ASTM F2777-23
(Test Method)Standard Test Method for Evaluating Knee Bearing (Tibial Insert) Endurance and Deformation Under High Flexion
Standard Test Method for Evaluating Knee Bearing (Tibial Insert) Endurance and Deformation Under High Flexion
SIGNIFICANCE AND USE
4.1 This test method can be used to describe the effects of materials, manufacturing, and design variables on the fatigue/cyclic creep performance of UHMWPE bearing components subject to substantial rotation in the transverse plane (relative to the tibial tray) for a relatively large number of cycles.
4.2 The loading and kinematics of bearing component designs in vivo will, in general, differ from the loading and kinematics defined in this test method. The results obtained here cannot be used to directly predict in vivo performance. However, this test method is designed to enable comparisons between the fatigue performance of different bearing component designs when tested under similar conditions.
4.3 The test described is applicable to any bicompartmental knee design, including mobile bearing knees that have mechanisms in the tibial articulating component to constrain the posterior movement of the femoral component and a built-in retention mechanism to keep the articulating component on the tibial plate.
SCOPE
1.1 This standard specifies a test method for determining the endurance properties and deformation, under specified laboratory conditions, of ultra high molecular weight polyethylene (UHMWPE) tibial bearing components used in bicompartmental or tricompartmental knee prosthesis designs.
1.2 This test method is intended to simulate near posterior edge loading similar to the type of loading that would occur during high flexion motions such as squatting or kneeling.
1.3 Although the methodology described attempts to identify physiological orientations and loading conditions, the interpretation of results is limited to an in vitro comparison between knee prosthesis designs and their ability to resist deformation and fracture under stated test conditions.
1.4 This test method applies to bearing components manufactured from UHMWPE.
1.5 This test method could be adapted to address unicompartmental total knee replacement (TKR) systems, provided that the designs of the unicompartmental systems have sufficient constraint to allow use of this test method. This test method does not include instructions for testing two unicompartmental knees as a bicompartmental system.
1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Aug-2023
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.22 - Arthroplasty
Relations
- Effective Date
- 15-Jun-2020
- Effective Date
- 15-May-2014
- Refers
ASTM F1223-08(2012) - Standard Test Method for Determination of Total Knee Replacement Constraint - Effective Date
- 01-Dec-2012
- Effective Date
- 01-Dec-2011
- Effective Date
- 15-Apr-2010
- Effective Date
- 01-Jul-2008
- Effective Date
- 01-Jul-2008
- Effective Date
- 01-Jun-2008
- Effective Date
- 01-May-2008
- Effective Date
- 01-Aug-2007
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Aug-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 01-Apr-2005
- Effective Date
- 01-Nov-2004
Overview
ASTM F2777-23: Standard Test Method for Evaluating Knee Bearing (Tibial Insert) Endurance and Deformation Under High Flexion establishes a laboratory test procedure for assessing the endurance characteristics and deformation of ultra high molecular weight polyethylene (UHMWPE) tibial bearing components. It is specifically designed for bicompartmental and tricompartmental knee prosthesis designs. This method simulates high-flexion motions-such as squatting or kneeling-in order to determine how different materials, manufacturing processes, and design variables affect the fatigue and creep performance of UHMWPE knee bearings.
While this standard does not directly predict in vivo (clinical) outcomes due to the complex biomechanics of the human body, it robustly enables comparative analysis of different knee prosthesis designs under controlled laboratory conditions. The test results aid in identifying potential design weaknesses and guiding improvements for patient safety and implant longevity.
Key Topics
Test Objectives
- Measure endurance properties and deformation of UHMWPE tibial inserts under high-flexion, cyclic loading.
- Evaluate resistance to fatigue, fracture, and creep deformation in simulated worst-case scenarios.
- Compare the performance of different knee prosthesis designs in vitro.
Test Scope
- Applicable to bicompartmental and tricompartmental knee prostheses.
- May be adapted for unicompartmental total knee replacement (TKR) designs with sufficient constraint.
Methodology Highlights
- Controlled cyclic loading up to 2275 N simulating posterior edge loading found in high-flexion activities.
- Test performed at recommended posterior slope angles using manufacturer guidelines.
- Evaluation of bearing deformation using coordinate measuring machines (CMM) or non-contact 3D scanners.
- Testing environment maintained at 37°C using deionized water to mimic physiological conditions.
Comparative Analysis
- Results offer a reliable means to compare design features, materials, or manufacturing processes.
- Identifies failure modes such as fracture, deformation, or disassociation from the tibial tray.
Applications
Implant Design Validation
- Supports medical device manufacturers in evaluating new or modified UHMWPE knee bearing designs for endurance under physiologically relevant, high-flexion loads.
Quality Assurance & Regulatory Submissions
- Provides a recognized method for demonstrating fatigue resistance and deformation properties required by regulatory authorities.
- Enables benchmarking of products against industry standards.
Research and Development
- Assists in material selection, testing new sterilization or aging processes, and optimizing geometry for durability.
- Facilitates in vitro assessment of design modifications aimed at preventing known clinical failure modes.
Comparative Product Evaluation
- Allows standardized, reproducible testing, making it possible to choose the best-performing designs for specific patient populations or clinical needs.
Related Standards
- ASTM F1223: Test Method for Determination of Total Knee Replacement Constraint
- ASTM F2003: Practice for Accelerated Aging of UHMWPE After Gamma Irradiation
- ASTM F2083: Specification for Knee Replacement Prosthesis
- ISO 4965-1: Metallic Materials-Dynamic Force Calibration for Uniaxial Fatigue Testing
- ISO 5833: Implants for Surgery-Acrylic Resin Cements
- ISO 14243-1: Wear of Total Knee-joint Prostheses-Load Control Parameters
- ISO 14243-3: Wear of Total Knee-joint Prostheses-Displacement Control Parameters
ASTM F2777-23 is an essential resource for orthopedic implant developers, testing laboratories, and regulatory professionals working to optimize and qualify knee prosthesis bearing components for durability and patient safety.
Buy Documents
ASTM F2777-23 - Standard Test Method for Evaluating Knee Bearing (Tibial Insert) Endurance and Deformation Under High Flexion
REDLINE ASTM F2777-23 - Standard Test Method for Evaluating Knee Bearing (Tibial Insert) Endurance and Deformation Under High Flexion
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2777-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Evaluating Knee Bearing (Tibial Insert) Endurance and Deformation Under High Flexion". This standard covers: SIGNIFICANCE AND USE 4.1 This test method can be used to describe the effects of materials, manufacturing, and design variables on the fatigue/cyclic creep performance of UHMWPE bearing components subject to substantial rotation in the transverse plane (relative to the tibial tray) for a relatively large number of cycles. 4.2 The loading and kinematics of bearing component designs in vivo will, in general, differ from the loading and kinematics defined in this test method. The results obtained here cannot be used to directly predict in vivo performance. However, this test method is designed to enable comparisons between the fatigue performance of different bearing component designs when tested under similar conditions. 4.3 The test described is applicable to any bicompartmental knee design, including mobile bearing knees that have mechanisms in the tibial articulating component to constrain the posterior movement of the femoral component and a built-in retention mechanism to keep the articulating component on the tibial plate. SCOPE 1.1 This standard specifies a test method for determining the endurance properties and deformation, under specified laboratory conditions, of ultra high molecular weight polyethylene (UHMWPE) tibial bearing components used in bicompartmental or tricompartmental knee prosthesis designs. 1.2 This test method is intended to simulate near posterior edge loading similar to the type of loading that would occur during high flexion motions such as squatting or kneeling. 1.3 Although the methodology described attempts to identify physiological orientations and loading conditions, the interpretation of results is limited to an in vitro comparison between knee prosthesis designs and their ability to resist deformation and fracture under stated test conditions. 1.4 This test method applies to bearing components manufactured from UHMWPE. 1.5 This test method could be adapted to address unicompartmental total knee replacement (TKR) systems, provided that the designs of the unicompartmental systems have sufficient constraint to allow use of this test method. This test method does not include instructions for testing two unicompartmental knees as a bicompartmental system. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This test method can be used to describe the effects of materials, manufacturing, and design variables on the fatigue/cyclic creep performance of UHMWPE bearing components subject to substantial rotation in the transverse plane (relative to the tibial tray) for a relatively large number of cycles. 4.2 The loading and kinematics of bearing component designs in vivo will, in general, differ from the loading and kinematics defined in this test method. The results obtained here cannot be used to directly predict in vivo performance. However, this test method is designed to enable comparisons between the fatigue performance of different bearing component designs when tested under similar conditions. 4.3 The test described is applicable to any bicompartmental knee design, including mobile bearing knees that have mechanisms in the tibial articulating component to constrain the posterior movement of the femoral component and a built-in retention mechanism to keep the articulating component on the tibial plate. SCOPE 1.1 This standard specifies a test method for determining the endurance properties and deformation, under specified laboratory conditions, of ultra high molecular weight polyethylene (UHMWPE) tibial bearing components used in bicompartmental or tricompartmental knee prosthesis designs. 1.2 This test method is intended to simulate near posterior edge loading similar to the type of loading that would occur during high flexion motions such as squatting or kneeling. 1.3 Although the methodology described attempts to identify physiological orientations and loading conditions, the interpretation of results is limited to an in vitro comparison between knee prosthesis designs and their ability to resist deformation and fracture under stated test conditions. 1.4 This test method applies to bearing components manufactured from UHMWPE. 1.5 This test method could be adapted to address unicompartmental total knee replacement (TKR) systems, provided that the designs of the unicompartmental systems have sufficient constraint to allow use of this test method. This test method does not include instructions for testing two unicompartmental knees as a bicompartmental system. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2777-23 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2777-23 has the following relationships with other standards: It is inter standard links to ASTM F1223-20, ASTM F1223-14, ASTM F1223-08(2012), ASTM F2083-11, ASTM F2083-10, ASTM F2083-08e1, ASTM F2083-08, ASTM F1223-08, ASTM F2003-02(2008), ASTM F2083-07, ASTM F2083-06b, ASTM F2083-06a, ASTM F2083-06, ASTM F1223-05, ASTM F2083-04. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2777-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2777 − 23
Standard Test Method for
Evaluating Knee Bearing (Tibial Insert) Endurance and
Deformation Under High Flexion
This standard is issued under the fixed designation F2777; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
1.1 This standard specifies a test method for determining the
endurance properties and deformation, under specified labora-
2. Referenced Documents
tory conditions, of ultra high molecular weight polyethylene
2.1 ASTM Standards:
(UHMWPE) tibial bearing components used in bicompartmen-
F1223 Test Method for Determination of Total Knee Re-
tal or tricompartmental knee prosthesis designs.
placement Constraint
1.2 This test method is intended to simulate near posterior
F2003 Practice for Accelerated Aging of Ultra-High Mo-
edge loading similar to the type of loading that would occur
lecular Weight Polyethylene After Gamma Irradiation in
during high flexion motions such as squatting or kneeling.
Air
1.3 Although the methodology described attempts to iden-
F2083 Specification for Knee Replacement Prosthesis
tify physiological orientations and loading conditions, the 3
2.2 Other Standards:
interpretation of results is limited to an in vitro comparison
ISO 4965-1 Metallic Materials—Dynamic Force Calibration
between knee prosthesis designs and their ability to resist
for Uniaxial Fatigue Testing—Part 1: Testing System
deformation and fracture under stated test conditions.
ISO 5833 Implants for Surgery—Acrylic Resin Cements
1.4 This test method applies to bearing components manu-
ISO 14243-1 Implants for Surgery—Wear of Total Knee-
factured from UHMWPE.
joint Prostheses—Part 1: Loading and Displacement Pa-
rameters for Wear-testing Machines with Load Control
1.5 This test method could be adapted to address unicom-
and Corresponding Environmental Conditions for Test
partmental total knee replacement (TKR) systems, provided
ISO 14243-3 Implants for Surgery—Wear of Total Knee-
that the designs of the unicompartmental systems have suffi-
joint Prostheses—Part 3: Loading and Displacement Pa-
cient constraint to allow use of this test method. This test
rameters for Wear-testing Machines with Displacement
method does not include instructions for testing two unicom-
Control and Corresponding Environmental Conditions for
partmental knees as a bicompartmental system.
Test
1.6 The values stated in SI units are to be regarded as
standard. No other units of measurement are included in this
3. Terminology
standard.
3.1 Definitions:
1.7 This standard does not purport to address all of the
3.1.1 anatomic (mechanical) axis of the femur—the line
safety concerns, if any, associated with its use. It is the
between the center of the femoral head and the center of the
responsibility of the user of this standard to establish appro-
femoral knee.
priate safety, health, and environmental practices and deter-
3.1.2 bearing centerline—the line running anteroposterior
mine the applicability of regulatory limitations prior to use.
that is the mirror line of the tibial bearing (tibial insert). For
1.8 This international standard was developed in accor-
asymmetric tibial bearing designs, the appropriate tibial bear-
dance with internationally recognized principles on standard-
ing centerline shall be determined and reported along with the
ization established in the Decision on Principles for the
rationale for the location.
Development of International Standards, Guides and Recom-
1 2
This test method is under the jurisdiction of ASTM Committee F04 on Medical For referenced ASTM standards, visit the ASTM website, www.astm.org, or
and Surgical Materials and Devices and is the direct responsibility of Subcommittee contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
F04.22 on Arthroplasty. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved Sept. 1, 2023. Published September 2023. Originally the ASTM website.
approved in 2010. Last previous edition approved in 2016 as F2777 – 16. DOI: Available from American National Standards Institute (ANSI), 25 W. 43rd St.,
10.1520/F2777-23. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2777 − 23
3.1.3 bearing retention mechanism—mechanical means for 4. Significance and Use
preventing tibial tray/bearing disassociation.
4.1 This test method can be used to describe the effects of
3.1.4 femoral component centerline—a line running antero-
materials, manufacturing, and design variables on the fatigue/
posterior between the femoral condyles and parallel to the
cyclic creep performance of UHMWPE bearing components
femoral condyles. The line should be equidistant between the
subject to substantial rotation in the transverse plane (relative
condyles. For asymmetric or non-parallel condyles designs, the
to the tibial tray) for a relatively large number of cycles.
appropriate centerline shall be determined, and the rationale for
4.2 The loading and kinematics of bearing component
that location reported.
designs in vivo will, in general, differ from the loading and
3.1.5 fixed bearing system—a knee prosthesis system com-
kinematics defined in this test method. The results obtained
prised of a femoral component and a tibial component, where
here cannot be used to directly predict in vivo performance.
the tibial articulating surface is not intended to move relative to
However, this test method is designed to enable comparisons
the tibial tray.
between the fatigue performance of different bearing compo-
3.1.6 mobile bearing component—the component between nent designs when tested under similar conditions.
fixed femoral and tibial knee components with an articulating
4.3 The test described is applicable to any bicompartmental
surface on both the inferior and superior sides.
knee design, including mobile bearing knees that have mecha-
3.1.7 mobile bearing knee system—a knee prosthesis system
nisms in the tibial articulating component to constrain the
comprised of a femoral component, a tibial component, and a
posterior movement of the femoral component and a built-in
mobile bearing component that can rotate and/or translate
retention mechanism to keep the articulating component on the
relative to the tibial component.
tibial plate.
3.1.8 posterior slope—the angle that the perpendicular axis
5. Apparatus and Materials
of the tibial tray makes when it is tilted posteriorly away from
the tibial axis (see Fig. 1).
5.1 Testing Machine, with the following characteristics:
3.1.9 R value—the ratio of the minimum force to the
5.1.1 A sinusoidal, dynamic-forcing waveform.
maximum force (that is, R = minimum force/maximum force).
5.1.2 An error in applied force not greater than 62 % at the
maximum force magnitude (in accordance with ISO 4965-1).
3.1.10 tibial axis—nominal longitudinal axis of the tibia,
5.1.3 Axial force peak representative of what could occur
which corresponds with the central axis of the medullary cavity
during daily activities of high flexion (about 2275 N). During
of the proximal tibia.
the tests, the values of the maximum and minimum forces shall
3.1.11 tibial tray/bearing disassociation—unrecoverable
be maintained to an accuracy of 62 % of the maximum force.
physical separation of the tibial bearing and tibial tray compo-
The test shall be stopped if this accuracy is not maintained.
nents as a result of bearing distraction or tilting.
5.1.4 The forcing accuracy must be maintained as bearing
3.1.12 tibial tray centerline—a line running anteroposterior
component deformation occurs.
that is the mirror line of the tibial articulating surface. For
5.1.5 Instrumentation to record the number of cycles.
asymmetric bearing tibial tray designs, the appropriate tibial
5.2 Fixturing, with the following characteristics:
tray centerline shall be determined and reported along with the
5.2.1 Means of mounting and enclosing the test specimens
rationale for the location.
using a corrosion-resistant material that is capable of holding
the femoral component and tibial tray.
5.2.2 The fixtures shall maintain the tibial and femoral
components in their required orientations for the duration of
the test.
5.2.3 If necessary, bone cement (see ISO 5833) or a high-
strength epoxy may be used to lock the femoral and tibial
components in their fixtures.
5.2.4 The test apparatus or fixture should allow the force to
be applied through the center of the femoral component and
ensure equal force transmission through the medial and lateral
condyles or offset medially as given by ISO 14243-1 and ISO
14243-3.
5.2.5 The test apparatus or fixture shall allow for varus-
valgus self-alignment of the femoral or tibial component.
5.3 Fluid Medium:
5.3.1 The test assembly shall be immersed in deionized
water at 37 6 2 °C.
5.3.2 Deionized water should be added as necessary to keep
the test components at the test temperature for the duration of
FIG. 1 Incline of the Tibial Tray Relative to the Tibial Axis at the
Recommended Angle (Posterior Slope) the test.
F2777 − 23
6. Specimen Selection 7.4 Mount the tibial tray in the test machine. The main
proximal planar surface shall be inclined at the posterior slope
6.1 The metallic components shall follow the complete
recommended by the manufacturer (see Fig. 1). If more than
manufacturing process (machining, surface treatment, laser
one slope is recommended, the largest slope should be used.
marking, passivation, cleaning, and so forth) until the steril-
Mount the bearing component on the tibial tray using the
ization stage. Because sterilization has no known effect on the
method recommended by the manufacturer.
mechanical properties for metallic components, it is not nec-
NOTE 1—The tibial slope will generate a shear force and a resulting
essary for these to be sterilized. The UHMWPE components
bending moment on the test frame actuator. This may cause a significant
shall be sterilized in a manner consistent with the clinical use
error of the load cell, depending on the sensitivity of the load cell to
for such devices, except when the mechanical properties of the
off-axis loading. This should be addressed in the test setup.
UHMWPE have been proven not to be detrimentally affected
7.5 Measure vertical distraction (when appropriate for the
by the sterilization process.
design) and bearing tilt (Fig. 2).
7.5.1 To measure the vertical distraction, use appropriately
6.2 The UHMWPE component(s) shall be artificially aged
sized feeler gauges, one set under each condyle to lift the
according to Practice F2003, except when the mechanical
properties of the UHMWPE have been proven not to be bearing away from the tibial plate, keeping the posterior
surface of the bearing parallel to the superior surface of the
detrimentally affected by aging.
tibial plate, until the gauges will not fit easily in the gap. The
6.3 Most of the knee systems allow the tibial tray to be
thickness of the feeler gauges is the vertical distraction value.
upsized, size for size, or downsized relative to the bearing
7.5.2 To measure the posterior bearing tilt displacement,
component size. Consistent with the principle of this test
push the bearing posteriorly and raise the posterior edge of the
method, the smallest tibial tray compatible with a given
bearing by hand. Select a location on the posterior edge of the
bearing component size (according to the manufacturer) shall
bearing and measure the perpendicular distance from that
be used.
location to the tibial plate. Change in these displacements after
6.4 There may be some small variation in the amount of
testing may be useful as an indicator of damage.
cold flow of the bearing component depending on the tibial
7.6 Mount the femoral component in the test machine with
bearing thickness. However, the possible effect of the cold flow
an alignment such that the component is flexed in the sagittal
is worst on the thinnest bearing components. Consequently, the
plane at the maximum flexion angle (including the posterior
thinnest bearing component in the knee system scope shall be
slope angle) the manufacturer recommends (see Fig. 3) accord-
used in this test.
ing to the method in Section 6.2.3 of Specification F2083.
7. Procedure
7.7 The femoral component should be placed so that it
contacts the bearing component close to the posterior edge of
7.1 On all samples, make the initial measurements on the
the bearing. The specific contact points between components
bearing surface to characterize the subsequent amount of
should be recorded and justified. At minimum, it should be
bearing deformation after completion of the test. Use of a
demonstrated that the anterior-posterior placement of the
coordinate measuring machine (CMM) or non-contact 3D
components would permit flexion and rotation of the femur to
measuring machine (for example, laser scanner, structured
the prescribed angles without impingement between the femur
light scanner, etc.) are the recommended methods of making
and tibia.
the measurements. The measurements should be in the form of
NOTE 2—If the mobile bearing knee design allows anterior-posterior
a grid of points, referenced to a fixed plane on the UHMWPE
translation of the mobile bearing, translate the bearing component
bearing, at a maximum of 1.5 mm apart over the entire superior
posteriorly relative to the tibial tray (according to the maximum transla-
surface of the UHMWPE bearing. The measurements should
tion allowed by the knee system) to simulate a worst-case condition.
be made with the bearing at 20 6 2 °C.
7.8 Initially align all components in neutral rotation to set
7.2 On one representative sample, perform the “A-P Draw
the maximum flexion angle. In this position, the femoral
Test” (Section 9.2) and the “Rotary Laxity Test” (Section 9.4) component, the bearing component, and the tib
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2777 − 16 F2777 − 23
Standard Test Method for
Evaluating Knee Bearing (Tibial Insert) Endurance and
Deformation Under High Flexion
This standard is issued under the fixed designation F2777; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This standard specifies a test method for determining the endurance properties and deformation, under specified laboratory
conditions, of ultra high molecular weight polyethylene (UHMWPE) tibial bearing components used in bicompartmental or
tricompartmental knee prosthesis designs.
1.2 This test method is intended to simulate near posterior edge loading similar to the type of loading that would occur during high
flexion motions such as squatting or kneeling.
1.3 Although the methodology described attempts to identify physiological orientations and loading conditions, the interpretation
of results is limited to an in vitro comparison between knee prosthesis designs and their ability to resist deformation and fracture
under stated test conditions.
1.4 This test method applies to bearing components manufactured from UHMWPE.
1.5 This test method could be adapted to address unicompartmental total knee replacement (TKR) systems, provided that the
designs of the unicompartmental systems have sufficient constraint to allow use of this test method. This test method does not
include instructions for testing two unicompartmental knees as a bicompartmental system.
1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of
the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
This test method is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.22 on Arthroplasty.
Current edition approved July 1, 2016Sept. 1, 2023. Published August 2016September 2023. Originally approved in 2010. Last previous edition approved in 20102016
as F2777F2777 – 16.–10. DOI: 10.1520/F2777-16.10.1520/F2777-23.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2777 − 23
2. Referenced Documents
2.1 ASTM Standards:
F1223 Test Method for Determination of Total Knee Replacement Constraint
F2003 Practice for Accelerated Aging of Ultra-High Molecular Weight Polyethylene After Gamma Irradiation in Air
F2083 Specification for Knee Replacement Prosthesis
2.2 Other Standards:
ISO 4965–14965-1 Metallic materials—DynamicMaterials—Dynamic Force Calibration for uniaxial fatigue testing —Part 1:
Testing systemUniaxial Fatigue Testing—Part 1: Testing System
ISO 5833 Implants for Surgery—Acrylic Resin Cements
ISO 14243-1 Implants for Surgery—Wear of Total Knee-joint Prostheses—Part 1: Loading and Displacement Parameters for
Wear-testing Machines with Load Control and Corresponding Environmental Conditions for Test
ISO 14243-3 Implants for Surgery—Wear of Total Knee-joint Prostheses—Part 3: Loading and Displacement Parameters for
Wear-testing Machines with Displacement Control and Corresponding Environmental Conditions for Test
3. Terminology
3.1 Definitions:
3.1.1 anatomic (mechanical) axis of the femur—the line between the center of the femoral head and the center of the femoral knee.
3.1.2 bearing centerline—the line running anteroposterior that is the mirror line of the femoral articulating surface. For
asymmetric bearing tibial traytibial bearing (tibial insert). For asymmetric tibial bearing designs, the appropriate tibial traybearing
centerline shall be determined and reported along with the rationale for the location.
3.1.3 bearing retention mechanism—mechanical means for preventing tibial tray/bearing disassociation.
3.1.4 femoral component centerline—a line running anteroposterior between the femoral condyles and parallel to the femoral
condyles. The line should be equidistant between the condyles. For asymmetric or non-parallel condyles designs, the appropriate
centerline shall be determined, and the rationale for that location reported.
3.1.5 fixed bearing system—a knee prosthesis system comprised of a femoral component and a tibial component, where the tibial
articulating surface is not intended to move relative to the tibial tray.
3.1.6 mobile bearing—bearing component—the component between fixed femoral and tibial knee components with an articulating
surface on both the inferior and superior sides.
3.1.7 mobile bearing knee system—a knee prosthesis system comprised of a femoral component, a tibial component, and a mobile
bearing component that can rotate and/or translate relative to the tibial component.
3.1.8 posterior slope—the angle that the perpendicular axis of the tibial tray makes when it is tilted posteriorly away from the tibial
axis (see Fig. 1).
3.1.9 R value—the ratio of the minimum force to the maximum force (that is, R = minimum force/maximum force).
3.1.10 tibial axis—nominal longitudinal axis of the tibia, which corresponds with the central axis of the medullary cavity of the
proximal tibia.
3.1.11 tibial tray/bearing disassociation—unrecoverable physical separation of the tibial bearing and tibial tray components as a
result of bearing distraction or tilting.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
F2777 − 23
FIG. 1 Incline of the Tibial Tray Relative to the Tibial Axis at the Recommended Angle (Posterior Slope)
3.1.12 tibial tray centerline—a line running anteroposterior that is the mirror line of the tibial articulating surface. For asymmetric
bearing tibial tray designs, the appropriate tibial tray centerline shall be determined and reported along with the rationale for the
location.
4. Significance and Use
4.1 This test method can be used to describe the effects of materials, manufacturing, and design variables on the fatigue/cyclic
creep performance of UHMWPE bearing components subject to substantial rotation in the transverse plane (relative to the tibial
tray) for a relatively large number of cycles.
4.2 The loading and kinematics of bearing component designs in vivo will, in general, differ from the loading and kinematics
defined in this test method. The results obtained here cannot be used to directly predict in vivo performance. However, this test
method is designed to enable comparisons between the fatigue performance of different bearing component designs when tested
under similar conditions.
4.3 The test described is applicable to any bicompartmental knee design, including mobile bearing knees that have mechanisms
in the tibial articulating component to constrain the posterior movement of the femoral component and a built-in retention
mechanism to keep the articulating component on the tibial plate.
5. Apparatus and Materials
5.1 Testing Machine, with the following characteristics:
5.1.1 A sinusoidal, dynamic-forcing waveform.
5.1.2 An error in applied force not greater than 62 % at the maximum force magnitude (in accordance with ISO 4965–1).4965-1).
5.1.3 Axial force peak representative of what could occur during daily activities of high flexion (about 2275 N). During the tests,
the values of the maximum and minimum forces shall be maintained to an accuracy of 62 % of the maximum force. The test shall
be stopped if this accuracy is not maintained.
5.1.4 The forcing accuracy must be maintained as bearing component deformation occurs.
5.1.5 Instrumentation to record the number of cycles.
F2777 − 23
5.2 Fixturing, with the following characteristics:
5.2.1 Means of mounting and enclosing the test specimens using a corrosion-resistant material that is capable of holding the
femoral component and tibial tray.
5.2.2 The fixtures shall maintain the tibial and femoral components in their required orientations for the duration of the test.
5.2.3 If necessary, bone cement (see ISO 5833) or a high-strength epoxy may be used to lock the femoral and tibial components
in their fixtures.
5.2.4 The test apparatus or fixture should allow the force to be applied through the center of the femoral component and ensure
equal force transmission through the medial and lateral condyles.condyles or offset medially as given by ISO 14243-1 and ISO
14243-3.
5.2.5 The test apparatus or fixture shall allow for varus-valgus self-alignment of the femoral or tibial component.
5.3 Fluid Medium:
5.3.1 The test assembly shall be immersed in deionized water at 37 6 2°C.2 °C.
5.3.2 Deionized water should be added as necessary to keep the test components at the test temperature for the duration of the test.
6. Specimen Selection
6.1 The metallic components shall follow the complete manufacturing process (machining, surface treatment, laser marking,
passivation, cleaning, and so forth) until the sterilization stage. Because sterilization has no known effect on the mechanical
properties for metallic components, it is not necessary for these to be sterilized. The UHMWPE components shall be sterilized in
a manner consistent with the clinical use for such devices, as this may affect except when the mechanical properties of the
material.UHMWPE have been proven not to be detrimentally affected by the sterilization process.
6.2 The UHMWPE component(s) shall be artificially aged according to Practice F2003, except when the mechanical properties
of the UHMWPE have been proven not to be detrimentally affected by aging.
6.3 Most of the knee systems allow the tibial tray to be upsized, size for size, or downsized relative to the bearing component size.
Consistent with the principle of this test method, the smallest tibial tray compatible with a given bearing component size (according
to the manufacturer) shall be used.
6.4 There may be some small variation in the amount of cold flow of the bearing component depending on the tibial bearing
thickness. However, the possible effect of the cold flow is worst on the thinnest bearing components. Consequently, the thinnest
bearing component in the knee system scope shall be used in this test.
7. Procedure
7.1 On one representative sample, all samples, make the initial measurements on the bearing surface to characterize the subsequent
amount of bearing deformation after completion of the test. Use of a Coordinate Measuring Machine (CMM) is the recommended
methodcoordinate measuring machine (CMM) or non-contact 3D measuring machine (for example, laser scanner, structured light
scanner, etc.) are the recommended methods of making the measurements. The measurements should be in the form of a grid of
points, referenced to a fixed plane on the UHMWPE bearing, at a maximum of 1.5 mm apart over the entire superior surface of
the UHMWPE bearing. The measurements should be made with the bearing at 20 6 2°C.6 2 °C.
7.2 On one representative sample, perform the “A-P Draw Test” (Section 9.2) and the “Rotary Laxity Test” (Section 9.4) from Test
Method F1223 at the same flexion angle used in 7.6 of this test method.
7.3 Condition the UHMWPE bearing in a deionized water environment at 37 6 2°C2 °C prior to initiation of the test for a long
enough time to bring the bearing into equilibrium with the fluid temperature.
F2777 − 23
7.4 Mount the tibial tray in the test machine. The main proximal planar surface shall be inclined at the posterior slope
recommended by the manufacturer (see Fig. 1). If more than one slope is recommended, the largest slope should be used. Mount
the bearing component on the tibial tray using the method recommended by the manufacturer.
NOTE 1—The tibial slope will generate a shear force and a resulting bending moment on the test frame actuator. This may cause a significant error of
the load cell, depending on the sensitivity of the load cell to off-axis loading. This should be addressed in the test setup.
7.5 Measure vertical distraction (when appropriate for the design) and bearing tilt (Fig. 2).
7.5.1 To measure the vertical distraction, use appropriately sized feeler gauges, one set under each condyle to lift the bearing away
from the tibial plate, keeping the posterior surface of the bearing parallel to the superior surface of the tibial plate, until the gauges
will not fit easily in the gap. The thickness of the feeler gauges is the vertical distraction value.
7.5.2 To measure the posterior bearing tilt displacement, push the bearing posteriorly and raise the posterior edge of the bearing
by hand. Select a location on the posterior edge of the bearing and measure the perpendicular distance from that location to the
tibial plate. Change in these displacements after testing may be useful as an indicator of damage.
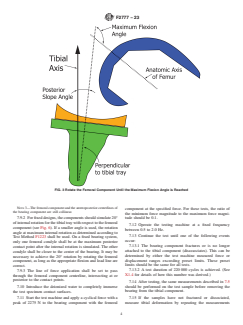
7.6 Mount the femoral component in the test machine with an alignment such that the component is flexed in the sagittal plane
at the maximum flexion angle (including the posterior slope angle) the manufacturer recommends (see Fig. 3) according to the
method in Section 6.2.3 of Specification F2083.
7.7 The femoral component should be placed so that it contacts the bearing component close to the posterior edge of the bearing.
The specific contact points between components should be recorded and justified. At minimum, it should be demonstrated that the
anterior-posterior placement of the components would permit flexion and rotation of the femur to the prescribed angles without
impingement between the femur and tibia.
NOTE 2—If the mobile bearing knee design allows anterior-posterior translation of the mobile bearing, translate the bearing component posteriorly relative
to the tibial tray (according to the maximum translation allowed by the knee system) to simulate a worst-case condition.
7.8 Initially align all components in neutral rotation to set the maximum flexion angle. In this position, the femoral component,
the bearing component, and the tibial tray should be aligned in the coronal plane according to the manufacturer’s intended neutral
alignment (see Fig. 4).
7.9 Rotational Alignment:
7.9.1 For mobile bearing knee system designs, simulate 20° of internal rotation for the tibial tray with respect to the femoral and
bearing components (see Fig. 5).
NOTE 3—The femoral component and the anteroposterior centerlines of the bearing component
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...