ASTM D8234-19
(Test Method)Standard Test Method for Anions in High Ionic Water by Ion Chromatography using Tandem Suppressed Conductivity and UV Detection
Standard Test Method for Anions in High Ionic Water by Ion Chromatography using Tandem Suppressed Conductivity and UV Detection
SIGNIFICANCE AND USE
5.1 Ion chromatography provides for both qualitative and quantitative determination of common anions, F −, NO2−, hydrogen phosphate ion (HPO4–2), Br −, NO3–, and SO4–2, in the milligram per litre range from a single analytical operation requiring only a few millilitres of sample for analysis.
5.2 Anion combinations such as chloride/bromide (Cl − /Br −) and nitrite/nitrate (NO2− /NO3–), which may be difficult to distinguish by other analytical methods, are readily separated by ion chromatography.
SCOPE
1.1 This test method covers the simultaneous determination of fluoride (F −), nitrite-N (NO2-N), ortho-phosphate-P (o-PO4-P), bromide (Br −), nitrate-N (NO3-N), and sulfate (SO4-2) ions in high saline water (up to 20 % sodium chloride (NaCl)) by suppressed ion chromatography and tandem ultra-violet (UV) detection.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.2.1 Exception—The inch-pound and SI units shown for pressure measurements are to be individually regarded as standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Jun-2019
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.05 - Inorganic Constituents in Water
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 01-Aug-2018
- Effective Date
- 01-Aug-2018
- Effective Date
- 15-Jun-2012
- Effective Date
- 15-Jun-2011
- Effective Date
- 01-May-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-Jan-2008
- Effective Date
- 01-Dec-2007
- Effective Date
- 15-Dec-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 15-Aug-2006
Overview
ASTM D8234-19 is the international standard test method for determining common anions in high ionic water using ion chromatography with tandem suppressed conductivity and UV detection. Developed by ASTM International, this method allows for the simultaneous qualitative and quantitative evaluation of fluoride, nitrite-N, ortho-phosphate-P, bromide, nitrate-N, and sulfate ions in saline water matrices containing up to 20% sodium chloride (NaCl). The procedure is widely adopted in water analysis to ensure reliable separation and detection where traditional methods may encounter interference or lack specificity, particularly in complex, high-salinity samples.
Key Topics
- Simultaneous Anion Analysis: The method enables the accurate measurement of multiple anions (F⁻, NO₂-N, o-PO₄-P, Br⁻, NO₃-N, and SO₄²⁻) in a single analytical run using only a small sample volume.
- Advanced Separation: Ion chromatography effectively resolves anions-even combinations such as chloride/bromide and nitrite/nitrate-that are challenging to differentiate with other analytical techniques.
- Detection Technologies: The test utilizes both suppressed conductivity and tandem UV detection, which enhances selectivity and sensitivity, especially in the presence of high chloride concentrations.
- Matrix Tolerance: The standard is optimized for samples with high ionic strength, tolerating up to 20% NaCl without significant sample preparation beyond dilution and filtration.
- Calibration and Quality Control: Extensive procedures for calibration, standard solution preparation, and quality control (QC) ensure precision and traceability across laboratories.
- Safety and Regulatory Compliance: The method complies with international standardization principles, addressing safety, health, and environmental practices to meet technical barriers to trade.
Applications
ASTM D8234-19 finds practical application across several sectors:
- Drinking Water Analysis: Ensuring the safety and regulatory compliance of potable water by monitoring anion concentrations.
- Wastewater and Produced Water Monitoring: Assessing industrial effluents and produced water from oil and gas operations, where high salt content is common.
- Environmental Testing: Supporting environmental laboratories in the determination of anions in natural water bodies that may have high ionic content due to geochemical or anthropogenic factors.
- Process Water Quality Control: Used in chemical manufacturing and power plants to monitor key anions and prevent scaling, corrosion, or process inefficiency.
- Research and Development: Providing a robust, reproducible methodology for academic and industrial research focused on water chemistry.
Related Standards
For comprehensive water analysis and method validation, ASTM D8234-19 references and complements a suite of other ASTM standards:
- ASTM D1129: Terminology Relating to Water
- ASTM D1193: Specification for Reagent Water
- ASTM D2777: Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
- ASTM D3370: Practices for Sampling Water from Flowing Process Streams
- ASTM D5810: Guide for Spiking into Aqueous Samples
- ASTM D5847: Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
These related standards support best practices in sampling, terminology, calibration, and quality control, thereby strengthening the overall reliability and international acceptance of testing results conducted in accordance with ASTM D8234-19.
Keywords: anion analysis, ion chromatography, high saline water, ion chromatograph, suppressed conductivity detection, UV detection, ASTM D8234, water quality, environmental testing, laboratory standard, calibration, fluoride, nitrite, nitrate, bromide, sulfate, phosphate, quality control, wastewater, drinking water, regulatory compliance.
Buy Documents
ASTM D8234-19 - Standard Test Method for Anions in High Ionic Water by Ion Chromatography using Tandem Suppressed Conductivity and UV Detection
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D8234-19 is a standard published by ASTM International. Its full title is "Standard Test Method for Anions in High Ionic Water by Ion Chromatography using Tandem Suppressed Conductivity and UV Detection". This standard covers: SIGNIFICANCE AND USE 5.1 Ion chromatography provides for both qualitative and quantitative determination of common anions, F −, NO2−, hydrogen phosphate ion (HPO4–2), Br −, NO3–, and SO4–2, in the milligram per litre range from a single analytical operation requiring only a few millilitres of sample for analysis. 5.2 Anion combinations such as chloride/bromide (Cl − /Br −) and nitrite/nitrate (NO2− /NO3–), which may be difficult to distinguish by other analytical methods, are readily separated by ion chromatography. SCOPE 1.1 This test method covers the simultaneous determination of fluoride (F −), nitrite-N (NO2-N), ortho-phosphate-P (o-PO4-P), bromide (Br −), nitrate-N (NO3-N), and sulfate (SO4-2) ions in high saline water (up to 20 % sodium chloride (NaCl)) by suppressed ion chromatography and tandem ultra-violet (UV) detection. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.2.1 Exception—The inch-pound and SI units shown for pressure measurements are to be individually regarded as standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Ion chromatography provides for both qualitative and quantitative determination of common anions, F −, NO2−, hydrogen phosphate ion (HPO4–2), Br −, NO3–, and SO4–2, in the milligram per litre range from a single analytical operation requiring only a few millilitres of sample for analysis. 5.2 Anion combinations such as chloride/bromide (Cl − /Br −) and nitrite/nitrate (NO2− /NO3–), which may be difficult to distinguish by other analytical methods, are readily separated by ion chromatography. SCOPE 1.1 This test method covers the simultaneous determination of fluoride (F −), nitrite-N (NO2-N), ortho-phosphate-P (o-PO4-P), bromide (Br −), nitrate-N (NO3-N), and sulfate (SO4-2) ions in high saline water (up to 20 % sodium chloride (NaCl)) by suppressed ion chromatography and tandem ultra-violet (UV) detection. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.2.1 Exception—The inch-pound and SI units shown for pressure measurements are to be individually regarded as standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D8234-19 is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D8234-19 has the following relationships with other standards: It is inter standard links to ASTM D1129-13(2020)e2, ASTM D1066-18e1, ASTM D1066-18, ASTM D2777-12, ASTM D1066-11, ASTM D5810-96(2011), ASTM D3370-10, ASTM D1129-10, ASTM D3370-08, ASTM D2777-08, ASTM D3370-07, ASTM D1066-06, ASTM D1129-06a, ASTM D1129-06ae1, ASTM D2777-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D8234-19 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D8234 − 19
Standard Test Method for
Anions in High Ionic Water by Ion Chromatography using
Tandem Suppressed Conductivity and UV Detection
This standard is issued under the fixed designation D8234; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope D3370Practices for Sampling Water from Flowing Process
Streams
1.1 This test method covers the simultaneous determination
− D5810Guide for Spiking into Aqueous Samples
of fluoride (F ), nitrite-N (NO -N), ortho-phosphate-P(o-PO -
2 4
− -2 D5847Practice for Writing Quality Control Specifications
P), bromide (Br ), nitrate-N (NO -N), and sulfate (SO ) ions
3 4
for Standard Test Methods for Water Analysis
in high saline water (up to 20% sodium chloride (NaCl)) by
suppressed ion chromatography and tandem ultra-violet (UV)
3. Terminology
detection.
3.1 Definitions:
1.2 The values stated in SI units are to be regarded as
3.1.1 For definitions of terms used in this standard, refer to
standard. No other units of measurement are included in this
Terminology D1129.
standard.
1.2.1 Exception—The inch-pound and SI units shown for
4. Summary of Test Method
pressure measurements are to be individually regarded as
−
4.1 This test method measures NO -N, NO -N, and Br in
2 3
standard.
presence of high level concentrations of chloride ion.
1.3 This standard does not purport to address all of the
5. Significance and Use
safety concerns, if any, associated with its use. It is the
responsibility of the user of this standard to establish appro-
5.1 Ion chromatography provides for both qualitative and
− −
priate safety, health, and environmental practices and deter-
quantitative determination of common anions, F,NO , hy-
–2 − – –2
mine the applicability of regulatory limitations prior to use.
drogen phosphate ion (HPO ), Br ,NO , and SO ,inthe
4 3 4
1.4 This international standard was developed in accor-
milligram per litre range from a single analytical operation
dance with internationally recognized principles on standard-
requiring only a few millilitres of sample for analysis.
ization established in the Decision on Principles for the
− −
5.2 Anioncombinationssuchaschloride/bromide(Cl /Br )
Development of International Standards, Guides and Recom-
− –
and nitrite/nitrate (NO /NO ), which may be difficult to
2 3
mendations issued by the World Trade Organization Technical
distinguish by other analytical methods, are readily separated
Barriers to Trade (TBT) Committee.
by ion chromatography.
2. Referenced Documents
6. Interferences
2.1 ASTM Standards:
6.1 Since chloride and nitrite elute very close together, they
D1066Practice for Sampling Steam
potentially interfere for each other. Low molecular weight
D1129Terminology Relating to Water
organic acid may interfere with fluoride ion. Selection of
D1193Specification for Reagent Water
proper analytical column will permit resolution of these
D2777Practice for Determination of Precision and Bias of
interfering compounds from fluoride ion.
Applicable Test Methods of Committee D19 on Water
6.2 As with other types of chromatography, if one of the
sample components is present at very high levels, it may
interfere by causing a very large peak on the chromatogram
This test method is under the jurisdiction ofASTM Committee D19 on Water
that could mask other peaks present. This type of interference
and is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents
is normally minimized by dilution of the sample and in some
in Water.
instances may be corrected if the concentration of that anion is
Current edition approved July 1, 2019. Published August 2019. DOI: 10.1520/
D8234-19.
of interest. However, care should be taken not to dilute the
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
analyteconcentrationbelowitsdetectablelimit.UseoftheUV
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
detector enables detection of nitrite nitrogen in presence of
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. high concentrations of chloride ion.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D8234 − 19
A
TABLE 1 Single Laboratory Detection Limits in HIW
7.9 Sample Loop—A loop on the injection valve that is
designed to contain an exact amount of the sample. For high
NOTE 1—n/a = not applicable.
saline waters use 10 µL. The sample volume injected onto the
Peak No. LOQ
Analyte
(See Fig. 1 and Fig. 2) mg/L
separatorcolumniscontrolledbythisloop.Useofalargersize
Fluoride 1 0.1 loopwillusuallycausepeakbroadeningandaloopsizegreater
Chloride 2 n/a (matrix)
than 100 µL may result in column overloading and nonlinear
Nitrite-N 3 0.03 (UV det)
response. The chromatogram in Fig. 1 and Fig. 2 uses a 10-µL
Bromide 4 0.1 (UV det)
Nitrate-N 5 0.02 (UV det)
size sample loop.
o-PO -P 6 0.1
7.10 When volumes larger than the sample loop size are
Sulfate 7 0.1
A
used to fill the loop, any volume above the sample loop size
goes to waste. It is considered good technique to flush the
Data provided by US EPA-Region 6 Laboratory, Houston, TX.
sample loop prior to injection by filling with 2 to 3 times the
B Column: as specified in 7.5.
sample loop volume.
Detector: as specified in 7.7.
Eluent: as specified in 8.3.
Pump rate: 0.7 mL/min. 8. Reagents and Materials
Sample loop: 10 µL.
8.1 Purity of Reagents—Reagent grade chemicals shall be
used in all tests. Unless otherwise indicated, it is intended that
all reagents shall conform to the specifications of the Commit-
teeonAnalyticalReagentsoftheAmericanChemicalSociety.
7. Apparatus
Other grades may be used, provided it is first ascertained that
7.1 Ion Chromatograph—The ion chromatograph should
the reagent is of sufficiently high purity to permit its use
have the components shown in Fig. 7.
without lessening the accuracy of the determination.
7.2 Eluent and Regenerant Containers.
8.2 Purity of Water—Unless otherwise indicated, reference
towatershallbeunderstoodtomeanreagentwaterconforming
7.3 Eluent Pump, capable of delivering 0.1 to 5 mL/min of
to Specification D1193,Type I. Other reagent water types may
eluent at a pressure of up to 20 000 kPa (4000 psi).
be used provided it is first ascertained that the water is of
7.4 Guard Column—Anion exchange column, typically of
sufficiently high purity to permit its use without adversely
the same anion exchange material used in the separator
affecting the precision and bias of this test method. Type II
column.The purpose of this column is to protect the analytical
water was specified at the time of round robin testing of this
column from particulate matter and irreversibly retained ma-
test method.
terials.
8.3 Eluent—Dissolve 0.742 g of sodium carbonate (3.5
7.5 Analytical Column—Anionexchangecolumncapableof
mM) in water and dilute to 2 L with 2% acetonitrile (ACN).
separating fluoride ion from the injection void volume, as well
Other eluents may also prove to be acceptable, provided they
−
as resolving the anions Cl through sulfate.
give the proper resolution between the component peaks. This
NOTE1—Anyanalyticalcolumnmaybeused.However,theusershould
eluentwillactasagrowthmediaforalgae.Forthisreason,the
be able to achieve the resolution and separation as shown in Figs. 1-5.
eluent should not be kept for longer than one month.
7.6 Suppressor Device—A suppressor device based upon
NOTE 3—Automatic eluent preparation systems (electrolytically or
cation-exchange principles. In this test method, a chemical
chemically) may be used.
exchange based suppressor device was used. An equivalent
8.4 Suppressor Regenerant Solution—Cautiously add 5.6
suppressor device may be used provided that comparable
mL of sulfuric acid (H SO ) (sp gr 1.84) to 1 L of water and
2 4
method detection limits are achieved and that adequate base-
also add 100 ml of acetonitrile solvent. Or, follow instrument
line stability at minimum of 0.2 µS/cm is attained. An
manufacture recommendations.
electrolytic suppressor device may also be used.
8.5 Stock Solutions:
7.7 Detector—Following detectors were used: −1
8.5.1 Bromide Stock Solution (1.00 mL = 1.00 mg Br )—
7.7.1 A low-volume, flow through, temperature-controlled
Dry approximately2gof sodium bromide (NaBr) for6hat
electrical conductivity cell equipped with a meter capable of
150°C and cool in a desiccator. Dissolve 1.2877 g of the dried
reading from 0 to 10 000 µS/cm on a linear scale or greater if
salt in water and dilute to 1 L with –matrix solution.
applicable.
Alternatively, certified bromide stock solutions with right
7.7.2 A low-volume UV detector capable of reading range
matrix may be commercially available through chemical sup-
from 190 to 900 nm wavelength with minimum four channels
ply vendors and may be used.
of data acquisition at one time.
NOTE 2—For this experiment, eight channel variable wavelength UV
detector was used.
Reagent Chemicals, American Chemical Society Specifications, American
Chemical Society, Washington, DC. For Suggestions on the testing of reagents not
7.8 Recorder, Integrator, Computer—A device compatible
listed by the American Chemical Society, see Annual Standards for Laboratory
withthedetectoroutputcapableofrecordingdetectorresponse
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
as a function of time for the purpose of measuring peak height
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
or area. MD.
D8234 − 19
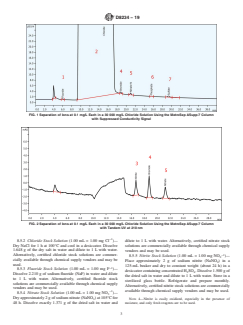
FIG. 1 Separation of Ions at 0.1 mg/L Each in a 30 000 mg/L Chloride Solution Using the MetroSep ASupp-7 Column
with Suppressed Conductivity Signal
FIG. 2 Separation of Ions at 0.1 mg/L Each in a 30 000 mg/L Chloride Solution Using the MetroSep ASupp-7 Column
with Tandem UV at 210 nm
−1
8.5.2 Chloride Stock Solution (1.00 mL = 1.00 mg Cl )— dilute to 1 L with water. Alternatively, certified nitrate stock
Dry NaCl for1hat 100°C and cool in a desiccator. Dissolve
solutions are commercially available through chemical supply
1.648 g of the dry salt in water and dilute to 1 L with water.
vendors and may be used.
−1
Alternatively, certified chloride stock solutions are commer-
8.5.5 Nitrite Stock Solution (1.00 mL = 1.00 mg NO )—
cially available through chemical supply vendors and may be
Place approximately2gof sodium nitrite (NaNO)ina
used.
125-mL beaker and dry to constant weight (about 24 h) in a
−1
8.5.3 Fluoride Stock Solution (1.00 mL = 1.00 mg F )—
desiccatorcontainingconcentratedH SO .Dissolve1.500gof
2 4
Dissolve 2.210 g of sodium fluoride (NaF) in water and dilute
the dried salt in water and dilute to 1 L with water. Store in a
to 1 L with water. Alternatively, certified fluoride stock
sterilized glass bottle. Refrigerate and prepare monthly.
solutions are commercially available through chemical supply
Alternatively, certified nitrite stock solutions are commercially
vendors and may be used.
available through chemical supply vendors and may be used.
−1
8.5.4 Nitrate Stock Solution (1.00 mL = 1.00 mg NO )—
Dryapproximately2gofsodiumnitrate(NaNO )at105°Cfor
NOTE 4—Nitrite is easily oxidized, especially in the presence of
48 h. Dissolve exactly 1.371 g of the dried salt in water and moisture, and only fresh reagents are to be used.
D8234 − 19
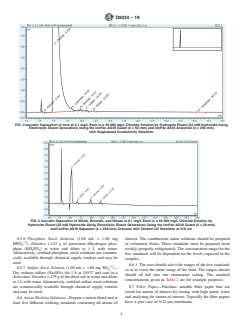
FIG. 3 Isocratic Separation of Ions at 0.1 mg/L Each in a 30 000 mg/L Chloride Solution by Hydroxide Eluent (23 mM Hydroxide Using
Electrolytic Eluent Generation) Using the IonPac AG18 Guard (4 × 50 mm) and IonPac AS18 Analytical (4 × 250 mm)
with Suppressed Conductivity Detection
FIG. 4 Isocratic Separation of Nitrite, Bromide, and Nitrate at 0.1 mg/L Each in a 30 000 mg/L Chloride Solution by
Hydroxide Eluent (23 mM Hydroxide Using Electrolytic Eluent Generation) Using the IonPac AG18 Guard (4 × 50 mm)
and IonPac AS18 Separator (4 × 250 mm) Columns with Tandem UV Detection at 210 nm
8.5.6 Phosphate Stock Solution (1.00 mL = 1.00 mg interest. The combination anion solutions should be prepared
−2
HPO )—Dissolve 1.433 g of potassium dihydrogen phos-
in volumetric flasks. These standards must be prepared fresh
phate (KH PO ) in water and dilute to 1 L with water.
weekly(properlyrefrigerated).Theconcentrationrangeforthe
2 4
Alternatively, certified phosphate stock solutions are commer-
five standards will be dependent on the levels expected in the
cially available through chemical supply vendors and may be
samples.
used.
8.6.1 The user should select the ranges of the five standards
−2
8.5.7 Sulfate Stock Solution (1.00 mL = 1.00 mg SO )—
so as to cover the entire range of the chart. The ranges chosen
Dry sodium sulfate (Na SO)for1hat 105°C and cool in a
2 4
should all fall into one attenuation setting. The standard
desiccator.Dissolve1.479gofthedriedsaltinwateranddilute
concentrations given in Table 2 are for example purposes.
to1Lwithwater.Alternatively,certifiedsulfatestocksolutions
are commercially available through chemical supply vendors
8.7 Filter Paper—Purchase suitable filter paper that are
and may be used.
tested for anions of interest by rinsing with high purity water
and analyzing for anions of interest. Typically the filter papers
8.6 Anion Working Solutions—Prepareamatrixblankandat
have a pore size of 0.22-µm membrane.
least five different working standards containing all anions of
D8234 − 19
FIG. 5 Gradient Separation of Nitrite, Bromide, Nitrate, and Phosphate (See the Peak in the Zoomed Window) at 0.1 mg/L
Each in a 30 000 mg/L Chloride Solution by Hydroxide Eluent Using Suppressed Conductivity Detection
Eluent: 23 mM hydroxide using electrolytic eluent
generation
Loop size: 5 µL
Column: IonPac AG18 (4 × 50 mm)
IonPac AS18 (4 × 250 mm)
Suppressor: AERS 500 electrolytic suppression
Detection: UV 210 nm
Gradient conditions:
23 mM (0–13 min)
23–40 mM (13–15 min)
40 mM (15–21 min)
23 mM (21–26 min)
FIG. 6 Gradient Separation of Nitrite, Bromide, Nitrate, and Phosphate at 0.1 mg/L Each in a 30 000 mg/L Chloride Solution
by Hydroxide Eluent Using UV Detection
NOTE5—Optionalin-linecrossflowfiltrationdevicemaybeusedusing
9. Sampling
commercially available 47-mm nylon filter paper. Syringe filters can also
9.1 Collect the sample in accordance with Practice D1066
be used provided they have been tested to confirm absence of anions of
interest. or D3370 as applicable.
D8234 − 19
FIG. 7 Schematic of Ion Chromatography System
TABLE 2 Calibration Ranges
Analyte Preservation Holding Time
NOTE 1—n/a = not applicable.
Sulfate Cool to 4°C 28 days
Analyte Low Standard High Standard
9.4 The method of preservation and the holding time for
Concentration, mg/L Concentration, mg/L
samples analyzed by this test method are determined by the
Fluoride 0.1 10
Chloride n/a (matrix) n/a (matrix) anionsofinterest.Inagivensample,theanionthatrequiresthe
Nitrite-N 0.03 3.3
most preservation treatment and the shortest holding time will
Bromide 0.1 10
determine the preservation treatment. It is recommended that
Nitrate-N 0.02 2.5
all samples be cooled to 4°C and held for no longer than 28
o-PO -P 0.03 3.3
Sulfate 0.1 10
days.
10. Calibration
9.2 Samplesshouldbecollectedinplasticorglassbottles.If
10.1 Determination of Retention Times:
fluoride is required, only use plastic bottles. Volume collected
10.1.1 The retention time for each anion is determined by
shouldbesufficienttoinsurearepresentativesample,allowfor
injecting a standard solution containing only the anion of
r
...


Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...