ASTM D6919-17
(Test Method)Standard Test Method for Determination of Dissolved Alkali and Alkaline Earth Cations and Ammonium in Water and Wastewater by Ion Chromatography
Standard Test Method for Determination of Dissolved Alkali and Alkaline Earth Cations and Ammonium in Water and Wastewater by Ion Chromatography
SIGNIFICANCE AND USE
5.1 This test method is applicable to the simultaneous determination of dissolved alkali and alkaline earth cations and ammonium in water and wastewaters. Alkali and alkaline earth cations are traditionally determined by using spectroscopic techniques, such as AAS or ICP; whereas ammonium can be measured by using a variety of wet chemical methods, including colorimetry, ammonia-selective electrode, and titrimetry. However, ion chromatography provides a relatively straightforward method for the simultaneous determination of cations, such as lithium, sodium, potassium, calcium, magnesium, and ammonium, in fewer than 20–30 min.
SCOPE
1.1 This test method is valid for the simultaneous determination of the inorganic alkali and alkaline earth cations, lithium, sodium, potassium, magnesium, and calcium, as well as the ammonium cation in reagent water, drinking water, and wastewaters by suppressed and nonsuppressed ion chromatography.
1.2 The anticipated range of the test method is 0.05–200 mg/L. The specific concentration ranges tested for this test method for each cation were as follows (measured in mg/L):
Lithium
0.4–10.0
Sodium
4.0–40.0
Ammonium
0.4–10.0
Potassium
1.2–20.0
Magnesium
2.4–20.0
Calcium
4.0–40.0
1.2.1 The upper limits may be extended by appropriate dilution or by the use of a smaller injection volume. In some cases, using a larger injection loop may extend the lower limits. It is the responsibility of the user to ensure the validity of this test method for concentrations if the range is extended.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 It is the user’s responsibility to ensure the validity of these test methods for waters of untested matrices.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. For hazards statements specific to this test method, see 8.3.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-May-2017
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.05 - Inorganic Constituents in Water
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 01-Jan-2013
- Effective Date
- 15-Jun-2012
- Refers
ASTM D3856-11 - Standard Guide for Management Systems in Laboratories Engaged in Analysis of Water - Effective Date
- 15-Nov-2011
- Effective Date
- 01-May-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-Jul-2008
- Effective Date
- 15-Jan-2008
- Effective Date
- 01-Dec-2007
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 15-Aug-2006
- Effective Date
- 15-Aug-2006
Overview
ASTM D6919-17 is the internationally recognized standard test method for the determination of dissolved alkali and alkaline earth cations (lithium, sodium, potassium, magnesium, calcium) and ammonium in reagent water, drinking water, and wastewaters using ion chromatography (IC). Published by ASTM International, this method is under the jurisdiction of ASTM Committee D19 on Water and provides laboratories and water quality professionals with a straightforward analytical approach. Its core value lies in facilitating the simultaneous, rapid, and accurate quantification of multiple cations essential for water quality assessment.
Key Topics
Scope of Application
- Determines lithium, sodium, potassium, magnesium, calcium, and ammonium in water and wastewater
- Suitable for a concentration range of approximately 0.05–200 mg/L for total cations
- Applicable to drinking water, reagent water, and wastewaters
Method Summary
- Employs ion chromatography using cation-exchange columns and conductivity detection
- Supports both suppressed and nonsuppressed detection techniques
- Achieves simultaneous separation and quantification, often in under 30 minutes
Precision and Quality Control
- Defined procedures for calibration, method validation, and ongoing verification
- Guidance on controlling potential interferences and ensuring data reliability
Analytical Range
- Lithium: 0.4–10.0 mg/L
- Sodium: 4.0–40.0 mg/L
- Ammonium: 0.4–10.0 mg/L
- Potassium: 1.2–20.0 mg/L
- Magnesium: 2.4–20.0 mg/L
- Calcium: 4.0–40.0 mg/L
- Technique allows ranges to be extended via dilution or injection volume adjustment
Interferences and Precautions
- Addresses potential interference from organic amines and high cation/acidity levels
- Emphasizes strict contamination control, particularly for sodium analysis
Applications
ASTM D6919-17 plays a vital role in water analysis across various sectors:
- Drinking Water Monitoring: Ensures regulatory compliance for cation content, supporting public health and safety.
- Wastewater Treatment: Monitors the removal efficiency and environmental discharge compliance by tracking key cations.
- Industrial Processes: Assesses water quality for processes sensitive to specific ionic compositions, such as boiler feedwater or food and beverage manufacturing.
- Environmental Assessment: Supports studies of natural waters, pollutant tracing, and remediation effectiveness.
Laboratories use this method to streamline workflows by consolidating the measurement of multiple analytes into a single, rapid procedure, reducing equipment and labor overhead compared to traditional spectroscopic and wet chemistry techniques.
Related Standards
For optimal results and compliance, these related ASTM and international standards are commonly referenced with ASTM D6919-17:
- ASTM D1129: Terminology Relating to Water
- ASTM D1193: Specification for Reagent Water
- ASTM D3370: Practices for Sampling Water from Closed Conduits
- ASTM D2777: Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19
- ASTM D3856: Guide for Management Systems in Laboratories Engaged in Analysis of Water
- ISO 14911: Water Quality – Determination of dissolved Li+, Na+, NH4+, K+, Mn²⁺, Ca²⁺, Mg²⁺, Sr²⁺, and Ba²⁺ using ion chromatography
Practical Value
ASTM D6919-17 is essential for professionals seeking reliable, efficient, and standardized means to measure critical cations and ammonium in water matrices. Its adoption contributes to improved data credibility, regulatory compliance, and quality assurance in water testing laboratories worldwide. By following this standard, users benefit from increased analytical throughput, lower risk of interference, and harmonized quality control processes-all crucial for robust water quality management and environmental stewardship.
Buy Documents
ASTM D6919-17 - Standard Test Method for Determination of Dissolved Alkali and Alkaline Earth Cations and Ammonium in Water and Wastewater by Ion Chromatography
REDLINE ASTM D6919-17 - Standard Test Method for Determination of Dissolved Alkali and Alkaline Earth Cations and Ammonium in Water and Wastewater by Ion Chromatography
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D6919-17 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Dissolved Alkali and Alkaline Earth Cations and Ammonium in Water and Wastewater by Ion Chromatography". This standard covers: SIGNIFICANCE AND USE 5.1 This test method is applicable to the simultaneous determination of dissolved alkali and alkaline earth cations and ammonium in water and wastewaters. Alkali and alkaline earth cations are traditionally determined by using spectroscopic techniques, such as AAS or ICP; whereas ammonium can be measured by using a variety of wet chemical methods, including colorimetry, ammonia-selective electrode, and titrimetry. However, ion chromatography provides a relatively straightforward method for the simultaneous determination of cations, such as lithium, sodium, potassium, calcium, magnesium, and ammonium, in fewer than 20–30 min. SCOPE 1.1 This test method is valid for the simultaneous determination of the inorganic alkali and alkaline earth cations, lithium, sodium, potassium, magnesium, and calcium, as well as the ammonium cation in reagent water, drinking water, and wastewaters by suppressed and nonsuppressed ion chromatography. 1.2 The anticipated range of the test method is 0.05–200 mg/L. The specific concentration ranges tested for this test method for each cation were as follows (measured in mg/L): Lithium 0.4–10.0 Sodium 4.0–40.0 Ammonium 0.4–10.0 Potassium 1.2–20.0 Magnesium 2.4–20.0 Calcium 4.0–40.0 1.2.1 The upper limits may be extended by appropriate dilution or by the use of a smaller injection volume. In some cases, using a larger injection loop may extend the lower limits. It is the responsibility of the user to ensure the validity of this test method for concentrations if the range is extended. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 It is the user’s responsibility to ensure the validity of these test methods for waters of untested matrices. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. For hazards statements specific to this test method, see 8.3. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method is applicable to the simultaneous determination of dissolved alkali and alkaline earth cations and ammonium in water and wastewaters. Alkali and alkaline earth cations are traditionally determined by using spectroscopic techniques, such as AAS or ICP; whereas ammonium can be measured by using a variety of wet chemical methods, including colorimetry, ammonia-selective electrode, and titrimetry. However, ion chromatography provides a relatively straightforward method for the simultaneous determination of cations, such as lithium, sodium, potassium, calcium, magnesium, and ammonium, in fewer than 20–30 min. SCOPE 1.1 This test method is valid for the simultaneous determination of the inorganic alkali and alkaline earth cations, lithium, sodium, potassium, magnesium, and calcium, as well as the ammonium cation in reagent water, drinking water, and wastewaters by suppressed and nonsuppressed ion chromatography. 1.2 The anticipated range of the test method is 0.05–200 mg/L. The specific concentration ranges tested for this test method for each cation were as follows (measured in mg/L): Lithium 0.4–10.0 Sodium 4.0–40.0 Ammonium 0.4–10.0 Potassium 1.2–20.0 Magnesium 2.4–20.0 Calcium 4.0–40.0 1.2.1 The upper limits may be extended by appropriate dilution or by the use of a smaller injection volume. In some cases, using a larger injection loop may extend the lower limits. It is the responsibility of the user to ensure the validity of this test method for concentrations if the range is extended. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 It is the user’s responsibility to ensure the validity of these test methods for waters of untested matrices. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. For hazards statements specific to this test method, see 8.3. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D6919-17 is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D6919-17 has the following relationships with other standards: It is inter standard links to ASTM D1129-13(2020)e2, ASTM D5905-98(2013), ASTM D2777-12, ASTM D3856-11, ASTM D5810-96(2011), ASTM D3370-10, ASTM D1129-10, ASTM D3370-08, ASTM D5905-98(2008), ASTM D2777-08, ASTM D3370-07, ASTM D1129-06ae1, ASTM D1129-06a, ASTM D2777-06, ASTM D5810-96(2006). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D6919-17 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D6919 − 17
Standard Test Method for
Determination of Dissolved Alkali and Alkaline Earth
Cations and Ammonium in Water and Wastewater by Ion
Chromatography
This standard is issued under the fixed designation D6919; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
1.1 This test method is valid for the simultaneous determi-
mendations issued by the World Trade Organization Technical
nation of the inorganic alkali and alkaline earth cations,
Barriers to Trade (TBT) Committee.
lithium, sodium, potassium, magnesium, and calcium, as well
as the ammonium cation in reagent water, drinking water, and
2. Referenced Documents
wastewaters by suppressed and nonsuppressed ion chromatog-
2.1 ASTM Standards:
raphy.
D1129Terminology Relating to Water
1.2 The anticipated range of the test method is 0.05–200
D1193Specification for Reagent Water
mg/L. The specific concentration ranges tested for this test
D2777Practice for Determination of Precision and Bias of
method for each cation were as follows (measured in mg/L):
Applicable Test Methods of Committee D19 on Water
Lithium 0.4–10.0
D3370Practices for Sampling Water from Closed Conduits
Sodium 4.0–40.0
D3856Guide for Management Systems in Laboratories
Ammonium 0.4–10.0
Potassium 1.2–20.0
Engaged in Analysis of Water
Magnesium 2.4–20.0
D4210Practice for Intralaboratory Quality Control Proce-
Calcium 4.0–40.0
dures and a Discussion on Reporting Low-Level Data
1.2.1 The upper limits may be extended by appropriate 3
(Withdrawn 2002)
dilution or by the use of a smaller injection volume. In some
D5810Guide for Spiking into Aqueous Samples
cases,usingalargerinjectionloopmayextendthelowerlimits.
D5847Practice for Writing Quality Control Specifications
It is the responsibility of the user to ensure the validity of this
for Standard Test Methods for Water Analysis
test method for concentrations if the range is extended.
D5905Practice for the Preparation of SubstituteWastewater
1.3 The values stated in SI units are to be regarded as
3. Terminology
standard. No other units of measurement are included in this
standard.
3.1 Definitions:
3.1.1 For definitions of terms used in this standard, refer to
1.4 It is the user’s responsibility to ensure the validity of
Terminology D1129.
these test methods for waters of untested matrices.
3.2 Definitions of Terms Specific to This Standard:
1.5 This standard does not purport to address all of the
3.2.1 continuing calibration blank, n—asolutioncontaining
safety concerns, if any, associated with its use. It is the
no analytes (of interest) which is used to verify blank response
responsibility of the user of this standard to establish appro-
and freedom from carryover.
priate safety and health practices and determine the applica-
bility of regulatory limitations prior to use. For hazards
3.2.2 continuing calibration verification, n—a solution (or
statements specific to this test method, see 8.3.
setofsolutions)ofknownconcentrationusedtoverifyfreedom
1.6 This international standard was developed in accor-
fromexcessiveinstrumentaldrift;theconcentrationistocover
dance with internationally recognized principles on standard-
the range of calibration curve.
1 2
This test method is under the jurisdiction ofASTM Committee D19 on Water For referenced ASTM standards, visit the ASTM website, www.astm.org, or
and is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
in Water. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved June 1, 2017. Published June 2017. Originally the ASTM website.
approved in 2003. Last previous edition approved in 2009 as D6919–09. DOI: The last approved version of this historical standard is referenced on
10.1520/D6919-17. www.astm.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6919 − 17
4. Summary of Test Method 6.1.1 High concentrations of analyte cations can interfere
with the determination of low concentrations of other analyte
4.1 Inorganic cations and the ammonium cation, hereafter
cationswithsimilarretentiontimes.Forinstance,highlevelsof
referred to as ammonium, are determined by ion chromatog-
sodium can interfere with the determination of low levels of
raphy in water and wastewater samples from a fixed sample
ammonium (that is, at ratios >1000:1).
volume, typically 10–50 µL. The cationic analytes are sepa-
6.1.2 Highlevelsofsampleacidity,thatis,lowpH,canalso
rated using a cation-exchange material, which is packed into
interfere with this analysis by overloading the column, leading
guardandanalyticalcolumns.Adiluteacidsolutionistypically
to poor peak shape and loss of resolution.The pH at which the
used as the eluent.
chromatographic separation begins to exhibit poor peak shape
4.1.1 The separated cations are detected by using conduc-
depends upon the ion-exchange capacity of the column. It is
tivity detection. To achieve sensitive conductivity detection, it
recommendedthatcolumnsusedforanalysisofacidicsamples
is essential that the background signal arising from the eluent
inconjunctionwiththesuppressedconductivityversionofthis
have low baseline noise. One means to achieve low back-
testmethodbeabletotolerateacidconcentrationsupto50mM
ground noise is to combine the conductivity detector with a
+ 5
H (pH 1.3), such as the IonPac CS16 column. The columns
suppressor device that will reduce the conductance of the
used with nonsuppressed conductivity detection typically have
eluent, hence background noise, and also transform the sepa-
4 lower capacity and can tolerate acid concentrations up to 10
rated cations into their more conductive corresponding bases.
+
mM H (pH 2.0), such as the IC-Pak C/MD column.
4.1.2 Detection can also be achieved without chemical
suppression, whereby the difference between the equivalent 6.2 A slight decrease or increase in eluent strength often
ionic conductance of the eluent and analyte cation is measured allows interferences to elute after or before the peak of
directly after the analytical column. This test method will concern.
consider both suppressed and nonsuppressed detection tech-
6.3 Sodium is a common contaminant from many sources
nologies. The conductivity data is plotted to produce a chro-
such as fingers, water, detergents, glassware, and other inci-
matogram that is used to determine peak areas. A chromato-
dental sources.As a precaution, the user of this test method is
graphic integrator or appropriate computer-based data system
advised to wear plastic gloves and use plasticware for all
is typically used for data presentation.
solutions,standards,andpreparedsamples.Inaddition,method
4.2 The cations are identified based on their retention times blanks should be monitored for background sodium contami-
compared to known standards. Quantification is accomplished nation.
by measuring cation peak areas and comparing them to the
7. Apparatus
areas generated from known standards. The results are calcu-
lated using a standard curve based on peak areas of known 7.1 Ion ChromatographyApparatus,analyticalsystemcom-
plete with all required accessories, including eluent pump,
concentrations of standards in reagent water.
injector, syringes, columns, suppressor (if used), conductivity
5. Significance and Use detector, data system, and compressed gasses (if required).
7.1.1 Eluent Pump,capableofdelivering0.25–5mL/minof
5.1 This test method is applicable to the simultaneous
eluent at a pressure of up to 4000 psi.
determinationofdissolvedalkaliandalkalineearthcationsand
7.1.2 Injection Valve, a low dead-volume switching valve
ammoniuminwaterandwastewaters.Alkaliandalkalineearth
that allows the loading of a sample into a sample loop and
cations are traditionally determined by using spectroscopic
subsequent injection of the loop contents into the eluent
techniques, such as AAS or ICP; whereas ammonium can be
stream.
measured by using a variety of wet chemical methods, includ-
7.1.3 Guard Column, cation-exchange column typically
ing colorimetry, ammonia-selective electrode, and titrimetry.
packed with the same material used in the analytical column.
However, ion chromatography provides a relatively straight-
The purpose of this column is to protect the analytical column
forward method for the simultaneous determination of cations,
from particulate matter and irreversibly retained material.
such as lithium, sodium, potassium, calcium, magnesium, and
7.1.4 Analytical Column, separator column, packed with a
ammonium, in fewer than 20–30 min.
weak acid functionalized cation-exchange material, capable of
separating the ions of interest from each other, and from other
6. Interferences
ions that commonly occur in the sample matrix. The chosen
6.1 Noindividualinterferenceshavebeenestablished,butit
column must give separations equivalent to those shown in
is possible that some low-molecular-weight organic bases
Figs. 1 and 2.
(amines) may have similar retention times to analytes of
7.1.5 Suppressor Device—If using the suppressed conduc-
interest, particularly later-eluting solutes, such as potassium,
tivity detection mode, the suppressor must provide peak-to-
magnesium, and calcium. Potential interferences include
peak noise of <2 nS per minute of monitored baseline.
amines such as mono-, di-, and trimethylamines; mono-, di-,
7.1.6 Conductivity Detector, a low-volume, flow-through,
and triethylamines; and alkanolamines.
temperature-controlled (typically at 35°C) conductivity cell
equipped with a meter capable of reading 0–1000 µS/cm on a
linear scale.
InternationalStandardISO14911,Waterquality—Determinationofdissolved
Li+, Na+, NH4+, K+, Mn2+, Ca2+, Mg2+, Sr2+ and Ba2+ using ion chromatog-
raphy — Method for water and waste water. IonPac is a trademark of Dionex Corporation, Sunnyvale, CA, 94088.
D6919 − 17
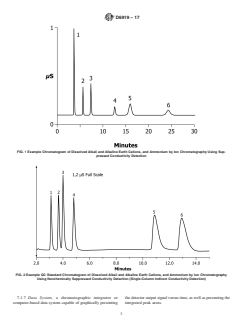
FIG. 1 Example Chromatogram of Dissolved Alkali and Alkaline Earth Cations, and Ammonium by Ion Chromatography Using Sup-
pressed Conductivity Detection
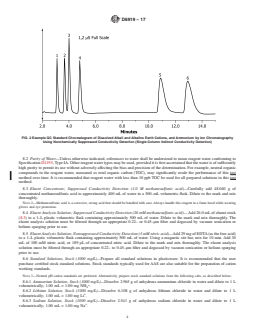
FIG. 2 Example QC Standard Chromatogram of Dissolved Alkali and Alkaline Earth Cations, and Ammonium by Ion Chromatography
Using Nonchemically Suppressed Conductivity Detection (Single-Column Indirect Conductivity Detection)
7.1.7 Data System, a chromatographic integrator or thedetectoroutputsignalversustime,aswellaspresentingthe
computer-based data system capable of graphically presenting integrated peak areas.
D6919 − 17
NOTE 3—Neutral pH cation standards are preferred. Alternatively,
8. Reagents and Materials
prepare stock standard solutions from the following salts, as described
8.1 Purity of Reagents—Reagent-grade chemicals shall be
below:
used in all tests. Unless otherwise indicated, all reagents shall
8.6.1 Ammonium Solution, Stock (1000 mg/L)—Dissolve
conform to the specifications of the Committee on Analytical
2.965 g of anhydrous ammonium chloride in water and dilute
Reagents of the American Chemical Society, where such
+
to 1 L volumetrically; 1.00 mL = 1.00 mg NH .
specifications are available. Other grades may be used, pro-
8.6.2 Lithium Solution, Stock (1000 mg/L)—Dissolve 6.108
vided it is first ascertained that the reagent is of sufficiently
g of anhydrous lithium chloride in water and dilute to 1 L
high purity to permit its use without reducing the accuracy of
+
volumetrically; 1.00 mL = 1.00 mg Li .
the determination.
8.6.3 Sodium Solution, Stock (1000 mg/L)—Dissolve 2.541
NOTE 1—Prepare all reagents, standards, and samples in plasticware.
g of anhydrous sodium chloride in water and dilute to 1 L
Sodium will leach from glassware and bias the quantification of sodium.
+
volumetrically; 1.00 mL = 1.00 mg Na .
8.2 Purity of Water—Unless otherwise indicated, references
8.6.4 Potassium Solution, Stock (1000 mg/L)—Dissolve
towatershallbeunderstoodtomeanreagentwaterconforming
3.481gofanhydrouspotassiumphosphatemonobasicinwater
to Specification D1193, Type IA. Other reagent water types
+
and dilute to 1 L volumetrically; 1.00 mL = 1.00 mg K .
may be used, provided it is first ascertained that the water is of
8.6.5 Magnesium Solution, Stock (1000 mg/L)—Dissolve
sufficiently high purity to permit its use without adversely
10.144 g of magnesium sulfate hetpahydrate in water and
affecting the bias and precision of the determination. For
2+
dilute to 1 L volumetrically; 1.00 mL = 1.00 mg Mg .
example, neutral organic compounds in the reagent water,
8.6.6 Calcium Solution, Stock e(1000 mg/L)—Dissolve
measured as total organic carbon (TOC), may significantly
3.668 g of calcium chloride dihydrate in water and dilute to 1
erode the performance of this test method over time. It is
2+
L volumetrically; 1.00 mL = 1.00 mg Ca .
recommendedthatreagentwaterwithlessthan10ppbTOCbe
8.7 Cation Working Standards—All calibration standards
used for all prepared solutions in this test method.
and standards used for analysis should be prepared in 100–mL
8.3 Eluent Concentrate; Suppressed Conductivity Detection
volumetric flasks, as described below.
(1.0 M methanesulfonic acid)—Carefully add 48.040 g of
Standardconcentration mg/L 5
~ !
concentratedmethanesulfonicacidtoapproximately400mLof
stockvolumeadded mL ·1000mg/L
~ ~ ! !
water in a 500–mL volumetric flask. Dilute to the mark and
100 ~mL!
mix thoroughly.
NOTE2—Methanesulfonicacidisacorrosive,strongacidthatshouldbe
Example:
handled with care. Always handle this reagent in a fume hood while
wearing gloves and eye protection. 1 mLNastock·1000mg/L Na
~ !
10mg/L Na 5
100 ~mL!
8.4 Eluent Analysis Solution; Suppressed Conductivity De-
tection (26 mM methanesulfonic acid)—Add26.0mLofeluent
8.8 Blank—Theblankstandardisaportionofthewaterused
stock (8.3) to a 1–L plastic volumetric flask containing to prepare the cation working solutions.
approximately 500 mL of water. Dilute to the mark and mix
8.9 Filter Paper—Purchase suitable filter paper. Typically
thoroughly. The eluent analysis solution must be filtered
the filter papers have a pore size of 0.22-µm or 0.45-µm
throughanappropriate0.22–or0.45–µmfilteranddegassedby
membrane. Material such as fine-textured, ashless paper, or
vacuum sonication or helium sparging prior to use.
glass fiber paper are acceptable. The user must first ascertain
8.5 Eluent Analysis Solution; Nonsuppressed Conductivity that the filter paper is of sufficient purity to use without
Detection (3 mM nitric acid)—Add29mgofEDTA(asthefree
adversely affecting the bias and precision of the test method.
acid) to a 1–L plastic volumetric flask containing approxi-
9. Precautions
mately 500 mLof water. Using a magnetic stir bar, mix for 10
min. Add 30 mL of 100 mM nitric acid, or 189 µL of
9.1 These methods address the determination of low con-
concentrated nitric acid. Dilute to the mark and mix thor-
centrationsofcations.Accordingly,everyprecautionshouldbe
oughly.Theeluentanalysissolutionmustbefilteredthroughan
takentoensurethecleanlinessofsamplecontainers,aswellas
appropriate 0.22– to 0.45–µm filter and degassed by vacuum
other materials and apparatus that come in contact with the
sonication or helium sparging prior to use.
sample.
8.6 Standard Solutions, Stock (1000 mg/L)—Prepare all
10. Sampling and Sample Preservation
standard solutions in plasticware. It is recommended that the
10.1 Collect the sample in accordance with Practices
user purchase certified stock standard solutions. Stock stan-
D3370, as applicable.
dards typically used for AAS are also suitable for the prepa-
ration of cation working standards.
10.2 Samplesmustbecollectedinplasticcontainersthatare
clean and free of artifacts and interferences. The suitability of
the containers must be demonstrated for each new lot by
Reagent Chemicals, American Chemical Society Specifications, American
performingacontainerblankandlaboratoryfortifiedcontainer
Chemical Society, Washington, DC. For Suggestions on the testing of reagents not
listed by the American Chemical Society, see Annual Standards for Laboratory blank.
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
10.3 Samplesthatwillnotbeanalyzedimmediatelymustbe
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
MD. preserved with sulfuric acid to a pH of 2. Whereas samples to
D6919 − 17
TABLE 1 Instrument Conditions for the Analysis of Dissolved
curve, prepare calibration standards, at a minimum of three
Alkali and Alkaline Earth Cations and Ammonium by Ion
concentration levels, by accurately adding measured volumes
Chromatography Using Suppressed Conductivity Detection
ofthestockstandards(8.6)toavolumetricflask(s)anddiluting
Eluent: 26 mM methanesulfonic acid
to volume with water.Aminimum of five concentration levels
Flow rate: 1.5 mL/min
is recommended if the curve covers two orders of magnitude.
Column: Dionex IonPac CG16/CS16
Sample Loop: 10 µL
12.2 The order of peak elution and typical retention times
Detection: Suppressed conductivity
are shown in Figs. 1 and 2 for suppressed and nonsuppressed
Suppressor: CSRS ULTRA
Background: ~2 µS
conductivitydetection,respectively.Theretentiontimeofeach
Solutes: 1 = lithium (1.0 mg/L), 2 = sodium (1.0 mg/L),
analyte can vary with the type and state of the guard and
3 = ammonium (1.0 mg/L), 4 = potassium (1.0 mg/L),
5 = magnesium (1.0 mg/L), 6 = calcium (1.0 mg/L) analytical columns and the eluent concentration, but should
remain consistent within a given analysis batch.
12.3 To establish the calibration curve, analyze a reagent
TABLE 2 Instrument Conditions for the Analysis of Dissolved
blank (8.8) and the calibration standards (8.7) in accordance
Alkali and Alkaline Earth Cations and Ammonium by Ion
withtheprocedureinSection13.Tabulatepeakarearesponses
Chromatography Using Nonsuppressed Conductivity Detection
(Single-Column Indirect Conductivity Detection)
against concentration. These results are used to prepare a
calibration curve using a linear least-squares fit for each
Eluent: 3 mM nitric acid / 0.05 mM EDTA
Flow rate: 1.0 mL/min
analyte (with the exception of ammonium). The squared
Column: Waters IC-Pak™C/MD,
correlation coefficient of determination (r ) should be ≥0.995
Injection 20 µL
Volume: for accurate results. Ammonia is a weak base; hence the
Detection: Nonsuppressed conductivity, indirect conductivity
ammonium cation does not give linear response with sup-
Background: ~1300 µS
pressedconductivitydetectionandshouldbecalibratedusinga
Analytes: 1 = lithium (1.0 mg/L), 2 = sodium (4.0 mg/L),
3 = ammonium (5.0 mg/L), 4 = potassium (10.0 mg/L), quadratic fit when using this detection method. However,
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D6919 − 09 D6919 − 17
Standard Test Method for
Determination of Dissolved Alkali and Alkaline Earth
Cations and Ammonium in Water and Wastewater by Ion
Chromatography
This standard is issued under the fixed designation D6919; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method is valid for the simultaneous determination of the inorganic alkali and alkaline earth cations, lithium,
sodium, potassium, magnesium, and calcium, as well as the ammonium cation in reagent water, drinking water, and wastewaters
by suppressed and nonsuppressed ion chromatography.
1.2 The anticipated range of the test method is 0.05–200 mg/L. The specific concentration ranges tested for this test method for
each cation were as follows (measured in mg/L):
Lithium 0.4–10.0
Sodium 4.0–40.0
Ammonium 0.4–10.0
Potassium 1.2–20.0
Magnesium 2.4–20.0
Calcium 4.0–40.0
1.2.1 The upper limits may be extended by appropriate dilution or by the use of a smaller injection volume. In some cases, using
a larger injection loop may extend the lower limits. It is the responsibility of the user to ensure the validity of this test method for
concentrations if the range is extended.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 It is the user’suser’s responsibility to ensure the validity of these test methods for waters of untested matrices.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use. For hazards statements specific to this test method, see 8.3.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D3370 Practices for Sampling Water from Closed Conduits
D3856 Guide for Management Systems in Laboratories Engaged in Analysis of Water
D4210 Practice for Intralaboratory Quality Control Procedures and a Discussion on Reporting Low-Level Data (Withdrawn
2002)
D5810 Guide for Spiking into Aqueous Samples
D5847 Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
This test method is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents in Water.
Current edition approved May 15, 2009June 1, 2017. Published May 2009June 2017. Originally approved in 2003. Last previous edition approved in 20032009 as
D6919 – 03.D6919 – 09. DOI: 10.1520/D6919-09.10.1520/D6919-17.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6919 − 17
D5905 Practice for the Preparation of Substitute Wastewater
3. Terminology
3.1 Definitions—Definitions: For definitions of terms used in this test method, refer to Terminology D1129.
3.1.1 For definitions of terms used in this standard, refer to Terminology D1129.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 continuing calibration blank, n—a solution containing no analytes (of interest) which is used to verify blank response and
freedom from carryover.
3.2.2 continuing calibration verification, n—a solution (or set of solutions) of known concentration used to verify freedom from
excessive instrumental drift; the concentration is to cover the range of calibration curve.
4. Summary of Test Method
4.1 Inorganic cations and the ammonium cation, hereafter referred to as ammonium, are determined by ion chromatography in
water and wastewater samples from a fixed sample volume, typically 10–50 μL. The cationic analytes are separated using a
cation-exchange material, which is packed into guard and analytical columns. A dilute acid solution is typically used as the eluent.
4.1.1 The separated cations are detected by using conductivity detection. To achieve sensitive conductivity detection, it is
essential that the background signal arising from the eluent have low baseline noise. One means to achieve low background noise
is to combine the conductivity detector with a suppressor device that will reduce the conductance of the eluent, hence background
noise, and also transform the separated cations into their more conductive corresponding bases.
4.1.2 Detection can also be achieved without chemical suppression, whereby the difference between the equivalent ionic
conductance of the eluent and analyte cation is measured directly after the analytical column. This test method will consider both
suppressed and nonsuppressed detection technologies. The conductivity data is plotted to produce a chromatogram that is used to
determine peak areas. A chromatographic integrator or appropriate computer-based data system is typically used for data
presentation.
4.2 The cations are identified based on their retention times compared to known standards. Quantification is accomplished by
measuring cation peak areas and comparing them to the areas generated from known standards. The results are calculated using
a standard curve based on peak areas of known concentrations of standards in reagent water.
5. Significance and Use
5.1 This test method is applicable to the simultaneous determination of dissolved alkali and alkaline earth cations and
ammonium in water and wastewaters. Alkali and alkaline earth cations are traditionally determined by using spectroscopic
techniques, such as AAS or ICP; whereas ammonium can be measured by using a variety of wet chemical methods, including
colorimetry, ammonia-selective electrode, and titrimetry. However, ion chromatography provides a relatively straightforward
method for the simultaneous determination of cations, such as lithium, sodium, potassium, calcium, magnesium, and ammonium,
in fewer than 20–30 min.
6. Interferences
6.1 No individual interferences have been established, but it is possible that some low-molecular-weight organic bases (amines)
may have similar retention times to analytes of interest, particularly later-eluting solutes, such as potassium, magnesium, and
calcium. Potential interferences include amines such as mono-, di-, and trimethylamines; mono-, di-, and triethylamines; and
alkanolamines.
6.1.1 High concentrations of analyte cations can interfere with the determination of low concentrations of other analyte cations
with similar retention times. For instance, high levels of sodium can interfere with the determination of low levels of ammonium
(that is, at ratios >1000:1).
6.1.2 High levels of sample acidity, that is, low pH, can also interfere with this analysis by overloading the column, leading to
poor peak shape and loss of resolution. The pH at which the chromatographic separation begins to exhibit poor peak shape depends
upon the ion-exchange capacity of the column. It is recommended that columns used for analysis of acidic samples in conjunction
+
with the suppressed conductivity version of this test method be able to tolerate acid concentrations up to 50 mM H (pH 1.3), such
as the IonPac®IonPac CS16 column. The columns used with nonsuppressed conductivity detection typically have lower capacity
+
and can tolerate acid concentrations up to 10 mM H (pH 2.0), such as the trademarked IC-Pak C/MD column.
6.2 A slight decrease or increase in eluent strength often allows interferences to elute after or before the peak of concern.
6.3 Sodium is a common contaminant from many sources such as fingers, water, detergents, glassware, and other incidental
sources. As a precaution, the user of this test method is advised to wear plastic gloves and use plasticware for all solutions,
standards, and prepared samples. In addition, method blanks should be monitored for background sodium contamination.
International Standard ISO 14911.International Standard ISO 14911, Water quality — Determination of dissolved Li+, Na+, NH4+, K+, Mn2+, Ca2+, Mg2+, Sr2+ and
Ba2+ using ion chromatography — Method for water and waste water.
IonPac is a trademark of Dionex Corporation, Sunnyvale, CA, 94088.
D6919 − 17
7. Apparatus
7.1 Ion Chromatography Apparatus , Apparatus, analytical system complete with all required accessories, including eluent
pump, injector, syringes, columns, suppressor (if used), conductivity detector, data system, and compressed gasses (if required).
7.1.1 Eluent Pump, capable of delivering 0.25–5 mL/min of eluent at a pressure of up to 4000 psi.
7.1.2 Injection Valve, a low dead-volume switching valve that allows the loading of a sample into a sample loop and subsequent
injection of the loop contents into the eluent stream.
7.1.3 Guard Column, cation-exchange column typically packed with the same material used in the analytical column. The
purpose of this column is to protect the analytical column from particulate matter and irreversibly retained material.
7.1.4 Analytical Column, separator column, packed with a weak acid functionalized cation-exchange material, capable of
separating the ions of interest from each other, and from other ions that commonly occur in the sample matrix. The chosen column
must give separations equivalent to those shown in Figs. 1 and 2.
7.1.5 Suppressor Device—If using the suppressed conductivity detection mode, the suppressor must provide peak-to-peak noise
of <2 nS per minute of monitored baseline.
7.1.6 Conductivity Detector, a low-volume, flow-through, temperature-controlled (typically at 35°C) conductivity cell equipped
with a meter capable of reading 0–1000 μS/cm on a linear scale.
7.1.7 Data System, a chromatographic integrator or computer-based data system capable of graphically presenting the detector
output signal versus time, as well as presenting the integrated peak areas.
8. Reagents and Materials
8.1 Purity of Reagents—Reagent-grade chemicals shall be used in all tests. Unless otherwise indicated, all reagents shall
conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where such
specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high purity
to permit its use without reducing the accuracy of the determination.
NOTE 1—Prepare all reagents, standards, and samples in plasticware. Sodium will leach from glassware and bias the quantification of sodium.
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For Suggestions on the testing of reagents not listed by
the American Chemical Society, see Annual Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
FIG. 1 Example Chromatogram of Dissolved Alkali and Alkaline Earth Cations, and Ammonium by Ion Chromatography Using Sup-
pressed Conductivity Detection
D6919 − 17
FIG. 2 Example QC Standard Chromatogram of Dissolved Alkali and Alkaline Earth Cations, and Ammonium by Ion Chromatography
Using Nonchemically Suppressed Conductivity Detection (Single-Column Indirect Conductivity Detection)
8.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean reagent water conforming to
Specification D1193, Type IA. Other reagent water types may be used, provided it is first ascertained that the water is of sufficiently
high purity to permit its use without adversely affecting the bias and precision of the determination. For example, neutral organic
compounds in the reagent water, measured as total organic carbon (TOC), may significantly erode the performance of this test
method over time. It is recommended that reagent water with less than 10 ppb TOC be used for all prepared solutions in this test
method.
8.3 Eluent Concentrate; Suppressed Conductivity Detection (1.0 M methanesulfonic acid)—Carefully add 48.040 g of
concentrated methanesulfonic acid to approximately 400 mL of water in a 500–mL volumetric flask. Dilute to the mark and mix
thoroughly.
NOTE 2—Methanesulfonic acid is a corrosive, strong acid that should be handled with care. Always handle this reagent in a fume hood while wearing
gloves and eye protection.
8.4 Eluent Analysis Solution; Suppressed Conductivity Detection (26 mM methanesulfonic acid)— Add 26.0 mL of eluent stock
(8.3) to a 1–L plastic volumetric flask containing approximately 500 mL of water. Dilute to the mark and mix thoroughly. The
eluent analysis solution must be filtered through an appropriate 0.22– or 0.45–μm filter and degassed by vacuum sonication or
helium sparging prior to use.
8.5 Eluent Analysis Solution; Nonsuppressed Conductivity Detection (3 mM nitric acid)—Add 29 mg of EDTA (as the free acid)
to a 1–L plastic volumetric flask containing approximately 500 mL of water. Using a magnetic stir bar, mix for 10 min. Add 30
mL of 100 mM nitric acid, or 189 μL of concentrated nitric acid. Dilute to the mark and mix thoroughly. The eluent analysis
solution must be filtered through an appropriate 0.22– to 0.45–μm filter and degassed by vacuum sonication or helium sparging
prior to use.
8.6 Standard Solutions, Stock (1000 mg/L)—Prepare all standard solutions in plasticware. It is recommended that the user
purchase certified stock standard solutions. Stock standards typically used for AAS are also suitable for the preparation of cation
working standards.
NOTE 3—Neutral pH cation standards are preferred. Alternatively, prepare stock standard solutions from the following salts, as described below:
8.6.1 Ammonium Solution, Stock (1000 mg/L)—Dissolve 2.965 g of anhydrous ammonium chloride in water and dilute to 1 L
+
volumetrically; 1.00 mL = 1.00 mg NH .
8.6.2 Lithium Solution, Stock (1000 mg/L)—Dissolve 6.108 g of anhydrous lithium chloride in water and dilute to 1 L
+
volumetrically; 1.00 mL = 1.00 mg Li .
8.6.3 Sodium Solution, Stock (1000 mg/L)—Dissolve 2.541 g of anhydrous sodium chloride in water and dilute to 1 L
+
volumetrically; 1.00 mL = 1.00 mg Na .
D6919 − 17
8.6.4 Potassium Solution, Stock (1000 mg/L)—Dissolve 3.481 g of anhydrous potassium phosphate monobasic in water and
+
dilute to 1 L volumetrically; 1.00 mL = 1.00 mg K .
8.6.5 Magnesium Solution, Stock (1000 mg/L)—Dissolve 10.144 g of magnesium sulfate hetpahydrate in water and dilute to 1
2+
L volumetrically; 1.00 mL = 1.00 mg Mg .
8.6.6 Calcium Solution, Stock e(1000 mg/L)—Dissolve 3.668 g of calcium chloride dihydrate in water and dilute to 1 L
2+
volumetrically; 1.00 mL = 1.00 mg Ca .
8.7 Cation Working Standards—All calibration standards and standards used for analysis should be prepared in 100–mL
volumetric flasks, as described below.
Standard concentration mg/L 5
~ !
~stock volume added ~mL!·1000 mg/L!
100 mL
~ !
Example:
~1 mL Na stock·1000 mg/L Na!
10 mg/L Na 5
100 mL
~ !
8.8 Blank—The blank standard is a portion of the water used to prepare the cation working solutions.
8.9 Filter Paper—Purchase suitable filter paper. Typically the filter papers have a pore size of 0.22-μm or 0.45-μm membrane.
Material such as fine-textured, ashless paper, or glass fiber paper are acceptable. The user must first ascertain that the filter paper
is of sufficient purity to use without adversely affecting the bias and precision of the test method.
9. Precautions
9.1 These methods address the determination of low concentrations of cations. Accordingly, every precaution should be taken
to ensure the cleanliness of sample containers, as well as other materials and apparatus that come in contact with the sample.
10. Sampling and Sample Preservation
10.1 Collect the sample in accordance with PracticePractices D3370, as applicable.
10.2 Samples must be collected in plastic containers that are clean and free of artifacts and interferences. The suitability of the
containers must be demonstrated for each new lot by performing a container blank and laboratory fortified container blank.
10.3 Samples that will not be analyzed immediately must be preserved with sulfuric acid to a pH of 2. Whereas samples to be
analyzed for cations are typically preserved with nitric acid, sulfuric acid is recommended for ammonium. Add 0.8 mL
concentrated H SO /L of sample and store at 4°C. The pH of samples preserved in this manner should be between 1.5 and 2,
2 4
although some wastewaters may require more concentrated H SO to achieve this pH. This pH increases the holding time to 28
2 4
days.
11. Preparation of Apparatus
11.1 Set up the ion chromatograph according to the manufacturer’s instructions.
11.2 Typical operating conditions for the ion chromatograph, in both suppressed and nonsuppressed conductivity modes, are
summarized in Tables 1 and 2. A10–μL A 10–μL sample injection is recommended for calibration and sample analysis when using
chemically suppressed conductivity detection. A larger injection volume, that is, up to 50 μL, may be required when using
nonsuppressed conductivity detection.
Standard Methods for the Examination of Water and Wastewater, APHA, Washington, DC, 1992.
TABLE 1 Instrument Conditions for the Analysis of Dissolved
Alkali and Alkaline Earth Cations and Ammonium by Ion
Chromatography Using Suppressed Conductivity Detection
Eluent: 26 mM methanesulfonic acid
Flow rate: 1.5 mL/min
Column: Dionex IonPac CG16/CS16
Sample Loop: 10 μL
Detection: Suppressed conductivity
Suppressor: CSRS ULTRA
Background: ~2 μS
Solutes: 1 = lithium (1.0 mg/L), 2 = sodium (1.0 mg/L),
3 = ammonium (1.0 mg/L), 4 = potassium (1.0 mg/L),
5 = magnesium (1.0 mg/L), 6 = calcium (1.0 mg/L)
D6919 − 17
TABLE 2 Instrument Conditions for the Analysis of Dissolved
Alkali and Alkaline Earth Cations and Ammonium by Ion
Chromatography Using Nonsuppressed Conductivity Detection
(Single-Column Indirect Conductivity Detection)
Eluent: 3 mM nitric acid / 0.05 mM EDTA
Flow rate: 1.0 mL/min
Column: Waters IC-Pak™C/MD,
Injection 20 μL
Volume:
Detection: Nonsuppressed conductivity, indirect conductivity
Background: ~1300 μS
Analytes: 1 = lithium (1.0 mg/L), 2 = sodium (4.0 mg/L),
3 = ammonium (5.0 mg/L), 4 = potassium (10.0 mg/L),
5 = magnesium (5.0 mg/L), 6 = calcium (10.0 mg/L)
11.3 The detector ranges are variable. Choose a range consistent with the concentration range in the expected samples and
within the operating requirements of the chromatographic system used.
11.4 Equilibrate the chromatographic system prior to use by pumping the eluent (8.4 or 8.5) through the system until a stable
baseline is obtained (approximately 30 min).
12. Calibration and Standardization
12.1 Determine the range of concentrations that will be quantified for each analyte. For each individual calibration curve,
prepare calibration standards, at a minimum of three concentration levels, by accurately adding measured volumes of the stock
standards (8.6) to a volumetric flask(s) and diluting to volume with water. A minimum of five concentration levels is recommended
if the curve covers two orders of magnitude.
12.2 The order of peak elution and typical retention times are shown in Figs. 1 and 2 for suppressed and nonsuppressed
conductivity detection, respectively. The retention time of each analyte can vary with the type and state of the guard and analytical
columns and the eluent concentration, but should remain consistent within a given analysis batch.
12.3 To establish the calibration curve, analyze a reagent blank (8.8) and the calibration standards (8.7) in accordance with the
procedure in Section 13. Tabulate peak area responses against concentration. These results are used to prepare a calibration curve
using a linear least-squares fit for each analyte (with the exception of ammonium). The squared correlation coefficient of
determination (r ) should be ≥0.995 for accurate results. Ammonia is a weak base; hence the ammonium cation does not give linear
response with suppressed conductivity detection and should be calibrated using a quadratic fit when using this detection method.
However, a linear least-squares curve fit can be suitably used for calibration of ammonium using the nonsuppressed version of this
method. Once the calibration curves have been established, verification must be performed on each analysis day, whenever fresh
eluent is prepared, and once for each batch of samples.
13. Procedure
13.1 Inject the reagent blank, calibration standard, or sample into the eluent stream and record the chromatogram. With a
fixed-loop manual injector, flush an excess of the sample (approximately 5× loop volume) through the sample injection port using
a syringe prior to injection. Examples of method chromatograms are shown in Figs. 1 and 2 for the suppressed and nonsuppressed
conductivity detection, ion chromatographic separation of the target cation analytes.
14. Calculations
14.1 Compare the peak areas for the cations in the sample to the calibration curves prepared in 12.3. Calculate and report the
concentration of each cation in mg/L by comparing the analyte response to that of the standard curve according to the following
equation:
Cation Concentration, mg/L5 A 3F (1)
where:
A = reading from the appropriate calibration plot, in mg/L
A = reading from the appropriate calibration plot, in mg/L, and
F = dilution factor if the sample was diluted prior to analysis
14.1.1 Computing integrators and computer-based chromatographic data systems can be programmed to perform these
calculations automatically.
15. Report
15.1 Report results as the constituent cation in mg/L. Ammonium results are frequently reported as mg of NH -N/L. To convert
to mg of NH -N/L, multiply the ammonium result by a factor of
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...