ASTM D8001-16e1
(Test Method)Standard Test Method for Determination of Total Nitrogen, Total Kjeldahl Nitrogen by Calculation, and Total Phosphorus in Water, Wastewater by Ion Chromatography
Standard Test Method for Determination of Total Nitrogen, Total Kjeldahl Nitrogen by Calculation, and Total Phosphorus in Water, Wastewater by Ion Chromatography
SIGNIFICANCE AND USE
5.1 This test method allows the simultaneous determination of total nitrogen and total phosphorous from one sample digestion step.
5.2 This test method measures oxidized ammonia and organic nitrogen (as nitrate) and soluble nitrate simultaneously. By subtracting the nitrate + nitrite value from a non-digested sample gives a TKN:
When using this test method:
where:
TN = total nitrogen, and TKN = total Kjeldahl nitrogen.
SCOPE
1.1 This test method is applicable for the analysis total nitrogen (organic nitrogen + ammonia-N + nitrate-N + nitrite-N) as nitrate and total phosphorus as orthophosphate in unfiltered water samples by alkaline persulfate digestion followed by ion chromatography (IC).
1.2 Total Kjeldahl nitrogen (TKN) is determined by the calculation. To determine TKN subtract the nitrate-N and nitrite-N in a digested sample from a non-digested sample (see Section 4, Summary of Test Method).
1.3 The limit of detection (LOD), limit of quantitation (LOQ), and reporting range in Table 1 are based on the two-step process for this test method: digestion and analytical step. Because the digestion step requires a sample dilution, the LOD and LOQ are higher than undigested samples. The reporting range, LOD, and LOQ can be modified and perhaps improved depending on several factors (see Section 6, Interferences). (A) Ten calibration levels, each injected in duplicate.(B) LOD calculated as 3 × S/N.(C) LOQ calculated as 10 × S/N.(D)
where:
A = the average method blank concentration, = the student’s t-value for the single-tailed 99th percentile t statistic a standard deviation estimate with n – 1 degrees of freedom, and Sb = the sample standard deviation of the replicate blank analyses. (E) Phosphate LOD/LOQ was calculated based on a dilution factor of 15× relative to the system concentrations.
1.4 The method detection limits (MDL) are shown for reference. The digestion reagent contains background nitrate and results in higher detection limits. MDL will be shown after the interlaboratory study (ILS) is completed.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 14-Jul-2016
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.06 - Methods for Analysis for Organic Substances in Water
Relations
- Effective Date
- 15-Jul-2016
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-May-2020
- Effective Date
- 15-Dec-2017
- Effective Date
- 15-Nov-2017
- Effective Date
- 01-Jan-2017
- Effective Date
- 01-Oct-2013
- Effective Date
- 15-Jun-2012
- Refers
ASTM D3856-11 - Standard Guide for Management Systems in Laboratories Engaged in Analysis of Water - Effective Date
- 15-Nov-2011
- Effective Date
- 01-May-2011
- Effective Date
- 01-Mar-2011
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Mar-2010
Overview
ASTM D8001-16e1 is a standard test method developed by ASTM International for the determination of total nitrogen (TN), total Kjeldahl nitrogen (TKN) by calculation, and total phosphorus (TP) in water and wastewater using ion chromatography (IC) after alkaline persulfate digestion. This method allows for accurate and simultaneous measurement of nitrogen and phosphorus forms from a single sample, supporting comprehensive water quality analysis crucial for environmental monitoring and regulatory compliance.
The standard is designed for use with unfiltered water samples, encompassing organic nitrogen, ammonia-nitrogen, nitrate-nitrogen, and nitrite-nitrogen as nitrate, and total phosphorus as orthophosphate. The TKN is determined by calculation, offering a modern and efficient alternative to traditional, more labor-intensive wet chemistry methods.
Key Topics
- Scope of Method: Applicable to the determination of TN, TKN (by calculation), and TP in unfiltered water and wastewater samples.
- Sample Digestion: Utilizes alkaline persulfate digestion to convert all nitrogen to nitrate and phosphorus to orthophosphate, prior to analysis by IC.
- Simultaneous Detection: Enables measurement of multiple nitrogen and phosphorus species from a single sample digestion step.
- Detection Limits & Reporting Range: Detection and quantitation limits are influenced by sample dilution during digestion and analytical procedures. Adjustments can be made depending on specific application needs and interferences encountered.
- Quality Control: Incorporates the use of method blanks, spike recoveries, and matrix spikes to ensure accuracy and precision within laboratory quality assurance protocols.
- Calculation of TKN: TKN is obtained by subtracting the nitrate and nitrite concentrations in a non-digested sample from the digested sample's total nitrogen value.
Applications
The ASTM D8001-16e1 standard finds broad application in:
- Environmental Monitoring: Critical for assessing nutrient loading in surface water, groundwater, and wastewater, supporting efforts to control eutrophication and monitor compliance with water quality standards.
- Wastewater Treatment Evaluation: Provides wastewater treatment plants with a rigorous method for tracking nitrogen and phosphorus removal efficiency, aiding in process optimization and regulatory reporting.
- Research and Laboratory Analysis: Suitable for academic, industrial, and government laboratories requiring reliable, standardized measurements of nitrogen and phosphorus species in water samples.
- Water Quality Compliance: Supports regulatory frameworks that mandate the monitoring of nutrient pollutants for the protection of aquatic environments.
The standardized approach and detailed quality assurance provisions make this method robust for use in routine laboratory settings, as well as in research and regulatory environments where data integrity is paramount.
Related Standards
ASTM D8001-16e1 is part of a suite of standards concerning water analysis and laboratory practices, including:
- ASTM D1129 - Terminology Relating to Water
- ASTM D1193 - Specification for Reagent Water
- ASTM D4327 - Test Method for Anions in Water by Suppressed Ion Chromatography
- ASTM D2777 - Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
- ASTM D3856 - Guide for Management Systems in Laboratories Engaged in Analysis of Water
- ASTM D5847 - Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
Utilizing these related standards alongside ASTM D8001-16e1 enhances laboratory best practices, ensuring accuracy, comparability, and reliability in water quality testing.
Keywords: ASTM D8001, total nitrogen, total phosphorus, total Kjeldahl nitrogen, nitrate, nitrite, ammonia, orthophosphate, ion chromatography, alkaline persulfate digestion, water analysis, wastewater testing, nutrient monitoring, environmental standards.
Buy Documents
ASTM D8001-16e1 - Standard Test Method for Determination of Total Nitrogen, Total Kjeldahl Nitrogen by Calculation, and Total Phosphorus in Water, Wastewater by Ion Chromatography
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D8001-16e1 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Total Nitrogen, Total Kjeldahl Nitrogen by Calculation, and Total Phosphorus in Water, Wastewater by Ion Chromatography". This standard covers: SIGNIFICANCE AND USE 5.1 This test method allows the simultaneous determination of total nitrogen and total phosphorous from one sample digestion step. 5.2 This test method measures oxidized ammonia and organic nitrogen (as nitrate) and soluble nitrate simultaneously. By subtracting the nitrate + nitrite value from a non-digested sample gives a TKN: When using this test method: where: TN = total nitrogen, and TKN = total Kjeldahl nitrogen. SCOPE 1.1 This test method is applicable for the analysis total nitrogen (organic nitrogen + ammonia-N + nitrate-N + nitrite-N) as nitrate and total phosphorus as orthophosphate in unfiltered water samples by alkaline persulfate digestion followed by ion chromatography (IC). 1.2 Total Kjeldahl nitrogen (TKN) is determined by the calculation. To determine TKN subtract the nitrate-N and nitrite-N in a digested sample from a non-digested sample (see Section 4, Summary of Test Method). 1.3 The limit of detection (LOD), limit of quantitation (LOQ), and reporting range in Table 1 are based on the two-step process for this test method: digestion and analytical step. Because the digestion step requires a sample dilution, the LOD and LOQ are higher than undigested samples. The reporting range, LOD, and LOQ can be modified and perhaps improved depending on several factors (see Section 6, Interferences). (A) Ten calibration levels, each injected in duplicate.(B) LOD calculated as 3 × S/N.(C) LOQ calculated as 10 × S/N.(D) where: A = the average method blank concentration, = the student’s t-value for the single-tailed 99th percentile t statistic a standard deviation estimate with n – 1 degrees of freedom, and Sb = the sample standard deviation of the replicate blank analyses. (E) Phosphate LOD/LOQ was calculated based on a dilution factor of 15× relative to the system concentrations. 1.4 The method detection limits (MDL) are shown for reference. The digestion reagent contains background nitrate and results in higher detection limits. MDL will be shown after the interlaboratory study (ILS) is completed. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 5.1 This test method allows the simultaneous determination of total nitrogen and total phosphorous from one sample digestion step. 5.2 This test method measures oxidized ammonia and organic nitrogen (as nitrate) and soluble nitrate simultaneously. By subtracting the nitrate + nitrite value from a non-digested sample gives a TKN: When using this test method: where: TN = total nitrogen, and TKN = total Kjeldahl nitrogen. SCOPE 1.1 This test method is applicable for the analysis total nitrogen (organic nitrogen + ammonia-N + nitrate-N + nitrite-N) as nitrate and total phosphorus as orthophosphate in unfiltered water samples by alkaline persulfate digestion followed by ion chromatography (IC). 1.2 Total Kjeldahl nitrogen (TKN) is determined by the calculation. To determine TKN subtract the nitrate-N and nitrite-N in a digested sample from a non-digested sample (see Section 4, Summary of Test Method). 1.3 The limit of detection (LOD), limit of quantitation (LOQ), and reporting range in Table 1 are based on the two-step process for this test method: digestion and analytical step. Because the digestion step requires a sample dilution, the LOD and LOQ are higher than undigested samples. The reporting range, LOD, and LOQ can be modified and perhaps improved depending on several factors (see Section 6, Interferences). (A) Ten calibration levels, each injected in duplicate.(B) LOD calculated as 3 × S/N.(C) LOQ calculated as 10 × S/N.(D) where: A = the average method blank concentration, = the student’s t-value for the single-tailed 99th percentile t statistic a standard deviation estimate with n – 1 degrees of freedom, and Sb = the sample standard deviation of the replicate blank analyses. (E) Phosphate LOD/LOQ was calculated based on a dilution factor of 15× relative to the system concentrations. 1.4 The method detection limits (MDL) are shown for reference. The digestion reagent contains background nitrate and results in higher detection limits. MDL will be shown after the interlaboratory study (ILS) is completed. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM D8001-16e1 is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D8001-16e1 has the following relationships with other standards: It is inter standard links to ASTM D8001-16, ASTM D6299-23a, ASTM D6792-23c, ASTM D6792-23b, ASTM D1129-13(2020)e2, ASTM D6299-17b, ASTM D6299-17a, ASTM D6299-17, ASTM D6299-13e1, ASTM D2777-12, ASTM D3856-11, ASTM D5810-96(2011), ASTM D4327-11, ASTM D6299-10e2, ASTM D6299-10. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D8001-16e1 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
´1
Designation: D8001 − 16
Standard Test Method for
Determination of Total Nitrogen, Total Kjeldahl Nitrogen by
Calculation, and Total Phosphorus in Water, Wastewater by
Ion Chromatography
This standard is issued under the fixed designation D8001; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Editorial changes were made throughout in September 2016.
1. Scope 2. Referenced Documents
2.1 ASTM Standards:
1.1 This test method is applicable for the analysis total
D1129Terminology Relating to Water
nitrogen (organic nitrogen + ammonia-N + nitrate-N +
D1193Specification for Reagent Water
nitrite-N) as nitrate and total phosphorus as orthophosphate in
D2777Practice for Determination of Precision and Bias of
unfiltered water samples by alkaline persulfate digestion fol-
Applicable Test Methods of Committee D19 on Water
lowed by ion chromatography (IC).
D3856Guide for Management Systems in Laboratories
1.2 Total Kjeldahl nitrogen (TKN) is determined by the
Engaged in Analysis of Water
calculation. To determine TKN subtract the nitrate-N and
D4327Test Method forAnions in Water by Suppressed Ion
nitrite-N in a digested sample from a non-digested sample (see
Chromatography
Section 4, Summary of Test Method).
D5847Practice for Writing Quality Control Specifications
for Standard Test Methods for Water Analysis
1.3 The limit of detection (LOD), limit of quantitation
D5810Guide for Spiking into Aqueous Samples
(LOQ), and reporting range in Table 1 are based on the
D6299Practice for Applying Statistical Quality Assurance
two-step process for this test method: digestion and analytical
and Control Charting Techniques to Evaluate Analytical
step. Because the digestion step requires a sample dilution, the
Measurement System Performance
LOD and LOQ are higher than undigested samples. The
D6792Practice for Quality System in Petroleum Products
reporting range, LOD, and LOQ can be modified and perhaps
and Lubricants Testing Laboratories
improved depending on several factors (see Section 6, Inter-
ferences).
3. Terminology
1.4 The method detection limits (MDL) are shown for
3.1 Definitions:
reference. The digestion reagent contains background nitrate
3.1.1 For definitions of terms used in this standard, refer to
andresultsinhigherdetectionlimits.MDLwillbeshownafter
Terminology D1129.
the interlaboratory study (ILS) is completed.
3.2 Definitions of Terms Specific to This Standard:
1.5 The values stated in SI units are to be regarded as
3.2.1 total Kjeldahl nitrogen (TKN), n—the sum of organic
standard. No other units of measurement are included in this
nitrogen plus ammonia (NH ).
standard.
3.2.2 total nitrogen (TN), n—the sum of all nitrate, nitrite,
1.6 This standard does not purport to address all of the
ammonia, and organic nitrogen, as N, in water or wastewater
safety concerns, if any, associated with its use. It is the
samples.
responsibility of the user of this standard to establish appro-
3.2.3 total phosphorus (TP), n—the sum of
priate safety and health practices and determine the applica-
orthophosphates, polyphosphates, and organically bound
bility of regulatory limitations prior to use.
phosphates, as P, in water or wastewater samples.
This test method is under the jurisdiction ofASTM Committee D19 on Water
andisthedirectresponsibilityofSubcommitteeD19.06onMethodsforAnalysisfor For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Organic Substances in Water. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Current edition approved July 15, 2016. Published August 2016. DOI: 10.1520/ Standards volume information, refer to the standard’s Document Summary page on
D8001-16E01. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
´1
D8001 − 16
TABLE 1 Calibration, Linearity, Limits of Detection, and Quantitation from the Single Lab Validation Study
System Digested Sample
A
Calibration Range
Linearity
B C
Analyte LOD LOQ
2 LOD LOQ
(r )
(µg/L)
(µg/L) (µg/L)
(µg/L) (µg/L)
Nitrite-N 2.5–300 0.9999 0.76 2.5 — —
D
Nitrate-N† 2.5–300 0.9999 1.0 3.4 171 171
E E
Phosphate-P 2.5–300 0.9998† 1.3 4.2 19.5 63
† Editorially corrected.
A
Ten calibration levels, each injected in duplicate.
B
LODcalculatedas3×S/N.
C
LOQ calculated as 10 × S/N.
D
Nitrate MDL 5 A1t S
b n 21,1 2 α 5 0.99 b
s d
where:
A = the average method blank concentration,
= the student’s t-value for the single-tailed 99th percentile t statistic a standard deviation estimate with n – 1 degrees of freedom, and
t
sn 21,1 2 α 5 0.99d
S = the sample standard deviation of the replicate blank analyses.
b
E
Phosphate LOD/LOQ was calculated based on a dilution factor of 15× relative to the system concentrations.
4. Summary of Test Method phosphate peaks. The use of columns with high capacity is
required to overcome these limitations.
4.1 Awater sample is digested with alkaline persulfate at a
6.2 Samples high in chloride from brackish, seawater and
2:1 ratio, the initial pH is >12. This sample is heated at 120°C
brines may also result in column overloading. Chloride is also
for 60 min. Initial alkaline conditions oxidize dissolved/
oxidized to chlorate during the digestion step, and thus
suspended nitrogen to nitrate. Over time the solution becomes
contributes to depletion of the persulfate digestion reagent.
acidic according to the following calculation:
These can either be diluted or pre-treated to remove excess
22 2 + -
S O 1H O→2HSO 1 O (1) chloride. Pretreatment usingAg precipitation or the use of Cl
2 8 2 4 2
removal cartridges are accepted for this test method. Dilution
The acidic conditions (pH ~2) result in the hydrolysis of
will increase the detection limits for total nitrogen and phos-
dissolved/suspended phosphorus to orthophosphate.
phate. The use of pretreatment cartridges may remove particu-
lates if performed prior to the digestion step, giving a possible
4.2 The determinative step using IC is equivalent to Test
negative bias.
Method D4327.
6.3 If very low µg/L concentrations are required, blank
5. Significance and Use
subtractionmaybeusedprovidedthespikerecoveriesmeetthe
methods detection limits. Approximately 92.5 µg/L nitrate
5.1 This test method allows the simultaneous determination
were found in the potassium persulfate digestion chemical.
of total nitrogen and total phosphorous from one sample
This test method provides an MDLcalculation where a peak is
digestion step.
foundintheblanksamples/digestionreagent.(SeeSection13.)
5.2 This test method measures oxidized ammonia and or-
6.4 High levels of organic carbon concentrations greater
ganic nitrogen (as nitrate) and soluble nitrate simultaneously.
than 800 mg/L of TOC, reducing agents, reduced forms of
By subtracting the nitrate + nitrite value from a non-digested
metals, etc. will consume the oxidative reagent that may limit
sample gives a TKN:
oxidationofreducednitrogenandphosphorous.(SeeFig.6and
TN 5TKN1 NO -N 1 NO -N (2)
~ ! ~ !
3 2
Table 6.)
TKN 5NH -N1OrganicN (3)
7. Instrumentation
When using this test method:
7.1 Digestion Step—Many techniques exist for heated di-
TKN 5DigestedSample 2Non-DigestedSample (4)
gestion of water samples. Regardless of the instrumentation
used, such as UV or microwave, the digestion must proceed
TKN 5TN 2 @NO -N 1 NO -N# (5)
3 2
long enough to consume all persulfate.
where:
7.1.1 Autoclave or heating block or alternative, capable of
TN = total nitrogen, and
120°C for 60 minutes.
TKN = total Kjeldahl nitrogen.
7.2 Digestion Tubes—OD × L: 16 × 125 mm disposable
glass tubes with screw caps.
6. Interferences
7.3 Analytical balance, capable of weighing up to 200 g
6.1 Interferences can be caused by substances with similar
accurately to 60. 01 g.
ion chromatographic retention times, especially if they are in
highconcentrationcomparedtotheanalyteofinterest.Follow- 7.4 Pipettes or Volumetric Transfer—1- and 5-mL Class A
ing digestion, samples contain high concentrations of sulfate volumetric pipettes or calibrated variable volume automatic
that can cause column overloading and obscure nitrate and pipettes fitted with disposable polypropylene tips.
´1
D8001 − 16
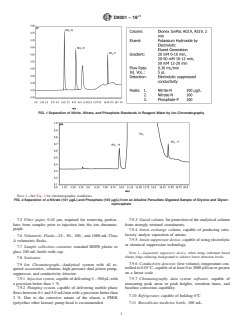
FIG. 1 Separation of Nitrite, Nitrate, and Phosphate Standards in Reagent Water by Ion Chromatography
NOTE 1—See Fig. 1 for chromatographic conditions.
FIG. 2 Separation of a Nitrate (101 µg/L) and Phosphate (105 µg/L) from an Alkaline Persulfate Digested Sample of Glycine and Glycer-
olphosphate
7.5 Filter paper, 0.45 µm, required for removing particu- 7.9.3 Guard column, for protection of the analytical column
lates from samples prior to injection into the ion chromato- from strongly retained constituents.
graph.
7.9.4 Anion exchange column, capable of producing satis-
factory analyte separation of anions.
7.6 Volumetric Flasks—25-, 50-, 100-, and 1000-mL Class
A volumetric flasks. 7.9.5 Anion suppressor device, capable of using electrolytic
or chemical suppression technology.
7.7 Sample collection container, standard HDPE plastic or
glass 100-mL bottle with cap.
NOTE 1—Sequential suppressor device, when using carbonate based
eluent, helps reducing background to achieve lower detection levels.
7.8 Sonicator.
7.9.6 Conductivity detector,(lowvolume),temperaturecon-
7.9 Ion Chromatograph—Analytical system with all re-
trolledto0.01°C,capableofatleast0to3000µS/cmorgreater
quired accessories, columns, high-pressure dual piston pump,
on a linear scale.
suppressor, and conductivity detector.
7.9.7 Chromatography data system software, capable of
7.9.1 Injectionsystem,capableofdelivering5–500µLwith
a precision better than 1 %. measuring peak areas or peak heights, retention times, and
baseline correction capability.
7.9.2 Pumping system, capable of delivering mobile phase
flows between 0.1 and 5.0 mL/min with a precision better than
7.10 Refrigerator, capable of holding 6°C
2 %. Due to the corrosive nature of the eluent, a PEEK
(polyether ether ketone) pump head is recommended. 7.11 Borosilicate medicine bottle, 100 mL.
´1
D8001 − 16
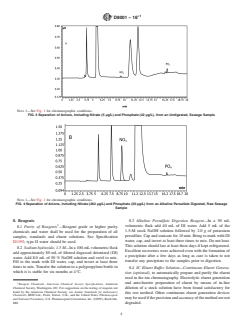
NOTE 1—See Fig. 1 for chromatographic conditions.
FIG. 3 Separation of Anions, Including Nitrate (5 µg/L) and Phosphate (22 µg/L), from an Undigested, Sewage Sample
NOTE 1—See Fig. 1 for chromatographic conditions.
FIG. 4 Separation of Anions, Including Nitrate (262 µg/L) and Phosphate (30 µg/L) from an Alkaline Persulfate Digested, Raw Sewage
Sample
8. Reagents 8.3 Alkaline Persulfate Digestion Reagent—Ina50mL
volumetric flask add 40 mL of DI water. Add 5 mL of the
8.1 Purity of Reagents —Reagent grade or higher purity
1.5-M stock NaOH solution followed by 2.0 g of potassium
chemicals and water shall be used for the preparation of all
persulfate.Capandsonicatefor10min.BringtomarkwithDI
samples, standards and eluent solutions. See Specification
water, cap, and invert at least three times to mix. Do not heat.
D1193; type II water should be used.
Thissolutionshouldlastatleastthreedaysifkeptrefrigerated.
8.2 Sodium hydroxide, 1.5 M—Ina100-mLvolumetricflask
Excellent recoveries were achieved even with the formation of
add approximately 80 mL of filtered degassed deionized (DI)
a precipitate after a few days as long as care is taken to not
water. Add 8.0 mL of 50 % NaOH solution and swirl to mix.
transfer any precipitate to the samples prior to digestion.
Fill to the mark with DI water, cap, and invert at least three
times to mix.Transfer the solution to a polypropylene bottle in
8.4 IC Eluent Buffer Solution—Continuous Eluent Genera-
which it is stable for six months at 4°C.
tion (optional), to automatically prepare and purify the eluent
used in the ion chromatography. Electrolytic eluent generation
and auto-burette preparation of eluent by means of in-line
Reagent Chemicals, American Chemical Society Specifications, American
Chemical Society, Washington, DC. For suggestions on the testing of reagents not dilution of a stock solution have been found satisfactory for
listed by the American Chemical Society, see Analar Standards for Laboratory
this test method. Other continuous eluent generation devices
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
maybeusediftheprecisionandaccuracyofthemethodarenot
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
MD. degraded.
´1
D8001 − 16
FIG. 5 Isocratic Separation of Nitrate (25 µg/L) and Phosphate (25 µg/L) from an Alkaline Persulfate Digested with Carbonate/
Bicarbonate Eluent
NOTE 1—See Fig. 1 for chromatographic conditions.
FIG. 6 Separation of Nitrate and Phosphate in the Presence of 1000 mg/L Chloride
8.5 IC Eluent Suppression Anion Suppressor Device, re- 9.1.1 Potassium Nitrate Stock Calibrant Solution, 1 mL
duces the background conductivity of the eluent after separa- =1.0 mg-N—Dissolve 0.72 g of potassium nitrate (KNO,FW
tionbytheanionseparatorcolumn.Bothchemical(sequential) = 101.1) in about 80 mL of DI water in a 100-mL volumetric
and continuous electrolytic suppressors have been found sat- flask. Dilute this solution to the mark with DI water and mix it
isfactory for this test method. Other anion suppressor devices thoroughly by manual inversion and shaking. Transfer the
may be used as long as the precision and accuracy of the stock calibrant to a 100-mLborosilicate media bottle in which
method are not degraded. it is stable for 6 months at 4°C (see Note 2).
9.1.2 Potassium Di-Hydrogen Phosphate Stock Calibrant
8.6 Suppressor Regeneration Solution (if needed)—Prepare
Solution, 1 mL =1.0 mg-P—Dissolve 0.44 g potassium di-
0.5-MSulfuricAcidSolutionbyadding28mLofconcentrated
hydrogen phosphate (KH PO , FW = 136.09) in about 80 mL
2 4
sulfuric acid into 1 L of DI water. Sulfuric acid is not needed
of DI water in a 100-mL volumetric flask. Dilute this solution
when using electrolytic eluent generation. (Warning—
to the mark with DI water and mix it thoroughly by manual
Solution will get hot, so use proper PPE while preparing this
inversion.Transferthestockcalibranttoa100-mLborosilicate
solution.)Alternatively, commercially available 0.5 M sulfuric
mediabottleinwhichitisstablefor6monthsat4°C(seeNote
acid may be used.
3).
9. Preparation of Standard Solutions
9.1.3 Mixed IC Calibration Stock Solution 10 mg/L N and
9.1 Solutions used to calibrate the IC system: 10 mg/L P—In a 100 mL volumetric flask add 1.0 mL of the
´1
D8001 − 16
FIG. 7 Graph of Total Nitrate in River Water Samples from Alkaline Persulfate Digested Samples Using Colorimetric (Vanadomolydo-
phosphoric Acid) and Ion Chromatography Determinative Steps
TABLE 2 Concentration Levels and Dilutions for Total N and Total P
Amount of Mixed Calibration PO -N Final Concentration
Final Volume NO -N Final Concentration
Concentration Level Solution (µg P/L)
(mL) (µg/L)
(10 mg/L each P and N)
1 0.025 100 2.5 2.5
2 0.05 100 5 5
3 0.1 100 10 10
4 0.25 100 25 25
5 0.5 100 50 50
6 1 100 100 100
7 2 100 200 200
8 3 100 300 300
TABLE 3 Spike Recoveries of Digestion Check Standards
Expected Conc. Found Conc.
Nitrogen Compounds Recovery
(mg N/L) (mg N/L)
Nicotinic Acid 0.1289 0.1291 100.1
Urea 0.1335 0.1274 95.4
Glycine 0.0985 0.0941 95.6
Ammonium Chloride 0.1367 0.1273 93.1
Expected Conc. Found Conc.
Phosphorous Compounds Recovery
(mg N/L) (mg N/L)
Glucose-1-Phosphate 0.1290 0.1253 97.1
Adenosine Triphosphate 0.1162 0.099 85.2
Phytic Acid 0.1232 0.1052 85.4
Glycerolphosphate 0.1077 0.107 99.3
Section 11 for additional information.)
stock N and 1.0 mL of the stock P solution. Fill to the mark
with DI water, cap and mix thoroughly by manual inversion.
9.1.4 Working Calibration Solutions—Use the amounts in
Prepare this solution fresh each time calibration solutions are
Table 2 to prepare working calibration solutions.
prepared.
9.2 Digest-Check Stock Solutions—Total Nitrogen—(it is
NOTE 2—Alternatively, commercial stock calibration solutions can be
recommended to use at least one of the digest check com-
used, provided that the solutions are traceable to primary stock solutions
pounds):
or certified reference materials, and are free from other analytes.
9.2.1 Glycine (1 mL = 1.0 mg-N)—Dissolve 3.98 g glycine
NOTE 3—In case of trace level of phosphorous contamination in
(C H NO -HCL, FW= 111.5) in about 400 mLof DI water in
reagents, it is highly recommended to matrix match the standards 2 5 2
preparation in order to nullify the effect of phosphorous in reagent. (See a 500-mL volumetric flask. Dilute this solution to the mark
´1
D8001 − 16
TABLE 4 Total Nitrogen and Total Phosphate from a Domestic Wastewater Treatment Plant
Nitrogen Phosphorous
Sample Undigested Digested Undigested Digested
(mg N/L) (mg N/L) (mg N/L) (mg N/L)
Effluent 1 6.52 8.56 2.85 3.20
Primary Effluent 1 0.31 36.52 2.02 3.87
Filtered Effluent 2 11.02 13.01 0.23 0.43
Pre-Effluent 2 11.69 13.43 0.25 0.39
Raw Sewage 2 0.55 39.87 2.19 4.50
TABLE 5 Calculated Total Kjeldahl Nitrogen from Table 4 of Wastewater Samples Collected from a Domestic Wastewater Treatment
Plant
Digested Undigested
Sample Calculated TKN
(mg N/L) (mg N/L)
Effluent 1 8.5 6.52 2.03
Primary Effluent 1 36.52 0.31 36.22
Filtered Effluent 2 11.01 11.02 1.99
Pre-Effluent 2 13.43 11.69 1.75
Raw Sewage 2 39.87 0.55 39.32
TABLE 6 Recoveries of Nitrogen and Phosphorous Test Compounds in the Presence of Increasing Amounts of Chloride
Chloride Conc. NO or PO Ret. Time Nominal Conc.
3 4
Test Compound % Rec.
(mg/L) (min) (mg N or P/L)
Glycine-N 9.10 1.49 104.5
Glycerolphosphate-P 16.8 1.63 96.2
Glycine-N 8.90 1.49 101.2
Glycerolphosphate-P 16.7 1.63 94.0
Glycine-N 8.70 1.49 87.5
Glycerolphosphate-P 16.7 1.63 88.9
with DI water and mix it thoroughly by manual inversion and 9.3.2 Adenosine Triphosphate (ATP) (1 mL = 0.1 mg-P)—
shaking. Transfer the stock digest-check solution to a 500-mL
Dissolve 0.06 gATP(C H N O P , FW = 551.15) in about
10 16 5 13 3
borosilicate media bottle in which it is stable for 6 months at
50 mL of DI water in a 100-mL volumetric flask. Dilute this
6°C
solution to the mark with DI water and mix it thoroughly by
9.2.2 Nicotinic Acid (1 mL = 1 mg-N)—Dissolve 0.88 g
manual inversion and shaking. The final concentration of this
nicotinic acid (C H NO , FW = 123.1) in about 50 mL of DI
6 5 2 solution was adjusted for the percent water content. Store this
water in a 100-mLvolumetric flask. Dilute this solution to the
solution at 6°C.
markwithDIwaterandmixitthoroughlybymanualin
...


Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...