ASTM A630-16a(2024)
(Test Method)Standard Test Methods for Determination of Tin Coating Weights for Electrolytic Tin Plate
Standard Test Methods for Determination of Tin Coating Weights for Electrolytic Tin Plate
SIGNIFICANCE AND USE
20.1 This test method covers determination of the total tin in the sample tested and does not apportion the tin to one or the other side of the test specimen. The calculations appearing in Section 27 assume uniform distribution of tin over the two surfaces.
20.2 This test method does not differentiate between free tin on the tinplate surface, tin combined with iron in the intermediate alloy layer, or tin alloyed with the steel as a residual tramp element.
SCOPE
1.1 These test methods include four methods for the determination of tin coating weights for electrolytic tin plate as follows:
Test Method
Sections
A—Bendix Test Method
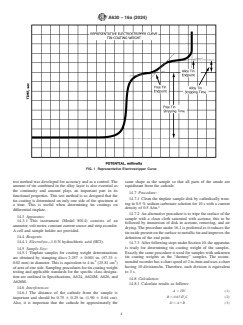
3 to 9
B—Constant-Current, Electrolytic Test Method (Referee Method)
10 to 17
C—Sellar's Test Method
18 to 27
D—Titration Test Method
28 to 36
1.2 The values stated in inch-pound units are to be regarded as standard. The values given in parentheses are mathematical conversions to SI units that are provided for information only and are not considered standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 29-Feb-2024
- Technical Committee
- A01 - Steel, Stainless Steel and Related Alloys
- Drafting Committee
- A01.20 - Tin Mill Products
Relations
- Effective Date
- 01-Mar-2024
- Effective Date
- 01-Mar-2024
- Effective Date
- 01-Mar-2024
- Effective Date
- 01-Mar-2024
Overview
ASTM A630-16a(2024), Standard Test Methods for Determination of Tin Coating Weights for Electrolytic Tin Plate, outlines internationally recognized procedures for measuring tin coating weights on electrolytic tin plate. This standard provides four primary test methods, ensuring accuracy and consistency in quality assurance, manufacturing, and regulatory compliance. Used broadly by steel producers, quality control laboratories, and end users in the packaging, food, and beverage industries, ASTM A630 helps guarantee the reliability and durability of tin-coated steel products.
Key Topics
- Purpose and Scope: Covers the determination of total tin content on electrolytic tin plate, without differentiating between tin forms (free tin, alloyed tin, or residual tramp element). Assumes uniform tin distribution on both sides of the sample.
- Test Methods Included:
- Bendix Test Method: Involves dissolution and titration processes for total tin measurement.
- Constant-Current, Electrolytic Test Method (Referee Method): Anodic stripping at constant current to determine both free and alloyed tin; recommended for referee purposes in case of disputes. Suitable for one-sided or differential coatings.
- Sellar’s Test Method: Dissolution in acid under controlled atmosphere followed by titration.
- Titration Test Method: Uses chemical dissolution and titration for tin quantification, especially applicable to higher tin contents.
- Unit Standards: Results are stated in inch-pound units, with SI conversions provided for informational purposes.
- Significance and Use: Designed to ensure production efficiency, product safety, and adherence to regulatory and trade requirements.
Applications
ASTM A630-16a(2024) is crucial for:
- Steel Manufacturers: Standardizes tin coating weight determination for consistent batch quality and compliance with industrial specifications such as ASTM A624/A624M and A626/A626M.
- Quality Control Laboratories: Provides reliable and reproducible methods for verifying tinplate coatings, vital for product validation and audit trails.
- Packaging Industry: Ensures tinplate used for food and beverage cans meets integrity standards for corrosion resistance and product protection.
- Regulatory Compliance: Supports international and domestic requirements, including those set by the World Trade Organization's Technical Barriers to Trade (TBT) Committee.
- Research and Development: Assists in developing new tinplate compositions, process optimization, and testing alternative measurement techniques.
Related Standards
For comprehensive tinplate testing and specification, refer to:
- ASTM A624/A624M: Specification for Tin Mill Products, Electrolytic Tin Plate, Single Reduced.
- ASTM A626/A626M: Specification for Tin Mill Products, Electrolytic Tin Plate, Double Reduced.
- ASTM D1125: Test Methods for Electrical Conductivity and Resistivity of Water - relevant for quality of reagents.
- ISO 2177: Electrochemical methods for measuring metallic coatings.
Practical Value
Implementing ASTM A630-16a(2024) ensures:
- Consistency in Tin Coating Measurements: By following recognized test methods, labs and manufacturers achieve comparable results regardless of location.
- Improved Product Performance: Accurate tin weight measurement directly impacts the corrosion resistance and service life of tinplate products.
- Quality Assurance: Minimizes the risk of non-compliance with customer or regulatory specifications.
- Dispute Resolution: Provides a clear referee method in case of conflicting results between suppliers and consumers.
By adhering to ASTM A630-16a(2024), organizations demonstrate commitment to best practices in tinplate production and quality control, maintain market reputation, and meet stringent material requirements for high-performance steel packaging solutions.
Buy Documents
ASTM A630-16a(2024) - Standard Test Methods for Determination of Tin Coating Weights for Electrolytic Tin Plate
Get Certified
Connect with accredited certification bodies for this standard

National Aerospace and Defense Contractors Accreditation Program (NADCAP)
Global cooperative program for special process quality in aerospace.

CARES (UK Certification Authority for Reinforcing Steels)
UK certification for reinforcing steels and construction.

DVS-ZERT GmbH
German welding certification society.
Sponsored listings
Frequently Asked Questions
ASTM A630-16a(2024) is a standard published by ASTM International. Its full title is "Standard Test Methods for Determination of Tin Coating Weights for Electrolytic Tin Plate". This standard covers: SIGNIFICANCE AND USE 20.1 This test method covers determination of the total tin in the sample tested and does not apportion the tin to one or the other side of the test specimen. The calculations appearing in Section 27 assume uniform distribution of tin over the two surfaces. 20.2 This test method does not differentiate between free tin on the tinplate surface, tin combined with iron in the intermediate alloy layer, or tin alloyed with the steel as a residual tramp element. SCOPE 1.1 These test methods include four methods for the determination of tin coating weights for electrolytic tin plate as follows: Test Method Sections A—Bendix Test Method 3 to 9 B—Constant-Current, Electrolytic Test Method (Referee Method) 10 to 17 C—Sellar's Test Method 18 to 27 D—Titration Test Method 28 to 36 1.2 The values stated in inch-pound units are to be regarded as standard. The values given in parentheses are mathematical conversions to SI units that are provided for information only and are not considered standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 20.1 This test method covers determination of the total tin in the sample tested and does not apportion the tin to one or the other side of the test specimen. The calculations appearing in Section 27 assume uniform distribution of tin over the two surfaces. 20.2 This test method does not differentiate between free tin on the tinplate surface, tin combined with iron in the intermediate alloy layer, or tin alloyed with the steel as a residual tramp element. SCOPE 1.1 These test methods include four methods for the determination of tin coating weights for electrolytic tin plate as follows: Test Method Sections A—Bendix Test Method 3 to 9 B—Constant-Current, Electrolytic Test Method (Referee Method) 10 to 17 C—Sellar's Test Method 18 to 27 D—Titration Test Method 28 to 36 1.2 The values stated in inch-pound units are to be regarded as standard. The values given in parentheses are mathematical conversions to SI units that are provided for information only and are not considered standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM A630-16a(2024) is classified under the following ICS (International Classification for Standards) categories: 25.220.40 - Metallic coatings. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM A630-16a(2024) has the following relationships with other standards: It is inter standard links to ASTM A630-16ae1, ASTM A624/A624M-22, ASTM A599/A599M-07(2017), ASTM A626/A626M-22. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM A630-16a(2024) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: A630 − 16a (Reapproved 2024)
Standard Test Methods for
Determination of Tin Coating Weights for Electrolytic Tin
Plate
This standard is issued under the fixed designation A630; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
INTRODUCTION
Four test methods for determination of tin coating weights are described. These are typical methods
and represent those most commonly used in the tin plate industry. Publication of these test methods
is not intended to preclude the use of any other methods such as X-ray fluorescence measurement
systems for control purposes by the consumer or supplier. However, in case of dispute, the referee
method is to be used to determine conformance to Specification A624/A624M and Specification
A626/A626M.
Sampling procedures for tin coating-weight testing and applicable standards for the specific class
designation are outlined in Specification A624/A624M and Specification A626/A626M.
1. Scope* 2. Referenced Documents
1.1 These test methods include four methods for the deter- 2.1 ASTM Standards:
mination of tin coating weights for electrolytic tin plate as
A624/A624M Specification for Tin Mill Products, Electro-
follows: lytic Tin Plate, Single Reduced
A626/A626M Specification for Tin Mill Products, Electro-
Test Method Sections
A—Bendix Test Method 3 to 9
lytic Tin Plate, Double Reduced
B—Constant-Current, Electrolytic Test Method (Referee Method) 10 to 17
D1125 Test Methods for Electrical Conductivity and Resis-
C—Sellar’s Test Method 18 to 27
tivity of Water
D—Titration Test Method 28 to 36
METHOD A—DETERMINATION OF THE TIN
1.2 The values stated in inch-pound units are to be regarded
COATING WEIGHTS BY THE BENDIX TEST
as standard. The values given in parentheses are mathematical
METHOD
conversions to SI units that are provided for information only
and are not considered standard.
3. Scope
1.3 This standard does not purport to address all of the
3.1 This test method covers the determination of tin coating
safety concerns, if any, associated with its use. It is the
weights on steel plate.
responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter-
4. Summary of Test Method
mine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accor-
4.1 The procedure involves dissolution of tin from a tin
dance with internationally recognized principles on standard-
plate anode in a dilute hydrochloric acid solution containing a
ization established in the Decision on Principles for the
measured excess of standard potassium iodate-potassium io-
Development of International Standards, Guides and Recom-
dide solution. Excess iodine from the iodate-iodide solution is
mendations issued by the World Trade Organization Technical
back titrated with standard sodium thiosulfate using a starch
Barriers to Trade (TBT) Committee.
indicator.
These test methods are under the jurisdiction of ASTM Committee A01 on
Steel, Stainless Steel and Related Alloys and are the direct responsibility of
Subcommittee A01.20 on Tin Mill Products. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved March 1, 2024. Published March 2024. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
ɛ1
approved in 1968. Last previous edition approved in 2016 as A630 – 16a . DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/A0630-16AR24. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
A630 − 16a (2024)
5. Apparatus the flask to a carbon dioxide (CO ) system and displace the air
in the flask with CO . While continuing the flow of CO , heat
2 2
5.1 The detinning cell consists essentially of two cathodes
the flask but do not boil violently. After the tin has dissolved
of carbon rod, the sample that serves as an anode, and a beaker
add 0.5 g of antimony and 2 g of high-purity aluminum. Heat
of dilute hydrochloric acid electrolyte. The carbon rods (6 by
until the aluminum is completely dissolved and digest for an
0.25 in. or 152 by 6.35 mm, encased in porous porcelain
additional 10 min. Cool the flask to room temperature in
thimbles) are suspended from a suitable frame about 1 in.
running water while maintaining an atmosphere of CO .
(25 mm) apart. A small glass-enclosed magnet is attached to
Disconnect from the CO system and titrate with the KIO -KI
2 3
the frame in such a manner that it will hold the sample
solution using starch as an indicator. Calculate the tin titer, T,
suspended midway between the two cathodes. A movable
as follows:
platform permits the beaker of electrolyte to be brought up
around the assembly so that the sample will be completely T 5 A/B
immersed.
where:
5.2 A source of direct current that can be regulated to supply
A = tin used (0.2700 g),
up to 3 A at 3 to 5 V through the deplater is required.
B = KIO -KI solution used for titration, mL, and
T = tin titer for above KIO -KI solution, (grams of tin/mL),
5.3 Although regular laboratory glassware can be used, it is 3
2 2
T = 17.28 = lb/base box (bb)/mL, for a 4-in. (25.81-cm )
advisable to use automatic dispensing pipets or burets, a
sample.
motor-driven stirrer for titrations, and a timing switch when
large numbers of determinations are to be made.
6.5 Sodium Thiosulfate, Standard Solution (for coatings
over 0.50 lb/bb)—Dissolve 15.11 g of Na S O ·5 H O and 1.11
5.4 Precautions—The apparatus must be kept in continuous
2 2 3 2
g of NaOH in 1 litre of distilled water in a light-proof bottle.
operation to prevent iron in the solution adhering to the porous
Allow this solution to age for 72 h, remix, and standardize as
cells from oxidizing and subsequently liberating iodine from
follows: Connect the bottle to the Bendix apparatus and titrate
the potassium iodate-potassium iodide solution. If the instru-
20 mL of the standardized KIO -KI solution with the thiosul-
ment has been idle for some time, it is necessary to remove the 3
fate solution using the same procedure as is used for making
ferric iron by running a disk of tinplate through the regular
weight of coating determinations, but ignore the stripping unit.
procedure before test samples are run.
The tin equivalent of the Na S O solution in pounds per base
2 2 3
box is equal to: 20/A × T × 17.28, where A = millilitres of
6. Reagents
sodium thiosulfate solution used in titrating 20 mL of standard
6.1 Purity of Reagents—Reagent grade chemicals shall be
KIO -KI solution. A chart can be prepared showing lb/bb/mL
used in all tests. Unless otherwise indicated, it is intended that
of thiosulfate.
all reagents shall conform to the specifications of the Commit-
tee on Analytical Reagents of the American Chemical Society 6.6 Sodium Thiosulfate, Standard Solution (for coatings
where such specifications are available. Other grades may be
0.50 lb/bb and under)—Dissolve 6.57 g of Na S O ·5 H O and
2 2 3 2
used, provided it is first ascertained that the reagent is of 2.78 g of NaOH in 1 litre of distilled water in a light-proof
sufficiently high purity to permit its use without lessening the
bottle. Larger quantities in the same proportions may be
accuracy of the determination. prepared if desired. Allow that solution to age for 72 h, remix,
and standardize by the same procedure used for the stronger
6.2 Water—Deionized or distilled water having a volume
Na S O solution but use only 10 mL of potassium iodate-
2 2 3
resistivity greater than 1 MΩ·cm at 25 °C as determined by
potassium iodide solution.
Nonreferee Method of Test Methods D1125.
6.7 Starch Solution—Heat 200 mL of distilled water to
6.3 Hydrochloric Acid (1.7 to 2.0 N)—Add 1 part of
boiling in a Florence flask and slowly add 2.5 g of soluble
concentrated hydrochloric acid (HCl, sp gr 1.19, 36.5 to
starch paste while the solution is agitated. Add the hot starch
38.0 %) to 5 parts of water and mix well.
solution to 500 mL of distilled water containing 2.5 g of
6.4 Potassium Iodate-Potassium Iodide, Standard Solution
NaOH. Dilute to 1 L and thoroughly mix.
(0.0975 N)—Dissolve 3.48 g of KIO , 21.74 g of KI, and 1.21
g of NaOH in 1 L of distilled water. Standardize as follows:
7. Test Sample
Transfer 0.2700 g of National Bureau of Standards tin to a
7.1 Tin plate samples for coating weight determinations are
500-mL Erlenmeyer flask. Add 200 mL of HCl (1+1). Connect
obtained by stamping disks 2.257 6 0.001 in. (57.33 6 0.02
2 2
mm) in diameter which is equivalent to 4 in. (25.81 cm ) of
2 2
area (8 in. (52 cm ) of surface area). Recommended methods
Complete details and drawings of the apparatus are contained in U. S. Patent
No. 2,455,726 entitled “Method for Electrolytic Stripping and Determination of
of obtaining representative samples are described in the Tin
Plating Metals.” A suitable commercial supplier of the apparatus has been found to
Mill Products sections of the American Iron and Steel Insti-
be the Wilkens-Anderson Company of Chicago.
4 tute’s Steel Products Manual.
Reagent Chemicals, American Chemical Society Specifications, American
Chemical Society, Washington, DC. For suggestions on the testing of reagents not
listed by the American Chemical Society, see Analar Standards for Laboratory
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, Available from American Iron and Steel Institute (AISI), 1140 Connecticut
MD. Ave., NW, Suite 705, Washington, DC 20036, http://www.steel.org.
A630 − 16a (2024)
8. Procedure tin coating which is present as free tin and that part which is
present in the alloyed form.
8.1 Make required electrical connections.
8.2 Add HCl (1+1) to the porous tubes containing the
11. Summary of Test Method
carbon cathodes.
11.1 In this test method, the tin is stripped from a sample of
8.3 Suspend the sample disk of tin plate from the magnetic
tin-plate anodically at constant current in an electrolyte of the
holder.
hydrochloric acid. The potential difference developed between
the sample and a reference electrode is plotted against time on
NOTE 1—If it is desired to strip on one side only, mask the opposite side
a strip chart recorder or an electric digital readout. The time
and reduce the current to half its normal value. If a heavy oxide film has
developed on the tin plate during storage, the plate must be cathodically
required for stripping the free tin and alloyed tin, respectively,
cleaned prior to testing.
are read from the resulting chart (see Fig. 1) or a digital
8.4 Place a measured quantity of standardized KIO -KI readout. Since the stripping current has been preset, the free-tin
and alloy-tin coating weights are calculated by employing
solution into a 400-mL beaker (see 8.10). Simultaneously add
Faraday’s law of electrolysis.
250 mL of dilute HCl and mix thoroughly.
8.5 Raise the beaker so that the sample and porous cells are
12. Calibration and Standardization
immersed.
12.1 Determine the weight loss of pure tin specimens
8.6 Turn on d-c current and adjust to give 0.5 A/in. of
electrolyzed for a given time interval, expressing the results as
sample.
milligrams or pounds of tin per base box per second.
8.7 Time for complete removal of tin (see 8.10).
12.2 The test specimen should be a 4-in. disk of pure tin
8.8 Remove the beaker and add approximately 5 mL of
approximately 0.20 in. (5.1 mm) thick.
starch indicator solution.
12.3 The milliammeter should be frequently checked using
8.9 Titrate with standardized Na S O solution to the dis-
a precision milliammeter.
2 2 3
appearance of the blue color.
13. Available Constant Current Procedures
8.10 The stripping times and amounts of KIO -KI solution
13.1 There are three commercially available constant
to use are as follows:
current, electrolytic units that are in common use. Either of the
Amount of
Stripping KIO -KI following, or equivalent equipment, can be used as an accept-
Product Time, s Solution, mL
able referee method:
Electrolytic 100 90 20
13.1.1 Willey & Kunze Coulometric Test Method—Supplier
Electrolytic 75 75 20
Electrolytic 50 60 10
will provide set up, start up and running procedures.
Electrolytic 25 60 10
13.1.2 The Stannomatic Test Method—Supplier will provide
Electrolytic 10 45 10
set up, start up and running procedures.
Stripping time should not be longer than is required to
13.1.3 The Donart Test Method—Supplier will provide
remove all of the tin. Results will be high by approximately 9
setup, start, and running procedures.
0.01 lb/bb for each minute of over-stripping.
14. Willey & Kunze Coulometric Test Method
9. Reproducibility of Results
14.1 In this test method, the tin is electrolytically removed
9.1 Arbitrary maximum spreads in intermill check tests 2 2
from a 4 in. (25.807 cm ) circular sample of tinplate anodi-
show the reproducibility of test results by the Bendix Method
cally at constant current in an electrolyte of 1.0 N hydrochloric
to be as follows: 60.02 lb/bb for 0.25–lb coatings, 60.03 lb/bb
acid. The potential difference developed between the sample
for 0.75-lb coatings, and 60.04 lb/bb for 1.25-lb coatings. Data
and a glass calomel reference electrode is plotted against time
have not been developed for 0.10-lb coatings.
on a strip chart recorder. The time required for stripping the
free tin and alloy tin, respectively are read from the resulting
METHOD B—DETERMINATION OF THE TIN
chart (see Fig. 1) or on an electronic digital readout. Since the
COATING WEIGHTS BY THE CONSTANT
stripping current is constant and preset, the free tin and alloy
CURRENT, ELECTROLYTIC METHOD (REFEREE
tin coating weights are calculated by employing Faraday’s law
METHOD)
of electrolysis.
10. Scope
14.2 Significance and Use:
14.2.1 The amount of tin coating is directly associated with
10.1 This test method may be used to determine not only
the economics of producing tinplate and the performance of the
the total tin coating weight but also to determine that part of the
container or part for which such plate is used. Therefore, the
See Kunze, C. T., and Willey, A. R., “Electrolytic Determination of Tin and
Tin-Iron Alloy Coating Weights on Tin Plate,” Journal, Electrochemical Society, Available from Coulometric Systems, Bergholtz, OH.
Vol 99, No. 9, September 1952, pp. 354–359; and “Methods for Determination of Available from Products Distribution Service Division, Wilkens Anderson Co.,
Coating Weights of Tin Plate,” American Iron and Steel Institute, December 1959, 4525 West Division St., Chicago, IL 60651.
pp. 19–26. Available from Donart Electronics, 1005 Robinson Hwy, McDonald PA, 15057.
A630 − 16a (2024)
FIG. 1 Representative Electrostripper Curve
test method was developed for accuracy and as a control. The same shape as the sample so that all parts of the anode are
amount of tin combined in the alloy layer is also essential as equidistant from the cathode.
the continuity and amount plays an important part in its
14.7 Procedure:
functional properties. This test method is so designed that the
14.7.1 Clean the tinplate sample disk by cathodically treat-
tin coating is determined on only one side of the specimen at
ing in 0.5 % sodium carbonate solution for 10 s with a current
a time. This is useful when determining tin coatings on
density of 0.5 A/in.
differential tinplate.
14.7.2 An alternative procedure is to wipe the surface of the
14.3 Apparatus:
sample with a clean cloth saturated with acetone, this to be
14.3.1 This instrument (Model 8014) consists of an
followed by immersion of disk in acetone, removing, and air
ammeter, volt meter, constant current source and strip recorder.
drying. The procedure under 16.1 is preferred as it reduces the
A cell and sample holder are provided.
tin oxide present on the surface to metallic tin and improves the
14.4 Reagents: definition of the end point.
14.4.1 Electrolyte—1.0 N hydrochloric acid (HCl).
14.7.3 After following steps under Section 10, the apparatus
is ready for determining tin coating weight of the samples.
14.5 Sample Size:
Exactly the same procedure is used for samples with unknown
14.5.1 Tinplate samples for coating weight determinations
tin coating weights as the “dummy” samples. The recom-
are obtained by stamping discs 2.257 6 0.001 in. (57.33 6
2 2
mended recorder has a chart speed of 2 in./min and uses a chart
0.02 mm) in diameter. This is equivalent to 4 in. (25.81 cm )
having 10 divisions/in. Therefore, each division is equivalent
of area of one side. Sampling procedures for tin coating weight
to 3 s.
testing and applicable standards for the specific class designa-
tion are outlined in Specifications, A624, A624M, A626, and
14.8 Calculation:
A626M.
14.8.1 Calculate results as follows:
14.6 Interferences:
A 5 ITC (1)
14.6.1 The distance of the cathode from the sample is
B 5 0.65 IT C (2)
important and should be 0.75 6 0.25 in. (1.90 6 0.64 cm).
Also, it is important that the cathode be approximately the D 5 A1B (3)
A630 − 16a (2024)
where: the proper setting, the alloy counter will become inoperative,
and the automatic program will stop after the first layer is
A = free tin coating weight, lb/bb
completely removed.
I = stripping current, A,
T = time required for the removal of free tin, s, 15.6.2 Automatic prepolarization and light electro-cleaning
C = 0.02126 or the electrochemical equivalent of tin of cathodes is performed during the first 3 s after each start of
2 2
expressed in lb/bb for a 4-in. or 25.81-cm area of
the Stannomatic III or later model.
tinplate, 15.6.2.1 During this initial sequence the auxiliary anodes
B = alloy tin coating weight, lb/bb,
are activated against the cathodes. This operation may be
0.65 = that portion of the current required for removal of
extended by pressing the start button for several seconds.
the alloyed tin. Since the tin-iron alloy composition
15.6.2.2 Failure of this function may cause ghost layers and
is FeSn and these elements enter the solution
subsequent premature switch over of the sequence.
stoichiometrically with a two-electron loss for each
15.6.3 One condition for reliable results and a well-defined
atom of tin and iron, only two thirds of the current is
inflection point displayed on the instrument meters is a uniform
dissipated for the dissolution of tin. Consequently,
stripping of the whole surface.
the total alloy time must be corrected to compensate
15.6.3.1 After completion of the measurement the test
for the electrolysis of the iron,
surface should be examined visually. If the dissolution of the
T = time required for the removal of the alloy layer, s,
1 coating is not complete the measurement should be discarded
and
and repeated.
D = total tin coating weight, lb/bb.
15.6.4 The following factors influence the uniformity of
stripping:
15. The Stannomatic Test Method
15.6.4.1 Contaminated or dirty cathode.
15.6.4.2 Cathode not well fitted and located.
15.1 Principle of Equipment Operation:
15.6.4.3 Cathode and fixing part substituted with non-
15.1.1 The free tin and the alloy tin covering the surface of
original parts.
the tin plate to be examined are dissolved electrolytically. The
15.6.4.4 Sample punched too near the edges of the metal
test size is exactly defined on both sides of the sample by two
strip may have a non-uniform coating.
precision gaskets. The free tin and iron-tin alloy results are a
15.6.4.5 Sample dirty, corroded, partially or totally coated
function of the amount of current (Coulombs) used for strip-
with organic coating.
ping and are displayed on a numeric readout.
15.6.4.6 Sample with a thick passivation layer.
15.2 Apparatus:
15.2.1 This instrument consists of a stripping cell with a
16. The Donart Electronics Test Method
standardized sample size control. The stripping controller has a
16.1 Principle of Equipment Operation:
numeric readout which displays separately free tin and alloy tin
16.1.1 The Model 3300 Coulometric Tester is an advanced
on both sides of the test sample.
instrument for measuring tin coating weights for electrolytic tin
15.3 Reagent:
according to ASTM Standard A630 and ISO 2177. The Model
15.3.1 Electrolyte—As specified by equipment manufac-
3300 is based on the constant current, electrolytic method. By
turer.
controlling the diameter of the area to be stripped, and the use
of a precision constant current source, the Model 3300 Coulo-
15.4 Sample Size:
metric Tester can accurately calculate tin and alloy coating
15.4.1 Tinplate samples can be of various dimensions but
weights.
must be large enough to afford a test area at least 1 ⁄2 in. in
diameter. The test area must be reasonably flat. Sampling 16.2 Apparatus:
procedures for tin coating weight testing and applicable stan- 16.2.1 The unit consists of two electrolyte cells fitted with
dards for the specific class designations are outlined in Speci-
cathodes and anodes. A precision gasket is used to uniformly
fications A624/A624M and A626/A626M. seal the sample. The electrolytic cells have provisions for
filling the cell. Each cell is designed with an automatic
15.5 Preparing Sample:
decavitation cell to eliminate air bubbles from being trapped on
15.5.1 The surface of the tested sample shall be clean. Oil,
the surface of the sample that could cause errors in measure-
grease and organic coating such as lacquer shall be removed
ment. Redundant grounding contacts are used to ensure good
with suitable solvents. Remove oxides, passivation oxide
grounding of sample.
coatings, and corrosi
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...