ASTM E3275-21
(Guide)Standard Guide for Visualization and Identification of Nanomaterials in Biological and Nonbiological Matrices Using Darkfield Microscopy/Hyperspectral Imaging (DFM/HSI) Analysis
Standard Guide for Visualization and Identification of Nanomaterials in Biological and Nonbiological Matrices Using Darkfield Microscopy/Hyperspectral Imaging (DFM/HSI) Analysis
SIGNIFICANCE AND USE
5.1 The information and recommendations in this guide are relevant for imaging and identifying ENMs in cells and other biological (for example, fixed tissues, whole plants) and nonbiological (for example, drug formulations, filter media, soil, and wastewater) matrices after appropriate sample preparation procedures have been performed (3-5). DFM/HSI is a recently developed analytical tool; however, the relative simplicity of sample preparation combined with the potential to acquire high-contrast ENM images and high-content ENM spectral responses facilitates the increasing use of the tool for diverse applications in drug delivery, toxicology, environmental science, biology, and medicine.
5.2 Verification of the uptake and spatial distribution of ENMs in cells, for example, is necessary for evaluating and understanding the biological effects of ENMs on living systems. Similarly, the closeness of the spatial distribution of ENMs in complex drug formulations can be an important criterion in establishing physicochemical similarity between formulations (6). Complex products are described in the most recent version of the Generic Drug User Fee Act (GDUFA) reauthorization commitment letter: (7). This guide covers the criteria and general considerations for performing DFM/HSI analyses on samples of biological and nonbiological origins containing ENMs (for example, metal and metal oxide nanoparticles, or carbon nanotubes, or both). This guide does not cover or address provisions for imaging or identifying, or both, non-engineered (natural) nanoparticles/nanomaterials in cells or other matrices, nor does this guide describe or discuss the application of DFM/HSI for determining the dimensions of ENMs.
SCOPE
1.1 This guide has been prepared to familiarize laboratory scientists with the background information and technical content necessary to image and identify engineered nanomaterials (ENMs) in cells via darkfield microscopy/hyperspectral imaging (DFM/HSI) methodology.
1.2 DFM/HSI is a hyphenated bioanalytical technique/tool that combines optical microscopy with high-resolution spectral imaging to both spatially localize the distribution of and identify ENMs within a suitably prepared test sample.
1.2.1 In the context of mammalian cells, ENMs will have distinctive light-scattering properties in comparison to subcellular organelles and cell structural features, which can allow one to discriminate between the spectral profiles of ENMs and cellular components.
1.2.2 The light-scattering properties of ENMs in other test samples, such as fixed tissues, plants, complex drug product formulations, filter media, and so forth, will also be different from the native matrix component scattering signals inherent to these other types of samples, thus allowing for ENM visualization and identification.
1.3 This guide is applicable to the use of DFM/HSI for identifying ENMs in the matrices mentioned.
1.4 This guide describes and discusses basic practices for setting up and using DFM/HSI instrumentation, sample imaging techniques, considerations for optics, image analysis, and the use of reference spectral libraries (RSLs). DFM/HSI is routinely used in industry, academia, and government as a research and development and quality control tool in diverse areas of nanotechnology.
1.5 The values stated in SI units are to be regarded as the standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of Internati...
General Information
- Status
- Published
- Publication Date
- 31-May-2021
- Technical Committee
- E56 - Nanotechnology
- Drafting Committee
- E56.08 - Nano-Enabled Medical Products
Relations
- Effective Date
- 01-Oct-2006
Overview
ASTM E3275-21 provides a comprehensive guide for laboratory scientists and researchers on the visualization and identification of engineered nanomaterials (ENMs) within both biological and nonbiological matrices. Using darkfield microscopy and hyperspectral imaging (DFM/HSI) techniques, this standard outlines key methods for localizing and identifying ENMs such as metal and metal oxide nanoparticles, and carbon nanotubes within a variety of complex test samples. The guide is applicable to areas like biological tissues, whole plants, pharmaceutical formulations, filter media, soil, and wastewater, supporting advances in nanotechnology research, product development, and quality control.
Key Topics
- DFM/HSI Methodology: Explanation of the combined analytical approach using optical microscopy with high-resolution spectral imaging to spatially and spectrally identify nanomaterials within test samples.
- Sample Preparation: Best practices for preparing biological and nonbiological samples to ensure accurate darkfield microscopy and hyperspectral imaging results.
- Instrument Setup and Calibration: Guidance on setting up DFM/HSI instrumentation, including microscope configuration, light sources, camera choices, and spectral calibration.
- Imaging Techniques: Methods for capturing high-contrast ENM images and ensuring optimal spatial resolution using both upright and inverted microscopes.
- Image Analysis: Using reference spectral libraries (RSLs) for classification and mapping of ENMs, along with considerations for system calibration and spectral normalization.
- Quality Control Considerations: Recommendations for maintaining quality assurance and addressing background signals or artifacts in imaging ENMs.
Applications
ASTM E3275-21 is widely relevant across industry, academia, and government settings. Primary applications include:
- Pharmaceutical Research and Drug Delivery: Imaging and mapping the distribution of ENMs in complex drug formulations, crucial for assessing physicochemical similarity and quality control.
- Toxicology and Environmental Science: Detecting ENMs in biological tissues, plants, soil, and wastewater to evaluate uptake, distribution, and potential impacts on living systems and the environment.
- Biological and Medical Research: Studying the interactions, cellular uptake, and spatial localization of nanomaterials to better understand biological effects and inform safety assessments.
- Nanotechnology and Material Science: Supporting R&D in the design, synthesis, and functional evaluation of engineered nanomaterials in diverse matrices.
- Quality Assurance and Regulatory Compliance: Assisting laboratories in developing standardized imaging protocols aligned with international standards, aiding compliance with regulations and guidelines.
Related Standards
For comprehensive implementation, ASTM E3275-21 references several related standards and terminology:
- ASTM E2456: Terminology Relating to Nanotechnology
- ASTM E3255: Practice for Quality Assurance of Forensic Science Service Providers Performing Forensic Chemical Analysis
- ISO 20473: Optics and Photonics-Spectral Bands
- ISO/TS 80004-1: Nanotechnologies-Vocabulary-Part 1: Core Terms
These documents assist users in adopting consistent terminology and methodologies across nanotechnology and spectroscopy fields.
Keywords: ASTM E3275-21, engineered nanomaterials, darkfield microscopy, hyperspectral imaging, nanomaterial identification, ENMs, biological matrices, nonbiological matrices, nanotechnology standards, spectral imaging, quality control, pharmaceutical formulations, environmental science, reference spectral library.
Buy Documents
ASTM E3275-21 - Standard Guide for Visualization and Identification of Nanomaterials in Biological and Nonbiological Matrices Using Darkfield Microscopy/Hyperspectral Imaging (DFM/HSI) Analysis
Frequently Asked Questions
ASTM E3275-21 is a guide published by ASTM International. Its full title is "Standard Guide for Visualization and Identification of Nanomaterials in Biological and Nonbiological Matrices Using Darkfield Microscopy/Hyperspectral Imaging (DFM/HSI) Analysis". This standard covers: SIGNIFICANCE AND USE 5.1 The information and recommendations in this guide are relevant for imaging and identifying ENMs in cells and other biological (for example, fixed tissues, whole plants) and nonbiological (for example, drug formulations, filter media, soil, and wastewater) matrices after appropriate sample preparation procedures have been performed (3-5). DFM/HSI is a recently developed analytical tool; however, the relative simplicity of sample preparation combined with the potential to acquire high-contrast ENM images and high-content ENM spectral responses facilitates the increasing use of the tool for diverse applications in drug delivery, toxicology, environmental science, biology, and medicine. 5.2 Verification of the uptake and spatial distribution of ENMs in cells, for example, is necessary for evaluating and understanding the biological effects of ENMs on living systems. Similarly, the closeness of the spatial distribution of ENMs in complex drug formulations can be an important criterion in establishing physicochemical similarity between formulations (6). Complex products are described in the most recent version of the Generic Drug User Fee Act (GDUFA) reauthorization commitment letter: (7). This guide covers the criteria and general considerations for performing DFM/HSI analyses on samples of biological and nonbiological origins containing ENMs (for example, metal and metal oxide nanoparticles, or carbon nanotubes, or both). This guide does not cover or address provisions for imaging or identifying, or both, non-engineered (natural) nanoparticles/nanomaterials in cells or other matrices, nor does this guide describe or discuss the application of DFM/HSI for determining the dimensions of ENMs. SCOPE 1.1 This guide has been prepared to familiarize laboratory scientists with the background information and technical content necessary to image and identify engineered nanomaterials (ENMs) in cells via darkfield microscopy/hyperspectral imaging (DFM/HSI) methodology. 1.2 DFM/HSI is a hyphenated bioanalytical technique/tool that combines optical microscopy with high-resolution spectral imaging to both spatially localize the distribution of and identify ENMs within a suitably prepared test sample. 1.2.1 In the context of mammalian cells, ENMs will have distinctive light-scattering properties in comparison to subcellular organelles and cell structural features, which can allow one to discriminate between the spectral profiles of ENMs and cellular components. 1.2.2 The light-scattering properties of ENMs in other test samples, such as fixed tissues, plants, complex drug product formulations, filter media, and so forth, will also be different from the native matrix component scattering signals inherent to these other types of samples, thus allowing for ENM visualization and identification. 1.3 This guide is applicable to the use of DFM/HSI for identifying ENMs in the matrices mentioned. 1.4 This guide describes and discusses basic practices for setting up and using DFM/HSI instrumentation, sample imaging techniques, considerations for optics, image analysis, and the use of reference spectral libraries (RSLs). DFM/HSI is routinely used in industry, academia, and government as a research and development and quality control tool in diverse areas of nanotechnology. 1.5 The values stated in SI units are to be regarded as the standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of Internati...
SIGNIFICANCE AND USE 5.1 The information and recommendations in this guide are relevant for imaging and identifying ENMs in cells and other biological (for example, fixed tissues, whole plants) and nonbiological (for example, drug formulations, filter media, soil, and wastewater) matrices after appropriate sample preparation procedures have been performed (3-5). DFM/HSI is a recently developed analytical tool; however, the relative simplicity of sample preparation combined with the potential to acquire high-contrast ENM images and high-content ENM spectral responses facilitates the increasing use of the tool for diverse applications in drug delivery, toxicology, environmental science, biology, and medicine. 5.2 Verification of the uptake and spatial distribution of ENMs in cells, for example, is necessary for evaluating and understanding the biological effects of ENMs on living systems. Similarly, the closeness of the spatial distribution of ENMs in complex drug formulations can be an important criterion in establishing physicochemical similarity between formulations (6). Complex products are described in the most recent version of the Generic Drug User Fee Act (GDUFA) reauthorization commitment letter: (7). This guide covers the criteria and general considerations for performing DFM/HSI analyses on samples of biological and nonbiological origins containing ENMs (for example, metal and metal oxide nanoparticles, or carbon nanotubes, or both). This guide does not cover or address provisions for imaging or identifying, or both, non-engineered (natural) nanoparticles/nanomaterials in cells or other matrices, nor does this guide describe or discuss the application of DFM/HSI for determining the dimensions of ENMs. SCOPE 1.1 This guide has been prepared to familiarize laboratory scientists with the background information and technical content necessary to image and identify engineered nanomaterials (ENMs) in cells via darkfield microscopy/hyperspectral imaging (DFM/HSI) methodology. 1.2 DFM/HSI is a hyphenated bioanalytical technique/tool that combines optical microscopy with high-resolution spectral imaging to both spatially localize the distribution of and identify ENMs within a suitably prepared test sample. 1.2.1 In the context of mammalian cells, ENMs will have distinctive light-scattering properties in comparison to subcellular organelles and cell structural features, which can allow one to discriminate between the spectral profiles of ENMs and cellular components. 1.2.2 The light-scattering properties of ENMs in other test samples, such as fixed tissues, plants, complex drug product formulations, filter media, and so forth, will also be different from the native matrix component scattering signals inherent to these other types of samples, thus allowing for ENM visualization and identification. 1.3 This guide is applicable to the use of DFM/HSI for identifying ENMs in the matrices mentioned. 1.4 This guide describes and discusses basic practices for setting up and using DFM/HSI instrumentation, sample imaging techniques, considerations for optics, image analysis, and the use of reference spectral libraries (RSLs). DFM/HSI is routinely used in industry, academia, and government as a research and development and quality control tool in diverse areas of nanotechnology. 1.5 The values stated in SI units are to be regarded as the standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of Internati...
ASTM E3275-21 is classified under the following ICS (International Classification for Standards) categories: 07.120 - Nanotechnologies. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E3275-21 has the following relationships with other standards: It is inter standard links to ASTM E2456-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E3275-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:E3275 −21
Standard Guide for
Visualization and Identification of Nanomaterials in
Biological and Nonbiological Matrices Using Darkfield
Microscopy/Hyperspectral Imaging (DFM/HSI) Analysis
This standard is issued under the fixed designation E3275; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 1.6 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
1.1 This guide has been prepared to familiarize laboratory
responsibility of the user of this standard to establish appro-
scientists with the background information and technical con-
priate safety, health, and environmental practices and deter-
tent necessary to image and identify engineered nanomaterials
mine the applicability of regulatory limitations prior to use.
(ENMs) in cells via darkfield microscopy/hyperspectral imag-
1.7 This international standard was developed in accor-
ing (DFM/HSI) methodology.
dance with internationally recognized principles on standard-
1.2 DFM/HSI is a hyphenated bioanalytical technique/tool
ization established in the Decision on Principles for the
thatcombinesopticalmicroscopywithhigh-resolutionspectral
Development of International Standards, Guides and Recom-
imaging to both spatially localize the distribution of and
mendations issued by the World Trade Organization Technical
identify ENMs within a suitably prepared test sample.
Barriers to Trade (TBT) Committee.
1.2.1 In the context of mammalian cells, ENMs will have
distinctive light-scattering properties in comparison to subcel-
2. Referenced Documents
lular organelles and cell structural features, which can allow
2.1 ASTM Standards:
one to discriminate between the spectral profiles of ENMs and
E2456Terminology Relating to Nanotechnology
cellular components.
E3255Practice for Quality Assurance of Forensic Science
1.2.2 The light-scattering properties of ENMs in other test
ServiceProvidersPerformingForensicChemicalAnalysis
samples, such as fixed tissues, plants, complex drug product
2.2 ISO Standards:
formulations, filter media, and so forth, will also be different
ISO 20473Optics and photonics—Spectral bands
fromthenativematrixcomponentscatteringsignalsinherentto
ISO/TS 80004-1Nanotechnologies—Vocabulary—Part 1:
these other types of samples, thus allowing for ENM visual-
Core terms
ization and identification.
1.3 This guide is applicable to the use of DFM/HSI for
3. Terminology
identifying ENMs in the matrices mentioned.
3.1 Definitions:
1.4 This guide describes and discusses basic practices for
3.1.1 agglomerate, n—group of particles held together by
setting up and using DFM/HSI instrumentation, sample imag-
relatively weak forces (for example, van der Waals or capil-
ing techniques, considerations for optics, image analysis, and
lary) that may break apart into smaller particles upon
the use of reference spectral libraries (RSLs). DFM/HSI is
processing. E2456
routinely used in industry, academia, and government as a
3.1.2 aggregate, n—discrete group of particles in which the
research and development and quality control tool in diverse
various individual components are not easily broken apart,
areas of nanotechnology.
such as the case of primary particles that are strongly bonded
1.5 The values stated in SI units are to be regarded as the
together (for example, fused, sintered, or metallically bonded
standard. No other units of measurement are included in this
particles). E2456
standard.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
This guide is under the jurisdiction of ASTM Committee E56 on Nanotech- contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
nology and is the direct responsibility of Subcommittee E56.08 on Nano-Enabled Standards volume information, refer to the standard’s Document Summary page on
Medical Products. the ASTM website.
Current edition approved June 1, 2021. Published July 2021. DOI: 10.1520/ Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
E3275-21. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E3275−21
3.1.3 blank sample, n—sample matrix carried through all or 3.1.16 image cube, n—hyperspectral image composed of
part of the analytical process, where the analyte is not present, concatenated two-dimensional spatial images at a single spec-
or where the analyte response is suppressed. tral frequency.
3.1.17 nanomaterial, n—material with any external dimen-
3.1.4 blocking negative ligand control sample, n—control
sion in the nanoscale or having internal structure or surface
sample that is treated with free ligand at an appropriate
structure in the nanoscale. ISO/TS 80004-1
concentration to reduce the number of available binding sites
for the targeted ENM.
3.1.18 nanoparticle, n—classificationofanultrafineparticle
with lengths in two or three dimensions greater than 1nm and
3.1.5 camera gain, n—conversion ratio between the de-
-
smallerthanapproximately100nmandwhichmayormaynot
tected electrons (e) and the digital number of the digitized
exhibit a size-related intensive property. E2456
signal output by the camera.
3.1.19 near ultraviolet, near UV, n—electromagnetic spec-
3.1.5.1 Discussion—For example, a gain of 4 means that it
-
takes4e toproduceoneanalog-to-digitalunit(one“count”)in trum containing the wavelength range 315nm to 380nm.
ISO 20473:2007
the camera output.
3.1.20 negative control, n—a material of established origin
3.1.6 dark current, n—rateofincreaseofunwantedelectron
-
that is used to confirm that a procedure does not produce an
signalthatgetaddedtoapixel’ssignal,usuallyexpressedine
unintended result. E3255
per second per pixel.
3.1.6.1 Discussion—Longer acquisition times result in
3.1.21 quantum effıciency, QE, n—ratio of the number of
larger dark signal and more contribution to noise. Higher photons detected as signal to the total number of photons
temperaturealsoincreasesthedarkcurrent,whichiswhymost
striking the camera sensor.
scientific cameras are cooled (sometimes down to -100°C) to
3.1.21.1 Discussion—Photons detected as signal are the
minimize dark current and the associated contribution to total
ones that get converted to electrons and then read out as signal
noise.
by the sensor electronics. The higher the QE of the camera
sensor, the shorter the measurement time for the same total
3.1.7 darkfield condenser, n—device that provides sample
number of detected photons and therefore the higher the signal
illumination that is only detected by the detection objective in
to noise per unit time. QE is wavelength-dependent, so it is
the presence of a scattering object in the sample and is usually
important to verify that the camera sensor has sufficiently high
achieved by oblique illumination of the sample.
QE over all wavelengths of interest.
3.1.8 darkfield microscopy, DFM, n—high-contrast light
3.1.22 read noise, n—error that gets added to each pixel
microscopy technique that excludes unscattered light in which
signaluponreadoutofacamerasensor,usuallygiveninterms
scattering bodies appear bright against a black background.
-
of electrons (e).
3.1.9 data cube, n—array that consists of data, which
3.1.22.1 Discussion—The lower the readout noise, the less
includes spatial (x, y) and spectral information.
it affects the quality of the signal.
3.1.10 engineered nanomaterial, ENM, n—nanomaterialde-
3.1.23 reference spectral library, RSL, n—collection of the
signed for a specific purpose or function. ISO/TS 80004-1
full absorption, emission, or reflection of light from known
materials combined into a spectral library used to identify and
3.1.11 field of view, FOV, n—spatial view of the sample that
map unknown materials within a hyperspectral image.
is obtained after acquisition.
3.1.24 short wave infrared, SWIR, n—electromagneticspec-
3.1.11.1 Discussion—The FOV is instrument dependent.
trum containing the wavelength range 1400nm to 3000nm.
3.1.12 full well, n—maximum number of electrons that can
ISO 20473:2007
be held in a single pixel of a camera sensor.
3.1.25 shot noise, n—statistical error caused by the discrete
3.1.12.1 Discussion—If more photons strike a full pixel
nature of the photons striking a sensor.
duringanexposure,thesignaloutputremainsthesameandthe
3.1.25.1 Discussion—This noise is inherent to the physical
pixel is said to be saturated.
nature of light and cannot be reduced by improving the quality
3.1.13 hyperspectral imaging, HSI, n—imaging technique
of the camera electronics.
in which the full absorption, emission, or reflection of light is
3.1.26 spectral library, n—collection of the full absorption,
collected for each spectral band or wavelength.
emission, or reflection of light of known materials, including
3.1.14 hyperspectral mapping, n—classification of points
those within specific matrices, collected by a hyperspectral
within a field of view using similarities between the spectra
system that may show matrix-dependent spectral features.
within the reference spectral library and with the spectrum of
3.1.27 spectrum cube, n—a 3-dimensional array of simulta-
the unknown material.
neously acquired 2-D images with spectral information for
3.1.15 hyperspectral system, n—systemconsistingofappro- each image contained in the third dimension; a spectrum cube
priate light sources and detectors that can measure a range of consistsofaseriesofindependentacquisitions,rangingfrom1
wavelengths typically including visible near infrared (VNIR), to n where n is defined as an unlimited field of view, in which
short wave infrared (SWIR), and sometimes near ultraviolet all spatially resolved spectral information is obtained simulta-
(UV). neously.
E3275−21
3.1.27.1 Discussion—The third dimension is the distance of tems. Similarly, the closeness of the spatial distribution of
the segment along the field of view. The final hyperspectral ENMs in complex drug formulations can be an important
image is dependent upon the data processing software utilized. criterion in establishing physicochemical similarity between
formulations (6). Complex products are described in the most
3.1.28 targeted ENMs, n—nanomaterials that have a target-
recent version of the Generic Drug User Fee Act (GDUFA)
ing ligand attached on the outer surface.
reauthorization commitment letter: (7). This guide covers the
3.1.29 treated sample, n—sample that has been exposed to
criteria and general considerations for performing DFM/HSI
one or more types of ENMs.
analyses on samples of biological and nonbiological origins
3.1.30 treated negative ligand control sample, n—a cell-
containing ENMs (for example, metal and metal oxide
based sample which does not contain a targeting ligand; this
nanoparticles, or carbon nanotubes, or both). This guide does
control sample has been exposed to one or more types of
not cover or address provisions for imaging or identifying, or
ENMs.
both, non-engineered (natural) nanoparticles/nanomaterials in
cells or other matrices, nor does this guide describe or discuss
3.1.31 visible near infrared, VNIR, n—electromagnetic
spectrumcontainingthewavelengthrange380nmto1400nm. theapplicationofDFM/HSIfordeterminingthedimensionsof
ISO 20473:2007 ENMs.
4. Summary of Guide
6. Instrumentation
4.1 A test sample containing ENMs is placed onto a glass 6.1 General Instrument Description:
microscope slide and protected with a coverslip. In addition,
6.1.1 A DFM system is composed of an optical imaging
samples that have not been exposed to ENMs (negative microscope with a series of direct light blocks and high angle
controls) and samples that have been exposed to known types
mirrorswhichonlyallowforlightscatteredathighanglestobe
and concentrations of ENMs (positive controls) are prepared detected. The imaged areas where the sample is absent appear
on corresponding microscope slides. To detect, identify, and dark; hence, the name darkfield imaging. A DFM/HSI system
determine the spatial distribution of ENMs in the test sample, detects the scattering spectrum of the sample at each spatial
the sample is illuminated at an oblique angle using a darkfield point of the image.
microscope. Scattered light from the ENMs in the sample is 6.1.2 Sample illumination is typically performed using a
captured in the form of high-contrast microscopy images that broad-spectrum light source, and the light scattered by the
enable precise localization of the ENMs. Next, an HSI system sample is then separated by wavelengths before reaching the
is used to obtain and record spectral data for each pixel in the image sensor.
DFM sample images within a specified range, commonly
6.2 Microscope Type:
within the VNIR or SWIR ranges. In this manner, spectral
6.2.1 DFM is typically performed on upright or inverted
libraries are prepared for each test sample.Appropriate control
microscopesthatdifferfromeachotherintheorientationofthe
measurements, light corrections, and system calibration proce-
illumination, sample, and objective.
dures to reduce the incidence and impact of matrix artifacts
6.2.2 In the case of upright microscopes, the transmitted
shall be performed with the negative and positive control
illumination is directed upwards from underneath the sample
samples to build a reference spectral library (RSL) (1, 2) .The
and the microscope objective is located above the sample.
RSL is used in a mapping process to positively identify and
Imagefocusistypicallyperformedbymovingthesamplestage
determinethespatialdistributionoftheENMsinatestsample.
vertically with the objective lenses in a fixed position while
focusing.
5. Significance and Use
6.2.3 In the case of inverted microscopes, the transmitted
5.1 The information and recommendations in this guide are
illumination is located above the sample and pointed down-
relevant for imaging and identifying ENMs in cells and other
wards.Theobjectiveislocatedunderthesample.Theheightof
biological (for example, fixed tissues, whole plants) and
the sample stage is fixed and focusing is performed by moving
nonbiological (for example, drug formulations, filter media,
the objective vertically.
soil, and wastewater) matrices after appropriate sample prepa-
6.3 Light Sources:
ration procedures have been performed (3-5). DFM/HSI is a
6.3.1 AtypicallightsourceforDFM/HSIshallbebrightand
recently developed analytical tool; however, the relative sim-
coverthewavelengthrangeofinterest.Ideally,thespectrumof
plicity of sample preparation combined with the potential to
lightemittedfromthesourceshouldbeflatanddevoidofsharp
acquire high-contrast ENM images and high-content ENM
spectral features to facilitate subsequent data processing.Typi-
spectral responses facilitates the increasing use of the tool for
cal examples of light sources for darkfield imaging are given
diverse applications in drug delivery, toxicology, environmen-
below.
tal science, biology, and medicine.
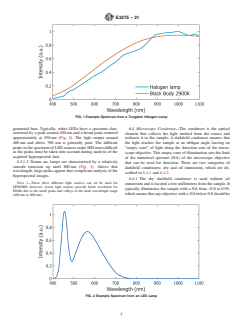
6.3.1.1 Halogen lamps, because of their low cost, are
5.2 Verification of the uptake and spatial distribution of
standard lighting sources found on most microscopes. The
ENMs in cells, for example, is necessary for evaluating and
typical spectrum from a halogen lamp (Fig. 1) is devoid of
understanding the biological effects of ENMs on living sys-
peaks and approximately follows the spectrum of a blackbody
radiator of equivalent temperature.
6.3.1.2 Light-emitting diode (LED) lamps are increasingly
The boldface numbers in parentheses refer to a list of references at the end of
this standard. popular for their electrical efficiency and small amount of
E3275−21
FIG. 1Example Spectrum from a Tungsten Halogen Lamp
generated heat. Typically, white LEDs have a spectrum char- 6.4 Microscope Condenser—The condenser is the optical
acterized by a peak around 450nm and a broad peak centered element that collects the light emitted from the source and
approximately at 550nm (Fig. 2). The light output around
redirects it to the sample. A darkfield condenser ensures that
400nm and above 700 nm is generally poor. The different
the light reaches the sample at an oblique angle leaving an
peaksinthespectrumofLEDsourcesmakeHSImoredifficult
“empty cone” of light along the detection axis of the micro-
as the peaks must be taken into account during analysis of the
scope objective. This empty cone of illumination sets the limit
acquired hyperspectral data.
of the numerical aperture (NA) of the microscope objective
6.3.1.3 Xenon arc lamps are characterized by a relatively
that can be used for detection. There are two categories of
smooth emission up until 800nm (Fig. 3). Above that
darkfield condensers: dry and oil immersion, which are de-
wavelength, large peaks appear that complicate analysis of the
scribed in 6.4.1 and 6.4.2.
hyperspectral images.
6.4.1 The dry darkfield condenser is used without oil
NOTE 1—These three different light sources can all be used for
immersionandislocatedafewmillimetresfromthesample.It
DFM/HSI; however, xenon light sources provide better resolution for
typically illuminates the sample with a NAfrom ~0.8 to 0.95,
ENMs due to the small peaks and valleys in the short wavelength range
whichmeansthatanyobjectivewithaNAbelow0.8shouldbe
(400nm to 800nm).
FIG. 2Example Spectrum from an LED Lamp
E3275−21
FIG. 3Example Spectrum from a Xenon Arc Lamp
suitablefordarkfieldimaging(inpractice,NAs~0.7andlower 6.5.2 The numerical aperture indicates the resolving power
are recommended to avoid parasitic light caused by potential of the objective and also its light-gathering capacity. Larger
system misalignment).
NA objectives (~0.5 to 1.2) provide better spatial resolution
6.4.2 The oil immersion darkfield condenser illuminates the
and better light collection efficiency. However, larger NA
sample with a much larger NA, typically above 1.2, which
objectives result in shallower depths of field, which may be
permits the use of objectives with a larger NA than allowable
undesirable for non-flat samples such as cells. Care shall also
with the dry condenser. However, this condenser uses immer-
betakentoensurethattheNAoftheobjectiveislowerthanthe
sion oil between the sample and the condenser, which can
limit set by the darkfield condenser that is used.
affect some samples and requires frequent cleaning.
6.5.3 Working distance (WD) is the distance between the
6.5 Microscope Objectives—Microscope objectives are a
front of the objective and the sample. The user should choose
central part of the instrument.The components of two types of
an objective with a sufficiently large WD to account for the
objectivesareshowninFig.4.Theusershouldunderstandand
thickness of the coverslip or the microscope slide thickness,
review all the characteristics of the objective to ensure optimal
depending on the sample orientation.
performance of the instrument.
6.5.4 Correcting ring is a feature found on some objectives
6.5.1 Magnification is the size of the sample’s image in the
that is used to vary its aberration correction to account for
instrument image plane relative to the real size of the sample.
different coverslip thicknesses.
The objective magnification fixes the FOV of the instrument
6.5.5 Iris ring is a feature found on some objectives that is
andthecorrespondingsizeoftheimagingpixelsonthesample.
used to vary the NA value to control the spatial resolution,
Ingeneral,theuserchoosesthemagnificationasacompromise
depth of field, and light collection efficiency. In DFM, this
between the desired size of the FOV and the desired spatial
feature is useful to reduce the NA of the objective to a value
resolution, that is, the larger the FOV, the poorer the spatial
resolution. lower than the limit set by the condenser.
FIG. 4Important Physical Characteristics of a Microscope Objective
E3275−21
6.5.6 Immersion media are required by some objectives to are many specifications for cameras that the user should
work correctly. The immersion media used will depend upon understand to ensure optimal camera choice and performance
theanalyzedsamplesandobjectives.Foridealillumination,the metrics such as quantum efficiency, gain, full-well capacity,
immersion media should be matched with the refractive index read noise, and dark current. See Section 3 for more details on
of the condenser and sample matrix. these metrics.
6.5.7 Objective Type—Microscope objectives are designed
NOTE 2—It is not practical to operate a commercial microscope at
to correct for different optical aberrations to increase image
wavelengths less than 360nm.
quality. Plan achromatic and apochromatic (APO) objectives
correctforchromaticandfieldcurvatureaberrationswithAPO
7. Image Capturing/Optics
objectives providing better chromatic aberration correction.
7.1 Optical Methodologies—HSI systems used in ENM
These corrections are crucial for HSI of ENMs in which two
characterization are presently available in two formats.
ENMsthatmayscatteratdifferentwavelengthsshallbothbein
7.1.1 Wavelength Dispersive Models:
focus at the same time within the field of view.
7.1.1.1 Instruments based on wavelength dispersive models
6.6 Widefield Tunable Filter—Awidefield tunable filter is a
use a diffraction grating or prism to disperse the spectrum and
bandpass-imaging filter for which the central wavelength can
are also referred to as “line-scan” or “push-broom” systems.
be selected by the user. For HSI, a sequence of images is
The FOV is imaged onto the entrance slit of a wavelength
acquired at different wavelengths to construct an image cube.
dispersivespectrometer(WDS)thatusesa2Ddetectorsuchas
See Section 7 for more details. Important specifications of the
a complementary metal-oxide semiconductor (CMOS) or
tunable filter are its NA, spectral width of the bandpass filter,
charge-coupled device (CCD) camera. The only light that
and the range of wavelength accordability.
passes through the WDS is the slice of the FOV that passes
6.7 Imaging Spectrometer—An imaging spectrometer im-
through the entrance slit of the WDS (see Fig. 5). Spectra are
agesalineonthesampleanddispersesthewavelengthsonthe
dispersed along rows of pixels and spatial information along
axis perpendicular to the line on a two-dimensional (2D)
columns of pixels. Each acquisition is an independent 2D
sensor. For HSI, a sequence of lines is acquired at different
mapping and contains spatially resolved spectra of each point
positions on the sample to construct a spectrum cube; see
along the entrance slit. These “points” along the entrance slit
Section 7 for more details. Important specifications of the
correspond to “points” along the segment of the FOV. Each
imaging spectrometer are its spectral resolution and its
acquisition can be thought of as a “spectral snapshot.”
f-number (F#, which is related to its light-gathering capacity).
7.1.1.2 The sample is moved perpendicular to the entrance
6.8 Camera—HSI typically uses monochromatic cameras. slit by a translation stage under computer control. This action
For ENM imaging, the visible and NIR wavelengths are of is arbitrarily repeated sequentially as often as required to
interest,withsilicon-basedimagesensorscoveringthe200nm complete a user-defined FOV. The spatial y-axis is determined
to 1100 nm wavelength range. Although less often by the height of the entrance slit. To create a hyperspectral
encountered, the 900nm to 1700nm range can be probed with image, independent spectral snapshots are concatenated to-
indium gallium arsenide (InGaAs) cameras. In all cases, there gethertoforma“spectrumcube;”seeFig.6.Animagecubeis
FIG. 5Example of Wavelength Dispersive HSI System Used for Interrogating the Presence of ENMs
in Biological and Nonbiological Matrices
E3275−21
FIG. 6Example of Wavelength Dispersive HSI System Used for Interrogating the Presence of ENMs
in Biological and Nonbiological Matrices
created when a fixed FOV is measured at a series of indepen- image sensor and are often referred to as “staring” devices. In
dent sequential wavelengths and shall be completed to acquire this model, sequential acquisitions are made of the same FOV
full spectral content. A single acquisition or element in the at different wavelengths through a tunable imaging bandpass
image cube produces a “picture” of the entire FOV at just one filter.Ascanof100acquisitionswillresultin100imagesofthe
wavelength.Conversely,aspectrumcubeisgeneratedbyWDS FOVat each wavelength. Each pixel of the camera measures a
systems, and each single acquisition presents a spatially spectrum of one point in the FOV one wavelength after
resolved slice of a FOV at all wavelengths simultaneously. A another. Once completed, the output from this process is
slice or “segment of the FOV” is defined as the height and termedan“imagecube;”seeFig.7.NotranslationoftheFOV
width of the entrance slit divided by the magnification of the is necessary in the case of these “staring” devices. Movements
microscope objective. Spatially resolved spectra produced of the FOV or objects within it can be compensated by
along the slit are captured along rows of pixels in the camera post-processing registration algorithms. Software to convert
consistent with the image of the height of the entrance slit. spatiallyresolvedspectraldataintoanimagecanbespecificto
Various file formats can be used specific to either data cube the instrument developer or obtained by commercial “off-the-
format including, but not limited to, tagged image format file shelf” packages. There are also open-source tools built on a
(TIF) with extended metadata, file information tool set (FITS), varietyofplatformssuchasPython,Octave,andRthatmaybe
and hierarchical data format (HDF) HDF4, and HDF5. All of customized to specific needs of advanced users.
these formats can be read by various image analysis software
8. Sample Imaging
for further processing. Software to convert spatially resolved
spectral data into an image can be specific to the instrument 8.1 General Considerations—Foreveryinstrument,theuser
developer or procured by commercial packages. should set up the microscope for darkfield observation of the
7.1.2 Tunable Imaging Filter Models—Instruments based sample before hyperspectral measurements can be started.The
on tunable image filter models use acousto-optical tunable microscope setup differs slightly depending on whether the
filters (AOTF), liquid-crystal tunable filters (LCTF), microscope is upright or inverted, and whether the condenser,
interferometers, or hologram-based wavelength tunable de- objective or both are dry or oil immersion. Generally, an
vices to produce a filtered image of the FOVcaptured by a 2D uprightmicroscopeispreferablefordarkfieldillumination.Not
E3275−21
FIG. 7Illustration of a “Staring” Wavelength Scanning System in which the Hyperspectral Image of an Object
(1) is Acquired through Sequential Acquisitions of Monochromatic Images at Different Wavelengths
(3) by Using a Tunable Imaging Bandpass
FilterImages courtesy of Valéry Simard, Marc Verhaegen, and David Rioux
allmanufacturer’smicroscopescanbeusedathighpowerwith 8.2.5 The sample is usually focused before the condenser is
an oil condenser. For higher power objectives (40× to 60× and adjusted.However,someinvestigatorsmayswitchtheorderof
100×), it is necessary to have an iris diaphragm built into the steps in the setup and adjust the condenser first. If using a dry
objectives to reduce the light and NA value. This lens feature
condenser, slowly raise the condenser until the light is opti-
is not currently offered by all microscope manufacturers. mized. In the case of an oil immersion condenser, slowly raise
Darkfield mode used to be standard in a condenser, but it is no
the condenser until the immersion oil contacts the slide and
longer and is not usually included in the condensers of newer, then proceed to focus on the sample and align the condenser.
infinity-corrected microscopes.
8.2.5.1 Slowly move the condenser up and down via the
focus knob until brightness and image quality are maximized.
8.2 Upright Microscope Setup—Refertothemanufacturer’s
It is advisable to adjust both the sample and condenser
instructionsfordetailed,instrument-specificsetupinstructions.
sequentially to get the best image and lighting conditions for
General setup steps should include:
darkfield observations.
8.2.1 Plug in the instrument and turn on the light source,
stage controller, optical camera, and hyperspectral camera.
8.2.5.2 Move the condenser focus up and down via the
Raisethestagetoitsmaximumheightandlowerthecondenser
condenser adjustment knob until maximum brightness is ob-
toitsminimumheight;connectthelightguidetothecondenser
tained. Create a bright central spot in the FOV.
for darkfield imaging.
8.2.5.3 Use the fine objective focus knob to bring the bright
8.2.2 Position the sample slide on the stage with the sample
spot (and the sample) into focus.
residing on top of the slide with a #1.5 thickness coverslip.
8.2.5.4 If a different magnification is required, place the
8.2.3 Raise the condenser to its operating position and
correct objective into place (the sample stage may need to be
partiallyalignitsothatlightcanbeseenthroughtheeyepieces
lowered) and establish focus. In the case of an oil immersion
or attached camera system. Focus the image of the sample on
objective, place a drop of immersion oil on the slide, avoiding
the slide using a 10× dry or 20× or 60× oil objective. In the
the formation of any bubbles, and slowly raise the stage until
caseofanoilimmersioncondenser,placethreetofivedropsof
the immersion oil contacts the objective; then establish focus.
immersion oil on the condenser lens avoiding the formation of
any bubbles. Make sure to use the microscope manufacturer’s 8.3 Inverted Microscope Setup—Generally, an upright mi-
recommended type of oil immersion medium because the oils croscope is preferable for darkfield imaging compared to an
have different viscosities. Also, it is best not to mix different inverted microscope. Not all manufacturer’s microscopes can
types of oils. be used easily with an oil condenser and some cannot be used
8.2.4 Put a low-magnification (for example, 10× to 20×) at all. The description in 8.3.1 – 8.3.7 is for new infinity-
objective in place. Examine the sample through the oculars, correctedmicroscopesthathavebeensuccessfullyusedwithan
focus on the sample, and align the condenser. oil condenser for DFM.
E3275−21
8.3.1 Plug in the instrument and turn on the light source, 9. Image Analysis
stage controller, optical camera, and hyperspectral camera.
9.1 Approaches to Imaging Software—Thesoftwareshallbe
8.3.2 Raise the condenser to its maximum height; this will
capable of performing curation and maintenance of RSLs,
resultinabrightfieldimagethatwillmakeiteasiertoestablish
background correction or accommodation, and spectral match-
focus.
ing of spectra presented by objects in the FOV using the RSL.
NOTE3—Withanoilimmersioncondenser,itislikelythatnolightwill
NOTE 5—This section should be read in association with Section 10.
come out of the condenser if there is no oil in contact with the slide.
8.3.3 Position the sample slide on the stage with the 9.2 System Calibration and Validation—Wavelength cali-
coverslip facing the objective. Generally, it is best to have the bration may be performed using commercially available low-
pressure ion emission lamps (LPIE) that emit spectral lines at
sample coverslip (#1.5 thickness) closest to the objective and
not facing the condenser. accurately known wavelengths. Depending on the spectral
range, LPIE lamps are commercially available with single or
8.3.4 Put a low-magnification objective in place. While
monitoring through the oculars, focus the sample using the multi-element combinations of mercury, argon, neon, and
edge of the microscope slide (or speckles of dust on the slide krypton. See Annex A1, Table A1.1 for a listing of useful
surface). emission wavelengths. The emission spectra of these lamps
8.3.5 By moving the microscope stage, navigate to an area enable an instrument user to validate wavelength accuracy and
of interest. actual spectral resolution at multiple wavelengths. If an LPIE
8.3.6 When using a dry condenser, while monitoring calibration lamp is unavailable, fluorescent room lights con-
through the oculars, slowly lower the condenser until the taining mercury may be used for wavelength calibration,
ENMs appear bright over a dark background and contrast is although it should be noted that such lights might present
optimized. significantly broadened spectral lines. HSI instruments may be
supplied with various cameras and optical systems with each
NOTE4—Ifusinganoilcondenser,placeadropofimmersionoilonthe
componenthavinguniquespecifications.Consequently,factors
slide and lower the condenser until the oil contacts the slide. In that case,
that are wavelength dependent such as camera quantum effi-
it is preferable to place the slide such that the sample is located on the
lower surface of the slide facing the objective. While monitoring through
ciency (QE), the wavelength diffraction/refraction efficiency
theocularsorusinglivedisplayfromthecameraoutput,slowlyadjustthe
profile of a wavelength dispersive element, or a filter shall be
condenser position until the ENMs appear bright over a dark background
accommodated by “normalizing” output spectra. System “nor-
and contrast is optimized. The best operating conditions will occur when
malization” can be performed in one of two ways described in
the NA values of all media are matched. This means using an oil
9.2.1.
condenser, an oil objective, and a mounting medium that are close to the
refractive index (1.52) of the oil. An oil condenser with an oil objective
9.2.1 Working in Relative Percent Reflectance,
will yield the best image quality for darkfield measurements.
Transmission, or Absorption:
8.3.7 Change the microscope objective to the objective of
9.2.1.1 ENMscattercanbetreatedasreflectionortransmis-
interest. Adjust the focus. If the objective is equipped with a
sion spectra. Consequently, all spectral data should be pre-
correction ring to compensate for glass slide thickness, adjust
sented as relative percent reflectance, transmission, or absorp-
the ring accordingly and focus using the fine focus adjustment
tion. This method eliminates wavelength-dependent issues
knob.Whenanoilimmersionobjectiveistobeused,lowerthe
such as camera QE differences in wavelength selection
objective and place a drop of immersion oil on the microscope
devices, lens coatings, and light source.
objective avoiding the formation of any bubbles. Slowly raise
9.2.1.2 Relative percent reflection measurements are made
the microscope objective until the immersion oil contacts the
by removing the DFM condenser and acquiring a spectrum of
slide and establish focus. The condenser position can be
the illuminant (a clean microscope slide may be recommended
adjusted for maximum brightness and field alignment. The iris
to account for contributions from the slide) just below saturat-
diaphragm contained in some objectives (usually with 40× to
ing the detector. This spectrum is then divided into spectra
100×lenses)mustbeadjustedtoachieveanNAvaluelessthan
acquired from the FOV.
0.9fordarkfieldimagingtooccur.Reflectionswilloccurwhen
9.2.1.3 Radiometric Calibration—HSI systems may also be
theNAvalueisabove0.9.ThesmallertheNAvalue,thebetter
radiometrically corrected using a National Institute of Stan-
the depth of field and the better the darkfield image quality.
dards andTechnology (NIST)-certified lamp.The digital spec-
However, the NA is directly related to the cutoff spatial
tral output presented by the instrument is compared to the
frequency, thus the spatial resolution will be lowered, and the
values stated in the certificate of the lamp and a correction
amount of transmitted light will be reduced. The values of the
factor is computed. This also has the effect of eliminating
irisdiaphragmneedtobebalancedtooptimizedarkfieldimage
contributions of camera QE and wavelength selection effi-
quality and spatial resolution. The essential rule is that one
ciency variations as a function of wavelength.
cannot exceed an NA value of 0.9 when using darkfield
illumination.
9.3 Classification of Materials in an Image:
8.4 Capturing Images with HSI Systems—Capture images 9.3.1 If a researcher needs evidence that the material clas-
according to the instrument operating manual. Instructions for sification was a success, ground truth spectral characterization
capturing hyperspectral data cubes strongly depends on the at the source will confirm the accuracy. Ground truth determi-
specific system, that is, push broom (WDS) and tunable nations are typically not possible without damaging the
imaging filter models. sample. In practice, scanning electron microscopy/electron/
E3275−21
energydispersiveX-ray(SEM/EDX)canpointtowardsground validation but may not offer reliable examples of reference
truth, but ground truth is rarely obtained. spectra that will occur in real cell samples. When ENMs are
9.3.2 ENMs associate with each other and matrix compo- associated with biological materials with some particles inter-
nents creating interdependent relationships that comingle with nalized and others in the media surrounding the cells or on the
scatter and diffusion. Spectral scatter from particles in biologi- cellsurface,spectralprofilescanbecomehighlyconvoluted.In
cal systems are particularly diverse because of variations in this case, the algorithms used to classify these spectra can be
pH, dissolution, protein binding, agglomeration, clustering, challenging.
and aggregation. These factors increase the complexity in
NOTE 7—The creation of an absolute RSLmay simply not be possible;
classificationofENMsinahyperspectralimage.Classification
however, a relative RSL can be robust and very useful if generated using
ofmaterialsinahyperspectralimageshouldbecarriedoutwith
linearity independent “matching” algorithms. Consult the instrument
great care using the algorithms recommended by the instru- manufacturer for more information.
mentmanufacturerthatarelinearityindependent(forexample,
10.1.2 Generating RSLs:
cross correlation). When working in percent transmission or
10.1.2.1 RSLs should be sample-specific. Variations in
percent reflectance mode, compensation for non-sample-
sample preparation and cell type will result in variations in
related background can be obtained. RSL spectra that include
spectral signatures. All samples of the same type, prepared
the evaluation of the sample matrix as well as identifiable
underthesame(standardized)conditions,shouldbeabletouse
cellular features (for example, subcellular organelles, lipid
the same RSL.
membranes, and so forth) can significantly enhance the char-
10.1.2.2 Use the instructions supplied by the manufacturer
acterization and positive identification of ENMs in cellular
to acquire and sort spectra into classes.
samples. The software and user guides provided by the
(1)Workinrelativereflectionmode(dividesamplespectra
instrument manufacturer will provide the best practices for
by the spectrum of the illuminant).
implementation for a particular system. Identifying the pres-
(2)Add the spectra presented by select target regions of
ence of ENMs that have been internalized by a cell with HSI
interest to the RSL. Include the spectrum of the illuminant and
will be dependent on: (1) an RSL that has been carefully
region of interest inside and outside a cell or of the sample
curated and (2) the use of appropriate algorithms that enable
respective matrix plus any other area in the FOV that offers
spectral classification and identification. In general, data pro-
interesting targets.
cessing and correlation with RSLspectra should be performed
10.1.3 Supervised and Unsupervised Spectral Classifica-
on raw data or data that have undergone minimal processing.
tion:
Data may then be classified according to the RSLtraining sets
10.1.3.1 Depending on the software being used, there are
using an algorithm recommended by the HSI instrument
two methods of classifying and selecting spectra to insert into
manufacturer.
a library:
10. Reference Spectral Libraries (RSLs) (1) Unsupervised Selection—Inunsupervisedclassification
NOTE 6—Refer to important background information in Section 9.
algorithms, the computer generates candidate spectra based on
a region of interest (ROI) without training sets. Classes are
10.1 Perspective and Purpose of an RSL—HSI is basically
generated based on spatial, spectral, or spatial-spectral pat-
an object recognition technique. When a specific reflection or
terns. An advantage of unsupervised selection is that all
absorption spectrum associated with a known object or mate-
spectral features are kept; however, this will lead to some
rial is found within an area of a sample, the known object is
irrelevant or redundant classes generated. The user will then
“recognized” within that sample. A library of spectral finger-
selectsome(orall)classestoaddtotheRSLbasedonS/Nratio
prints or spectral “classes” is referred to as an RSLand is used
or another relevant metric.
to identify the presence of objects in a FOV using the RSL as
(2) Supervised Selection—Generally, in supervised
a reference.
selection,atrainingsetisgeneratedfromtheRSL.Itiscritical
10.1.1 Nature of RSL Spectra—Some materials present
that the RSL was obtained and curated carefully in conditions
spectra that are spectral profiles and permit recognition of the
as similar to the sample conditions as possible. Materials that
materials presenting such spectra. For example, the low-
exhibitpolymorphic,size-dependent,orotherconditionalspec-
pressure wavelength calibration lamps described in Section 9
tral changes are particularly important to capture in as close to
emit fingerprint emission lines at wavelengths known to many
sample conditions as possible for the RSL. Supervised selec-
decimal places and can be found in NIST “Basic Atomic
tions should be used when there is a known spectral class
Spectroscopic Data” listings (8). HSI instruments validated
present and it is available with excellent S/N in the RSL.
with these light sources can be compared to any other spectral
10.1.3.2 In practice, both methods can be used as needed.
instrument similarly calibrated. While some materials present
spectrathatpermitinstantabsoluterecognition,relativespectra 10.1.4 Pseudo- or False-Colored Spectral Classes—Even
cannot be used for unequivocal object recognition out of thoughagiventestsampleofENMsmayundergoaggregation,
context. This is almost always the case for reflection/ agglomeration, structural adherence, or potential interactions,
transmission and fluorescence spectra of composite materials. orcombinationsthereof,withmatrixmaterialswithinaFOV,it
Samples containing ENMs will use relative library spectra is possible to acquire and classify the spectra from the FOV.
when acquired in relative percent reflection for insertion into Each class will include similar, but somewhat different, spec-
an RSL. Spectra of NIST gold NP reference materials RM- tra. This reduces or eliminates the selection of spectra that
8011, RM-8012, and RM-8013 may be excellent for system differ only as a function of shot noise (sampling noise),
E3275−21
background, matrix effects, and complex diversity. Once “subtracted” from sample spectra with ENMs because of
generated, an RSL enables recognition of similar spectral background heterogeneity.
profiles in unknown but similar samples. RSL spectra should
10.3.3 Libraries as a Function of Particle Size—ENMs that
be pseudo-colored so that a hyperspectral image, presenting
are present both inside and outside a cell have the potential to
objects that correlate with an RSL spectrum, can acquire its
besingleparticles,pairs,aggregates,clusters,oragglomerates,
pseudo-color. Raw hyperspectral data sets should include
or combinations thereof. If enough ENMs associate to form a
spe
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...