ASTM D3920-18
(Test Method)Standard Test Method for Strontium in Water
Standard Test Method for Strontium in Water
SIGNIFICANCE AND USE
5.1 Although most potable supplies contain little strontium, some well waters in the midwestern part of the United States have levels as high as 39 mg/L.3
5.2 This test method affords a reliable means of accurately determining strontium and correcting calcium results obtained by the methods cited in 6.2.
5.3 ICP-MS or ICP-AES may also be appropriate but at a higher instrument cost. See Test Methods D1976 and D5673.
SCOPE
1.1 This test method covers the determination of dissolved and total recoverable strontium in water and wastewater by atomic absorption spectroscopy.
1.2 The test method is applicable in the range from 0.1 to 1 mg/L of strontium. The range may be extended by dilution of the original sample.
1.3 Round-robin data were obtained in natural and reagent water matrices. It is the user's responsibility to ensure the validity of the test method for waters of untested matrices.
1.4 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific precautionary statements are given in 8.4 and 8.9.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Jan-2018
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.05 - Inorganic Constituents in Water
Relations
- Effective Date
- 01-Feb-2018
- Effective Date
- 01-May-2020
- Effective Date
- 01-May-2020
- Effective Date
- 01-Jul-2018
- Effective Date
- 01-Jul-2015
- Effective Date
- 01-Jan-2013
- Effective Date
- 01-Jan-2013
- Effective Date
- 15-Jun-2012
- Effective Date
- 01-Mar-2012
- Effective Date
- 01-Sep-2011
- Effective Date
- 01-May-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Aug-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Oct-2008
Overview
ASTM D3920-18 is the internationally recognized standard test method for the determination of strontium in water and wastewater. Developed by ASTM International, this method utilizes atomic absorption spectroscopy (AAS) to measure both dissolved and total recoverable strontium, offering reliable results in the concentration range of 0.1 to 1 mg/L. The method can be extended to higher concentrations through sample dilution. Its primary significance is in supporting water quality testing, especially where well waters may contain elevated levels of strontium, such as in certain regions of the midwestern United States. Accuracy is reinforced via strict quality control protocols and the ability to identify and correct interference from other elements.
Key Topics
- Test Method: Utilizes atomic absorption spectrophotometry, offering a practical balance of precision, cost, and operational simplicity for strontium analysis in water.
- Sample Types: Applicable to both dissolved and total recoverable strontium in natural waters and wastewaters.
- Concentration Range: 0.1–1 mg/L, with extension possible by dilution.
- Chemical Interferences: Managed by using lanthanum chloride and potassium chloride in sample preparation.
- Quality Control: Requires rigorous calibration, use of method blanks, matrix spikes, duplicates, and laboratory control samples to ensure accuracy and reproducibility.
- Instrument Requirements: Atomic absorption spectrophotometer with specific accessories, such as a strontium hollow-cathode lamp, is essential.
Applications
ASTM D3920-18 is widely used in the following sectors and situations:
- Municipal Water Testing: Ensuring compliance with safety standards by detecting strontium levels in drinking water supplies.
- Environmental Monitoring: Supporting regulatory and research activities by tracking strontium in surface water, groundwater, and wastewater effluent.
- Industrial Water Quality Management: Assisting industries that discharge wastewater containing inorganic constituents to monitor and control strontium concentrations.
- Correcting Calcium Analyses: Enabling the correction of calcium results affected by the presence of strontium, thereby improving the accuracy of broader water chemistry assessments.
- Laboratory Accreditation: Providing a peer-reviewed, globally recognized method essential for laboratory accreditation and proficiency testing in water analysis.
The robust quality control guidelines outlined in ASTM D3920-18 help laboratories demonstrate their performance and credibility, while the standard’s adaptability facilitates its use in a wide variety of water matrices.
Related Standards
To support comprehensive water analysis and maximize the utility of ASTM D3920-18, the following related ASTM standards are frequently referenced or recommended:
- ASTM D1193: Specification for Reagent Water
- ASTM D1976: Test Method for Elements in Water by ICP Atomic Emission Spectroscopy
- ASTM D5673: Test Method for Elements in Water by Inductively Coupled Plasma-Mass Spectrometry (ICP-MS)
- ASTM D3370: Practices for Sampling Water from Closed Conduits
- ASTM D4691: Practice for Measuring Elements in Water by Flame Atomic Absorption Spectrophotometry
- ASTM D2777: Practice for Determination of Precision and Bias
- ASTM D5810: Guide for Spiking into Aqueous Samples
Summary
ASTM D3920-18 provides environmental laboratories, municipal authorities, and industry with a robust, accurate, and cost-effective procedure for the quantification of strontium in water. By relying on well-established atomic absorption spectroscopy techniques and integrating essential quality control methods, it ensures both compliance and scientific integrity in water quality monitoring. This standard remains a critical reference in global water testing protocols, supporting the protection of public health and the environment.
Buy Documents
ASTM D3920-18 - Standard Test Method for Strontium in Water
REDLINE ASTM D3920-18 - Standard Test Method for Strontium in Water
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D3920-18 is a standard published by ASTM International. Its full title is "Standard Test Method for Strontium in Water". This standard covers: SIGNIFICANCE AND USE 5.1 Although most potable supplies contain little strontium, some well waters in the midwestern part of the United States have levels as high as 39 mg/L.3 5.2 This test method affords a reliable means of accurately determining strontium and correcting calcium results obtained by the methods cited in 6.2. 5.3 ICP-MS or ICP-AES may also be appropriate but at a higher instrument cost. See Test Methods D1976 and D5673. SCOPE 1.1 This test method covers the determination of dissolved and total recoverable strontium in water and wastewater by atomic absorption spectroscopy. 1.2 The test method is applicable in the range from 0.1 to 1 mg/L of strontium. The range may be extended by dilution of the original sample. 1.3 Round-robin data were obtained in natural and reagent water matrices. It is the user's responsibility to ensure the validity of the test method for waters of untested matrices. 1.4 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific precautionary statements are given in 8.4 and 8.9. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Although most potable supplies contain little strontium, some well waters in the midwestern part of the United States have levels as high as 39 mg/L.3 5.2 This test method affords a reliable means of accurately determining strontium and correcting calcium results obtained by the methods cited in 6.2. 5.3 ICP-MS or ICP-AES may also be appropriate but at a higher instrument cost. See Test Methods D1976 and D5673. SCOPE 1.1 This test method covers the determination of dissolved and total recoverable strontium in water and wastewater by atomic absorption spectroscopy. 1.2 The test method is applicable in the range from 0.1 to 1 mg/L of strontium. The range may be extended by dilution of the original sample. 1.3 Round-robin data were obtained in natural and reagent water matrices. It is the user's responsibility to ensure the validity of the test method for waters of untested matrices. 1.4 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific precautionary statements are given in 8.4 and 8.9. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D3920-18 is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D3920-18 has the following relationships with other standards: It is inter standard links to ASTM D3920-12, ASTM D1129-13(2020)e2, ASTM D1976-20, ASTM D1976-18, ASTM D5673-15, ASTM D4841-88(2013)e1, ASTM D4841-88(2013), ASTM D2777-12, ASTM D1976-12, ASTM D4691-11, ASTM D5810-96(2011), ASTM D3370-10, ASTM D5673-10, ASTM D1129-10, ASTM D3370-08. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D3920-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D3920 − 18
Standard Test Method for
Strontium in Water
This standard is issued under the fixed designation D3920; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* D2777 Practice for Determination of Precision and Bias of
Applicable Test Methods of Committee D19 on Water
1.1 This test method covers the determination of dissolved
D3370 Practices for Sampling Water from Closed Conduits
and total recoverable strontium in water and wastewater by
D4691 Practice for Measuring Elements in Water by Flame
atomic absorption spectroscopy.
Atomic Absorption Spectrophotometry
1.2 The test method is applicable in the range from 0.1 to 1
D4841 Practice for Estimation of Holding Time for Water
mg/L of strontium. The range may be extended by dilution of
Samples Containing Organic and Inorganic Constituents
the original sample.
D5673 Test Method for Elements in Water by Inductively
1.3 Round-robin data were obtained in natural and reagent Coupled Plasma—Mass Spectrometry
D5810 Guide for Spiking into Aqueous Samples
water matrices. It is the user’s responsibility to ensure the
validity of the test method for waters of untested matrices. D5847 Practice for Writing Quality Control Specifications
for Standard Test Methods for Water Analysis
1.4 The values stated in SI units are to be regarded as
standard. The values given in parentheses are mathematical
3. Terminology
conversions to inch-pound units that are provided for informa-
3.1 Definitions:
tion only and are not considered standard.
3.1.1 For definitions of terms used in this standard, refer to
1.5 This standard does not purport to address all of the
Terminology D1129.
safety concerns, if any, associated with its use. It is the
3.2 Definitions of Terms Specific to This Standard:
responsibility of the user of this standard to establish appro-
3.2.1 total recoverable strontium, n—a descriptive term
priate safety, health, and environmental practices and deter-
relating to the forms of strontium that are determinable by the
mine the applicability of regulatory limitations prior to use.
digestion procedure described in this test method.
Specific precautionary statements are given in 8.4 and 8.9.
1.6 This international standard was developed in accor- 3.2.2 laboratory control sample, n—a solution with a certi-
dance with internationally recognized principles on standard- fied concentration of the strontium.
ization established in the Decision on Principles for the
4. Summary of Test Method
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
4.1 Strontium is determined by atomic absorption spectro-
Barriers to Trade (TBT) Committee.
photometry.Thesampleisaspiratedintoanair-acetyleneflame
following the addition of lanthanum chloride/potassium chlo-
2. Referenced Documents
ride solution. Samples containing particulate matter that may
2.1 ASTM Standards: clogtheaspiratorcapillaryorburner,thusproducinginaccurate
D1129 Terminology Relating to Water results, are filtered through a 0.45-µm membrane filter prior to
D1193 Specification for Reagent Water testing.
D1976 Test Method for Elements in Water by Inductively-
5. Significance and Use
Coupled Argon Plasma Atomic Emission Spectroscopy
5.1 Although most potable supplies contain little strontium,
some well waters in the midwestern part of the United States
This test method is under the jurisdiction of ASTM Committee D19 on Water
have levels as high as 39 mg/L.
and is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents
in Water.
5.2 This test method affords a reliable means of accurately
Current edition approved Feb. 1, 2018. Published May 2018. Originally
determining strontium and correcting calcium results obtained
approved in 1980. Last previous edition approved in 2012 as D3920 – 12. DOI:
10.1520/D3920-18. by the methods cited in 6.2.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on Standard Method for the Examination of Water and Wastewater, 14th Ed.
the ASTM website. American Public Health Assn., Washington, DC 20005.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D3920 − 18
5.3 ICP-MS or ICP-AES may also be appropriate but at a (Warning—Add acid slowly and in small portion to control
higher instrument cost. See Test Methods D1976 and D5673. the reaction rate upon mixing.)
8.5 Nitric Acid (sp gr 1.42)—Concentrated HNO .
6. Interferences
8.6 Strontium Solution, Stock (1.0 mL = 1.0 mg Sr)—
6.1 Chemical interference caused by silicon, aluminum, and
Dissolve 2.415 g of strontium nitrate (Sr(NO ) ) in a 1-L flask
3 2
phosphate is controlled by adding lanthanum chloride. Potas-
containing 10 mL of concentrated HCl and 700 mL of water.
sium chloride is added to suppress the ionization of strontium.
Dilute to 1000 mL with water. A purchased strontium stock
NOTE1—Anitrousoxide-acetyleneflamehasbeenusedsuccessfullyby
solution of appropriate known purity is also acceptable.
some to remove chemical interferences.
8.7 Strontium Solution, Standard (1 mL = 0.010 mg Sr)—
6.2 Strontium chemically resembles calcium and causes a
Dilute 5.0 mL of strontium solution standard to 500 mL with
positive error in gravimetric and titrimetric methods for cal-
water.
cium determination.
8.8 Oxidant—Air that has been cleaned and dried through a
7. Apparatus
suitable filter to remove oil, water, and other foreign
substances, is the usual oxidant.
7.1 Atomic Absorption Spectrophotometer for use at 460.7
nm. A general guide for the use of flame atomic absorption
8.9 Fuel—Standard, commercially available acetylene is the
applications is given in Practice D4691.
usual fuel.Acetone, always present in acetylene cylinders, can
be prevented from entering the burner system by replacing the
NOTE 2—The manufacturer’s instructions should be followed for
setting instrumental parameters. cylinder when the pressure reaches 490 kPa (70 psig).
(Warning—Purified grade acetylene containing a special pro-
7.2 Strontium Hollow-Cathode Lamp.
prietary solvent other than acetone should not be used with
poly (vinyl chloride) tubing as weakening of the walls may
8. Reagents and Materials
result and cause a potentially hazardous situation.)
8.1 Purity of Reagents—Reagent grade chemicals shall be
8.10 Filter Paper—Purchase suitable filter paper. Typically
used in all tests. Unless otherwise indicated, it is intended that
the filter papers have a pore size of 0.45-µm membrane.
all reagents shall conform to the specifications of the Commit-
Material such as fine-textured, acid-washed, ashless paper, or
tee onAnalytical Reagents of theAmerican Chemical Society,
glass fiber paper are acceptable. The user must first ascertain
where such specifications are available. Other grades may be
that the filter paper is of sufficient purity to use without
used, provided it is first ascertained that the reagent is of
adversely affecting the bias and precision of the test method.
sufficiently high purity to permit its use without lessening the
accuracy of the determination.
9. Sampling
8.2 Purity of Water—Unless otherwise indicated, reference
towatershallbeunderstoodtomeanreagentwaterconforming
9.1 Collect the samples in accordance with instructions in
to Specification D1193, Type I, II, and III water. Type I is
Practices D3370. The holding time for the samples may be
preferred and more commonly used. Other reagent water types
calculated in accordance with Practice D4841.
may be used provided it is first ascertained that the water is of
9.2 To preserve the samples add concentrated HNO (sp gr
sufficiently high purity to permit its use without adversely
1.42) to a pH of 2 or less immediately at the time of collection;
affecting the precision and bias of the test method. Type II
normally about 2 mL/Lis required. If only dissolved strontium
water was specified at the time of round robin testing of this
is to be determined, filter (8.10) the samples at time of
test method.
collection through a 0.45-µm membrane filter before acidifica-
NOTE 3—The user must ensure the type of reagent water chosen is
tion.
sufficiently free of interferences. The water should be analyzed using the
test method. NOTE 4—Alternatively, the pH may be adjusted in the laboratory within
14 days of collection. This could reduce hazards of working with acids in
8.3 Hydrochloric Acid (sp gr 1.19)—Concentrated hydro-
the field when appropriate.
chloric acid (HCl).
8.4 Lanthanum Chloride/Potassium Chloride Solution—
10. Standardization
Dissolve 11.73 g of lanthanum oxide (La O ) in a minimum
2 3
10.1 An effective way to clean all glassware to be used for
amount of concentrated hydrochloric acid (approximately 50
preparation of standard solutions or in the digestion step, or
mL.).Add 1.91 g of potassium chloride (KCl).Allow solution
both, is by soaking the glassware for 2 h first with HNO
to cool to room temperature and dilute to 100 mL with water.
(1 + 1) and then rinsing with reagent water.
10.2 Prepare a blank and at least four working standards to
bracket the expected strontium concentration range of the
Reagent Chemicals, American Chemical Society Specifications, American
Chemical Society, Washington, DC. For Suggestions on the testing of reagents not
samples to be analyzed by diluting the standard strontium
listed by the American Chemical Society, see Annual Standards for Laboratory
solution (8.6) to the desired concentrations. Select concentra-
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
tions that will give a zero, middle, and maximum points for the
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
MD. analytical curve.
D3920 − 18
10.3 Pipette 10.0 mL of each standard into a 50-mL beaker 11.4 Pipette 10.0 mLof sample, or a dilution of the sample,
or flask and add 1.0 mL of lanthanum chloride/potassium into a 50-mL beaker or flask.
chloride solution. Mix well by swirling.
11.5 Add 1.0 mL of lanthanum chloride/potassium chloride
10.4 To test the suitability of reagents used in the analysis, solution (8.4) to each sample. Mix well by swirling.
zerotheinstrumentwhileaspiratingreagentwater.Aspiratethe
11.6 Aspirate each sample and record its absorbance or
zero standard and record the response. If the zero standard
concentration. Aspirate reagent water between each sample.
produces a response sufficient to affect the detection limit or
11.7 For instruments that do not readout directly in
accuracy, or both, of the test method, the contaminated reagent
concentration, determine the strontium concentration of the
should be identified and replaced with a grade of suitable
aspirated solution from the analytical curve (10.6).
quality before proceeding.
10.5 Aspirate the blank (zero standard) and adjust the
12. Calculation
instrument reading to zero. Aspirate standards and record the
12.1 Where dilutions have been made, calculate the concen-
instrument reading for each. Aspirate reagent water between
tration of strontium in the original sample as follows:
each standard.
B
Sr,mg/L 5 A 3 (1)
NOTE5—Bestresultshavebeenobtainedwithaslightlyfuelrichflame.
C
10.6 Read directly in concentration if this capability is
where:
provided with the instrument or measure the absorbance of the
A = Sr read from the analytical curve (10.6), mg/L,
standards or construct an analytical curve by plotting the
B = total final volume of the diluted sample, and
absorbance of standards versus milligrams of strontium per
C = volume of original sample diluted.
litre.
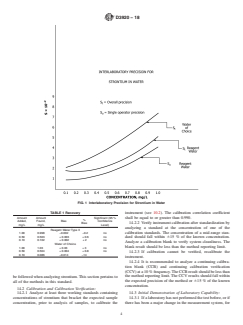
13. Precision and Bias
11. Procedure
13.1 The overall precision (reagent and waters of choice)
11.1 For total recoverable strontium, add 5 mL of concen-
and the single-operator precision for reagent water, based on
trated nitric acid (8.5) to 100 mL of the sample in a 250-mL
data from eight participating laboratories involving twelve
Erlenmeyer flask, and mix well. Heat the sample (between
operators, varies with the quantity measured within the desig-
65°C and 95°C) on a steam bath/hot plate below boiling in a
nated range of the test method as shown in Fig. 1.
well-ve
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D3920 − 12 D3920 − 18
Standard Test Method for
Strontium in Water
This standard is issued under the fixed designation D3920; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the determination of dissolved and total recoverable strontium in water and wastewater by atomic
absorption spectroscopy.
1.2 The test method is applicable in the range from 0.1 to 1 mg/L of strontium. The range may be extended by dilution of the
original sample.
1.3 Round-robin data were obtained in natural and reagent water matrices. It is the user’s responsibility to ensure the validity
of the test method for waters of untested matrices.
1.4 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values statedgiven
in each system parentheses are mathematical conversions and may not be exact equivalents; therefore, each system shall be used
independently of the other.to inch-pound units that are provided for information only and are not considered standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use. Specific precautionary statements are given in 8.4 and 8.9.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
D1976 Test Method for Elements in Water by Inductively-Coupled Argon Plasma Atomic Emission Spectroscopy
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D3370 Practices for Sampling Water from Closed Conduits
D4691 Practice for Measuring Elements in Water by Flame Atomic Absorption Spectrophotometry
D4841 Practice for Estimation of Holding Time for Water Samples Containing Organic and Inorganic Constituents
D5673 Test Method for Elements in Water by Inductively Coupled Plasma—Mass Spectrometry
D5810 Guide for Spiking into Aqueous Samples
D5847 Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
3. Terminology
3.1 Definitions—Definitions: For definitions of terms used in this test method, refer to Terminology D1129.
3.1.1 For definitions of terms used in this standard, refer to Terminology D1129.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 total recoverable strontium, n—an arbitrary analytical a descriptive term relating to the forms of strontium that are
determinable by the digestion procedure described in this test method.
3.2.2 laboratory control sample, n—a solution with a certified concentration of the strontium.
This test method is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents in Water.
Current edition approved Sept. 1, 2012Feb. 1, 2018. Published September 2012 May 2018. Originally approved in 1980. Last previous edition approved in 20072012 as
ɛ1
D3920 – 02 (2007)D3920 – 12. . DOI: 10.1520/D3920-12.10.1520/D3920-18.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D3920 − 18
4. Summary of Test Method
4.1 Strontium is determined by atomic absorption spectrophotometry. The sample is aspirated into an air-acetylene flame
following the addition of lanthanum chloride/potassium chloride solution. Samples containing particulate matter that may clog the
aspirator capillary or burner, thus producing inaccurate results, are filtered through a 0.45-μm membrane filter prior to testing.
5. Significance and Use
5.1 Although most potable supplies contain little strontium, some well waters in the midwestern part of the United States have
levels as high as 39 mg/L.
5.2 This test method affords a reliable means of accurately determining strontium and correcting calcium results obtained by
the methods cited in 6.2.
5.3 ICP-MS or ICP-AES may also be appropriate but at a higher instrument cost. See Test Methods D1976 and D5673.
6. Interferences
6.1 Chemical interference caused by silicon, aluminum, and phosphate is controlled by adding lanthanum chloride. Potassium
chloride is added to suppress the ionization of strontium.
NOTE 1—A nitrous oxide-acetylene flame has been used successfully by some to remove chemical interferences.
6.2 Strontium chemically resembles calcium and causes a positive error in gravimetric and titrimetric methods for calcium
determination.
7. Apparatus
7.1 Atomic Absorption Spectrophotometer for use at 460.7 nm. A general guide for the use of flame atomic absorption
applications is given in Practice D4691.
NOTE 2—The manufacturer’s instructions should be followed for setting instrumental parameters.
7.2 Strontium Hollow-Cathode Lamp.
8. Reagents and Materials
8.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where
such specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high
purity to permit its use without lessening the accuracy of the determination.
8.2 Purity of Water—Unless otherwise indicated, reference to water shall be understood to mean reagent water conforming to
Specification D1193, Type I, II, and III water. Type I is preferred and more commonly used. Other reagent water types may be used
provided it is first ascertained that the water is of sufficiently high purity to permit its use without adversely affecting the precision
and bias of the test method. Type II water was specified at the time of round robin testing of this test method.
NOTE 3—The user must ensure the type of reagent water chosen is sufficiently free of interferences. The water should be analyzed using the test method.
8.3 Hydrochloric Acid (sp gr 1.19)—Concentrated hydrochloric acid (HCl).
8.4 Lanthanum Chloride/Potassium Chloride Solution—Dissolve 11.73 g of lanthanum oxide (La O ) in a minimum amount of
2 3
concentrated hydrochloric acid (approximately 50 mL.). Add 1.91 g of potassium chloride (KCl). Allow solution to cool to room
temperature and dilute to 100 mL with water. (Warning—Add acid slowly and in small portion to control the reaction rate upon
mixing.Warning)—Add acid slowly and in small portion to control the reaction rate upon mixing.)
8.5 Nitric Acid (sp gr 1.42)—Concentrated HNO .
8.6 Strontium Solution, Stock (1.0 mL = 1.0 mg Sr)—Dissolve 2.415 g of strontium nitrate (Sr(NO ) ) in a 1-L flask containing
3 2
10 mL of concentrated HCl and 700 mL of water. Dilute to 1000 mL with water. A purchased strontium stock solution of
appropriate known purity is also acceptable.
8.7 Strontium Solution, Standard (1 mL = 0.010 mg Sr)—Dilute 5.0 mL of strontium solution standard to 500 mL with water.
8.8 Oxidant—Air that has been cleaned and dried through a suitable filter to remove oil, water, and other foreign substances,
is the usual oxidant.
Standard Method for the Examination of Water and Wastewater, 14th Ed. American Public Health Assn., Washington, DC 20005.
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For Suggestions on the testing of reagents not listed by
the American Chemical Society, see Annual Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
D3920 − 18
8.9 Fuel—Standard, commercially available acetylene is the usual fuel. Acetone, always present in acetylene cylinders, can be
prevented from entering the burner system by replacing the cylinder when the pressure reaches 490 kPa (70 psig).
(Warning—Purified grade acetylene containing a special proprietary solvent other than acetone should not be used with poly
(vinyl chloride) tubing as weakening of the walls may result and cause a potentially hazardous situation.Warning)—Purified grade
acetylene containing a special proprietary solvent other than acetone should not be used with poly (vinyl chloride) tubing as
weakening of the walls may result and cause a potentially hazardous situation.)
8.10 Filter Paper—Purchase suitable filter paper. Typically the filter papers have a pore size of 0.45-μm membrane. Material
such as fine-textured, acid-washed, ashless paper, or glass fiber paper are acceptable. The user must first ascertain that the filter
paper is of sufficient purity to use without adversely affecting the bias and precision of the test method.
9. Sampling
9.1 Collect the samples in accordance with instructions in Practices D3370. The holding time for the samples may be calculated
in accordance with Practice D4841.
9.2 To preserve the samples add concentrated HNO (sp gr 1.42) to a pH of 2 or less immediately at the time of collection;
normally about 2 mL/L is required. If only dissolved strontium is to be determined, filter (8.10) the samples at time of collection
through a 0.45-μm membrane filter before acidification.
NOTE 4—Alternatively, the pH may be adjusted in the laboratory if the sample is returned within 14 days. within 14 days of collection. This could
reduce hazards of working with acids in the field when appropriate.
10. Standardization
10.1 An effective way to clean all glassware to be used for preparation of standard solutions or in the digestion step, or both,
is by soaking the glassware for 2 h first with HNO (1 + 1) and then rinsing with reagent water.
10.2 Prepare a blank and at least four working standards to bracket the expected strontium concentration range of the samples
to be analyzed by diluting the standard strontium solution (8.6) to the desired concentrations. Select concentrations that will give
a zero, middle, and maximum points for the analytical curve.
10.3 Pipette 10.0 mL of each standard into a 50-mL beaker or flask and add 1.0 mL of lanthanum chloride/potassium chloride
solution. Mix well by swirling.
10.4 To test the suitability of reagents used in the analysis, zero the instrument while aspirating reagent water. Aspirate the zero
standard and record the response. If the zero standard produces a response sufficient to affect the detection limit or accuracy, or
both, of the test method, the contaminated reagent should be identified and replaced with a grade of suitable quality before
proceeding.
10.5 Aspirate the blank (zero standard) and adjust the instrument reading to zero. Aspirate standards and record the instrument
reading for each. Aspirate reagent water between each standard.
NOTE 5—Best results have been obtained with a slightly fuel rich flame.
10.6 Construct Read directly in concentration if this capability is provided with the instrument or measure the absorbance of
the standards or construct an analytical curve by plotting the absorbance of standards versus milligrams of strontium per litre.
Alternatively, read directly in concentration if this capability is provided in the instrument.
11. Procedure
11.1 For total recoverable strontium, add 5 mL of concentrated nitric acid (8.5)(8.5) to 100 mL of the sample in a 250-mL
Erlenmeyer flask, and mix well. Heat the sample at 95°C (between 65°C and 95°C) on a steam bath/hot plate below boiling in a
well-ventilated fume hood until the volume is reduced to 15 to 20 mL.mL, making certain that the samples do not boil.
NOTE 6—When testing samples of brine or samples containing a large amount of solids, the amount of reduction in volume is left to the discretion
of the analyst.
NOTE 7—Many laboratories have found block digestion systems a useful way to digest samples for trace metals analysis. Systems typically consist of
either a metal or graphite block with wells to hold digestion tubes. The block temperature controller must be able to maintain uniformity of temperature
across all positions of the block. The digestion block must be capable of maintaining a temperature between 65°C and 95°C. For trace metals analysis,
the digestion tubes should be constructed of polypropylene and have a volume accuracy of at least 0.5%. 0.5 %. All lots of tubes should come with a
certificate of analysis to demonstrate suitability for their intended purpose. If a block digestion system is used, reduced volumes of samples and reagents
will be required. Make sure that the reagent volumes maintain the same proportions as in the macro method.
11.2 If color in the digested solution indicates the presence of partially oxidized materials, add additional nitric acid and
approximately 90 mL of reagent water to the cooled solution and repeat the digestion as before. Repeat this step several times if
necessary.
11.3 Cool and filter the digested solution through a suitable filter (such as fine-textured, acid-washed ashless paper)paper; 8.10)
into a 100-mL volumetric flask. Wash the filter paper 2 to
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...