ASTM F3495-23
(Test Method)Standard Test Methods for Determining the Static Failure Load of Ceramic Knee Femoral Components
Standard Test Methods for Determining the Static Failure Load of Ceramic Knee Femoral Components
SIGNIFICANCE AND USE
5.1 These test methods are intended to determine the ultimate failure load of a ceramic femoral knee component. This information can be used for evaluation of different ceramic component designs or different ceramic materials, or for series production control.
5.2 Although the test methodology described attempts to identify physiologically relevant intraoperative and in vivo loading conditions, the interpretation of results is limited to an in vitro comparison between ceramic femoral component designs and materials regarding their static ultimate failure load under the stated test conditions.
SCOPE
1.1 The test methods included in this standard cover two procedures for static burst testing of a ceramic femoral component used in total knee replacement (TKR). The two procedures are used to determine the static ultimate failure load of a ceramic femoral knee component. Both procedures are simulating in vivo loading conditions. One of the procedures additionally simulates intraoperative loading conditions. The standard applies to cruciate retaining (CR) femoral components which cover both the medial and lateral condyles and the patellar surface of the femur. These test methods may require modifications to accommodate other femoral component designs.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Jul-2023
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.22 - Arthroplasty
Relations
- Effective Date
- 01-Feb-2016
- Effective Date
- 01-Aug-2013
- Effective Date
- 01-Dec-2011
- Effective Date
- 15-Apr-2010
- Effective Date
- 01-Jul-2008
- Effective Date
- 01-Jul-2008
- Effective Date
- 01-Jan-2008
- Effective Date
- 01-Jan-2008
- Effective Date
- 01-Aug-2007
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Aug-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 01-Nov-2004
- Effective Date
- 10-Apr-2003
- Effective Date
- 10-Oct-2002
Overview
ASTM F3495-23: Standard Test Methods for Determining the Static Failure Load of Ceramic Knee Femoral Components provides comprehensive procedures for assessing the ultimate failure load of ceramic femoral components used in total knee replacement (TKR). Developed by ASTM International, this standard enables comparison of different ceramic knee component designs and materials, supporting product development, quality control, and research efforts. The standard simulates physiologically relevant in vivo and intraoperative loading conditions with two specifically designed static burst test methods.
Key Topics
- **Static Failure Load Testing:**Describes two core test methods to determine the static ultimate failure load for ceramic femoral components:
- Tension Load Test: Simulates “opening” loading conditions, including those encountered during implant insertion (intraoperative) and specific articulation scenarios, focusing on stresses at the component’s inner contours and corners.
- Compression Load Test: Simulates “closing” loading conditions associated with normal knee articulation, measuring the failure load at the outer component surfaces.
- Component Compatibility: Procedures apply to cruciate retaining (CR) femoral components covering medial and lateral condyles plus the patellar surface, with suggestions for adaptation to other designs.
- Testing Apparatus: Details requirements for uniaxial testing machines, loading fixtures, force application, and alignment to ensure physiologically relevant and repeatable results.
- Reporting and Interpretation: Outlines requirements for documenting testing parameters, component specifications, loading rates, fracture locations, and statistical summary of results.
- Safety and Best Practices: Emphasizes the need for appropriate safety measures, health practices, and environmental considerations due to the high forces involved in the destructive testing of ceramic components.
Applications
- Material and Design Evaluation: Assists manufacturers, research institutions, and quality assurance teams in comparing different ceramic materials and component designs, enabling data-driven improvements in TKR prosthesis reliability and performance.
- Quality Control: Supports consistent production standards by enabling series production control and adherence to regulatory guidelines for implant components.
- Benchmarking and Research: Provides a reproducible framework for in vitro comparisons of ceramic knee femoral components, fostering innovation and safety in the development of advanced orthopedic implants.
- Regulatory Compliance: Aligns with internationally recognized principles on standardization, facilitating conformity with global health and medical device regulations. The test results serve as part of the technical documentation required by regulatory bodies.
- Risk Mitigation: By identifying static ultimate failure loads under controlled simulation of in vivo and intraoperative conditions, the standard helps mitigate risks associated with component failure during and after surgical implantation.
Related Standards
- ASTM C1161: Test Method for Flexural Strength of Advanced Ceramics at Ambient Temperature
- ASTM F2083: Specification for Knee Replacement Prosthesis
- ASTM F3161: Test Method for Finite Element Analysis (FEA) of Metallic Orthopaedic Total Knee Femoral Components under Closing Conditions
- ASTM F3210: Test Method for Fatigue Testing of Total Knee Femoral Components under Closing Conditions
- ISO 14704: Fine ceramics (advanced technical ceramics) - Test Method for Flexural Strength of Monolithic Ceramics at Room Temperature
These referenced standards complement ASTM F3495-23 by providing related test procedures, definitions, and guidance for the assessment of orthopedic ceramic and metallic components.
Keywords: ASTM F3495-23, ceramic knee femoral component, static failure load, total knee replacement, orthopedic implant testing, compression load test, tension load test, in vitro testing, prosthesis quality control, fracture analysis, knee arthroplasty standards.
Buy Documents
ASTM F3495-23 - Standard Test Methods for Determining the Static Failure Load of Ceramic Knee Femoral Components
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F3495-23 is a standard published by ASTM International. Its full title is "Standard Test Methods for Determining the Static Failure Load of Ceramic Knee Femoral Components". This standard covers: SIGNIFICANCE AND USE 5.1 These test methods are intended to determine the ultimate failure load of a ceramic femoral knee component. This information can be used for evaluation of different ceramic component designs or different ceramic materials, or for series production control. 5.2 Although the test methodology described attempts to identify physiologically relevant intraoperative and in vivo loading conditions, the interpretation of results is limited to an in vitro comparison between ceramic femoral component designs and materials regarding their static ultimate failure load under the stated test conditions. SCOPE 1.1 The test methods included in this standard cover two procedures for static burst testing of a ceramic femoral component used in total knee replacement (TKR). The two procedures are used to determine the static ultimate failure load of a ceramic femoral knee component. Both procedures are simulating in vivo loading conditions. One of the procedures additionally simulates intraoperative loading conditions. The standard applies to cruciate retaining (CR) femoral components which cover both the medial and lateral condyles and the patellar surface of the femur. These test methods may require modifications to accommodate other femoral component designs. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 These test methods are intended to determine the ultimate failure load of a ceramic femoral knee component. This information can be used for evaluation of different ceramic component designs or different ceramic materials, or for series production control. 5.2 Although the test methodology described attempts to identify physiologically relevant intraoperative and in vivo loading conditions, the interpretation of results is limited to an in vitro comparison between ceramic femoral component designs and materials regarding their static ultimate failure load under the stated test conditions. SCOPE 1.1 The test methods included in this standard cover two procedures for static burst testing of a ceramic femoral component used in total knee replacement (TKR). The two procedures are used to determine the static ultimate failure load of a ceramic femoral knee component. Both procedures are simulating in vivo loading conditions. One of the procedures additionally simulates intraoperative loading conditions. The standard applies to cruciate retaining (CR) femoral components which cover both the medial and lateral condyles and the patellar surface of the femur. These test methods may require modifications to accommodate other femoral component designs. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F3495-23 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3495-23 has the following relationships with other standards: It is inter standard links to ASTM F3161-16, ASTM C1161-13, ASTM F2083-11, ASTM F2083-10, ASTM F2083-08e1, ASTM F2083-08, ASTM C1161-02c(2008), ASTM C1161-02c(2008)e1, ASTM F2083-07, ASTM F2083-06b, ASTM F2083-06a, ASTM F2083-06, ASTM F2083-04, ASTM F2083-03, ASTM C1161-02ce1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F3495-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F3495 − 23
Standard Test Methods for
Determining the Static Failure Load of Ceramic Knee
Femoral Components
This standard is issued under the fixed designation F3495; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope F3161 Test Method for Finite Element Analysis (FEA) of
Metallic Orthopaedic Total Knee Femoral Components
1.1 The test methods included in this standard cover two
under Closing Conditions
procedures for static burst testing of a ceramic femoral
F3210 Test Method for Fatigue Testing of Total Knee
component used in total knee replacement (TKR). The two
Femoral Components Under Closing Conditions
procedures are used to determine the static ultimate failure load
2.2 Other Standards:
of a ceramic femoral knee component. Both procedures are
ISO 14704 Fine ceramics (advanced ceramics, advanced
simulating in vivo loading conditions. One of the procedures
technical ceramics)—Test method for flexural strength of
additionally simulates intraoperative loading conditions. The
monolithic ceramics at room temperature
standard applies to cruciate retaining (CR) femoral components
which cover both the medial and lateral condyles and the
3. Terminology
patellar surface of the femur. These test methods may require
3.1 Definitions—The definitions and terms of Specification
modifications to accommodate other femoral component de-
F2083 apply.
signs.
3.2 Definitions of Terms Specific to This Standard:
1.2 The values stated in SI units are to be regarded as
3.2.1 component size—size of femoral component as given
standard. No other units of measurement are included in this
by the manufacturer.
standard.
3.2.2 compression force—used for the compression load test
1.3 This standard does not purport to address all of the
and is referred to as F.
safety concerns, if any, associated with its use. It is the
3.2.3 corner—angular design feature of the inner contour of
responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter- a femoral component; to be seen in the sagittal view; see Fig.
mine the applicability of regulatory limitations prior to use. 1 for details.
1.4 This international standard was developed in accor-
3.2.4 counter force—used for the tension load test, is acting
dance with internationally recognized principles on standard-
along the force axis and is referred to as F .
ization established in the Decision on Principles for the
3.2.5 distal face—the distal face is part of the inner contour
Development of International Standards, Guides and Recom-
of the component. The distal face is touching the femoral bone
mendations issued by the World Trade Organization Technical
at the transverse resection plane. Typically the pegs are part of
Barriers to Trade (TBT) Committee.
the distal face; see Fig. 2 for details.
3.2.6 force axis—line of action of the tensile force F and
2. Referenced Documents
2 the counter force F applied to the femoral component.
2.1 ASTM Standards:
3.2.7 lower unit—bearing frame attached to the test machine
C1161 Test Method for Flexural Strength of Advanced
to apply the counter force.
Ceramics at Ambient Temperature
F2083 Specification for Knee Replacement Prosthesis
3.2.8 stroke rate—the rate of the stroke displacement of the
force applicator.
1 3.2.9 tensile force—used for the tension load test, is acting
These test methods are under the jurisdiction of ASTM Committee F04 on
Medical and Surgical Materials and Devices and are the direct responsibility of
along the force axis and is referred to as F .
Subcommittee F04.22 on Arthroplasty.
3.2.10 upper unit—bearing frame attached to test machine
Current edition approved July 15, 2023. Published August 2023. DOI: 10.1520/
F3495-23. to apply the tensile force.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on Available from American National Standards Institute (ANSI), 25 W. 43rd St.,
the ASTM website. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3495 − 23
4.3 The compression load test method provides a means to
measure the ultimate failure load of the outer contour of the
femoral component.
5. Significance and Use
5.1 These test methods are intended to determine the ulti-
mate failure load of a ceramic femoral knee component. This
information can be used for evaluation of different ceramic
component designs or different ceramic materials, or for series
production control.
5.2 Although the test methodology described attempts to
FIG. 1 Corners of a Femoral Component
identify physiologically relevant intraoperative and in vivo
loading conditions, the interpretation of results is limited to an
in vitro comparison between ceramic femoral component
designs and materials regarding their static ultimate failure
load under the stated test conditions.
6. Equipment Characteristics
6.1 Generally, the ultimate failure load tests should be
performed on uniaxial testing machines. Note: Mechanical
load frames with power screws are recommended, but all other
types of uniaxial testing machines with adequate load capacity
and stroke rate control may be used.
6.2 The loading fixtures should be capable of sustaining
forces up to the anticipated fracture level. Note: Add a safety
margin to the anticipated fracture level.
7. Apparatus
7.1 For the tension load test setup, the femoral knee
FIG. 2 Locations of the Anterior Flange, Distal Face, Condyles,
component shall be positioned in a way that the applied forces
and Peg of a Femoral Knee Component
F and F are acting along a common force axis when viewed
1 2
in the sagittal plane. Misalignment in the sagittal plane must be
avoided throughout the full loading cycle. As the femoral knee
component is subjected to deformation while loaded, the
4. Summary of Test Method
means of the tension load test setup shall incorporate moveable
parts to level the specimen in place and keep the forces F and
4.1 In this standard, two test methods are included to 1
F on their common force axis.
determine the static ultimate failure load of a ceramic femoral 2
knee component. Two test methods are included in this
7.2 The force is applied to the inner contour of the femoral
standard to represent the “opening” and “closing” loading
component in a way that the force is opening the femoral
conditions the femoral component experiences during use.
component. Depending on the position of the force axis, the
Opening loading conditions, meant to simulate both loading on
number of stressed corners may vary; see Figs. 1-4.
the articulating surface in proximity to a corner on the inner
contour of the femoral component which causes high tensile
stresses on the inner surface at the corner feature and intraop-
erative impaction, are simulated by the tension load test
method and closing loading conditions, meant to simulate
loading on the articulating surface not in proximity to a corner
or loading of the component after loss of supporting bone
which causes high tensile stresses on the articulating surface,
are simulated by the compression load test method in this
standard. Intraoperatively, the component is pounded onto the
prepared end of the femur under loading conditions which can
force it to open and again are simulated by the tension load test
method outlined in this standard.
4.2 The tension load test method provides a means to
measure the ultimate failure load of the inner contour of the
FIG. 3 Forces F and F Along the Force Axis Are Stressing Only
1 2
femoral component. Three Corners and the Pegs of the Femoral Component
F3495 − 23
7.2.3.1 The force for the tension load test will be applied
through the four loading points of the upper and the lower unit
to the femoral component. Note: Use tapered pins or dome-
shaped buttons 1 mm to 10 mm in diameter made of a
malleable metal, for example an aluminium alloy like AA-
7075-T6 (EN AW-7075-T6), to apply the load to the femoral
component and to secure the femoral component between the
upper and the lower unit. Large or plane loading points may
alter the stress distribution in the femoral component or will
lead to undesirable constraints in the femoral component.
7.2.4 The following constraints shall apply to the upper and
the lower unit. The constraints of the units are necessary for the
femoral component to move freely between the four loading
points.
7.2.4.1 The upper unit shall incorporate a ball joint to level
between the two condyles and ensure that all four loading
points are in full contact to the femoral component; see Figs. 5
FIG. 4 Forces F and F Along the Force Axis Are Stressing All
1 2
Four Corners and the Pegs of the Femoral Component and 6 for details.
7.2.4.2 The tensile force F is applied in the Z-direction
through the upper unit to the femoral component.
7.2.1 Note: Depending on the details of the femoral knee
7.2.4.3 The lower unit is connected to an X-Y table to move
component design, it is very likely that not all corners are
under force effect of F , to compensate any force constraints in
stressed to an equivalent level in vivo. As the tension load setup
the X- and Y-directions, and to act as the counter force F in the
is intended to simulate in vivo loading conditions, in vivo low
Z-direction.
stressed corners can be neglected in the tension load test.
7.2.5 The details of the tension load apparatus shall be
Please refer to Appendix X1 and Appendix X2 for further
designed to the requirements of the femoral component under
explanations.
consideration, meaning that the loading points shall fit and fix
7.2.2 The bearings for F and F shall be decoupled from
1 2
into the cement pockets or alternative fixtures. Note: For
each other. The apparatus shall consist of two separate units, an
further guidance, examples, and alternative fixtures, please
upper unit to apply the tensile force F to the condyles and a
refer to the appendixes.
lower unit as a counter bearing to apply the counter force F to
7.3 For the compression load test setup, the femoral com-
the anterior flange. Note: The forces F and F have to be on
1 2
ponent shall be positioned in a way that the distal face is
the same force axis when viewed in the sagittal plane.
However, the upper and the lower units may be mounted
inverted in the loading frame, as long as the forces F and F
1 2
remain on the same axis. Note: Please refer to the appendixes
for an example of the upper and lower units.
7.2.3 The upper unit has two loading points to the femoral
component condyles, while the lower unit has two loading
points to the femoral component anterior flange; see Fig. 5 for
details.
FIG. 6 Examples of an Upper and a Lower Unit to Apply
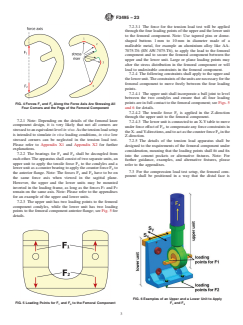
FIG. 5 Loading Points for F and F to the Femoral Component F and F
1 2 1 2
F3495 − 23
parallel to the back surface of the test rig. By applying the
compression force F, the outer contour of femoral component
is stressed; see Fig. 7 for details.
7.3.1 The corners of the femoral component shall be parallel
to the ground surface of the test rig; see Fig. 8 for details.
7.3.2 The details of compression load apparatus shall be
designed to the requirements of the femoral component under
consideration, meaning asymmetric condyles or an asymmetric
anterior flange have to be levelled to the parallel requirements.
7.3.3 The compression force F is applied to the apex of both
condyles. A rocker should be used to level the force between
the two condyles. The force should be applied evenly distrib-
uted to both condyles to avoid rocking of the specimen. Note:
FIG. 8 The Distal Face Shall Be Parallel to the Back Surface of
the Test Rig. The Corners Shall Be Parallel to the Ground Sur-
The fixation methods of the compression load setup are
face of the Test Rig. The Upper Unit Shall Incorporate a Rocker
different from the methods described in Test Methods F3161
to Level the Force Between the Two Condyles
and F3210. Test Methods F3161 and F3210 recommend
anterior flange potting, while the femoral knee component is
not clamped, fixed, or potted in any way in the compression
portfolio. The manufacturer shall justify the worst-case selec-
load test setup.
tion if not all sizes of the implant portfolio are tested.
7.3.4 The compression force F shall be applied to the center
9.2 Loading Rate—For tension load testing, a stroke rate of
of the medio-lateral width (MLW) of the femoral component;
5 mm/min is set as the default stroke rate. For compression
see Fig. 8 for details.
load testing, a stroke rate of 40 mm/min is set as the default
stroke rate. Note: It is suspected that slow crack growth is
8. Hazards
active during testing, therefore a fast testing rate should be
8.1 Due to the high forces anticipated in this type of
used so that the fracture of the ceramic specimens occurs
destructive test, appropriate shielding of the femoral compo-
within a 5 to 15 s interval. Please refer to Test Method C1161
nent test site is required.
and ISO 14704 for guidance. Depending on the ceramic
material and the femoral component design, other stroke rates
9. Sampling, Test Specimens, and Test Units
may be applicable. The user of this standard shall justify if
9.1 Number of Test Specimens—A minimum of five speci-
other stroke rates have been used as the default stroke rates.
mens is recommended per test group for each test method. A
test group shall represent one single size of the femoral implant 10. Procedure
10.1 Tension Load Test:
10.1.1 Following normal laboratory cleaning procedures to
remove any debris or other surface contaminants, the femoral
component is positioned in the upper unit of the test rig first.
10.1.2 Care should be taken to position the femoral compo-
nent to the loading points of the upper unit. The femoral
component shall be self-locking to the loading points of the
upper unit. Note: The femoral component may swing when
placed on the pins or buttons, respectively. Wait until the
femo
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...