ASTM F326-23
(Test Method)Standard Test Method for Electronic Measurement for Hydrogen Embrittlement From Cadmium-Electroplating Processes
Standard Test Method for Electronic Measurement for Hydrogen Embrittlement From Cadmium-Electroplating Processes
SIGNIFICANCE AND USE
5.1 Hydrogen is evolved during metal electrodeposition in aqueous baths. Some of this hydrogen enters parts during plating. If the absorbed hydrogen is at a level presenting embrittlement hazards to high-strength steel, it is removed by baking parts after plating to expel this hydrogen. However, the lack of plate porosity itself may block hydrogen egress. Thus, it becomes important to know both the relative amount of hydrogen absorbed and the plate porosity.
5.2 This test provides a quantitative control number for cadmium plate porosity that can be used to control a cadmium plating process and the status of cadmium-plated hardware. It can also be used for plating process troubleshooting and research and development to determine the effects on plate porosity by process variables, contaminants, and materials. When used to control a critical process, control numbers for plate porosity must be determined by correlation with stress rupture specimens or other acceptable standards.
5.3 There is no prime standard for plate porosity. For this reason, two ovens must be used, with tests alternated between ovens. Data from the ovens are compared to ensure no equipment change has occurred.
SCOPE
1.1 This test method covers an electronic hydrogen detection instrument procedure for measurement of plating permeability to hydrogen. This method measures a variable related to hydrogen absorbed by steel during plating and to the hydrogen permeability of the plate during post plate baking. A specific application of this method is controlling cadmium-plating processes in which the plate porosity relative to hydrogen is critical, such as cadmium on high-strength steel.
1.2 The values stated in SI units are to be regarded as the standard. The values given in parentheses are for information only.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statement, see Section 8.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Dec-2022
- Technical Committee
- F07 - Aerospace and Aircraft
- Drafting Committee
- F07.04 - Hydrogen Embrittlement
Relations
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Nov-2018
- Effective Date
- 01-Dec-2017
- Effective Date
- 01-Mar-2017
- Effective Date
- 01-Dec-2013
- Effective Date
- 15-Nov-2012
- Effective Date
- 01-Jun-2012
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-May-2008
- Effective Date
- 01-Jul-2006
- Effective Date
- 01-Jul-2006
- Effective Date
- 01-Jul-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 01-Apr-2005
- Effective Date
- 26-Jun-2000
Overview
ASTM F326-23: Standard Test Method for Electronic Measurement for Hydrogen Embrittlement From Cadmium-Electroplating Processes provides an established electronic test method for detecting hydrogen embrittlement risk in steel components subjected to cadmium electroplating. Hydrogen embrittlement, often a critical concern for high-strength steels, can lead to premature failure if not properly controlled during plating processes. This standard describes an instrument-based procedure for measuring hydrogen permeability and absorbed hydrogen in cadmium-plated parts, offering quantitative data to assess process quality and plating integrity.
The method is particularly suited for process control, quality assurance, and troubleshooting of cadmium electroplating operations where the hydrogen retention characteristics of the plated layer are critical. The test also plays a vital role in correlating plating porosity and hydrogen egress with embrittlement hazards, complementing mechanical test methods.
Key Topics
Hydrogen Absorption in Plating: The standard addresses hydrogen evolution and absorption during metal electrodeposition in aqueous cadmium plating baths. Absorbed hydrogen presents embrittlement risks, especially in high-strength steels.
Electronic Detection Method: Utilizes a hydrogen detection instrument and specially designed probe for quantitative measurement of hydrogen permeability through cadmium plating after process baking. The test quantifies hydrogen content and plating porosity.
Porosity Control: By determining a plate porosity control number, the method helps manage cadmium plating processes to minimize hydrogen entrapment and ensure optimum plating performance.
Process Troubleshooting: The test can identify process variables, contaminants, and material choices influencing the risk of hydrogen embrittlement, supporting research and development as well as continuous process improvement.
Calibration and Equipment Correlation: To ensure reliability, the method requires calibration of the hydrogen detection apparatus and cross-verification using duplicate ovens to maintain consistency in results.
Applications
ASTM F326-23 is widely implemented across industries where the durability and safety of cadmium-plated, high-strength steel components are paramount. Key areas of application include:
Aerospace and Defense: Used to assess and control hydrogen embrittlement risk in fasteners, landing gear, and other safety-critical hardware.
Automotive Industry: Applied to plated components exposed to high loads or stress where hydrogen-induced delayed failure is a concern.
Manufacturing and Maintenance: Adopted for process control in plating facilities, ensuring the quality and compliance of cadmium-plated products.
Research & Development: Utilized for evaluating new plating processes, alternative materials, and process variables impacting hydrogen egress and plate porosity.
Employers and quality managers rely on this standard to document process capability, investigate failures, or verify that plating baths and operating procedures do not promote excessive hydrogen entrapment.
Related Standards
- ASTM F519: Mechanical Hydrogen Embrittlement Evaluation of Plating/Coating Processes and Service Environments
- ASTM D1193: Specification for Reagent Water - used for solution preparation and rinsing
- ISO Standards on Hydrogen Embrittlement: Comparative international methods in hydrogen risk evaluation
Practical Value
- Quality Assurance: Provides a quantitative, reproducible method to monitor and control hydrogen embrittlement risks due to cadmium electroplating.
- Risk Mitigation: Helps prevent catastrophic failures in service by proactively identifying plating conditions that retain hazardous hydrogen levels.
- Process Optimization: Enables process adjustments based on empirical data, enhancing manufacturing reliability and component longevity.
- Compliance: Supports industry conformance with international quality and testing requirements for hydrogen embrittlement control.
Keywords
cadmium electroplating, hydrogen embrittlement, hydrogen detection, plating porosity, ASTM F326-23, quality control, process control, steel, high-strength steel, aerospace, automotive, plating process verification
Buy Documents
ASTM F326-23 - Standard Test Method for Electronic Measurement for Hydrogen Embrittlement From Cadmium-Electroplating Processes
REDLINE ASTM F326-23 - Standard Test Method for Electronic Measurement for Hydrogen Embrittlement From Cadmium-Electroplating Processes
Get Certified
Connect with accredited certification bodies for this standard

National Aerospace and Defense Contractors Accreditation Program (NADCAP)
Global cooperative program for special process quality in aerospace.

CARES (UK Certification Authority for Reinforcing Steels)
UK certification for reinforcing steels and construction.

DVS-ZERT GmbH
German welding certification society.
Sponsored listings
Frequently Asked Questions
ASTM F326-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Electronic Measurement for Hydrogen Embrittlement From Cadmium-Electroplating Processes". This standard covers: SIGNIFICANCE AND USE 5.1 Hydrogen is evolved during metal electrodeposition in aqueous baths. Some of this hydrogen enters parts during plating. If the absorbed hydrogen is at a level presenting embrittlement hazards to high-strength steel, it is removed by baking parts after plating to expel this hydrogen. However, the lack of plate porosity itself may block hydrogen egress. Thus, it becomes important to know both the relative amount of hydrogen absorbed and the plate porosity. 5.2 This test provides a quantitative control number for cadmium plate porosity that can be used to control a cadmium plating process and the status of cadmium-plated hardware. It can also be used for plating process troubleshooting and research and development to determine the effects on plate porosity by process variables, contaminants, and materials. When used to control a critical process, control numbers for plate porosity must be determined by correlation with stress rupture specimens or other acceptable standards. 5.3 There is no prime standard for plate porosity. For this reason, two ovens must be used, with tests alternated between ovens. Data from the ovens are compared to ensure no equipment change has occurred. SCOPE 1.1 This test method covers an electronic hydrogen detection instrument procedure for measurement of plating permeability to hydrogen. This method measures a variable related to hydrogen absorbed by steel during plating and to the hydrogen permeability of the plate during post plate baking. A specific application of this method is controlling cadmium-plating processes in which the plate porosity relative to hydrogen is critical, such as cadmium on high-strength steel. 1.2 The values stated in SI units are to be regarded as the standard. The values given in parentheses are for information only. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statement, see Section 8. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Hydrogen is evolved during metal electrodeposition in aqueous baths. Some of this hydrogen enters parts during plating. If the absorbed hydrogen is at a level presenting embrittlement hazards to high-strength steel, it is removed by baking parts after plating to expel this hydrogen. However, the lack of plate porosity itself may block hydrogen egress. Thus, it becomes important to know both the relative amount of hydrogen absorbed and the plate porosity. 5.2 This test provides a quantitative control number for cadmium plate porosity that can be used to control a cadmium plating process and the status of cadmium-plated hardware. It can also be used for plating process troubleshooting and research and development to determine the effects on plate porosity by process variables, contaminants, and materials. When used to control a critical process, control numbers for plate porosity must be determined by correlation with stress rupture specimens or other acceptable standards. 5.3 There is no prime standard for plate porosity. For this reason, two ovens must be used, with tests alternated between ovens. Data from the ovens are compared to ensure no equipment change has occurred. SCOPE 1.1 This test method covers an electronic hydrogen detection instrument procedure for measurement of plating permeability to hydrogen. This method measures a variable related to hydrogen absorbed by steel during plating and to the hydrogen permeability of the plate during post plate baking. A specific application of this method is controlling cadmium-plating processes in which the plate porosity relative to hydrogen is critical, such as cadmium on high-strength steel. 1.2 The values stated in SI units are to be regarded as the standard. The values given in parentheses are for information only. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statement, see Section 8. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F326-23 is classified under the following ICS (International Classification for Standards) categories: 25.220.40 - Metallic coatings. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F326-23 has the following relationships with other standards: It is inter standard links to ASTM F519-23, ASTM F519-18, ASTM F519-17a, ASTM F519-17, ASTM F519-13, ASTM F519-12a, ASTM F519-12, ASTM F519-10, ASTM F519-08, ASTM F519-06, ASTM F519-06e2, ASTM F519-06e1, ASTM D1193-06, ASTM F519-05, ASTM F519-97e2. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F326-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F326 − 23
Standard Test Method for
Electronic Measurement for Hydrogen Embrittlement From
Cadmium-Electroplating Processes
This standard is issued under the fixed designation F326; the number immediately following the designation indicates the year of original
adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A superscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 3. Terminology
3.1 Definitions of Terms Specific to This Standard:
1.1 This test method covers an electronic hydrogen detec-
tion instrument procedure for measurement of plating perme-
3.1.1 hydrogen pressure peak—the maximum hydrogen
ability to hydrogen. This method measures a variable related to
pressure value (see I ) obtained when the probe is heated
H
hydrogen absorbed by steel during plating and to the hydrogen
following calibration, plating, or fluid testing.
permeability of the plate during post plate baking. A specific
3.2 Symbols:
application of this method is controlling cadmium-plating
3.2.1 HP = calibration hydrogen pressure peak.
processes in which the plate porosity relative to hydrogen is
3.2.2 HP = plating hydrogen pressure peak.
p
critical, such as cadmium on high-strength steel.
3.2.3 I or I = probe cathode emission current.
E e
1.2 The values stated in SI units are to be regarded as the
3.2.4 I = probe hydrogen pressure.
H
standard. The values given in parentheses are for information
3.2.5 I = integral of I curve from probe on to HP.
γ H
only.
3.2.6 lambda = time in seconds for hydrogen pressure peak
1.3 This standard does not purport to address all of the
to drop to half its value.
safety concerns, if any, associated with its use. It is the
3.2.7 λ = lambda obtained from a calibration run.
responsibility of the user of this standard to establish appro-
3.2.8 λ = lambda obtained from a plating run.
p
priate safety, health, and environmental practices and deter-
3.2.9 λ = normalized test lambda, obtained as follows:
pc
mine the applicability of regulatory limitations prior to use.
For specific hazard statement, see Section 8. λ 5 λ 40/λ (1)
~ !
pc p
1.4 This international standard was developed in accor-
¯
3.2.10 λ = arithmetic average of normalized lambdas for a
pc
dance with internationally recognized principles on standard-
set of tests.
ization established in the Decision on Principles for the
3.2.11 range = difference between maximum λ and mini-
pc
Development of International Standards, Guides and Recom-
mum λ for a given set of tests.
pc
mendations issued by the World Trade Organization Technical
3.2.12 run = calibration or plating of a probe.
Barriers to Trade (TBT) Committee.
3.2.13 test = single evaluation of a plating solution for
hydrogen embrittlement determination; run using a previously
2. Referenced Documents
calibrated probe.
2.1 ASTM Standards:
3.2.14 set of tests—all consecutive tests on a plating solu-
D1193 Specification for Reagent Water
tion for a given operator-instrument-day evaluation.
F519 Test Method for Mechanical Hydrogen Embrittlement
3.2.15 window—test surface of a probe described in Fig.
Evaluation of Plating/Coating Processes and Service En-
vironments 1(A).
4. Summary of Test Method
This test method is under the jurisdiction of ASTM Committee F07 on
4.1 This method uses a metal-shelled vacuum probe as an
Aerospace and Aircraft and is the direct responsibility of Subcommittee F07.04 on
ion gage to evaluate electrodeposited cadmium characteristics
Hydrogen Embrittlement.
relative to hydrogen permeation. After calibration, a section of
Current edition approved Jan. 1, 2023. Published February 2023. Originally
the probe shell is electroplated at the lowest current density
approved in 1978. Last previous edition approved in 2017 as F326 – 17. DOI:
10.1520/F0326-23.
encountered in the cadmium electroplating process. During the
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
subsequent baking of the probe at a closely controlled
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
temperature, the probe ion current, proportional to hydrogen
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. pressure, is recorded as a function of time. From these data and
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F326 − 23
FIG. 1 Probe Configuration
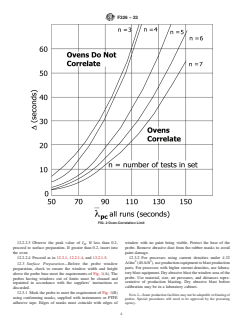
the calibration data of the probe, a number related to the ovens. Data from the ovens are compared to ensure no
porosity of the electroplated metal relative to hydrogen is equipment change has occurred.
obtained.
6. Apparatus
4.2 During the initial part of the bakeout, hydrogen contin-
6.1 Hydrogen Detection Instrument—A system consisting of
ues to diffuse through the metal shell of the probe and the ion
a control unit, two special ovens, auxiliary heater, recorder, test
current increases. Within a short time, however, a maximum
probes, and associated equipment.
current is observed and then falls off as hydrogen is driven out
of the system.
6.2 Oven—The oven warms the probe to increase the
hydrogen diffusion rate into the probe. Oven parameters are
4.3 Observations of the ion current-time curve indicate that
selected by apparatus manufacturer to provide a standard
the slope of the curve has an empirical relationship with failure
reading for all hydrogen detection instruments.
data on stress rupture specimens such as those in Test Method
F519. For this method, HP and λ variables (see Section 3) must
6.3 Oven Stopper—Stopper covering the oven opening.
be empirically correlated with results from the stress rupture
Remove 10 s before inserting the probe.
specimens. This gives a quick means of measuring ease of
6.4 Window—The window is the unpainted, bare steel
baking hydrogen out of cadmium-electroplated parts.
portion of the probe, 0.63 in. 6 0.03 in. in height, that is plated
4.4 Before an electroplating test, calibration is accom-
in the solution under test. The window is shown in Fig. 1.
plished by electrolyzing the probe in a standard solution and
6.5 Abrasive Blast—Abrasive blast window area in the same
baking it to determine HP and λ of the unplated steel shell of
way, using the same media, as used for the parts. Probe should
the probe.
be rotated while being blasted to provide uniform surface.
5. Significance and Use 6.6 Electronic Bakeout Unit—This heats the probe electri-
cally to remove hydrogen absorbed into the probe after testing.
5.1 Hydrogen is evolved during metal electrodeposition in
May be part of hydrogen detection instrument.
aqueous baths. Some of this hydrogen enters parts during
plating. If the absorbed hydrogen is at a level presenting
7. Reagents and Materials
embrittlement hazards to high-strength steel, it is removed by
7.1 Reagents:
baking parts after plating to expel this hydrogen. However, the
7.1.1 Purity of Reagents—Reagent grade chemicals shall be
lack of plate porosity itself may block hydrogen egress. Thus,
used in all tests. Unless otherwise indicated, it is intended that
it becomes important to know both the relative amount of
all reagents conform to the specifications of the Committee on
hydrogen absorbed and the plate porosity.
Analytical Reagents of the American Chemical Society where
5.2 This test provides a quantitative control number for
such specifications are available. Other grades may be used,
cadmium plate porosity that can be used to control a cadmium
provided it is first ascertained that the reagent is of sufficient
plating process and the status of cadmium-plated hardware. It
high purity to permit its use without lessening the accuracy of
can also be used for plating process troubleshooting and
the determination.
research and development to determine the effects on plate
porosity by process variables, contaminants, and materials.
When used to control a critical process, control numbers for
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
plate porosity must be determined by correlation with stress
Standard-Grade Reference Materials, American Chemical Society, Washington,
rupture specimens or other acceptable standards. DC. For suggestions on the testing of reagents not listed by the American Chemical
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
5.3 There is no prime standard for plate porosity. For this
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
reason, two ovens must be used, with tests alternated between copeial Convention, Inc. (USPC), Rockville, MD.
F326 − 23
−7
7.1.2 Acetone (C H O), technical. 11.5 Probe I : 1 I unit = 10 A
3 6 H H
7.1.3 Anode Cleaning Solution—Concentrated nitric acid Linearity, 62 % full scale within each
(HNO ), reagent grade. range, 1 to 10 000
7.1.4 Cadmium Stripping Solution—Ammonium Nitrate
11.6 Ovens—Ovens are calibrated by the manufacturers
(125 g/L)—Dissolve 125 g of ammonium nitrate (NH NO ,
4 3
against standard ovens that in turn were calibrated with
technical) in water and dilute to 1 L. Use at room temperature.
notched tension specimen data. Oven stability is checked by
7.1.5 Calibration Solution—Sodium Cyanide (50 g/L) Plus
comparing ovens against each other in duplicate tests.
Sodium Hydroxide (50 g/L)—Dissolve 50 g of sodium hydrox-
11.7 Correlation of Ovens—To correlate ovens, determine
ide (NaOH) in water. Add 50 g of sodium cyanide (NaCN) and
¯
λ for all tests of a set (except tests discarded in accordance
dissolve. Dilute to 1 L. Use at 18 °C to 27 °C (65 °F to 80 °F).
pc
¯
with 13.4.4). From λ and the number of tests, determine Δ
7.1.6 Water, Distilled or Deionized, minimum electrical
pc
¯
from Fig. 2. Separate data and compute λ for each oven. Let
resistivity 50 000 Ω·cm (for example, Specification D1193).
pc
¯ ¯
λ (A) be the higher value and λ (B) the lower value. Where
pc pc
7.2 Materials:
¯ ¯
λ (A) − λ (B) is less than Δ, the ovens are comparable.
pc pc
7.2.1 Anodes (Calibration), solid-carbon arc rods, 5.1 mm
¯ ¯
Where λ (A) − λ (B) is greater than Δ, the ovens are not
pc pc
to 12.7 mm (0.20 in. to 0.50 in.) diameter.
comparable.
7.2.2 Anodes (Plating), cadmium rods, A-A-51126 6.4 mm
to 12.7 mm (0.25 in. to 0.50 in.) thick, round or square.
12. Procedure
7.2.3 Polytetrafluoroethylene (PTFE) Tape—The tape
should be appropriate for use in solution, width about 12 mm 12.1 Bakeout of Probe:
to 19 mm, thickness small enough to seal.
12.1.1 Strip cadmium-plated probes in stripping solution
7.2.4 Glass 1-L Beaker.
(7.1.4) and rinse in 50 °C (122 °F) water for 2 min before
bakeout.
8. Hazards
12.1.2 Insert a probe into the socket of an electronic bakeout
unit.
8.1 Sodium cyanide, cyanide, cadmium, nitric acid, and
acetone can be health hazards. Use adequate face, hands, and 12.1.3 Within 30 s, the Bakeout cathode current should
respiratory protection commensurate with standards estab- reach 86.5 mA 6 16.5 mA. If the Bakeout cathode current does
lished by American Conference of Government and Industrial not maintain this current, the probe may not be used.
Hygiene for these chemicals.
12.1.4 Bake out the probe for the time required to meet the
limits in 12.2. Do not continuously bake out probes for longer
9. Sampling
than 2 h to preclude damaging paint.
9.1 Stir plating bath to ensure homogeneity. The plating
12.2 Probe Checkout—Probes that are new, or have been
bath sample must be representative of the bath. Obtain the
calibrated or plated and stripped, need to be baked out to meet
sample from beneath the surface of the bath, not by skimming
checkout requirements as follows:
the surface. Chemical constituents must be within normal
12.2.1 Hot Probe:
operating range.
12.2.1.1 Set the range to 10.
NOTE 1—Here and throughout the specification, range settings are for
10. Preparation of Apparatus
full-scale reading.
10.1 Plug in instrument and allow sufficient time for war-
12.2.1.2 Remove the probe from the electronic bakeout unit;
mup.
plug into the socket assembly and 15 s 6 1 s after removal
10.2 Turn on the oven and allow 4 h for warmup.
from the bakeout unit, turn the probe on.
10.3 Leave the instrument on continuously.
12.2.1.3 Observe the peak value of I . If less than 1,
H
proceed with surface activation. If it is greater than 1.0, screw
10.4 Clean contaminated anodes in cleaning solution,
on the cap and insert probe into the oven.
(7.1.3) until heavy gassing is observed. (Warning—See Sec-
12.2.1.4 If I is 0.5 or less within 5 min of inserting the
tion 8.)
H
probe into the oven, proceed to surface preparation. If the
probe does not drop to I = 0.5 or less with 5 min, bake out
11. Calibration of Apparatus
H
2 2 2 again. If three successive bakeouts do not reduce I to 0.5 or
H
11.1 Calibration Position, 1.08 A/dm 6 0.2 A/dm (10 A/ft
less within 5 min of insertion into the oven, discard the probe.
6 2 A/ft )—Use nominal dimensions of Fig. 1(A) for current
12.2.1.5 Set the instrument to read I . Probe I should read
E E
calculations.
6.0 mA 6 0.2 mA. If I does not read or cannot be adjusted to
E
11.2 Plating Position, 62 % of Current—Set plating current
this, the probe or the instrument is defective. Check the
density at the minimum value allowed by the plating specifi-
instrument with other probes to determine which is defective.
cation.
Discard defective probes.
11.3 Probe Current, I , 6 mA 6 0.2 mA.
12.2.2 Cold Probe:
E
12.2.2.1 Set the range to 1.0.
11.4 Electronic Probe Bakeout, Cathode current, 80 mA to
103 mA. 12.2.2.2 Plug the probe into socket assembly and turn on.
F326 − 23
FIG. 2 Oven-Correlation Limit
12.2.2.3 Observe the peak value of I . If less than 0.2, window with no paint being visible. Protect the base of the
H
proceed to surface preparation. If greater than 0.2, insert into probe. Remove abrasive dust from the rubber masks to avoid
the oven.
paint damage.
12.2.2.4 Proceed as in 12.2.1, 12.2.1.4, and 12.2.1.5.
12.3.2 For processes using current densities under 4.32
2 2
A/dm (40 A/ft ), use production equipment to blast production
12.3 Surface Preparation—Before the probe window
parts. For processes with higher current densities, use labora-
preparation, check to ensure the window width and height
tory blast equipment. Dry abrasive blast the window area of the
above the probe base meet the requirements of Fig. 1(A). The
probe. Use material, size, air pressures, and distances repre-
probes having windows out of limits must be cleaned and
sentative of production blasting. Dry abrasive blast before
repainted in accordance with the suppliers’ instructions or
calibration may be in a laboratory cabinet.
discarded.
12.3.1 Mask the probe to meet the requirement of Fig. 1(B)
NOTE 2—Some production facilities may not be adaptable to blasting of
using conforming masks, supplied with instruments or PTFE
probes. Special procedures will need to be approved by the procuring
adhesive tape. Edges of masks must coincide with edges of agency.
F326 − 23
12.3.3 Remove conformal blasting masks, ensuring that the time the probe is on. Insert the probe into the oven 10 s from
window area is not touched. Remove loose
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F326 − 17 F326 − 23
Standard Test Method for
Electronic Measurement for Hydrogen Embrittlement From
Cadmium-Electroplating Processes
This standard is issued under the fixed designation F326; the number immediately following the designation indicates the year of original
adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A superscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method covers an electronic hydrogen detection instrument procedure for measurement of plating permeability to
hydrogen. This method measures a variable related to hydrogen absorbed by steel during plating and to the hydrogen permeability
of the plate during post plate baking. A specific application of this method is controlling cadmium-plating processes in which the
plate porosity relative to hydrogen is critical, such as cadmium on high-strength steel.
1.2 The values stated in SI units are to be regarded as the standard. The values given in parentheses are for information only.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. For specific hazard statement, see Section 8.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
F519 Test Method for Mechanical Hydrogen Embrittlement Evaluation of Plating/Coating Processes and Service Environments
3. Terminology
3.1 Definitions of Terms Specific to This Standard:
3.1.1 hydrogen pressure peak—the maximum hydrogen pressure value (see I ) obtained when the probe is heated following
H
calibration, plating, or fluid testing.
3.2 Symbols:
3.2.1 HP = calibration hydrogen pressure peak.
3.2.2 HP = plating hydrogen pressure peak.
p
This test method is under the jurisdiction of ASTM Committee F07 on Aerospace and Aircraft and is the direct responsibility of Subcommittee F07.04 on Hydrogen
Embrittlement.
Current edition approved Dec. 1, 2017Jan. 1, 2023. Published January 2018February 2023. Originally approved in 1978. Last previous edition approved in 20122017 as
F326 – 96 (2012).F326 – 17. DOI: 10.1520/F0326-17.10.1520/F0326-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F326 − 23
3.2.3 I or I = probe cathode emission current.
E e
3.2.4 I = probe hydrogen pressure.
H
3.2.5 I = integral of I curve from probe on to HP.
γ H
3.2.6 lambda = time in seconds for hydrogen pressure peak to drop to half its value.
3.2.7 λ = lambda obtained from a calibration run.
3.2.8 λ = lambda obtained from a plating run.
p
3.2.9 λ = normalized test lambda, obtained as follows:
pc
λ 5 λ ~40/λ! (1)
pc p
¯
3.2.10 λ = arithmetic average of normalized lambdas for a set of tests.
pc
3.2.11 range = difference between maximum λ and minimum λ for a given set of tests.
pc pc
3.2.12 run = calibration or plating of a probe.
3.2.13 test = single evaluation of a plating solution for hydrogen embrittlement determination; run using a previously calibrated
probe.
3.2.14 set of tests—all consecutive tests on a plating solution for a given operator-instrument-day evaluation.
3.2.15 window—test surface of a probe described in Fig. 1(A).
4. Summary of Test Method
4.1 This method uses a metal-shelled vacuum probe as an ion gage to evaluate electrodeposited cadmium characteristics relative
to hydrogen permeation. After calibration, a section of the probe shell is electroplated at the lowest current density encountered
in the cadmium electroplating process. During the subsequent baking of the probe at a closely controlled temperature, the probe
ion current, proportional to hydrogen pressure, is recorded as a function of time. From these data and the calibration data of the
probe, a number related to the porosity of the electroplated metal relative to hydrogen is obtained.
4.2 During the initial part of the bakeout, hydrogen continues to diffuse through the metal shell of the probe and the ion current
increases. Within a short time, however, a maximum current is observed and then falls off as hydrogen is driven out of the system.
4.3 Observations of the ion current-time curve indicate that the slope of the curve has an empirical relationship with failure data
FIG. 1 Probe Configuration
F326 − 23
on stress rupture specimens such as those in Test Method F519. For this method, IHP and λ variables (see Section 3) must be
γ
empirically correlated with results from the stress rupture specimens. This gives a quick means of measuring ease of baking
hydrogen out of cadmium-electroplated parts.
4.4 Before an electroplating test, calibration is accomplished by electrolyzing the probe in a standard solution and baking it to
determine IHP and λ of the unplated steel shell of the probe.
γ
5. Significance and Use
5.1 Hydrogen is evolved during metal electrodeposition in aqueous baths. Some of this hydrogen enters parts during plating. If
the absorbed hydrogen is at a level presenting embrittlement hazards to high-strength steel, it is removed by baking parts after
plating to expel this hydrogen. However, the lack of plate porosity itself may block hydrogen egress. Thus, it becomes important
to know both the relative amount of hydrogen absorbed and the plate porosity.
5.2 This test provides a quantitative control number for cadmium plate porosity that can be used to control a cadmium plating
process and the status of cadmium-plated hardware. It can also be used for plating process troubleshooting and research and
development to determine the effects on plate porosity by process variables, contaminants, and materials. When used to control
a critical process, control numbers for plate porosity must be determined by correlation with stress rupture specimens or other
acceptable standards.
5.3 There is no prime standard for plate porosity. For this reason, two ovens must be used, with tests alternated between ovens.
Data from the ovens are compared to ensure no equipment change has occurred.
6. Apparatus
6.1 Hydrogen Detection Instrument—A system consisting of a control unit, two special ovens, auxiliary heater, recorder, test
probes, and associated equipment.
6.2 Oven—The oven warms the probe to increase the hydrogen diffusion rate into the probe. Oven parameters are selected by
apparatus manufacturer to provide a standard reading for all hydrogen detection instruments.
6.3 Oven Stopper—Stopper covering the oven opening. Remove 10 s 10 s before inserting the probe.
6.4 Window—The window is the unpainted, bare steel portion of the probe, 0.63 6 0.03 in. 0.63 in. 6 0.03 in. in height, that is
plated in the solution under test. The window is shown in Fig. 1.
6.5 Abrasive Blast—Abrasive blast window area in the same way, using the same media, as used for the parts. Probe should be
rotated while being blasted to provide uniform surface.
6.6 Electronic Bakeout Unit—This heats the probe electrically to remove hydrogen absorbed into the probe after testing. May be
part of hydrogen detection instrument.
7. Reagents and Materials
7.1 Reagents:
7.1.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society where such
specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficient high purity
to permit its use without lessening the accuracy of the determination.
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by
the American Chemical Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade
Reference Materials, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society, see Analar
Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc.
(USPC), Rockville, MD.
F326 − 23
7.1.2 Acetone (C H O), technical.
3 6
7.1.3 Anode Cleaning Solution—Concentrated nitric acid (HNO ), reagent grade.
7.1.4 Cadmium Stripping Solution—Ammonium Nitrate (125 g/L)—Dissolve 125 g 125 g of ammonium nitrate (NH NO ,
4 3
technical) in water and dilute to 1 L. 1 L. Use at room temperature.
7.1.5 Calibration Solution—Sodium Cyanide (50 g/L) Plus Sodium Hydroxide (50 g/L)—Dissolve 50 g 50 g of sodium hydroxide
(NaOH) in water. Add 50 g of sodium cyanide (NaCN) and dissolve. Dilute to 1 L. Use at 18 to 27°C (65 to 80°F).18 °C to 27 °C
(65 °F to 80 °F).
7.1.6 Water, Distilled or Deionized, minimum electrical resistivity 50 000 Ω·cm (for example, Specification D1193).
7.2 Materials:
7.2.1 Anodes (Calibration), solid-carbon arc rods, 5.1 to 12.7 mm (0.20 to 0.50 in.) 5.1 mm to 12.7 mm (0.20 in. to 0.50 in.)
diameter.
7.2.2 Anodes (Plating), cadmium rods, A-A-51126 6.4 to 12.7 mm (0.25 to 0.50 in.) 6.4 mm to 12.7 mm (0.25 in. to 0.50 in.) thick,
round or square.
7.2.3 Polytetrafluoroethylene (PTFE) Tape—The tape should be appropriate for use in solution, width about 12 to 19 mm, 12 mm
to 19 mm, thickness small enough to seal.
7.2.4 Glass 1-L Beaker.
8. Hazards
8.1 Sodium cyanide, cyanide, cadmium, nitric acid, and acetone can be health hazards. Use adequate face, hands, and respiratory
protection commensurate with standards established by American Conference of Government and Industrial Hygiene for these
chemicals.
9. Sampling
9.1 Stir plating bath to ensure homogeneity. The plating bath sample must be representative of the bath. Obtain the sample from
beneath the surface of the bath, not by skimming the surface. Chemical constituents must be within normal operating range.
10. Preparation of Apparatus
10.1 Plug in instrument and allow sufficient time for warmup.
10.2 Turn on the oven and allow 4 h 4 h for warmup.
10.3 Leave the instrument on continuously.
10.4 Clean contaminated anodes in cleaning solution, (7.1.3) until heavy gassing is observed. (Warning—See Section 8.)
11. Calibration of Apparatus
2 2 2 2
11.1 Calibration Position, 1.08 A/dm 6 0.2 A/dm (10 A/ft 6 2 A/ft )—Use nominal dimensions of Fig. 1(A) for current
calculations.
11.2 Plating Position, 62 % of Current—Set plating current density at the minimum value allowed by the plating specification.
11.3 Probe Current, I , 6 mA 6 0.2 mA.
E
F326 − 23
11.4 Electronic Probe Bakeout, Cathode current, 100 6 1080 mA to 103 mA.
−7
11.5 Probe I : 1 I unit = 10 A
H H
Linearity, 62 % full scale within each
range, 1 to 10 000
11.6 Ovens—Ovens are calibrated by the manufacturers against standard ovens that in turn were calibrated with notched tension
specimen data. Oven stability is checked by comparing ovens against each other in duplicate tests.
¯
11.7 Correlation of Ovens—To correlate ovens, determine λ for all tests of a set (except tests discarded in accordance with
pc
¯ ¯ ¯
13.4.4). From λ and the number of tests, determine Δ from Fig. 2. Separate data and compute λ for each oven. Let λ (A) be
pc pc pc
¯ ¯ ¯ ¯
the higher value and λ (B) the lower value. Where λ (A) − λ (B) is less than Δ, the ovens are comparable. Where λ (A) −
pc pc pc pc
¯
λ (B) is greater than Δ, the ovens are not comparable.
pc
FIG. 2 Oven-Correlation Limit
F326 − 23
12. Procedure
12.1 Bakeout of Probe:
12.1.1 Strip cadmium-plated probes in stripping solution (7.1.4) and rinse in 50°C (122°F)50 °C (122 °F) water for 2 min before
bakeout.
12.1.2 Insert a probe into the socket of an electronic bakeout unit.
12.1.3 Within 30 s, the heater should stabilize or be adjusted to 86.5 Bakeout cathode current should reach 86.5 mA 6 16.5 mA.
If the heater Bakeout cathode current does not register maintain this current, the probe is defective and must be discarded.may not
be used.
12.1.4 Bake out the probe for the time required to meet the limits in 12.2. Do not continuously bake out probes for longer than
2 h to preclude damaging paint.
12.2 Probe Checkout—Probes that are new, or have been calibrated or plated and stripped, need to be baked out to meet checkout
requirements as follows:
12.2.1 Hot Probe:
12.2.1.1 Set the range to 10.
NOTE 1—Here and throughout the specification, range settings are for full-scale reading.
12.2.1.2 Remove the probe from the electronic bakeout unit; plug into the socket assembly and 15 s 6 1 s after removal from the
bakeout unit, turn the probe on.
12.2.1.3 Observe the peak value of I . If less than 1, proceed with surface activation. If it is greater than 1.0, screw on the cap
H
and insert probe into the oven.
12.2.1.4 If I is 0.5 or less within 5 min of inserting the probe into the oven, proceed to surface preparation. If the probe does
H
not drop to I = 0.5 or less with 5 min, bake out again. If three successive bakeouts do not reduce I to 0.5 or less within 5 min
H H
of insertion into the oven, discard the probe.
12.2.1.5 Set the instrument to read I . Probe I should read 6.0 6 0.2 mA. 6.0 mA 6 0.2 mA. If I does not read or cannot be
E E E
adjusted to this, the probe or the instrument is defective. Check the instrument with other probes to determine which is defective.
Discard defective probes.
12.2.2 Cold Probe:
12.2.2.1 Set the range to 1.0.
12.2.2.2 Plug the probe into socket assembly and turn on.
12.2.2.3 Observe the peak value of I . If less than 0.2, proceed to surface preparation. If greater than 0.2, insert into the oven.
H
12.2.2.4 Proceed as in 12.2.1, 12.2.1.4, and 12.2.1.5.
12.3 Surface Preparation—Before the probe window preparation, check to ensure the window width and height above the probe
base meet the requirements of Fig. 1(A). The probes having windows out of limits must be cleaned and repainted in accordance
with the suppliers’ instructions or discarded.
12.3.1 Mask the probe to meet the requirement of Fig. 1(B) using conforming masks, supplied with instruments or PTFE adhesive
tape. Edges of masks must coincide with edges of window with no paint being visible. Protect the base of the probe. Remove
abrasive dust from the rubber masks to avoid paint damage.
F326 − 23
2 2
12.3.2 For processes using current densities under 4.32 A/dm (40 A/ft ), use production equipment to blast production parts. For
processes with higher current densities, use laboratory blast equipment. Dry abrasive blast the window area of the probe. Use
material, size, air pressures, and distances representative of production blasting. Dry abrasive blast before calibration may be in
a laboratory cabinet.
NOTE 2—Some production
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...