ASTM C761-96
(Test Method)Standard Test Methods for Chemical, Mass Spectrometric, Spectrochemical, Nuclear, and Radiochemical Analysis of Uranium Hexafluoride
Standard Test Methods for Chemical, Mass Spectrometric, Spectrochemical, Nuclear, and Radiochemical Analysis of Uranium Hexafluoride
SCOPE
1.1 These test methods cover procedures for subsampling and for chemical, mass spectrometric, spectrochemical, nuclear, and radiochemical analysis of uranium hexafluoride (UF6). All of these test methods are in routine use to determine conformance to UF6 specifications in the Department of Energy (DOE) gaseous diffusion plants or at other DOE installations.
1.2 The analytical procedures in this document appear
1.3 Additional test methods have been developed and are included in Appendix X 1.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. (For specific safeguard and safety consideration statements, see Section 6.)
General Information
Relations
Standards Content (Sample)
NOTICE: This standard has either been superseded and replaced by a new version or discontinued.
Contact ASTM International (www.astm.org) for the latest information.
Designation: C 761 – 96
Standard Test Methods for
Chemical, Mass Spectrometric, Spectrochemical, Nuclear,
and Radiochemical Analysis of Uranium Hexafluoride
This standard is issued under the fixed designation C 761; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (e) indicates an editorial change since the last revision or reapproval.
1. Scope
Determination of Fission Product Activity by Beta and Gamma 192 to 198
Counting
1.1 These test methods cover procedures for subsampling
Determination of Plutonium by Ion Exchange and Alpha Count- 199 to 203
and for chemical, mass spectrometric, spectrochemical, ing
Determination of Plutonium by Extraction and Alpha Counting 204 to 211
nuclear, and radiochemical analysis of uranium hexafluoride
Determination of Neptunium by Extraction and Alpha Counting 212 to 219
(UF ). All of these test methods are in routine use to determine
Atomic Absorption Determination of Chromium Soluble In Ura- 220 to 226
conformance to UF specifications in the Department of nium Hexafluoride
Atomic Absorption Determination of Chromium Insoluble In Ura- 227 to 233
Energy (DOE) gaseous diffusion plants or at other DOE
nium Hexafluoride
installations.
Determination of Technetium-99 In Uranium Hexafluoride 234 to 242
1.2 The analytical procedures in this document appear in the
Method for the Determiation of Gamma-Energy Emission Rate 243 to 250
from Fission Products in Uranium Hexafluoride
following order:
Metallic Impurities by ICP-AES 251 to 260
Sections
Molybdenum, Niobium, Tantalum, Titanium, and Tungsten by 261 to 270
Subsampling of Uranium Hexafluoride 7 to 10
ICP-AES
Gravimetric Determination of Uranium 11 to 19
Titrimetric Determination of Uranium 20 to 27 1.3 Additional test methods have been developed and are
Preparation of High-Purity U O 28 to 34
3 8
included in Appendix X1.
Isotopic Analysis by Double-Standard Mass-Spectrometer 35 to 40
1.4 This standard does not purport to address all of the
Method
Isotopic Analysis by Single-Standard Mass-Spectrometer 41 to 46
safety concerns, if any, associated with its use. It is the
Method
responsibility of the user of this standard to establish appro-
Determination of Hydrocarbons, Chlorocarbons, and Partially 47 to 53
priate safety and health practices and determine the applica-
Substituted Halohydrocarbons
Atomic Absorption Determination of Antimony 54 to 60
bility of regulatory limitations prior to use. (For specific
Spectrophotometric Determination of Bromine 61 to 68
safeguard and safety consideration statements, see Section 6.)
Titrimetric Determination of Chlorine 69 to 75
Spectrophotometric Determination of Silicon and Phosphorus 76 to 82
2. Referenced Documents
Spectrographic Determination of Boron and Silicon 83 to 90
Atomic Absorption Determination of Ruthenium 91 to 97
2.1 The following documents of the issue in effect on date
Spectrographic Determination of Ruthenium 98 to 103
of material procurement form a part of this specification to the
Spectrophotometric Determination of Titanium and Vanadium 104 to 111
Spectrographic Determination of Metallic Impurities by Carrier 112to118
extent referenced herein:
Distillation
2.2 ASTM Standards:
Spectrographic Determination of Hafnium, Molybdenum, Nio- 119to125
C 696 Test Methods for Chemical, Mass Spectrometric, and
bium, Tantalum, Titanium, Tungsten, and Zirconium After Sepa-
ration from UF with BPHA
6 Spectrochemical Analysis of Nuclear-Grade Uranium Di-
Spectrographic Determination of Hafnium, Molybdenum, Nio- 126 to 132
oxide Powders and Pellets
bium, Tantalum, Titanium, Tungsten, Vanadium, and Zirconium
C 753 Specification for Nuclear-Grade, Sinterable Uranium
After Separation from UF as Cupferrides
Spectrophotometric Determination of Tungsten 133 to 139
Dioxide Powder
Spectrophotometric Determination of Thorium 140 to 145
C 787 Specification for Uranium Hexafluoride for Enrich-
Spectrographic Determination of Thorium and Rare Earths 146 to 152
ment
Spectrophotometric Determination of Molybdenum 153 to 158
Atomic Absorption Determination of Metallic Impurities 159 to 164
C 1219 Test Methods for Arsenic in Uranium Hexafluoride
Impurity Determination by Spark-Source Mass Spectrography 165 to 173
C 1287 Test Method for Determination of Impurities in
Determination of Boron-Equivalent Neutron Cross Section 174 to 177
Uranium Dioxide by Inductively Coupled Plasma Mass
Determination of Uranium-233 Abundance by Thermal Ioniza- 178 to 184
tion Mass Spectrometry
Spectrometry
Determination of Uranium-232 by Alpha Spectrometry 185 to 191
C 1295 Test Method for Gamma Energy Emission from
Fission Products in Uranium Hexafluoride
D 1193 Specification for Reagent Water
These test methods are under the jurisdiction of ASTM Committee C-26 on
E 60 Practice for Photometric and Spectrophotometric
Nuclear Fuel Cycle and are the direct responsibility of Subcommittee C 26.05on
Methods of Test.
Current edition approved Dec. 10, 1996. Published February 1997. Originally Annual Book of ASTM Standards, Vol 12.01.
published as C761 – 73. Last previous edition C761 – 91. Annual Book of ASTM Standards, Vol 11.01.
Copyright © ASTM, 100 Barr Harbor Drive, West Conshohocken, PA 19428-2959, United States.
C 761
Methods for Chemical Analysis of Metals especially with reducing substances and moisture (see Uranium
E 115 Practice for Photographic Processing in Optical Hexafluoride: Handling Procedures and Container Criteria),
Emission Spectrographic Analysis appropriate facilities and practices for sampling and analysis
E 130 Practice for Designation of Shapes and Sizes of must be provided.
Graphite Electrodes 6.2 Committee C-26 Safeguards Statement:
2.3 U.S. Department of Energy Specifications: 6.2.1 The material (uranium hexafluoride) to which these
Uranium Hexafluoride: Base Charges, Use Charges, Spe- test methods apply, is subject to nuclear safeguards regulations
cial Charges, Table of Enriching Services, Specifications, governing its possession and use. The following analytical
and Packaging procedures in these test methods have been designated as
Uranium Hexafluoride: Handling Procedures and Container technically acceptable for generating safeguards accountability
Criteria measurement data: Gravimetric Determination of Uranium;
2.4 American Chemical Society Specification: Titrimetric Determination of Uranium; Isotopic Analysis by
Reagent Chemicals Double Standard Mass-Spectrometer Method; Isotopic Analy-
sis by Single Standard Mass-Spectrometer Method.
3. Significance and Use
6.2.2 When used in conjunction with appropriate certified
3.1 Uranium hexafluoride is a basic material used to prepare
Reference Materials (CRMs), these procedures can demon-
nuclear reactor fuel. To be suitable for this purpose the material
strate traceability to the national measurement base. However,
must meet criteria for uranium content, isotopic composition,
adherence to these procedures does not automatically guaran-
metallic impurities, hydrocarbon and partially substituted ha-
tee regulatory acceptance of the resulting safeguards measure-
lohydrocarbon content. These test methods are designed to
ments. It remains the sole responsibility of the user of these test
determine whether the material meets the requirements de-
methods to assure that its application to safeguards has the
scribed in Specification C 787.
approval of the proper regulatory authorities.
3.1.1 The material is analyzed to determine whether it
contains the uranium content specified. SUBSAMPLING OF URANIUM HEXAFLUORIDE
3.1.2 The isotopic content of the material is measured to (1, 2)
determine whether it contains the isotopic content specified.
7. Scope
3.1.3 The metallic impurity content is determined to ensure
that the maximum concentration limit of specified impurity 7.1 This test method is applicable to the subsampling (3) of
elements is not exceeded. UF from bulk sample containers into smaller containers
3.1.4 The hydrocarbon and partially substituted halohydro- suitable for laboratory analyses. The procedure includes
carbon contents are measured to ensure that the maximum sample filtration that facilitates determination of both soluble
concentration limits are not exceeded. and insoluble chromium compounds.
4. Reagents
8. Summary of Test Method
4.1 Purity of Reagents—Reagent grade chemicals shall be
8.1 The UF in the bulk sample container is liquefied and
used in all procedures. Unless otherwise indicated, all reagents
homogenized by vigorous shaking. The container is inverted
shall conform to the specifications of the Committee on
and connected to the top of a heated vacuum-manifold system,
Analytical Reagents of the American Chemical Society, where
and the subsample container is attached to the appropriate port
such specifications are available. Other grades may be used,
of the system. The system is evacuated, and the liquid UF is
provided that it is first established that the reagent to be used is
allowed to flow by gravity into the subsample container.
of sufficiently high purity to permit its use without lessening
the accuracy of the determination. 9. Apparatus
4.2 Purity of Water—Unless otherwise indicated, references
9.1 Hot Water Bath.
to water shall mean reagent water conforming to Specification
9.2 Heated Vacuum Manifold with Liquid Nitrogen Cold
D 1193.
Trap (Fig. 1).
9.3 Isotopic Abundance Sample Tube (Fig. 2).
5. Rejection
9.4 Fluorothene Sample Tube (Fig. 3).
5.1 Rejection or acceptance shall be by lot, a lot consisting
9.5 Fluorothene Knockout Cylinder (Fig. 4), closed with a
of the contents of a shipping container.
Cajon M-16 VCR-1 female nut and an M-16 VCR-4 male nut,
6. Safety Considerations or equivalent.
9.6 Nickel Filter Disk, porous, 2-μm, free of chromium (Fig.
6.1 Since UF is radioactive, toxic, and highly reactive,
5). Mott Metallurgical Corp. or equivalent.
9.7 Gas Sample Cylinder, 0.5 L.
Annual Book of ASTM Standards, Vol 03.05.
United States Department of Energy, Oak Ridge, TN 37830.
“Reagent Chemicals, American Chemical Society Specifications,” Washington,
DC. For suggestions on the testing of reagents not listed by the American Chemical Brand names mentioned in this procedure are intended to be typical, not
Society, See “Reagent Chemicals and Standards,” by Joseph Rosin, D. Van Nostrand limiting. Another brand of comparable characteristics could perform equally well.
Co., Inc., New York, NY, and the “United States Pharmacopeia.” The filter disk should weigh approximately 1 g and be 16 mm in diameter by
The boldface numbers in parentheses refer to a list of references at the end of 0.6 m thick. It should be of nickel powder produced from carbonyl nickel and
these test methods. formed by the no pressure sintering method in graphite or ceramic molds.
C 761
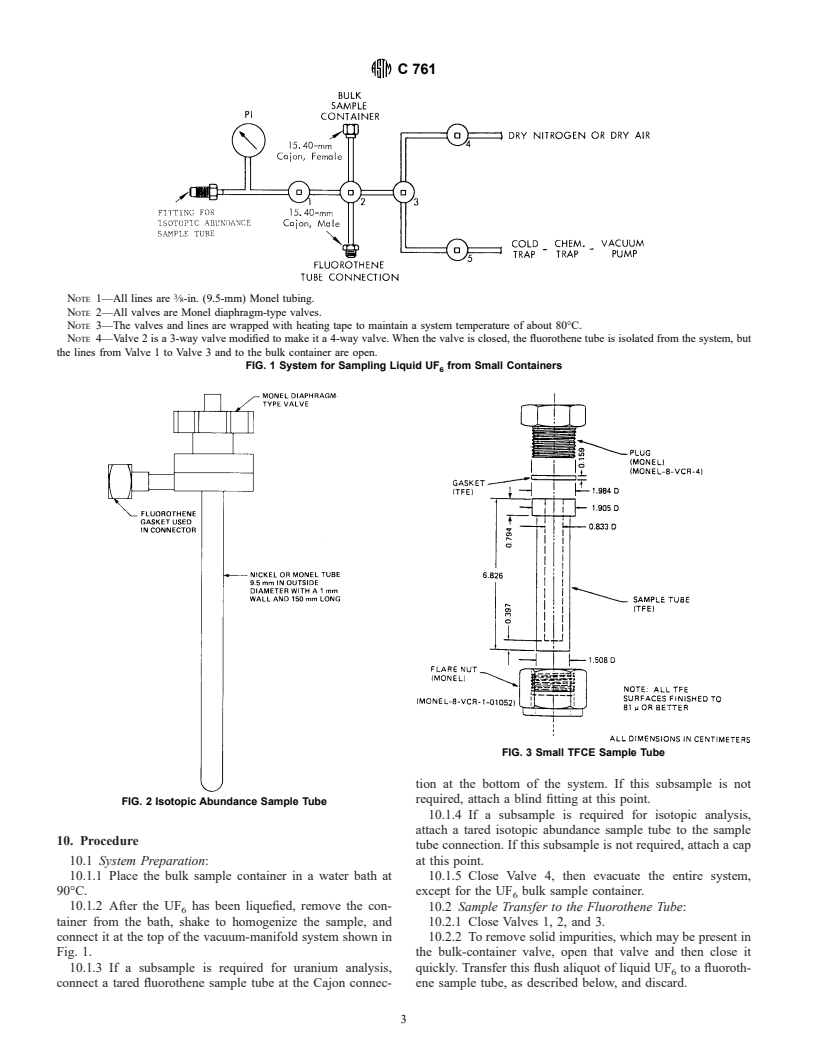
NOTE 1—All lines are ⁄8-in. (9.5-mm) Monel tubing.
NOTE 2—All valves are Monel diaphragm-type valves.
NOTE 3—The valves and lines are wrapped with heating tape to maintain a system temperature of about 80°C.
NOTE 4—Valve 2 is a 3-way valve modified to make it a 4-way valve. When the valve is closed, the fluorothene tube is isolated from the system, but
the lines from Valve 1 to Valve 3 and to the bulk container are open.
FIG. 1 System for Sampling Liquid UF from Small Containers
FIG. 3 Small TFCE Sample Tube
tion at the bottom of the system. If this subsample is not
required, attach a blind fitting at this point.
FIG. 2 Isotopic Abundance Sample Tube
10.1.4 If a subsample is required for isotopic analysis,
attach a tared isotopic abundance sample tube to the sample
10. Procedure
tube connection. If this subsample is not required, attach a cap
10.1 System Preparation: at this point.
10.1.1 Place the bulk sample container in a water bath at 10.1.5 Close Valve 4, then evacuate the entire system,
90°C. except for the UF bulk sample container.
10.1.2 After the UF has been liquefied, remove the con- 10.2 Sample Transfer to the Fluorothene Tube:
tainer from the bath, shake to homogenize the sample, and 10.2.1 Close Valves 1, 2, and 3.
connect it at the top of the vacuum-manifold system shown in 10.2.2 To remove solid impurities, which may be present in
Fig. 1. the bulk-container valve, open that valve and then close it
10.1.3 If a subsample is required for uranium analysis, quickly. Transfer this flush aliquot of liquid UF to a fluoroth-
connect a tared fluorothene sample tube at the Cajon connec- ene sample tube, as described below, and discard.
C 761
FIG. 4 Fluorothene Knockout Cylinder
FIG. 5 Filter Disk Unit
10.2.3 Open the bulk-container valve; then open Valve 2 10.2.6 Close Valve 3.
slowly, allowing liquid UF to flow into the fluorothene tube. 10.2.7 Immerse the fluorothene tube in liquid nitrogen for 6
When the tube is half full of liquid UF , close Valve 2. min.
10.2.4 Close the bulk-container valve. 10.2.8 Open Valve 2 to ensure that the sample does not exert
10.2.5 Open Valve 3 to remove UF from the system. Open a detectable vapor pressure.
Valve 1 to ensure that the system is evacuated. 10.2.9 Close Valve 5.
C 761
10.2.10 Open Valves 3 and 4, and admit dry nitrogen or dry 15 min, and remove the metal fittings and cover gasket.
air until a pressure slightly above 1 atm is reached. Transfer the sample to a tared, 2-L polypropylene beaker
10.2.11 Disconnect the fluorothene tube, seal with a fluo- chilled in ice water, by inverting the knockout cylinder over the
beaker and rapping the bottom of the knockout cylinder with a
rothene gasket and a Monel plug, and weigh the tube assembly.
10.2.12 Cap the manifold port and close Valves 2 and 4. rubber mallet.
10.6.5 Immediately add chilled nitric acid (HNO ), 1 part in
10.3 Sample Transfer to an Isotopic Abundance Tube:
4 parts distilled water, to form a solution of approximately 0.1
10.3.1 Open Valves 1, 3, and 5, as well as the isotopic
g U/g of solution.
abundance tube valve, to evacuate the tube.
10.6.6 Allow the solution to reach ambient temperature
10.3.2 Close Valves 1, 2, and 3.
while stirring periodically with a polypropylene stirring rod
10.3.3 Immerse the lower half of the metal isotopic abun-
until all of the solid has dissolved.
dance tube, as shown in Fig. 2, in liquid nitrogen for 1 or 2 min.
10.6.7 Weigh the solution and determine the uranium con-
Immerse plastic tubing in ice water and observe desublimed
centration per gram of solution.
UF .
10.6.8 Dispense aliquots of the solution for analysis accord-
10.3.4 Remove
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.