ASTM F1841-19e1
(Practice)Standard Practice for Assessment of Hemolysis in Continuous Flow Blood Pumps
Standard Practice for Assessment of Hemolysis in Continuous Flow Blood Pumps
SIGNIFICANCE AND USE
6.1 The objective of this practice is to standardize the evaluation method for assessing the hemolytic effect of a blood pump used in extracorporeal circulation and/or circulatory assistance. By comparing the hemolysis results between a subject device and a comparator device through paired testing, a relative evaluation of hemolysis for the subject device can be made.
SCOPE
1.1 This practice covers a protocol for the assessment of the hemolytic properties of continuous, intermittent, and pulsatile flow blood pumps used in circulatory assist, including extracorporeal, percutaneous, and implantable devices. An assessment is made based on the pump's effects on the erythrocytes over a certain period of time. Adopting current practices for this assessment, a 6-hour in vitro test is performed on a pump placed in a device-specific recirculating blood loop that mimics the pressure and flow conditions of the expected worst-case clinical use of the device. If the ultimate goal of the testing is to evaluate the blood damage potential of a pump for clinical use, it is suggested that paired testing between the subject blood pump and a legally marketed comparator device be conducted using the same blood pool in a matched blood test loop so that a relative hemolysis comparison can be made.
1.2 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the two systems may result in non-conformance with the standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Aug-2019
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.30 - Cardiovascular Standards
Relations
- Effective Date
- 01-Sep-2019
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Mar-2013
- Effective Date
- 01-Mar-2005
- Refers
ASTM F1830-97 - Standard Practice for Selection of Blood for In Vitro Evaluation of Blood Pumps - Effective Date
- 01-Jan-1997
Overview
ASTM F1841-19e1: Standard Practice for Assessment of Hemolysis in Continuous Flow Blood Pumps establishes a standardized protocol for evaluating the hemolytic properties of blood pumps used in extracorporeal circulation and circulatory support. Developed by ASTM International, this practice focuses on assessing how different blood pump designs - including continuous, intermittent, and pulsatile flow pumps - can cause hemolysis, or damage to red blood cells (erythrocytes), during operation. Ensuring minimized hemolysis is critical to patient safety in clinical applications such as cardiopulmonary bypass, ventricular assist devices, percutaneous support, and extracorporeal membrane oxygenation (ECMO).
This standard promotes consistency in in vitro hemolysis testing, enabling meaningful hemolysis comparisons between a subject device and a comparator device. By following the prescribed methodology, manufacturers, researchers, and regulators can directly compare hemolytic performance and support the safety and efficacy of blood pumps intended for clinical use.
Key Topics
- Hemolysis Indices: The standard defines methods for calculating the Normalized Index of Hemolysis (NIH) and Modified Index of Hemolysis (MIH) to quantify blood cell damage in recirculating blood loops.
- Test Protocol: A 6-hour in vitro test evaluates pumps under worst-case clinical conditions by recirculating blood through a custom blood loop designed to simulate real-world flow and pressure.
- Paired Testing: The document emphasizes comparative assessment of a subject blood pump and a comparator device using the same blood pool, allowing for relative hemolysis evaluation while controlling for blood variability.
- Sample Collection and Reporting: Regular sampling for plasma free hemoglobin (PfH), hematocrit, and total hemoglobin is required, along with data plotting and statistical analysis to interpret hemolysis results.
- Blood Source and Handling: Clear procedures for blood collection, anticoagulation, hematocrit adjustment, storage, and physiological parameter maintenance help ensure test reliability.
- Clinical Relevance: Test loops are to be tailored to simulate the specific clinical application and configuration for each pump, reflecting maximum expected flow rates and pressures.
Applications
- Medical Device Development: Manufacturers use ASTM F1841-19e1 to validate the hemolytic safety of new and existing blood pumps, ensuring compliance with regulatory and marketplace expectations.
- Regulatory Submissions: Standardized hemolysis assessments performed in accordance with this practice can be submitted to regulatory agencies as part of premarket approval for extracorporeal and circulatory support devices.
- Comparative Product Assessment: Clinicians and stakeholders can compare hemolytic risk across different pumps using results derived from the same methodology.
- Quality Assurance: Consistent test protocols facilitate quality control in manufacturing and routine assessment of product batches.
- Research and Innovation: The standard enables investigators to design hemolysis studies for new blood pump technologies and to compare them to existing, legally marketed devices.
Related Standards
- ASTM F1830: Practice for Collection and Preparation of Blood for Dynamic in vitro Evaluation of Hemolysis in Blood Pumps.
- ISO 14708-5: Implants for surgery – Active implantable medical devices – Part 5: Circulatory support devices.
- Good Laboratory Practices (GLP): For blood component measurement and assay validation.
Practical Value
Implementing ASTM F1841-19e1 ensures that hemolysis data is robust, reproducible, and clinically relevant. By standardizing the assessment method, the standard improves patient safety, supports regulatory compliance, and underpins the development of innovative circulatory assist devices with lower risks of blood trauma. This results in enhanced device quality, streamlined regulatory review, and better clinical outcomes in the use of continuous flow blood pumps.
Buy Documents
ASTM F1841-19e1 - Standard Practice for Assessment of Hemolysis in Continuous Flow Blood Pumps
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F1841-19e1 is a standard published by ASTM International. Its full title is "Standard Practice for Assessment of Hemolysis in Continuous Flow Blood Pumps". This standard covers: SIGNIFICANCE AND USE 6.1 The objective of this practice is to standardize the evaluation method for assessing the hemolytic effect of a blood pump used in extracorporeal circulation and/or circulatory assistance. By comparing the hemolysis results between a subject device and a comparator device through paired testing, a relative evaluation of hemolysis for the subject device can be made. SCOPE 1.1 This practice covers a protocol for the assessment of the hemolytic properties of continuous, intermittent, and pulsatile flow blood pumps used in circulatory assist, including extracorporeal, percutaneous, and implantable devices. An assessment is made based on the pump's effects on the erythrocytes over a certain period of time. Adopting current practices for this assessment, a 6-hour in vitro test is performed on a pump placed in a device-specific recirculating blood loop that mimics the pressure and flow conditions of the expected worst-case clinical use of the device. If the ultimate goal of the testing is to evaluate the blood damage potential of a pump for clinical use, it is suggested that paired testing between the subject blood pump and a legally marketed comparator device be conducted using the same blood pool in a matched blood test loop so that a relative hemolysis comparison can be made. 1.2 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the two systems may result in non-conformance with the standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 6.1 The objective of this practice is to standardize the evaluation method for assessing the hemolytic effect of a blood pump used in extracorporeal circulation and/or circulatory assistance. By comparing the hemolysis results between a subject device and a comparator device through paired testing, a relative evaluation of hemolysis for the subject device can be made. SCOPE 1.1 This practice covers a protocol for the assessment of the hemolytic properties of continuous, intermittent, and pulsatile flow blood pumps used in circulatory assist, including extracorporeal, percutaneous, and implantable devices. An assessment is made based on the pump's effects on the erythrocytes over a certain period of time. Adopting current practices for this assessment, a 6-hour in vitro test is performed on a pump placed in a device-specific recirculating blood loop that mimics the pressure and flow conditions of the expected worst-case clinical use of the device. If the ultimate goal of the testing is to evaluate the blood damage potential of a pump for clinical use, it is suggested that paired testing between the subject blood pump and a legally marketed comparator device be conducted using the same blood pool in a matched blood test loop so that a relative hemolysis comparison can be made. 1.2 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the two systems may result in non-conformance with the standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F1841-19e1 is classified under the following ICS (International Classification for Standards) categories: 11.100 - Laboratory medicine. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F1841-19e1 has the following relationships with other standards: It is inter standard links to ASTM F1830-19, ASTM F1830-97(2017), ASTM F1830-97(2013), ASTM F1830-97(2005), ASTM F1830-97. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F1841-19e1 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
´1
Designation: F1841 − 19
Standard Practice for
Assessment of Hemolysis in Continuous Flow Blood
Pumps
This standard is issued under the fixed designation F1841; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Editorial corrections were made to 4.1 and 4.2.1 in May 2021.
INTRODUCTION
The goal of blood pump development is to replace or supplement the function of the human heart
to circulate blood. In practice, blood pumps are commonly used in cardiopulmonary bypass during
routine cardiac surgery, and for ventricular assist, percutaneous cardiopulmonary support, and
extracorporeal membrane oxygenation applications.
Many investigators have attempted to develop an atraumatic blood pump. Hemolysis is one of the
most important parameters of blood trauma induced by blood pumps and can occur from mechanical,
thermal, or chemical sources in these devices. Dynamic in vitro hemolysis testing is an essential
componentoftheassessmentofdevices,asitevaluatesthebiologicalresponseduringoperationofthe
device under worst-case clinical use conditions. Directly comparing the reported results of in vitro
hemolysis testing between laboratories is of limited utility, however, due to the lack of uniformity of
thetestmethodsemployed,andvariabilityinthefragilityofthetestbloodandthemeasurementassays
used to assess hemolysis. Thus, it is necessary to provide standardization of the methods for
performingandreportingdynamic in vitrohemolysistestsintheevaluationofpotentialclinicalblood
pumps.Asthereisadiverserangeofdevicetechnologiesandclinicalpumpapplications,thisstandard
proposes methodology for evaluating a blood pump under its simulated clinical use conditions and in
relation to a relevant comparator device.
1. Scope clinical use, it is suggested that paired testing between the
subject blood pump and a legally marketed comparator device
1.1 Thispracticecoversaprotocolfortheassessmentofthe
be conducted using the same blood pool in a matched blood
hemolytic properties of continuous, intermittent, and pulsatile
test loop so that a relative hemolysis comparison can be made.
flow blood pumps used in circulatory assist, including
extracorporeal, percutaneous, and implantable devices. An 1.2 The values stated in either SI units or inch-pound units
assessment is made based on the pump’s effects on the are to be regarded separately as standard. The values stated in
erythrocytes over a certain period of time. Adopting current each system may not be exact equivalents; therefore, each
practicesforthisassessment,a6-hour in vitrotestisperformed system shall be used independently of the other. Combining
on a pump placed in a device-specific recirculating blood loop values from the two systems may result in non-conformance
that mimics the pressure and flow conditions of the expected with the standard.
worst-caseclinicaluseofthedevice.Iftheultimategoalofthe
1.3 This standard does not purport to address all of the
testing is to evaluate the blood damage potential of a pump for
safety concerns, if any, associated with its use. It is the
responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter-
ThispracticeisunderthejurisdictionofASTMCommitteeF04onMedicaland
Surgical Materials and Devices and is the direct responsibility of Subcommittee
mine the applicability of regulatory limitations prior to use.
F04.30 on Cardiovascular Standards.
1.4 This international standard was developed in accor-
Current edition approved Sept. 1, 2019. Published December 2019. Originally
dance with internationally recognized principles on standard-
approved in 1997. Last previous edition approved in 2017 as F1841–97(2017).
DOI: 10.1520/F1841-19E01. ization established in the Decision on Principles for the
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
´1
F1841 − 19
Development of International Standards, Guides and Recom- 4.2 Modified Index of Hemolysis (MIH):
mendations issued by the World Trade Organization Technical 4.2.1 Modified index of hemolysis (MIH) (5, 6) can be
Barriers to Trade (TBT) Committee. written with no units or as (mg of hemoglobin released into
plasma / mg of total hemoglobin pumped through the loop) ×
6 6
2. Referenced Documents
10 factor.The10 factorisintroducedtoreducethenumberof
decimal places (5) and is accounted for in the following
2.1 ASTM Standards:
equation when the appropriate parameter units are used (see
F1830Practice for Collection and Preparation of Blood for
Appendix X2):
Dynamic in vitro Evaluation of Hemolysis in Blood
Pumps
~100 2 Hct!
∆PfH *V*
2.2 ISO Standards: 100
MIH 5 (2)
ISO 14708-5 Implants for surgery—Active implantable
Q * ∆T * Hgb
medical devices—Part 5: Circulatory support devices
where:
Hgb = total blood hemoglobin concentration at time zero
3. Terminology
(g/dL).
3.1 Definitions:
4.3 Differences Between NIH and MIH—While the NIH
3.1.1 comparator device—a legally marketed blood pump
value has historically been reported for blood pumps because
intended for a similar application and flow rate range as the
of its simplicity, this index is limited as it does not account for
subject device (e.g. adult implantable blood pump providing
the total hemoglobin concentration of the blood (6). For this
full circulatory support), which can be tested in parallel with
reason, the MIH equation is often used as it considers both
the subject device using the same blood pool so that a relative
blood hematocrit and hemoglobin concentration directly (5).
hemolysis comparison can be made between the devices.
Thus, along with the PfH values, it is recommended that both
3.1.2 hemolysis—damage to erythrocytes resulting in the
the NIH and MIH indices be reported to express the degree of
liberation of hemoglobin into the plasma.
hemolysis caused by a blood pump tested in a recirculating
3.1.3 plasma free hemoglobin (PfH)—the amount of hemo-
flow system. Example calculations for NIH and MIH are
globin (iron or heme-containing protein) in plasma.
included in Appendix X2.
3.1.4 subject device—a blood pump to be evaluated for its
5. Summary of Practice
hemolysis potential prior to clinical use.
3.2 Index of Hemolysis: 5.1 The hemolytic potential of a subject blood pump is
assessedbyoperatingitinadevice-specificrecirculatingblood
3.2.1 normalized index of hemolysis (NIH)—increase in
grams of plasma free hemoglobin per 100 Lof blood pumped, loop that mimics the pressure and flow conditions of the
expected clinical use of the device. Blood is recirculated for 6
corrected for plasma volume using hematocrit and normalized
by flow rate and circulation time. h and the hemolysis is assessed by measuring the plasma free
hemoglobin concentration (PfH) at periodic time points. If the
3.2.2 modified index of hemolysis (MIH)—mass of hemo-
goal of the testing is to evaluate the blood damage potential of
globin released into plasma normalized by the total mass of
a pump intended for clinical use, it is suggested that paired
hemoglobin pumped through the loop (multiplied by 10 ).
testing between the finalized subject blood pump and a legally
marketed comparator device be concurrently conducted using
4. Formulas (see Appendix X2 for units and sample
the same blood pool in a matched blood test loop so that a
calculations)
relative hemolysis comparison can be made. This standard
4.1 Normalized Index of Hemolysis (NIH) (1, 2, 3, 4) :
provides methodology for preparing blood, designing a recir-
~100 2 Hct!
culating flow loop, conducting replicate pump testing, and
∆PfH * V *
evaluating and reporting the results (see Table in Appendix
NIH ~g/100L!5 (1)
Q * ∆T * 1000
X4).
where:
6. Significance and Use
∆PfH = change in plasma free hemoglobin concentration
6.1 The objective of this practice is to standardize the
(mg/dL) over the sampling time interval,
evaluationmethodforassessingthehemolyticeffectofablood
V = blood volume in the loop (mL),
pump used in extracorporeal circulation and/or circulatory
Q = flow rate (L/min),
assistance. By comparing the hemolysis results between a
Hct = hematocrit (%), and
subject device and a comparator device through paired testing,
∆T = sampling time interval (min).
arelativeevaluationofhemolysisforthesubjectdevicecanbe
made.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
7. Preparation of Hemolysis Test
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on
7.1 Blood for Testing—See Practice F1830 for details on
the ASTM website.
obtaining and preparing blood for testing. Briefly, the blood is
The boldface numbers given in parentheses refer to a list of references at the
end of the text. obtained from animals or human volunteers having afebrile
´1
F1841 − 19
body temperatures, no physical signs of disease, and an tion. For cardiopulmonary support applications (i.e. extracor-
acceptable range of hematological values. Human, bovine, poreal or percutaneous), a pressure head in the range of 325 to
porcine, and ovine blood have been used as the primary 500mmHg may be appropriate (7).
sources of blood in the evaluation of pumps. Because the level Anultrasonicorelectromagneticflowprobeistypicallyused
of trauma-induced hemolysis may be different based on the to monitor the blood flow rate. The flow meter should be
source of blood, it is necessary to identify the source of blood calibrated using blood at the proper hematocrit and tempera-
when reporting the indices of hemolysis. It is preferable that ture. As pumping may cause a temperature rise in the blood,
animal blood be collected by controlled venipuncture to thermal control should be considered in the form of a water
minimize the risk of contamination with debris or fluids other bathorheatexchangertomaintainanappropriateandconstant
than blood. The blood from an abattoir can be used if it is blood temperature during the experiments. Blood temperature
obtained in such a way as to minimize contamination and should be monitored and recorded throughout the testing. As
excessivetraumatotheblood(seePracticeF1830).Commonly local thermal heating within pumps can damage blood, testing
used blood anticoagulants include heparin (4000 to 6000 USP should be performed at 37 6 2°C for pump applications in
unitsperliterofcollectedblood),ACD-A(anticoagulantcitrate which the patient is expected to be normothermic. For cardio-
dextrose solution A; volume ratio of ACD-A to blood is pulmonary bypass applications, it may be appropriate to
typically 1:5 to 1:8), and CPDA-1 (citrate phosphate dextrose maintain the blood during the testing at 23 6 2 °C.
adenine anticoagulant solution; volume ratio of CPDA-1 to All necessary blood-contacting components and provided
blood is typically 1:7). Blood obtained for device testing is accessories to be used with the subject device (e.g. a device-
generally used within 48 h of blood draw.As a quality control specific cannula) should be included in the testing loop as they
measure, an initial plasma free hemoglobin concentration could impact the hemolysis results. Moreover, pumps being
shouldgenerallybelessthan50mg/dL.Inordertostandardize evaluatedforclinicalusemaybesubjectedtopre-conditioning
the blood trauma testing, the blood subjected to the test should such as sterilization and aging (if sensitive to possible effects).
havethehematocritvalueadjustedtobewithintherangeof35 See ISO 14708-5 for details on pre-conditioning.
6 2% by hemodilution (with phosphate buffered saline) or 7.2.2 Preparing the Loop for Blood Testing—Since all test
hemoconcentration (via minimal centrifugation).Alternate he- runs are of a 6-h duration, sterility is generally considered not
matocrit values may be used if clinically relevant (e.g. lower necessary. Prior to testing with blood, the loop should be filled
for diluted blood, or higher for pediatric application). Blood with phosphate buffered saline (PBS) that is recirculated for
temperature during storage is typically kept between 2 and approximately 5 to 10 min to rinse and wet all the blood-
8°C, and warmed to testing temperature prior to use. It is contacting surfaces.
recommended that acceptable values for physiological blood
7.3 Pump Conditions—The pump flow rate for the testing
parameters(e.g.pH,glucose)bemaintainedpriortoandduring
should be determined based on the expected clinical use
testing, as appropriate (5).
conditions of the subject device. Testing should be conducted
at the proposed maximum limit for the pump (i.e. worst-case
7.2 Recirculating Blood Test Loop:
clinical use condition) by considering the maximum flow rate
7.2.1 Loop Configuration—As there is a diverse range of
and pump speed at a clinically-relevant pressure head. Addi-
device technologies and clinical pump applications, a device-
tional testing may also include the “nominal” and minimum
specific flow loop should be used to test the blood pump under
flow rates for pump operation as appropriate.
conditions which simulate its worst-case clinical use. For
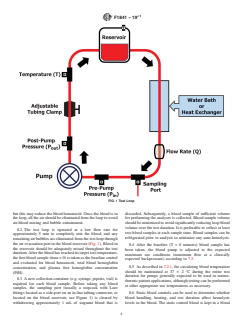
illustrative purposes, Figure 1 shows the components of a 7.4 Concurrent Testing of Subject and Comparator Devices:
typicalflowloopfortestinganextracorporealbloodpumpthat 7.4.1 Paired Device Testing—If the goal of the testing is to
may be used for cardiopulmonary bypass procedures, or a evaluate the blood damage potential of a pump intended for
pumpwhichmaybeusedforventricularassist.Forothertypes clinical use, it is recommended that paired testing between the
of blood pump designs or placements, the test loop should be subject bl
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...