ASTM E2036-24
(Test Method)Standard Test Method for Nitrogen Trichloride in Liquid Chlorine by High Performance Liquid Chromatography (HPLC)
Standard Test Method for Nitrogen Trichloride in Liquid Chlorine by High Performance Liquid Chromatography (HPLC)
SIGNIFICANCE AND USE
4.1 This test method was developed for the determination of nitrogen trichloride in samples of carbon tetrachloride liquid taken from the compressor suction chiller bottoms of chlorine production plants and other places in the plants that may collect and concentrate nitrogen trichloride to levels that could explode. The test method was then modified to measure the lower levels of nitrogen trichloride observed in product liquid chlorine. This test method is sensitive enough to measure the levels of nitrogen trichloride observed in the normal production of liquid chlorine.
4.2 This test method for nitrogen trichloride will require the dilution (100:1) of highly concentrated in-process samples to bring them within the working range of the analysis.
SCOPE
1.1 This test method uses high performance liquid chromatography (HPLC) to determine nitrogen trichloride levels in liquid chlorine at the 0.1 μg/g (ppm) to 600 μg/g (ppm) range. Solvent samples from chlorine production facilities containing very high concentrations of nitrogen trichloride may be analyzed by dilution with methanol.
1.2 Review the current safety data sheet (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 The following applies for the purposes of determining the conformance of the test results using this test method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazard statements are given in Section 8.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 29-Feb-2024
- Technical Committee
- D16 - Aromatic, Industrial, Specialty and Related Chemicals
- Drafting Committee
- D16.13 - Chlorine
Relations
- Effective Date
- 01-Mar-2024
Overview
ASTM E2036-24 is the internationally recognized standard test method for determining nitrogen trichloride content in liquid chlorine using high performance liquid chromatography (HPLC). This test method is essential in the chlorine production industry, where controlling nitrogen trichloride concentrations is crucial for plant safety and product quality. Nitrogen trichloride can accumulate in various plant locations and may reach hazardous levels, making accurate detection and quantification vital to prevent incidents such as explosions and ensure regulatory compliance.
Key Topics
- High Performance Liquid Chromatography (HPLC): The standard outlines the use of HPLC for precise measurement of nitrogen trichloride (NCl₃) within a working range of 0.1 μg/g (ppm) to 600 μg/g (ppm).
- Sample Preparation: The method details procedures for preparing both product liquid chlorine samples and highly concentrated in-process solvent samples through appropriate dilution, typically using methanol.
- Safety Considerations: Handling chlorine and nitrogen trichloride involves significant safety hazards. The standard stresses the importance of following safety data sheets (SDS), occupational regulations, and plant procedures, as well as using appropriate laboratory equipment and protective gear.
- Quality Assurance: Laboratories utilizing this method are required to maintain rigorous quality control systems, including routine calibration, use of control samples, and interlaboratory comparisons to ensure reliability and comparability of results.
- Results and Reporting: Results are calculated with attention to precision, using defined statistical controls. The method adheres to established rounding practices for reporting test data and supports consistency with relevant regulatory requirements.
Applications
- Chlorine Production Facilities: With its sensitivity and selectivity, ASTM E2036-24 is indispensable for monitoring nitrogen trichloride in liquid chlorine during manufacturing, storage, and distribution.
- Process Safety Management: By providing early detection of potentially hazardous NCl₃ concentrations, the method assists in process optimization and in preventing unsafe accumulation of this explosive compound.
- Regulatory Compliance: This standard supports adherence to environmental, health, and safety regulations related to chlorine and its impurities, as required by industry best practices and national transportation codes.
- Quality Control: The test method enables producers and buyers of liquid chlorine to ensure product purity and consistent quality, building customer confidence and market acceptance.
Related Standards
This standard references and aligns with several other key ASTM and regulatory documents for analytical procedures, laboratory safety, and data accuracy:
- ASTM E29: Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications.
- ASTM D6809: Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials.
- ASTM E806: Test Method for Carbon Tetrachloride and Chloroform in Liquid Chlorine by Direct Injection (Gas Chromatographic Procedure).
- ASTM E691: Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method.
- CFR Title 49, Part 173: U.S. Department of Transportation regulations for handling compressed gases, including chlorine.
- Chlorine Institute Pamphlet No. 152: Guidelines for the safe handling of chlorine containing nitrogen trichloride.
Practical Value
ASTM E2036-24 is critical for chemical producers, laboratory analysts, and safety professionals in the chlorine supply chain. It provides:
- A reliable method for routine monitoring of nitrogen trichloride to prevent safety incidents.
- A framework for laboratory best practices in testing and data reporting.
- Ease of adoption internationally, being developed under World Trade Organization recognized principles.
For the best results and compliance, laboratories should ensure all staff are properly trained and equipped, and all work is reviewed against the current standard and referenced safety documents.
Buy Documents
ASTM E2036-24 - Standard Test Method for Nitrogen Trichloride in Liquid Chlorine by High Performance Liquid Chromatography (HPLC)
REDLINE ASTM E2036-24 - Standard Test Method for Nitrogen Trichloride in Liquid Chlorine by High Performance Liquid Chromatography (HPLC)
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E2036-24 is a standard published by ASTM International. Its full title is "Standard Test Method for Nitrogen Trichloride in Liquid Chlorine by High Performance Liquid Chromatography (HPLC)". This standard covers: SIGNIFICANCE AND USE 4.1 This test method was developed for the determination of nitrogen trichloride in samples of carbon tetrachloride liquid taken from the compressor suction chiller bottoms of chlorine production plants and other places in the plants that may collect and concentrate nitrogen trichloride to levels that could explode. The test method was then modified to measure the lower levels of nitrogen trichloride observed in product liquid chlorine. This test method is sensitive enough to measure the levels of nitrogen trichloride observed in the normal production of liquid chlorine. 4.2 This test method for nitrogen trichloride will require the dilution (100:1) of highly concentrated in-process samples to bring them within the working range of the analysis. SCOPE 1.1 This test method uses high performance liquid chromatography (HPLC) to determine nitrogen trichloride levels in liquid chlorine at the 0.1 μg/g (ppm) to 600 μg/g (ppm) range. Solvent samples from chlorine production facilities containing very high concentrations of nitrogen trichloride may be analyzed by dilution with methanol. 1.2 Review the current safety data sheet (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 The following applies for the purposes of determining the conformance of the test results using this test method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazard statements are given in Section 8. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This test method was developed for the determination of nitrogen trichloride in samples of carbon tetrachloride liquid taken from the compressor suction chiller bottoms of chlorine production plants and other places in the plants that may collect and concentrate nitrogen trichloride to levels that could explode. The test method was then modified to measure the lower levels of nitrogen trichloride observed in product liquid chlorine. This test method is sensitive enough to measure the levels of nitrogen trichloride observed in the normal production of liquid chlorine. 4.2 This test method for nitrogen trichloride will require the dilution (100:1) of highly concentrated in-process samples to bring them within the working range of the analysis. SCOPE 1.1 This test method uses high performance liquid chromatography (HPLC) to determine nitrogen trichloride levels in liquid chlorine at the 0.1 μg/g (ppm) to 600 μg/g (ppm) range. Solvent samples from chlorine production facilities containing very high concentrations of nitrogen trichloride may be analyzed by dilution with methanol. 1.2 Review the current safety data sheet (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 The following applies for the purposes of determining the conformance of the test results using this test method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazard statements are given in Section 8. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2036-24 is classified under the following ICS (International Classification for Standards) categories: 71.040.30 - Chemical reagents. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2036-24 has the following relationships with other standards: It is inter standard links to ASTM E2036-15. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2036-24 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2036 − 24
Standard Test Method for
Nitrogen Trichloride in Liquid Chlorine by High Performance
Liquid Chromatography (HPLC)
This standard is issued under the fixed designation E2036; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 2. Referenced Documents
2.1 ASTM Standards:
1.1 This test method uses high performance liquid chroma-
D6809 Guide for Quality Control and Quality Assurance
tography (HPLC) to determine nitrogen trichloride levels in
Procedures for Aromatic Hydrocarbons and Related Ma-
liquid chlorine at the 0.1 μg ⁄g (ppm) to 600 μg/g (ppm) range.
terials
Solvent samples from chlorine production facilities containing
E29 Practice for Using Significant Digits in Test Data to
very high concentrations of nitrogen trichloride may be ana-
Determine Conformance with Specifications
lyzed by dilution with methanol.
E180 Practice for Determining the Precision of ASTM
1.2 Review the current safety data sheet (SDS) for detailed
Methods for Analysis and Testing of Industrial and Spe-
information concerning toxicity, first aid procedures, and safety cialty Chemicals (Withdrawn 2009)
precautions. E691 Practice for Conducting an Interlaboratory Study to
Determine the Precision of a Test Method
1.3 The values stated in SI units are to be regarded as
E806 Test Method for Carbon Tetrachloride and Chloroform
standard. No other units of measurement are included in this
in Liquid Chlorine by Direct Injection (Gas Chromato-
standard.
graphic Procedure)
1.4 The following applies for the purposes of determining 2.2 Federal Standards:
the conformance of the test results using this test method to CFR 173 Title 49 Transportation; Shippers’ General Re-
applicable specifications, results shall be rounded off in accor- quirements for Shipments and Packaging, including Sec-
dance with the rounding-off method of Practice E29. tions:
173.304 Charging of Cylinders with Liquefied Compressed
1.5 This standard does not purport to address all of the
Gas
safety concerns, if any, associated with its use. It is the
173.314 Requirements for Compressed Gases in Tank Cars
responsibility of the user of this standard to establish appro-
173.315 Compressed Gases in Cargo Tanks and Portable
priate safety, health, and environmental practices and deter-
Tank Containers
mine the applicability of regulatory limitations prior to use.
2.3 Other Document:
Specific hazard statements are given in Section 8.
Chlorine Institute Pamphlet No. 152 Safe Handling of Chlo-
1.6 This international standard was developed in accor-
rine Containing Nitrogen Trichloride
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the
3. Summary of Test Method
Development of International Standards, Guides and Recom-
3.1 Weighed samples of chlorine are delivered into a cooled
mendations issued by the World Trade Organization Technical
graduated centrifuge tube. One mL of cooled HPLC eluent is
Barriers to Trade (TBT) Committee.
added before the chlorine is allowed to evaporate. After the
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website.
1 3
This test method is under the jurisdiction of ASTM Committee D16 on The last approved version of this historical standard is referenced on
Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsi- www.astm.org.
bility of Subcommittee D16.13 on Chlorine. Available from U.S. Government Publishing Office, 732 N. Capitol St., NW,
Current edition approved March 1, 2024. Published April 2024. Originally Washington, DC 20401-0001, http://www.gpo.gov.
approved in 1999. Last previous edition approved in 2015 as E2036 – 15. DOI: Available from The Chlorine Institute (CI), 1300 Wilson Blvd., Ste. 525,
10.1520/E2036-24. Arlington, VA 22209, http://www.chlorineinstitute.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2036 − 24
chlorine has evaporated the remaining eluent is analyzed 7. Reagents
directly on the HPLC for nitrogen trichloride concentration.
7.1 Purity of Reagents—Unless otherwise indicated, it is
3.2 In-process solvent samples from chlorine production intended that all reagents shall conform to the specifications of
facility may be analyzed by dilution in methanol followed by the Committee on Analytical Reagents of the American Chemi-
direct HPLC analysis of the diluted solution. cal Society, where such specifications are available. Other
grades may be used, provided it is first ascertained that the
4. Significance and Use reagent is of sufficiently high purity to permit its use without
lessening the accuracy of the determination.
4.1 This test method was developed for the determination of
nitrogen trichloride in samples of carbon tetrachloride liquid 7.2 Water, HPLC grade.
taken from the compressor suction chiller bottoms of chlorine
7.3 Methanol, HPLC grade. CAS# 67-56-1.
production plants and other places in the plants that may collect
7.4 Sodium Acetate, reagent grade. CAS# 127-09-3.
and concentrate nitrogen trichloride to levels that could ex-
plode. The test method was then modified to measure the lower
7.5 Glacial Acetic Acid, reagent grade. CAS# 64-19-7.
levels of nitrogen trichloride observed in product liquid chlo-
7.6 Dry Ice. CAS# 124-38-9.
rine. This test method is sensitive enough to measure the levels
7.7 Ammonium Sulfate, reagent grade. CAS# 7783-20-2.
of nitrogen trichloride observed in the normal production of
liquid chlorine.
4.2 This test method for nitrogen trichloride will require the
dilution (100:1) of highly concentrated in-process samples to
bring them within the working range of the analysis.
5. Interferences
FIG. 1 Ammonium Sulfate, reagent grade: CAS# 7783-20-2
5.1 This test method is selective for nitrogen trichloride. At
this time there is no known interference in the materials used
7.8 Chlorine, reagent grade. CAS# 7782-50-5.
in this test method.
7.9 Helium.
5.2 Contact with any metal surfaces should be avoided due
to the corrosive nature of the sample. 8. Hazards
8.1 Consult current OSHA regulations, suppliers’ Safety
5.3 Nitrogen trichloride is decomposed by UV light, heat or
strong acid. The presence of metal ions will increase the rate of Data Sheets, and local regulations for all materials used in this
test method.
decomposition. Special care should be taken to avoid exposure
of the samples to direct sunlight. Samples and standards should
8.2 Safety and Health Precautions—Exposure to all solvents
be analyzed immediately upon preparation. Samples not im-
used in this test method should be avoided.
mediately analyzed must be stored in dry ice until just before
8.3 Concentrations of nitrogen chloride over 2 % pose the
analysis.
risk of explosion and should be considered dangerous. Never
allow any solutions containing nitrogen chloride to evaporate,
6. Apparatus
concentrating the nitrogen chloride in the remaining residue.
6.1 A high performance liquid chromatograph (HPLC) com-
The concentrations of nitrogen trichloride synthesized in this
posed of the following:
test method are small and should not pose any significant risk
6.1.1 HPLC Pump, capable of 1 mL/min flow,
of explosion. All of the solutions prepared in this test method
6.1.2 HPLC UV Detector, capable of operating at 221 nm
can be disposed of by flushing them down any laboratory sink
with a 1 cm light path,
using a reasonable amount of water.
6.1.3 HPLC Injection Valve, 20 μL sample loop, all
8.4 Chlorine is a corrosive and toxic material. A well-
nonmetal, and
ventilated fume hood should be used to house all sample
6.1.4 HPLC Column, C18 reverse phase, 25 cm by 4.6 mm.
handling and to vent the test equipment when this product is
6.2 Plastic Syringes. 1 mL, 2 mL, 5 mL, 10 mL, 20 mL, and
analyzed in the laboratory.
60 mL.
8.5 This analysis should only be performed by persons who
6.3 Nonmetallic Syringe Needles.
are thoroughly familiar with the handling of chlorine, and even
6.4 Top Loader Balance, capable of 0.01 g resolution and 1 an experienced person should not work alone. The operator
must be provided with adequate eye protection and a respirator.
kg capacity.
6.5 TFE-fluorocarbon Tubing, 1.59 mm outside diameter.
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
6.6 Stainless Steel Sample Cylinder, with a needle valve on
Standard-Grade Reference Materials, American Chemical Society, Washington,
one end.
DC. For suggestions on the testing of reagents not listed by the American Chemical
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
6.7 Graduated Centrifuge Tube, 15 mL.
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
6.8 Reducing Tube Fitting, 6.35 mm to 1.59 mm tubing. copeial Convention, Inc. (USPC), Rockville, MD.
E2036 − 24
Splashes of liquid chlorine destroy clothing and, if such the 1.59 mm nut and ferrule. See Fig. 2. It may be helpful to cut
clothing is next to the skin, will produce irritations and burns. a 6.35 mm circle of fluoropolymer frit material and place it into
the reducing fitting prior to assembly to prevent plugging of the
8.6 If liquid samples are to be taken in cylinders, do not
1.59 mm tubing. Cut the length of the 1.59 mm tubing so that
allow the sample cylinder to become liquid full. Test Method
only 38.10 mm protrudes out of the fitting.
E806, 49 CFR 173.314, 173.315, and 173.304 advise that the
weight of the chlorine in the cylinder should not be more than
13. Standardization of the HPLC
125 % of the weight of the water that the cylinder could
13.1 Prepare a 1000 μg ⁄g (ppm) stock solution of ammo-
contain.
nium sulfate in water. Make a series of standards of ammonium
8.7 When sampling and working with chlorine out-of-doors,
sulfate by serial dilution covering the range between 1 μg ⁄g
people downwind from such an operation should be warned of
and 650 μg/g (ppm) nitrogen trichloride. Calculate the nitrogen
the possible release of chlorine vapors.
trichloride concentration of each standard from the ammonium
8.8 In the event chlorine is inhaled, first aid should be
sulfate concentration by multiplying by the ratio of the
summoned immediately and oxygen administered without
molecular weights, as follows:
delay.
2NCl / NH SO 5 2 × 120.5/132 5 1.826 (1)
@ ~ ! # ~ !
3 4 2 4
8.9 Store pressurized samples where involuntary release
13.2 Withdraw 0.5 mL of an ammonium sulfate standard
would not cause excessive risk to people or property.
into a 1 mL plastic syringe and then pull the plunger back to the
8.10 It is recommended that means be available for disposal 1 mL mark to fill the remainder of the syringe with air.
of excess chlorine in an environmentally safe and acceptable
13.3 Clamp a sample cylinder containing reagent chlorine
manner. If chlorine cannot be disposed of in a chlorine
vertically in a ring stand and attach a 152.4 mm piece of 1.59
consuming process, a chlorine absorption system should be
mm outside diameter fluoropolymer tubing to the valve. Take
provided. When the analysis and sampling regimen requires an
a second 1 mL plastic syringe and pull the plunger back to the
initial purging of chlorine from a container, the purged chlorine
1 mL mark. Insert the 1.59 mm fluoropolymer tubing into the
should be similarly handled. Purging to the atmosphere should
second syringe and gently purge chlorine through the syringe
be avoided.
filling this second syringe with reagent chlorine gas. Remove
the fluoropolymer tubing from the syringe and attach a
9. Typical Instrument Parameters
nonmetal needle to the luer tip.
9.1 Adjust the chromatograph in accordance with the fol-
13.4 Holding the first syringe pointed upward, use the
lowing parameters and allow the instrument to equilibrate until
second syringe equipped with the nonmetal needle to bubble
a steady baseline is obtained:
the reagent chlorine gas through the ammonium sulfate stan-
9.1.1 Column—C18 reverse phase ODS (C18) 25 cm by 4.6
dard solution, converting the ammonium ions to nitrogen
mm, 10 μm,
trichloride. Use of a 1 mL syringe should avoid overchlorina-
9.1.2 Eluent—60 % by volume methanol, 40 % by volume
tion. See Fig. 3.
0.1 mol/L (M) acetate buffer, pH 4.5, helium sparged,
13.5 Use the entire 0.5 mL standard to flush the sample
9.1.3 Flow Rate—1 mL/min,
valve and fill the sample loop. Immediately inject this standard
9.1.4 Injection Volume—20 μl sample loop, and
into the HPLC for analysis. The nitrogen trichloride peak will
9.1.5 Detector Wavelength—UV at 221 nm.
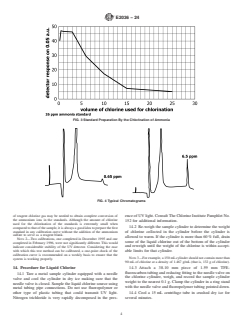
elute between 11 min and 12 min in the chromatogram. Se
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E2036 − 15 E2036 − 24
Standard Test Method for
Nitrogen Trichloride in Liquid Chlorine by High Performance
Liquid Chromatography (HPLC)
This standard is issued under the fixed designation E2036; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Scope*
1.1 This test method uses high performance liquid chromatography (HPLC) to determine nitrogen trichloride levels in liquid
chlorine at the 0.10.1 μg ⁄g (ppm) to 600 μg/g (ppm) range. Solvent samples from chlorine production facilities containing very
high concentrations of nitrogen trichloride may be analyzed by dilution with methanol.
1.2 Review the current safety data sheet (SDS) for detailed information concerning toxicity, first aid procedures, and safety
precautions.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 The following applies for the purposes of determining the conformance of the test results using this test method to applicable
specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. Specific hazard statements are given in Section 8.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D6809 Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials
E29 Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications
E180 Practice for Determining the Precision of ASTM Methods for Analysis and Testing of Industrial and Specialty Chemicals
(Withdrawn 2009)
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E806 Test Method for Carbon Tetrachloride and Chloroform in Liquid Chlorine by Direct Injection (Gas Chromatographic
Procedure)
This test method is under the jurisdiction of ASTM Committee D16 on Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsibility of
Subcommittee D16.13 on Chlorine.
Current edition approved June 1, 2015March 1, 2024. Published June 2015April 2024. Originally approved in 1999. Last previous edition approved in 20072015 as
E2036E2036 – 15.– 07. DOI: 10.1520/E2036-15. DOI: 10.1520/E2036-24.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2036 − 24
2.2 Federal Standards:
CFR 173 Title 49 Transportation; Shippers’ General Requirements for Shipments and Packaging, including Sections:
173.304 Charging of Cylinders with Liquefied Compressed Gas
173.314 Requirements for Compressed Gases in Tank Cars
173.315 Compressed Gases in Cargo Tanks and Portable Tank Containers
2.3 Other Document:
Chlorine Institute Pamphlet No. 152 Safe Handling of Chlorine Containing Nitrogen Trichloride
3. Summary of Test Method
3.1 Weighed samples of chlorine are delivered into a cooled graduated centrifuge tube. One mL of cooled HPLC eluent is added
before the chlorine is allowed to evaporate. After the chlorine has evaporated the remaining eluent is analyzed directly on the
HPLC for nitrogen trichloride concentration.
3.2 In-process solvent samples from chlorine production facility may be analyzed by dilution in methanol followed by direct
HPLC analysis of the diluted solution.
4. Significance and Use
4.1 This test method was developed for the determination of nitrogen trichloride in samples of carbon tetrachloride liquid taken
from the compressor suction chiller bottoms of chlorine production plants and other places in the plants that may collect and
concentrate nitrogen trichloride to levels that could explode. The test method was then modified to measure the lower levels of
nitrogen trichloride observed in product liquid chlorine. This test method is sensitive enough to measure the levels of nitrogen
trichloride observed in the normal production of liquid chlorine.
4.2 This test method for nitrogen trichloride will require the dilution (100:1) of highly concentrated in-process samples to bring
them within the working range of the analysis.
5. Interferences
5.1 This test method is selective for nitrogen trichloride. At this time there is no known interference in the materials used in this
test method.
5.2 Contact with any metal surfaces should be avoided due to the corrosive nature of the sample.
5.3 Nitrogen trichloride is decomposed by UV light, heat or strong acid. The presence of metal ions will increase the rate of
decomposition. Special care should be taken to avoid exposure of the samples to direct sunlight. Samples and standards should
be analyzed immediately upon preparation. Samples not immediately analyzed must be stored in dry ice until just before analysis.
6. Apparatus
6.1 A high performance liquid chromatograph (HPLC) composed of the following:
6.1.1 HPLC Pump, capable of 1 mL/min flow,
6.1.2 HPLC UV Detector, capable of operating at 221 nm with a 1-cm1 cm light path,
6.1.3 HPLC Injection Valve, 20 μL sample loop, all nonmetal, and
6.1.4 HPLC Column, C18 reverse phase, 25 cm by 4.6 mm.
6.2 Plastic Syringes. 1, 2, 5, 10, 20,1 mL, 2 mL, 5 mL, 10 mL, 20 mL, and 60 mL.
Available from U.S. Government Publishing Office, 732 N. Capitol St., NW, Washington, DC 20401-0001, http://www.gpo.gov.
Available from The Chlorine Institute (CI), 1300 Wilson Blvd., Ste. 525, Arlington, VA 22209, http://www.chlorineinstitute.org.
E2036 − 24
6.3 Nonmetallic Syringe Needles.
6.4 Top Loader Balance, capable of 0.01 g resolution and 1 kg capacity.
6.5 TFE-fluorocarbon Tubing, 1.59 mm outside diameter.
6.6 Stainless Steel Sample Cylinder, with a needle valve on one end.
6.7 Graduated Centrifuge Tube, 15 mL.
6.8 Reducing Tube Fitting, 6.35 mm to 1.59 mm tubing.
7. Reagents
7.1 Purity of Reagents—Unless otherwise indicated, it is intended that all reagents shall conform to the specifications of the
Committee on Analytical Reagents of the American Chemical Society, where such specifications are available. Other grades may
be used, provided it is first ascertained that the reagent is of sufficiently high purity to permit its use without lessening the accuracy
of the determination.
7.2 Water, HPLC grade.
7.3 Methanol, HPLC grade. CAS# 67-56-1.
7.4 Sodium Acetate, reagent grade. CAS# 127-09-3.
7.5 Glacial Acetic Acid, reagent grade. CAS# 64-19-7.
7.6 Dry Ice. CAS# 124-38-9.
7.7 Ammonium Sulfate,Sulfate, reagent grade. CAS# 7783-20-2.
FIG. 1 Ammonium Sulfate, reagent grade: CAS# 7783-20-2
7.8 Chlorine, reagent grade. CAS# 7782-50-5.
7.9 Helium.
8. Hazards
8.1 Consult current OSHA regulations, suppliers’ Safety Data Sheets, and local regulations for all materials used in this test
method.
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society, see Analar Standards for
Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC),
Rockville, MD.
E2036 − 24
8.2 Safety and Health Precautions—Exposure to all solvents used in this test method should be avoided.
8.3 Concentrations of nitrogen chloride over 2 % pose the risk of explosion and should be considered dangerous. Never allow any
solutions containing nitrogen chloride to evaporate, concentrating the nitrogen chloride in the remaining residue. The
concentrations of nitrogen trichloride synthesized in this test method are small and should not pose any significant risk of
explosion. All of the solutions prepared in this test method can be disposed of by flushing them down any laboratory sink using
a reasonable amount of water.
8.4 Chlorine is a corrosive and toxic material. A well-ventilated fume hood should be used to house all sample handling and to
vent the test equipment when this product is analyzed in the laboratory.
8.5 This analysis should only be performed by persons who are thoroughly familiar with the handling of chlorine, and even an
experienced person should not work alone. The operator must be provided with adequate eye protection and a respirator. Splashes
of liquid chlorine destroy clothing and, if such clothing is next to the skin, will produce irritations and burns.
8.6 If liquid samples are to be taken in cylinders, do not allow the sample cylinder to become liquid full. Test Method E806, 49
CFR 173.314, 173.315, and 173.304 advise that the weight of the chlorine in the cylinder should not be more than 125 % of the
weight of the water that the cylinder could contain.
8.7 When sampling and working with chlorine out-of-doors, people downwind from such an operation should be warned of the
possible release of chlorine vapors.
8.8 In the event chlorine is inhaled, first aid should be summoned immediately and oxygen administered without delay.
8.9 Store pressurized samples where involuntary release would not cause excessive risk to people or property.
8.10 It is recommended that means be available for disposal of excess chlorine in an environmentally safe and acceptable manner.
If chlorine cannot be disposed of in a chlorine consuming process, a chlorine absorption system should be provided. When the
analysis and sampling regimen requires an initial purging of chlorine from a container, the purged chlorine should be similarly
handled. Purging to the atmosphere should be avoided.
9. Typical Instrument Parameters
9.1 Adjust the chromatograph in accordance with the following parameters and allow the instrument to equilibrate until a steady
baseline is obtained:
9.1.1 Column—C18 reverse phase ODS (C18) 25 cm by 4.6 mm, 10 μm,
9.1.2 Eluent—60 % by volume methanol, 40 % by volume 0.1 mol/L (M) acetate buffer, pH 4.5, helium sparged,
9.1.3 Flow Rate—1 mL/min,
9.1.4 Injection Volume—20-μl20 μl sample loop, and
9.1.5 Detector Wavelength—UV at 221 nm.
10. Preparation of Buffer Solution
10.1 Sodium Acetate Buffer Stock Solution (1 mol/L (M))—Dissolve 136 g sodium acetate (NaOOCCH × 3H O) and 60 g glacial
3 2
acetic acid (HOOCCH ) in water and dilute to 1 L.
10.2 Sodium Acetate Buffer (0.1 mol/L (M))—Transfer 100 mL of the stock buffer solution into a 1-L1 L volumetric flask and dilute
to volume with water.
E2036 − 24
11. Preparation of Eluent
11.1 Add 600 mL methanol to 400 mL 0.1 M sodium acetate buffer solution and mix well. Before use purge the solution with
helium for 20 min to remove dissolved oxygen.
12. Preparation of the Sample Cylinder
12.1 Clamp the sample cylinder in a vertical position with the needle valve in the downward position. Insert the 6.35 mm end of
the reducing tube fitting into the needle valve and set the 6.35 mm nut and ferrule of the fitting. Insert the 1.59 mm fluoropolymer
tubing in the reducing tube fitting and tighten the 1.59 mm nut and ferrule. See Fig. 12. It may be helpful to cut a 6.35 mm circle
of fluoropolymer frit material and place it into the reducing fitting prior to assembly to prevent plugging of the 1.59 mm tubing.
Cut the length of the 1.59 mm tubing so that only 38.10 mm protrudes out of the fitting.
13. Standardization of the HPLC
13.1 Prepare a 1000-μg/g1000 μg ⁄g (ppm) stock solution of ammonium sulfate in water. Make a series of standards of ammonium
sulfate by serial dilution covering the range between 11 μg ⁄g and 650 μg/g (ppm) nitrogen trichloride. Calculate the nitrogen
trichloride concentration of each standard from the ammonium sulfate concentration by multiplying by the ratio of the molecular
weights, as follows:
@2NCl /~NH ! SO # 5 ~2 ×120.5/132!5 1.826 (1)
3 4 2 4
13.2 Withdraw 0.5 mL of an ammonium sulfate standard into a 1-mL1 mL plastic syringe and then pull the plunger back to the
1-mL1 mL mark to fill the remainder of the syringe with air.
13.3 Clamp a
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...