample
methylphosphonate where the two isopropyl moieties contain carry-over in the analysis of organophosphonates.
all H. 8
11.2 Mass Spectrometer Parameters:
8.15 Methylphosphonic acid-D (MPA-D3, Unlabeled CAS 11.2.1 In order to acquire the maximum number of data
# 993-13-5). points per SRM channel while maintaining adequate
8.15.1 MPA-D3 represents deuterium labeled methylphos- sensitivity, the tune parameters may be optimized according to
phonic acid where the methyl moiety contains all H. your instrument. Each peak requires at least 10 scans per peak
for adequate quantitation. This standard contains three surro-
8.16 Pinacolyl methylphosphonic acid- C (PMPA-13C6,
gates and six target compounds which are located in multiple
Unlabeled CAS # 616-52-4).
reaction monitoring (MRM) experiment windows. This test
8.16.1 PMPA-13C6 represents C labeled pinacolyl meth-
method does however require the analysis of the organophos-
ylphosphonic where all the trimethylpropyl carbon atoms are
phonatesandsurrogates,inmostcases,beanalyzedinboththe
uniformly labeled C.
ESI positive and negatives modes in order to obtain the
optimum results from the individual matrices tested. Depend-
9. Hazards
ing on your instrument, this may be accomplished in one
9.1 Normal laboratory safety applies to this test method.
analysis run if the instrument can switch between positive and
Analysts should wear safety glasses, gloves, and lab coats
negative mode fast enough without losing sensitivity and
when working in the lab. Analysts should review the Safety
maintainingatleast10scansperpeak.Ifyourinstrumentisnot
Data Sheets (SDS) for all reagents used in this test method.
able to switch between modes fast enough it will require two
analyses, one in ESI positive and one in ESI negative. For
10. Sampling
example, the newer instruments may be capable of switching
10.1 Sampling—Grabsamplesmustbecollectedin≥25-mL
betweenpositiveandnegativemodesin50millisecondswhich
pre-cleaned amber glass bottles withTeflon-lined caps demon-
would require one analysis run for this test method. The older
strated to be free of interferences. This test method requires a
instruments may be capable of switching between positive and
25-mL sample size per analysis. Conventional sampling prac-
negative modes in 300 milliseconds which will require two
tices should be followed. Refer to Guide D3856 and Practices
analysis runs to obtain maximum sensitivity and the adequate
D3694.
numberofscansperpeak.Thesinglelaboratorydatainthistest
10.2 Preservation—Store samples between 0°C and 6°C
method was generated using two analysis runs. Variable
from the time of collection until analysis.Analyze the sample
parameters regarding retention times, SRM transitions and
within 1 day of collection.
cone and collision energies are shown in Table 5.
The instrument is set in the Electrospray (+) positive and/or
11. Preparation of LC/MS/MS
(–)negative source setting
Capillary Voltage: 3.5 kV
11.1 LC Chromatograph Operating Conditions:
Cone: Variable depending on analyte (Table 3)
11.1.1 Injection volumes of all calibration standards and
Extractor: 2 Volts
RF Lens: 0.2 Volts
samples are 50 µL. The first sample analyzed after the
Source Temperature: 120°C
calibrationcurveisablanktoensurethereisnocarry-over.The
Desolvation Temperature: 300°C
gradient conditions for the liquid chromatograph are shown in
Desolvation Gas Flow: 500 L/hr
Cone Gas Flow: 25 L/hr
Table 4.
Low Mass Resolution 1: 14.5
11.1.2 Temperatures—Column, 30°C; Sample
High Mass Resolution 1: 14.5
compartment, 15°C.
Ion Energy 1: 0.5
11.1.3 Seal Wash—Solvent: 50% Acetonitrile/50% Water; Entrance Energy: –1
Collision Energy: Variable depending on analyte (Table 5)
Time: 5 minutes.
Exit Energy: 2
11.1.4 Needle Wash—Solvent: 50% Acetonitrile/50% Wa-
Low Mass Resolution 2: 15
High Mass resolution 2: 15
ter; Normal Wash, Approximately 13 second wash time.
Ion Energy 2: 0.5
Multiplier: 650
–3
TABLE 4 Gradient Conditions for Liquid Chromatography Gas Cell Pirani Gauge: 3.3 × 10 Torr
Inter-Channel Delay: 0.02 seconds
Percent
Inter-Scan Delay: 0.1 seconds in one ESI mode (0.3 seconds if
Time Flow Percent Percent 2% Formic
acquiring in ESI positive and negative mode in sameanalysis run
(min) (µL/min) CH CN Water Acid
on a Quattro microAPI mass spectrometer)
in Water
Repeats: 1
0 300 0 95 5
Span: 0 Daltons
4 300 0 95 5 Dwell: 0.1 seconds
5 300 45 50 5
9 300 45 50 5
12. Calibration and Standardization
10 300 95 0 5
13 300 95 0 5
12.1 The mass spectrometer must be calibrated per manu-
14 300 0 95 5
facturer specifications before analysis. In order that analytical
20 300 0 95 5
values obtained using this test method are valid and accurate
D7597−16 (2017)
TABLE 5 Retention Times, SRM Ions, and Analyte-Specific Mass Spectrometer Parameters
Retention Time SRM Mass Transition Cone Voltage Collision Energy
Analyte ESI Mode
(min) (Parent > Product) (Volts) (eV)
Diisopropyl methylphosphonate Positive 10.9 181.3 > 139.1 25 6
Ethyl hydrogen dimethylamidophosphate Negative 3.4 152.7 > 78.7 30 15
Ethyl hydrogen dimethylamidophosphate Positive 3.4 154.2 > 126 20 12
Ethyl methylphosphonic acid Negative 3.8 123.1 > 94.8 30 12
Ethyl methylphosphonic acid Positive 3.8 125 > 96.8 25 10
Isopropyl methylphosphonic acid Negative 8.9 137.1 > 94.8 32 13
Isopropyl methylphosphonic acid Positive 8.9 139.1 > 96.8 18 9
Methylphosphonic acid Negative 2.3 94.9 > 78.7 35 15
Methylphosphonic acid Positive 2.3 96.9 > 78.7 45 15
Pinacolyl methylphosphonic acid Negative 10.5 179.2 > 94.8 35 18
Pinacolyl methylphosphonic acid Positive 10.5 181.3 > 96.8 15 7
DIMP-D14 (Surrogate) Positive 10.9 195.3 > 147.2 25 7
PMPA-13C6 (Surrogate) Negative 10.5 185.3 > 94.8 35 18
MPA-D3 (Surrogate) Negative 2.3 97.9 > 78.7 35 15
MPA-D3 (Surrogate) Positive 2.3 99.8 > 81.8 45 15
within the confidence limits of the test method, the following ion, fragmenting it the product ion fragment, and also relating
proceduresmustbefollowedwhenperformingthetestmethod. it to the retention time in the calibration standard.
12.2.4 The calibration software manual should be consulted
12.2 Calibration and Standardization—To calibrate the
to use the software correctly.The quantitation method is set as
instrument, analyze seven calibration standards containing the
anexternalcalibrationusingthepeakareasinppborppmunits
seven concentration levels of the organophosphonates and
as long as the analyst is consistent. Concentrations may be
surrogates prior to analysis as shown in Table 2.Acalibration
calculated using the data system software to generate linear
stock standard solution is prepared from standard materials or
regression or quadratic calibration curves. Forcing the calibra-
purchased as certified solutions. Stock standard solution A
tion through the origin is not recommended.
(Level 7) containing the organophosphonates, diisopropyl
12.2.5 Linear calibration may be used if the coefficient of
methylphosphonate-D , pinacolyl methylphosphonic acid-
determination, r , is >0.98 for the analyte. The point of origin
C and methylphosphonic acid-D is prepared at Level 7
6 3
is excluded and a fit weighting of 1/X is used in order to give
concentration and aliquots of that solution are diluted to
more emphasis to the lower concentrations. If one of the
prepare Levels 1 through 6. The following steps will produce
calibration standards other than the high or low point causes
standards with the concentration values shown in Table 2. The
ther ofthecurvetobe<0.98,thispointmustbere-injectedor
analyst is responsible for recording initial component weights
anewcalibrationcurvemustberegenerated.Iftheloworhigh
carefully when working with pure materials and correctly
(or both) point is excluded, minimally a five point curve is
carrying the weights through the dilution calculations.
acceptable but the reporting range must be modified to reflect
12.2.1 Prepare stock standard solution A (Level 7) by
this change.
adding to a 100-mL volumetric flask individual methanol
12.2.6 Quadratic calibration may be used if the coefficient
solutions of the following: 750 µL of 0.1g/L solutions of
of determination, r , is >0.99 for the analyte. The point of
MPA-D3 and PMPA-13C6, 15 µLof 1 g/Lsolutions of DIMP,
originisexcludedandafitweightingof1/Xisusedinorderto
DIMP-D14 and EHDMAP and 150 µL of 1 g/L solutions of
give more emphasis to the lower concentrations. If one of the
EMPA, IMPA, MPA and PMPA then dilute to 100 mL with
calibration standards, other than the high or low, causes the
water. The preparation of the Level 7 standard can be accom-
curve to be <0.99 this point must be re-injected or a new
plished using different volumes and concentrations of stock
calibration curve must be regenerated. If the low or high point
solutions as is accustomed in the individual laboratory. De-
isexcluded,asixpointcurveisacceptableusingaquadraticfit.
pendingonstockconcentrationsprepared,thesolubilityatthat
An initial seven point curve over the calibration range is
concentration will have to be ensured.
suggested in the event that the low or high point must be
12.2.2 Aliquots of Sol
...


Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...