ASTM D6508-15
(Test Method)Standard Test Method for Determination of Dissolved Inorganic Anions in Aqueous Matrices Using Capillary Ion Electrophoresis and Chromate Electrolyte
Standard Test Method for Determination of Dissolved Inorganic Anions in Aqueous Matrices Using Capillary Ion Electrophoresis and Chromate Electrolyte
SIGNIFICANCE AND USE
5.1 Capillary ion electrophoresis provides a simultaneous separation and determination of several inorganic anions using nanolitres of sample in a single injection. All anions present in the sample matrix will be visualized yielding an anionic profile of the sample.
5.2 Analysis time is less than 5 minutes with sufficient sensitivity for drinking water and wastewater applications. Time between samplings is less than seven minutes allowing for high sample throughput.
5.3 Minimal sample preparation is necessary for drinking water and wastewater matrices. Typically, only a dilution with water is needed.
5.4 This test method is intended as an alternative to other multi-analyte methods and various wet chemistries for the determination of inorganic anions in water and wastewater. Compared to other multi-analyte methods the major benefits of CIE are speed of analysis, simplicity, and reduced reagent consumption and operating costs.
SCOPE
1.1 This test method covers the determination of the inorganic anions fluoride, bromide, chloride, nitrite, nitrate, ortho-phosphate, and sulfate in drinking water, wastewater, and other aqueous matrices using capillary ion electrophoresis (CIE) with indirect UV detection. See Figs. 1-6.
1.2 The test method uses a chromate-based electrolyte and indirect UV detection at 254 nm. It is applicable for the determination or inorganic anions in the range of 0.1 to 50 mg/L except for fluoride whose range is 0.1 to 25 mg/L.
1.3 It is the responsibility of the user to ensure the validity of this test method for other anion concentrations and untested aqueous matrices.
Note 1: The highest accepted anion concentration submitted for precision and bias extend the anion concentration range for the following anions: Chloride to 93 mg/L, Sulfate to 90 mg/L, Nitrate to 72 mg/L, and ortho-phosphate to 58 mg/L.
1.4 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9.
General Information
- Status
- Published
- Publication Date
- 30-Sep-2015
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.05 - Inorganic Constituents in Water
Relations
- Effective Date
- 01-Oct-2015
- Effective Date
- 01-May-2020
- Effective Date
- 01-Aug-2018
- Effective Date
- 01-Aug-2018
- Effective Date
- 01-Jan-2013
- Effective Date
- 15-Jun-2012
- Refers
ASTM D3856-11 - Standard Guide for Management Systems in Laboratories Engaged in Analysis of Water - Effective Date
- 15-Nov-2011
- Effective Date
- 15-Jun-2011
- Effective Date
- 01-May-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-Jul-2008
- Effective Date
- 15-Jan-2008
- Effective Date
- 01-Dec-2007
Overview
ASTM D6508-15 is a standardized test method developed by ASTM International for the determination of dissolved inorganic anions in aqueous matrices, such as drinking water and wastewater, using Capillary Ion Electrophoresis (CIE) with a chromate-based electrolyte. This test method enables the rapid, simultaneous separation and quantification of several inorganic anions in minimal sample volumes with high sensitivity and efficiency.
This method is recognized as a fast and cost-effective alternative to traditional wet chemistry and other multi-analyte methods. It offers practical value by reducing reagent consumption, minimizing sample preparation, and supporting high sample throughput in laboratory environments analyzing water quality.
Key Topics
- Target Analytes: Fluoride, bromide, chloride, nitrite, nitrate, ortho-phosphate, and sulfate in concentrations ranging from 0.1 to 50 mg/L (fluoride up to 25 mg/L).
- Analytical Technique: Capillary Ion Electrophoresis (CIE) with indirect UV detection at 254 nm, employing a chromate-based electrolyte for effective separation.
- Matrix Applicability: Suitable for drinking water, municipal and industrial wastewater, and other aqueous environmental samples.
- Speed and Sensitivity: Less than 5-minute analysis per sample, with minimal time required for turnover between samples (under 7 minutes), ideal for high-throughput laboratories.
- Sample Preparation: Streamlined process typically requires only dilution with reagent-grade water.
- Data Output: Generates a comprehensive anionic profile for each sample, aiding both routine analysis and investigative studies.
- Regulatory Compliance: Developed in line with international standardization principles and is applicable in compliance-driven water quality monitoring.
Applications
Water Quality Analysis
- Drinking Water Monitoring: Provides sensitive detection and quantification of health-relevant anions, supporting regulatory compliance and potable water safety assessments.
- Wastewater Treatment: Facilitates operational monitoring and regulatory reporting for municipal and industrial wastewater, ensuring effective effluent management.
- Environmental Studies: Offers a reliable method for profiling anionic contaminants in surface waters, groundwater, and process streams.
Laboratory Efficiency
- High Throughput Testing: Rapid analysis and minimal sample preparation enhance productivity in contract laboratories, municipal water labs, and industrial quality control settings.
- Cost Reduction: Lower reagent and material usage reduce operational expenses compared to traditional methods.
- Versatility: With broad applicability across water types and a robust detection range, the method supports diverse sample matrices.
Quality Assurance
- Precision and Bias Statements: Extensive precision and matrix recovery data support confidence in method accuracy across variable matrices.
- Traceability: Encourages use of certified reference standards and quality control solutions for method validation and ongoing performance verification.
Related Standards
- ASTM D1129: Terminology Relating to Water-establishes key definitions used throughout water analysis standards.
- ASTM D1193: Specification for Reagent Water-details requirements for water purity in laboratory testing.
- ASTM D2777: Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water.
- ASTM D3370: Practices for Sampling Water from Closed Conduits-guidelines for proper collection and handling of water samples.
- ASTM D3856: Guide for Management Systems in Laboratories Engaged in Analysis of Water.
- ASTM D5847: Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis.
By referencing these related ASTM standards, laboratories can ensure rigorous quality control, standardization, and regulatory alignment in the implementation of ASTM D6508-15.
Keywords: ASTM D6508-15, capillary ion electrophoresis, chromate electrolyte, inorganic anions, water quality analysis, drinking water, wastewater, environmental testing, laboratory standards, inorganic ion determination, UV detection, sample preparation, precision, bias, related ASTM standards.
Buy Documents
ASTM D6508-15 - Standard Test Method for Determination of Dissolved Inorganic Anions in Aqueous Matrices Using Capillary Ion Electrophoresis and Chromate Electrolyte
REDLINE ASTM D6508-15 - Standard Test Method for Determination of Dissolved Inorganic Anions in Aqueous Matrices Using Capillary Ion Electrophoresis and Chromate Electrolyte
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D6508-15 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Dissolved Inorganic Anions in Aqueous Matrices Using Capillary Ion Electrophoresis and Chromate Electrolyte". This standard covers: SIGNIFICANCE AND USE 5.1 Capillary ion electrophoresis provides a simultaneous separation and determination of several inorganic anions using nanolitres of sample in a single injection. All anions present in the sample matrix will be visualized yielding an anionic profile of the sample. 5.2 Analysis time is less than 5 minutes with sufficient sensitivity for drinking water and wastewater applications. Time between samplings is less than seven minutes allowing for high sample throughput. 5.3 Minimal sample preparation is necessary for drinking water and wastewater matrices. Typically, only a dilution with water is needed. 5.4 This test method is intended as an alternative to other multi-analyte methods and various wet chemistries for the determination of inorganic anions in water and wastewater. Compared to other multi-analyte methods the major benefits of CIE are speed of analysis, simplicity, and reduced reagent consumption and operating costs. SCOPE 1.1 This test method covers the determination of the inorganic anions fluoride, bromide, chloride, nitrite, nitrate, ortho-phosphate, and sulfate in drinking water, wastewater, and other aqueous matrices using capillary ion electrophoresis (CIE) with indirect UV detection. See Figs. 1-6. 1.2 The test method uses a chromate-based electrolyte and indirect UV detection at 254 nm. It is applicable for the determination or inorganic anions in the range of 0.1 to 50 mg/L except for fluoride whose range is 0.1 to 25 mg/L. 1.3 It is the responsibility of the user to ensure the validity of this test method for other anion concentrations and untested aqueous matrices. Note 1: The highest accepted anion concentration submitted for precision and bias extend the anion concentration range for the following anions: Chloride to 93 mg/L, Sulfate to 90 mg/L, Nitrate to 72 mg/L, and ortho-phosphate to 58 mg/L. 1.4 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9.
SIGNIFICANCE AND USE 5.1 Capillary ion electrophoresis provides a simultaneous separation and determination of several inorganic anions using nanolitres of sample in a single injection. All anions present in the sample matrix will be visualized yielding an anionic profile of the sample. 5.2 Analysis time is less than 5 minutes with sufficient sensitivity for drinking water and wastewater applications. Time between samplings is less than seven minutes allowing for high sample throughput. 5.3 Minimal sample preparation is necessary for drinking water and wastewater matrices. Typically, only a dilution with water is needed. 5.4 This test method is intended as an alternative to other multi-analyte methods and various wet chemistries for the determination of inorganic anions in water and wastewater. Compared to other multi-analyte methods the major benefits of CIE are speed of analysis, simplicity, and reduced reagent consumption and operating costs. SCOPE 1.1 This test method covers the determination of the inorganic anions fluoride, bromide, chloride, nitrite, nitrate, ortho-phosphate, and sulfate in drinking water, wastewater, and other aqueous matrices using capillary ion electrophoresis (CIE) with indirect UV detection. See Figs. 1-6. 1.2 The test method uses a chromate-based electrolyte and indirect UV detection at 254 nm. It is applicable for the determination or inorganic anions in the range of 0.1 to 50 mg/L except for fluoride whose range is 0.1 to 25 mg/L. 1.3 It is the responsibility of the user to ensure the validity of this test method for other anion concentrations and untested aqueous matrices. Note 1: The highest accepted anion concentration submitted for precision and bias extend the anion concentration range for the following anions: Chloride to 93 mg/L, Sulfate to 90 mg/L, Nitrate to 72 mg/L, and ortho-phosphate to 58 mg/L. 1.4 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9.
ASTM D6508-15 is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D6508-15 has the following relationships with other standards: It is inter standard links to ASTM D6508-10, ASTM D1129-13(2020)e2, ASTM D1066-18e1, ASTM D1066-18, ASTM D5905-98(2013), ASTM D2777-12, ASTM D3856-11, ASTM D1066-11, ASTM D5810-96(2011), ASTM D3370-10, ASTM D1129-10, ASTM D3370-08, ASTM D5905-98(2008), ASTM D2777-08, ASTM D3370-07. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D6508-15 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D6508 − 15
Standard Test Method for
Determination of Dissolved Inorganic Anions in Aqueous
Matrices Using Capillary Ion Electrophoresis and Chromate
Electrolyte
This standard is issued under the fixed designation D6508; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* D1129Terminology Relating to Water
D1193Specification for Reagent Water
1.1 This test method covers the determination of the inor-
D2777Practice for Determination of Precision and Bias of
ganic anions fluoride, bromide, chloride, nitrite, nitrate, ortho-
Applicable Test Methods of Committee D19 on Water
phosphate,andsulfateindrinkingwater,wastewater,andother
D3370Practices for Sampling Water from Closed Conduits
aqueous matrices using capillary ion electrophoresis (CIE)
D3856Guide for Management Systems in Laboratories
with indirect UV detection. See Figs. 1-6.
Engaged in Analysis of Water
1.2 The test method uses a chromate-based electrolyte and
D5810Guide for Spiking into Aqueous Samples
indirect UV detection at 254 nm. It is applicable for the
D5847Practice for Writing Quality Control Specifications
determination or inorganic anions in the range of 0.1 to 50
for Standard Test Methods for Water Analysis
mg/L except for fluoride whose range is 0.1 to 25 mg/L.
D5905Practice for the Preparation of SubstituteWastewater
F488Test Method for On-Site Screening of Heterotrophic
1.3 It is the responsibility of the user to ensure the validity
of this test method for other anion concentrations and untested Bacteria in Water (Withdrawn 2005)
aqueous matrices.
3. Terminology
NOTE 1—The highest accepted anion concentration submitted for
3.1 Definitions:
precision and bias extend the anion concentration range for the following
anions: Chloride to 93 mg/L, Sulfate to 90 mg/L, Nitrate to 72 mg/L, and 3.1.1 For definitions of terms used in this standard, refer to
ortho-phosphate to 58 mg/L.
Terminology D1129.
1.4 The values stated in SI units are to be regarded as
3.2 Definitions of Terms Specific to This Standard:
standard. The values given in parentheses are mathematical
3.2.1 capillary ion electrophoresis, n—an electrophoretic
conversions to inch-pound units that are provided for informa-
techniqueinwhichaUV-absorbingelectrolyteisplacedina50
tion only and are not considered standard.
µm to 75 µm fused-silica capillary.
1.5 This standard does not purport to address all of the 3.2.1.1 Discussion—Voltage is applied across the capillary
safety concerns, if any, associated with its use. It is the
causing electrolyte and anions to migrate towards the anode
responsibility of the user of this standard to establish appro- and through the capillary’s UV detector window. Anions are
priate safety and health practices and determine the applica-
separated based upon the differential rates of migration in the
bility of regulatory limitations prior to use. For specific hazard electrical field. Anion detection and quantitation are based
statements, see Section 9.
upon the principles of indirect UV detection.
3.2.2 electrolyte, n—a combination of a UV-absorbing salt
2. Referenced Documents
and an electroosmotic-flow modifier placed inside the
2.1 ASTM Standards:
capillary, used as a carrier for the analytes, and for detection
D1066Practice for Sampling Steam
and quantitation.
3.2.2.1 Discussion—The UV-absorbing portion of the salt
This test method is under the jurisdiction ofASTM Committee D19 on Water
mustbeanionicandhaveanelectrophoreticmobilitysimilarto
and is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents
the analyte anions of interest.
in Water.
Current edition approved Oct. 1, 2015. Published October 2015. Originally
3.2.3 electroosmotic flow (EOF), n—the direction and ve-
approved in 2000. Last previous edition approved in 2010 as D6508–10. DOI:
locityofelectrolyte-solutionflowwithinthecapillaryunderan
10.1520/D6508-15.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on The last approved version of this historical standard is referenced on
the ASTM website. www.astm.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6508 − 15
FIG. 4 Electropherogram of Drinking Water
FIG. 1 Electropherogram of Mixed Anion Working Solution and
Added Common Organic Acids
FIG. 5 Electropherogram of Municipal Wastewater Treatment
Plant Discharge
FIG. 2 Electropherogram of 0.2 mg/L Anions Used to Determine
MDL
FIG. 6 Electropherogram of Industrial Wastewater
FIG. 3 Electropherogram of Substitute Wastewater
3.2.4 electroosmotic-flow modifier (OFM), n—a cationic
quaternary amine in the electrolyte that dynamically coats the
appliedelectricalpotential(voltage);thevelocityanddirection negatively charged silica wall giving it a net positive charge.
of flow is determined by electrolyte chemistry, capillary-wall 3.2.4.1 Discussion—This modifier reverses the direction of
chemistry, and applied voltage. the electrolyte’s natural electroosmotic flow and directs it
D6508 − 15
towards the anode and detector. This modifier augments anion 3.2.10 migration time, n—the time required for a specific
migration and enhances speed of analysis. Its concentration analyte to migrate through the capillary to the detector.
secondarily affects anion selectivity and resolution, (see Fig. 3.2.10.1 Discussion—The migration time in capillary ion
7). electrophoresis is analogous to retention time in chromatogra-
phy.
3.2.5 electropherogram, n—a graphical presentation of UV-
3.2.11 time-corrected peak area, n—normalized peak area;
detector response versus time of analysis; the X-axis is
migration time, which is used to identify the anion peak area divided by migration time.
3.2.11.1 Discussion—CE principles state that peak area is
qualitatively, and the Y-axis is UV response, which can be
converted to time-corrected peak area for quantitation. dependent upon migration time, that is, for the same concen-
trationofanalyte,asmigrationtimeincreases(decreases)peak
3.2.6 electrophoretic mobility, n—the specific velocity of a
area increases (decreases). Time-corrected peak area accounts
charged analyte in the electrolyte under specific
for these changes.
electroosmotic-flow conditions.
3.2.6.1 Discussion—The mobility of an analyte is directly
4. Summary of Test Method
related to the analyte’s equivalent ionic conductance and
4.1 Capillary ion electrophoresis, see Figs. 7-10, is a free
applied voltage, and is the primary mechanism of separation.
zoneelectrophoretictechniqueoptimizedforthedetermination
3.2.7 hydrostatic sampling, n—a sample-introduction tech-
of anions with molecular weight less than 200. The anions
niqueinwhichthecapillarywithelectrolyteisimmersedinthe
migrate and are separated according to their mobility in the
sample, and both are elevated to a specific height, typically 10
electrolyte when an electrical field is applied through the open
cm, above the receiving-electrolyte reservoir for a preset
tubular fused silica capillary. The electrolyte’s electroosmotic
amount of time, typically less than 60 s.
low modifier dynamically coats the inner wall of the capillary
3.2.7.1 Discussion—Nanolitres of sample are siphoned into
changing the surface to a net positive charge. This reversal of
the capillary by differential head pressure and gravity.
wall charge reverses the natural EOF. The modified EOF in
3.2.8 indirect UV detection, n—a form of UV detection in combination with a negative power supply augments the
whichtheanalytedisplacesanequivalentnet-chargeamountof mobility of the analyte anions towards the anode and detector
the highly UV-absorbing component of the electrolyte causing achieving rapid analysis times. Cations migrate in the opposite
a net decrease in background absorbance. directiontowardsthecathodeandareremovedfromthesample
during analysis. Water and other neutral species move toward
3.2.8.1 Discussion—The magnitude of the decreased absor-
bance is directly proportional to analyte concentration. the detector at the same rate as the EOF. The neutral species
migrate slower than the analyte anions and do not interfere
Detector-output polarity is reversed in order to obtain a
positive mV response. with anion analysis (see Figs. 7 and 8).
3.2.9 midpoint of peak width, n—CIE peaks typically are 4.2 Thesampleisintroducedintothecapillaryusinghydro-
asymmetrical with the peak apex’s shifting with increasing staticsampling.Theinletofthecapillarycontainingelectrolyte
concentration, and the peak apex may not be indicative of true
is immersed in the sample and the height of the sample raised
analyte-migration time. 10 cm for 30 s where low nanolitre volumes are siphoned into
3.2.9.1 Discussion—Midpointofpeakwidthisthemidpoint
thecapillary.Aftersampleloading,thecapillaryisimmediately
between the analyte peak’s start and stop integration, or the immersed back into the electrolyte. The voltage is applied
peak center of gravity.
initiating the separation process.
FIG. 7 Pictorial Diagram of Anion Mobility and ElectroOsomotic Flow Modifier
D6508 − 15
5.3 Minimal sample preparation is necessary for drinking
water and wastewater matrices. Typically, only a dilution with
water is needed.
5.4 This test method is intended as an alternative to other
multi-analyte methods and various wet chemistries for the
determination of inorganic anions in water and wastewater.
Comparedtoothermulti-analytemethodsthemajorbenefitsof
CIE are speed of analysis, simplicity, and reduced reagent
consumption and operating costs.
6. Interferences
6.1 Analyte identification, quantitation, and possible comi-
gration occur when one anion is in significant excess to other
FIG. 8 Selectivity Diagram of Anion Mobility Using Capillary Ion
anions in the sample matrix. For two adjacent peaks, reliable
Electrophoresis
quantitation can be achieved when the concentration differen-
tial is less than 100:1. As the resolution between two anion
peaks increase so does the tolerated concentration differential.
In samples containing 1000 mg/L Cl, 1 mg/L SO can be
resolved and quantitated, however, the high Cl will interfere
with Br and NO quantitation.
-1
6.2 Dissolved carbonate, detected as HCO , is an anion
present in all aqueous samples, especially alkaline samples.
Carbonate concentrations greater than 500 mg/L will interfere
with PO quantitation.
6.3 Monovalent organic acids, except for formate, and
neutral organics commonly found in wastewater migrate later
in the electropherogram, after carbonate, and do not interfere.
Formate, a common organic acid found in environmental
samples, migrates shortly after fluoride but before phosphate.
Formate concentrations greater than 5 mg/Lwill interfere with
fluoride identification and quantitation. Inclusion of 2 mg/L
FIG. 9 Pictorial Diagram of Indirect UV Detection
formate into the mixed anion working solution aids in fluoride
and formate identification and quantitation.
4.3 Anion detection is based upon the principles of indirect
6.4 Divalent organic acids usually found in wastewater
UVdetection.TheUV-absorbingelectrolyteanionisdisplaced
migrate after phosphate. At high concentrations, greater than
charge-for-charge by the separated analyte anion. The analyte
10 mg/L, they may interfere with phosphate identification and
anion zone has a net decrease in background absorbance. This
quantitation.
decrease in UV absorbance in quantitatively proportional to
6.5 Chlorate also migrates after phosphate and at concen-
analyte anion concentration (see Fig. 9). Detector output
trations greater than 10 mg/L will interfere with phosphate
polarityisreversedtoprovidepositivemVresponsetothedata
identification and quantitation. Inclusion of 5 mg/L chlorate
system, and to make the negative absorbance peaks appear
into the mixed anion working solution aids in phosphate and
positive.
chlorate identification and quantitation.
4.4 The analysis is complete once the last anion of interest
6.6 As analyte concentration increases, analyte peak shape
is detected. The capillary is vacuum purged automatically by
becomes asymmetrical. If adjacent analyte peaks are not
thesystemofanyremainingsampleandreplenishedwithfresh
baseline resolved, the data system will drop a perpendicular
electrolyte. The system now is ready for the next analysis.
between them to the baseline. This causes a decrease in peak
areaforbothanalytepeaksandalowbiasforanalyteamounts.
5. Significance and Use
For optimal quantitation, insure that adjacent peaks are fully
5.1 Capillary ion electrophoresis provides a simultaneous
resolved, if they are not, dilute the sample 1:1 with water.
separation and determination of several inorganic anions using
nanolitres of sample in a single injection.All anions present in 7. Apparatus
thesamplematrixwillbevisualizedyieldingananionicprofile
7.1 Capillary Ion Electrophoresis System—Thesystemcon-
of the sample.
sists of the following components, as shown in Fig. 10 or
5.2 Analysis time is less than 5 minutes with sufficient equivalent:
sensitivity for drinking water and wastewater applications. 7.1.1 High Voltage Power Supply, capable of generating
Time between samplings is less than seven minutes allowing voltage (potential) between 0 and minus 30 kV relative to
for high sample throughput. groundwiththecapabilityworkinginaconstantcurrentmode.
D6508 − 15
FIG. 10 General Hardware Schematic of a Capillary Ion Electrophoresis System
7.1.2 Covered Sample Carousel, to prevent environmental 7.4 Plastic Syringe, 20-mL, disposable.
contamination of the samples and electrolytes during a multi-
7.5 Vacuum Filtration Apparatus, capable for filtering 100
sample batch analysis.
mL of reagent through a 0.45-µm aqueous filter (see 8.14).
7.1.3 Sample Introduction Mechanism, capable of hydro-
8. Reagents and Materials
static sampling technique, using gravity, positive pressure, or
equivalent.
8.1 Purity of Reagents—Unless otherwise indicated, it is
7.1.4 Capillary Purge Mechanism, to purge the capillary
intended that all reagents shall conform to the reagent grade
after every analysis with fresh electrolyte to eliminate any
specification of the Analytical Reagents of the American
interference from the previous sample matrix, and to clean the
Chemical Society, where such specifications are available.
capillary with other reagent, such as sodium hydroxide.
Other grades may be used, provided it is first ascertained that
7.1.5 UV Detector, having the capability of monitoring 254
the reagent is of sufficient high purity to permit its use without
nm, or equivalent, with a time constant of 0.3 s.
lessening the performance or accuracy of the determination.
7.1.6 Fused Silica Capillary—a 75 µm (inner diameter) ×
Reagent chemicals shall be used for all tests.
375 µm (outer diameter) × 60 cm (length) having a polymer
NOTE 3—Calibration and detection limits of this test method are biased
coating for flexibility, and noncoated section to act as the cell
by the purity of the reagents.
4,5
window for UV detection.
8.2 Purity of Water—Unless otherwise indicated, references
7.1.7 Constant Temperature Compartment, to keep the
to water shall be understood to mean Type I reagent water
samples, capillary, and electrolytes at constant temperature.
conforming or exceeding Specification D1193. Freshly drawn
7.2 Data System—Acomputer system that can acquire data
water should be used for preparation of all stock and working
at 20 points/s minimum, express migration time in minutes to
standards, electrolytes, and solutions. Performance and detec-
three decimal places, use midpoint of the analyte peak width,
tion limits of this test method are limited by the purity of
or center of gravity, to determine the analyte migration time,
reagent water, especially TOC. Other reagent water types may
use normalized migration times with respect to a reference
be used provided it is first ascertained that the water is of
peakforqualitativeidentification,usetimecorrectedpeakarea
sufficiently high purity to permit its use without adversely
response for analyte quantitation, and express results in con-
affecting the bias and precision of the test method.
centration units.
8.3 Reagent Blank—Reagent water, or any other solution,
NOTE 2—It is recommended that integrators or standard chromato-
used to preserve or dilute the sample.
graphic data processing not be used with this test method.
4,6
7.3 Anion Exchange Cartridges in the Hydroxide Form.
Reagent Chemicals, American Chemical Society Specifications, Am. Chem.
Soc., Washington, DC. For suggestions on the testing of reagents not listed by the
AmericanChemicalSociety,see Analar Standards for Laboratory Chemicals,BDH
The sole source of supply of the apparatus known to the committee at this time Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
is Waters Corp., 34 Maple St., Milford, MA 01757. Formulary, U.S. Pharmacopoeia Convention, Inc. (USPC), Rockville, Md.
5 8
If you are aware of alternative suppliers, please provide this information to AlthoughthereagentwatermayexceedSpecificationD1193,thereagentwater
ASTM International Headquarters. Your comments will receive careful consider- needs to be periodically tested for bacterial contamination. Bacteria and their waste
ation at a meeting of the responsible technical committee, which you may attend. products may adversely affect system performance. As a guide, ASTM Type IA
The sole source of supply of the apparatus known to the committee at this time water specifies a total bacteria count of 10 colonies/L. Refer to Test Method F488
is Alltech Associates, P/N 30254, 2051 Waukegan Rd., Deerfield, IL, 60015. for analysis procedure.
D6508 − 15
NOTE 9—Anions of no interest may be omitted.
8.4 Individual Anion Solution, Stock:
NOTE 10—The midrange mixed anion solution, working may be used
NOTE 4—It is suggested that certified individual 1000 mg/L anion
for the determination of migration times and resolution described in 12.1.
standards of appropriate known purity be purchased for use with this test
8.6 Calibration Verification Solution (CVS)—Asolutionfor-
method.
mulatedbythelaboratoryofmixedanalytesofknownconcen-
NOTE 5—All weights given are for anhydrous or dried salts. Reagent
puritymustbeaccountedforinordertocalculatetruevalueconcentration.
tration prepared in water. The CVS solution must be prepared
Certify against NIST traceable standards.
from a different source to the calibration standards.
8.4.1 Bromide Solution, Standard (1.0 mL = 1.00 mg
8.7 Performance Evaluation Solution (PES)—A solution
Bromide)—Dryapproximately0.5gofsodiumbromide(NaBr)
formulated by an independent source of mixed analytes of
for6hat 150°C and cool in a desiccator. Dissolve 0.128 g of
known concentration prepared in water. Ideally, the PES
the dry salt in a 100 mLvolumetric flask with water, and fill to
solution should be purchased from an independent source.
mark with water.
8.8 Quality Control Solution (QCS)—A solution of known
8.4.2 Chloride Solution, Standard (1.0 mL = 1.00 mg
analyteconcentrationsaddedtoasyntheticsamplematrixsuch
Chloride)—Dry approximately 0.5 g of sodium chloride
as substitute wastewater that sufficiently challenges the test
(NaCl) for1hat 100°C and cool in a desiccator. Dissolve
method.
0.165 g of the dry salt in a 100 mL a volumetric flask with
8.9 Buffer Solution (100 mM CHES/1 mM Calcium
water, and fill to mark with water.
8.4.3 Fluoride Solution, Standard (1.0 mL = 1.00 mg Gluconate)—Dissolve 20.73 g of CHES (2-[N-
Cyclohexylamino]-Ethane Sulfonic Acid) and 0.43 g of cal-
Fluoride)—Dry approximately 0.5 g of sodium fluoride (NaF)
for1hat 100°C and cool in a desiccator. Dissolve 0.221 g of ciumgluconateina1-Lvolumetricflaskwithwater,anddilute
to 1 L with water. This concentrate may be stored in a capped
the dry salt in a 100 mLvolumetric flask with water, and fill to
mark with water. glass or plastic container for up to one year.
8.4.4 Formate Solution, Standard (1.0 mL = 1.00 mg
8.10 Chromate Concentrate Solution (100 mM Sodium
Formate)—Dissolve 0.151 g of sodium formate in a 100-mL
Chromate)—Dissolve23.41gofsodiumchromatetetrahydrate
volumetric flask with water, and fill to mark with water.
(Na CrO ·4 H O) in a 1-L volumetric flask with water, and
2 4 2
8.4.5 Nitrate Solution, Standard (1.0 mL = 1.00 mg
dilute to 1 L with water. This concentrate may be stored in a
Nitrate)—Dry approximately 0.5 g of sodium nitrate (NaNO )
capped glass or plastic container for up to one year.
for 48 h at 105°C and cool in a desiccator. Dissolve 0.137 g of
8.11 OFM Concentrate Solution (100 mM Tetradecyltrim-
thedrysaltina100-mLvolumetricflaskwithwater,andfillto
ethyl Ammonium Bromide)—Dissolve 33.65 g of Tetradecylt-
mark with water.
rimethyl Ammonium Bromide (TTABr) in a 1-L volumetric
8.4.6 Nitrite Solution, Standard (1.0 mL = 1.00 mg
flask with water, and dilute to 1 L with water. Store this
Nitrite)—Dry approximately 0.5 g of sodium nitrite (NaNO )
solution in a capped glass or plastic container for up to one
for 24 h in a desiccator containing concentrated sulfuric acid.
year.
Dissolve 0.150 g of the dry salt in a 100-mL volumetric flask
with water, and fill to mark with water. Store in a sterilized NOTE 11—TTABr needs to be converted to the hydroxide form
(TTAOH)forusewiththistestmethod.TTAOHiscommerciallyavailable
glass bottle. Refrigerate and prepare monthly.
as 100 mM TTAOH, which is an equivalent substitute.
NOTE 6—Nitrite is easily oxidized, especially in the presence of
8.12 Sodium Hydroxide Solution (500 mM Sodium
moisture. Use only fresh reagent.
Hydroxide)—Dissolve 20 g of sodium hydroxide (NaOH) in a
NOTE 7—Prepare sterile bottles for storing nitrite solutions by heating
for1hat 170°C in an air oven. 1-L plastic volumetric flask with water, and dilute to 1 L with
water.
8.4.7 Ortho-Phosphate Solution, Standard (1.0 mL = 1.00
mg o-Phosphate)—Dissolve 0.150 g of anhydrous dibasic
8.13 Electrolyte Solution, Working (4.7 mM Chromate/4
4,9
sodium phosphate (Na HPO ) in a 100-mL volumetric flask
mM TTAOH/10 mM CHES/0.1 mM Calcium Gluconate) —
2 4
with water, and fill to mark with water.
Wash the anion exchange cartridge in the hydroxide form (see
8.4.8 Sulfate Solution, Standard (1.0 mL = 1.00 mg 7.3) using the 20-mL plastic syringe (see 7.4) with 10 mL of
Sulfate)—Dry approximately 0.5 g of anhydrous sodium sul-
500mMNaOH(see8.12)followedby10mLofwater.Discard
fate (Na SO ) for 1 h at 110°C and cool in a dessicator.
the washings. Slowly pass 4-mL of the 100 mM TTABr
2 4
Dissolve 0.148 g of the dry salt in a 100-mL volumetric flask
solution (see 8.11) through the cartridge into a 100-mL
with water, and fill to mark with water.
volumetric flask. Rinse the cartridge with 20 mL of water,
adding the washing to the volumetric flask.
8.5 Mixed Anion Solution, Working—Prepare at least three
different working standard concentrations for the analyte
NOTE 12—The above procedure is used to convert the TTABr to
anions of interest bracketing the desired range of analysis, TTAOH, which is used in the electrolyte. If using commercially available
100 mMTTAOH, the above conversion step is not necessary; substitute 4
typicallybetween0.1and50mg/L,andadd2mg/Lformateto
mL of 100 mM TTAOH and continue below.
all standards. Add an appropriate aliquot of Individual anion
stocksolution(see8.4)toaprerinsed100-mLvolumetricflask,
and dilute to 100 mL with water.
The sole source of supply of the apparatus known to the committee at this time
NOTE 8—Use 100 µL of Individual anion stock solution (see 8.4) per is Waters Corp., 34 Maple St., Milford, MA01757, as IonSelect High MobilityAn-
100 mL for 1 mg/L anion. ion Electrolyte, P/N 49385.
D6508 − 15
8.13.1 Into the 100-mL volumetric flask add 4.7 mL of 11.4 Apply 15 kV of voltage and test for current. The
chromate concentrate solution (see 8.10) and 10 mL of buffer current should be 14 6 1 µA. If no current is observed, then
solution (see 8.9). Mix and dilute to 100 mL with water. The there is a bubble, or blockage, or both, in the capillary. Degas
natural pH of the electrolyte should be 9 6 0.1. Filter and the chromate electrolyte working solution and retry. If still no
degas using the vacuum filtration apparatus. Store the any current, replace the capillary.
remaining electrolyte in a capped glass or plastic container at
11.5 SettheUVdetectorto254nmdetection,orequivalent.
ambient temperature. The electrolyte is stable for one year.
Zero the detector to 0.000 absorbance. UV offset is less than
8.14 Filter Paper—Purchase suitable filter paper. Typically
0.1 AU.
the filter papers have a pore size of 0.45-µm membrane.
11.6 Program the CE system for constant current of 14 µA.
Material such as fine-textured, acid-washed, ashless paper, or
glass fiber paper are acceptable. The user must first ascertain
11.7 Program the CE system for a hydrostatic sampling of
that the filter paper is of sufficient purity to use without 30 s. Approximately 37 nL of sample is siphoned into the
adversely affecting the bias and precision of the test method.
capillary. Different sampling times may be used provided that
the samples and standards are analyzed identically.
9. Precautions
11.8 Program the CE system for 1 minute purge with the
9.1 Chemicals used in this test method are typical of many
chromate electrolyte working solution between each analysis.
useful laboratory chemicals, reagents, and cleaning solutions,
Usinga103kPa(15psi)vacuumpurgemechanism,one60-cm
whichcanbehazardousifnothandledproperly.RefertoGuide
capillary volume can be displaced in 30 s.
D3856.
11.9 Program the data system for an acquisition rate of at
9.2 Itistheresponsibilityoftheusertoprepare,handle,and
least 20 points/s. Program the data system to identify analyte
disposeofchemicalsolutionsinaccordancewithallapplicable
peaks based upon normalized migration time using Cl as the
federal, state, and local regulations. (Warning—This capillary
reference peak, and to quantitate analyte peak response using
electrophoresis method uses high voltage as a means for
time corrected peak area.
separatingtheanalyteanions,andcanbehazardousifnotused
NOTE 13—Under the analysis conditions Cl is always the first peak in
properly.Useonlythoseinstrumentsthathaveallpropersafety
the electropherogram, and can be used as migration time reference peak.
features.)
12. Calibration
10. Sampling
12.1 Determination of Migration Times (Calibrate Daily)—
10.1 CollectsamplesinaccordancewithPracticesD3370or
The migration time of an anion is dependent upon the
D1066.
electrolyte composition, pH, capillary surface and length,
10.2 Rinse sample containers with sample and discard to
applied voltage, the ionic strength of the sample, and tempera-
eliminate any contamination from the container. Fill to over-
ture. For every fresh electrolyte determine the analyte migra-
flowing and cap to exclude air.
tion time, in min to the third decimal place, of the midrange
10.3 Analyze samples, as soon as possible, after collection.
mixed anion standard working solution (see 8.5), described in
For nitrite, nitrate, and phosphate refrigerate the sample at 4°C Section 11. Use the midpoint of analyte peak width as the
aftercollection.Warmtoroomtemperaturebeforedilutionand
determinant of analyte migration time.
analysis.
NOTE 14—Analyte peak apex may be used as the migration time
10.4 At the laboratory, filter samples containing suspended
determinant, but potential analyte misidentification may result with
solids through a prerinsed 0.45-µm aqueous compatible mem- asymmetrical peak shape at high analyte concentrations.
brane filter (8.14) before analysis.
12.2 Analyze the blank (see 8.3) and at least three working
10.5 If sample dilution is required to remain within the mg/Lsolutions (see 8.5), using the set-up described in Section
11. For each anion concentration (X-axis) plot time corrected
scope of this test method, dilute with water only.
peak area response (Y-axis). Determine the best linear calibra-
tion line through the data points, or use the linear regression
11. Preparation of Apparatus
calibration routine (linear through zero) available in the data
11.1 Set up the CE and data system according to the
system.
manufacturer’s instructions.
NOTE 15—Do not use peak height for calibration. Peak area is directly
11.2 Program the CE system to maintain a constant tem-
related to migration time, that is, for the same analyte concentration,
perature of 25 6 0.5°C, or 5°C above ambient laboratory
increasing migration time give increasing peak area.
temperature. Fill the electrolyte reservoirs with fresh chromate
12.2.1 Ther (coefficientofdetermination)valuesshouldbe
electrolyte working solution (see 8.13), and allow 10 minutes
greater than 0.995; typical r values obtained from the inter-
for thermal equilibration.
laboratory collaborative are given in Table A1.2.
11.3 Condition a new capillary (see 7.1.6) with 500 mM
NaOH solution (see 8.12) for 5 minutes followed by water for 12.3 Calibratedailyandwitheachchangeinelectrolyte,and
5 minutes. Purge the capillary with electrolyte (see 8.13) for 3 validate by analyzing the CVS solution (see 8.6) according to
minutes. procedure in 16.4.
D6508 − 15
12.4 After validation of linear multiple point calibration, a 16. Quality Control
single point calibration solution can be used between 0.1 and
16.1 Before this test method is applied to the analysis of
50 mg/Lfor recalibration provided the quality control require-
unknown samples, the analyst should establish control accord-
ments in 16.4 are met.
ing to procedures recommended in Practice D5847 and Guide
D5810.
13. Procedure
16.2 The laboratory using this test should perform an initial
13.1 Dilute the sample, if necessary with water, to remain
demonstrationoflaboratorycapabilityaccordingtoprocedures
within the scope (see 1.2 and 1.3) and calibration of this test
outlines in Practice D5847.
method. Refer to A1.5.1.
NOTE 18—Certified performance evaluation solutions (PES) and QC
13.2 Analyze all blanks (see 8.3), standards (see 8.5), and
solutions (QCS and CVS) are commercially available and recommended.
samples as described in Section 11 using the quality control
16.3 Initial Demonstration of Performance—Analyze seven
criteria described in 16.5 – 16.9. Refer to Figs. 1-6 for
replicatesofaperformanceevaluationsolution(PES)(see8.7).
representative anion standard, detection li
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D6508 − 10 D6508 − 15
Standard Test Method for
Determination of Dissolved Inorganic Anions in Aqueous
Matrices Using Capillary Ion Electrophoresis and Chromate
Electrolyte
This standard is issued under the fixed designation D6508; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the determination of the inorganic anions fluoride, bromide, chloride, nitrite, nitrate, ortho-
phosphate, and sulfate in drinking water, wastewater, and other aqueous matrices using capillary ion electrophoresis (CIE) with
indirect UV detection. See Figs. 1-6.
1.2 The test method uses a chromate-based electrolyte and indirect UV detection at 254 nm. It is applicable for the
determination or inorganic anions in the range of 0.1 to 50 mg/L except for fluoride whose range is 0.1 to 25 mg/L.
1.3 It is the responsibility of the user to ensure the validity of this test method for other anion concentrations and untested
aqueous matrices.
NOTE 1—The highest accepted anion concentration submitted for precision and bias extend the anion concentration range for the following anions:
Chloride to 93 mg/L, Sulfate to 90 mg/L, Nitrate to 72 mg/L, and ortho-phosphate to 58 mg/L.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this The values
given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not
considered standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use. For specific hazard statements, see Section 9.
2. Referenced Documents
2.1 ASTM Standards:
D1066 Practice for Sampling Steam
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D3370 Practices for Sampling Water from Closed Conduits
D3856 Guide for Management Systems in Laboratories Engaged in Analysis of Water
D5810 Guide for Spiking into Aqueous Samples
D5847 Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
D5905 Practice for the Preparation of Substitute Wastewater
F488 Test Method for On-Site Screening of Heterotrophic Bacteria in Water (Withdrawn 2005)
3. Terminology
3.1 Definitions—Definitions: For definitions of terms used in this test method, refer to Terminology D1129.
3.1.1 For definitions of terms used in this standard, refer to Terminology D1129.
3.2 Definitions of Terms Specific to This Standard:
This test method is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents in Water.
Current edition approved Sept. 1, 2010Oct. 1, 2015. Published October 2010October 2015. Originally approved in 2000. Last previous edition approved in 20052010 as
ε2
D6508 – 00D6508 – 10. (2005) . DOI: 10.1520/D6508-10.10.1520/D6508-15.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6508 − 15
FIG. 1 Electropherogram of Mixed Anion Working Solution and Added Common Organic Acids
FIG. 2 Electropherogram of 0.2 mg/L Anions Used to Determine MDL
FIG. 3 Electropherogram of Substitute Wastewater
3.2.1 capillary ion electrophoresis, n—an electrophoretic technique in which a UV-absorbing electrolyte is placed in a 50 μm
to 75 μm fused-silica capillary.
3.2.1.1 Discussion—
D6508 − 15
FIG. 4 Electropherogram of Drinking Water
FIG. 5 Electropherogram of Municipal Wastewater Treatment Plant Discharge
FIG. 6 Electropherogram of Industrial Wastewater
Voltage is applied across the capillary causing electrolyte and anions to migrate towards the anode and through the capillary’s UV
detector window. Anions are separated based upon the differential rates of migration in the electrical field. Anion detection and
quantitation are based upon the principles of indirect UV detection.
3.2.2 electrolyte, n—a combination of a UV-absorbing salt and an electroosmotic-flow modifier placed inside the capillary, used
as a carrier for the analytes, and for detection and quantitation.
3.2.2.1 Discussion—
The UV-absorbing portion of the salt must be anionic and have an electrophoretic mobility similar to the analyte anions of interest.
3.2.3 electroosmotic flow (EOF), n—the direction and velocity of electrolyte-solution flow within the capillary under an applied
electrical potential (voltage); the velocity and direction of flow is determined by electrolyte chemistry, capillary-wall chemistry,
and applied voltage.
3.2.4 electroosmotic-flow modifier (OFM), n—a cationic quaternary amine in the electrolyte that dynamically coats the
negatively charged silica wall giving it a net positive charge.
3.2.4.1 Discussion—
This modifier reverses the direction of the electrolyte’s natural electroosmotic flow and directs it towards the anode and detector.
This modifier augments anion migration and enhances speed of analysis. Its concentration secondarily affects anion selectivity and
resolution, (see Fig. 7).
D6508 − 15
FIG. 7 Pictorial Diagram of Anion Mobility and ElectroOsomotic Flow Modifier
3.2.5 electropherogram, n—a graphical presentation of UV-detector response versus time of analysis; the x-axisX-axis is
migration time, which is used to identify the anion qualitatively, and the y-axisY-axis is UV response, which can be converted to
time-corrected peak area for quantitation.
3.2.6 electrophoretic mobility, n—the specific velocity of a charged analyte in the electrolyte under specific electroosmotic-flow
conditions.
3.2.6.1 Discussion—
The mobility of an analyte is directly related to the analyte’s equivalent ionic conductance and applied voltage, and is the primary
mechanism of separation.
3.2.7 hydrostatic sampling, n—a sample-introduction technique in which the capillary with electrolyte is immersed in the
sample, and both are elevated to a specific height, typically 10 cm, above the receiving-electrolyte reservoir for a preset amount
of time, typically less than 60 s.
3.2.7.1 Discussion—
Nanolitres of sample are siphoned into the capillary by differential head pressure and gravity.
3.2.8 indirect UV detection, n—a form of UV detection in which the analyte displaces an equivalent net-charge amount of the
highly UV-absorbing component of the electrolyte causing a net decrease in background absorbance.
3.2.8.1 Discussion—
The magnitude of the decreased absorbance is directly proportional to analyte concentration. Detector-output polarity is reversed
in order to obtain a positive mV response.
3.2.9 midpoint of peak width, n—CIE peaks typically are asymmetrical with the peak apex’s shifting with increasing
concentration, and the peak apex may not be indicative of true analyte-migration time.
3.2.9.1 Discussion—
Midpoint of peak width is the midpoint between the analyte peak’s start and stop integration, or the peak center of gravity.
3.2.10 migration time, n—the time required for a specific analyte to migrate through the capillary to the detector.
3.2.10.1 Discussion—
The migration time in capillary ion electrophoresis is analogous to retention time in chromatography.
D6508 − 15
3.2.11 time-corrected peak area, n—normalized peak area; peak area divided by migration time.
3.2.11.1 Discussion—
CE principles state that peak area is dependent upon migration time, that is, for the same concentration of analyte, as migration
time increases (decreases) peak area increases (decreases). Time-corrected peak area accounts for these changes.
4. Summary of Test Method
4.1 Capillary ion electrophoresis, see Figs. 7-10, is a free zone electrophoretic technique optimized for the determination of
anions with molecular weight less than 200. The anions migrate and are separated according to their mobility in the electrolyte
when an electrical field is applied through the open tubular fused silica capillary. The electrolyte’s electroosmotic low modifier
dynamically coats the inner wall of the capillary changing the surface to a net positive charge. This reversal of wall charge reverses
the natural EOF. The modified EOF in combination with a negative power supply augments the mobility of the analyte anions
towards the anode and detector achieving rapid analysis times. Cations migrate in the opposite direction towards the cathode and
are removed from the sample during analysis. Water and other neutral species move toward the detector at the same rate as the
EOF. The neutral species migrate slower than the analyte anions and do not interfere with anion analysis (see Figs. 7 and 8).
4.2 The sample is introduced into the capillary using hydrostatic sampling. The inlet of the capillary containing electrolyte is
immersed in the sample and the height of the sample raised 10 cm for 30 s where low nanolitre volumes are siphoned into the
capillary. After sample loading, the capillary is immediately immersed back into the electrolyte. The voltage is applied initiating
the separation process.
4.3 Anion detection is based upon the principles of indirect UV detection. The UV-absorbing electrolyte anion is displaced
charge-for-charge by the separated analyte anion. The analyte anion zone has a net decrease in background absorbance. This
decrease in UV absorbance in quantitatively proportional to analyte anion concentration (see Fig. 9). Detector output polarity is
reversed to provide positive mV response to the data system, and to make the negative absorbance peaks appear positive.
4.4 The analysis is complete once the last anion of interest is detected. The capillary is vacuum purged automatically by the
system of any remaining sample and replenished with fresh electrolyte. The system now is ready for the next analysis.
5. Significance and Use
5.1 Capillary ion electrophoresis provides a simultaneous separation and determination of several inorganic anions using
nanolitres of sample in a single injection. All anions present in the sample matrix will be visualized yielding an anionic profile of
the sample.
5.2 Analysis time is less than 5 minutes with sufficient sensitivity for drinking water and wastewater applications. Time between
samplings is less than seven minutes allowing for high sample throughput.
5.3 Minimal sample preparation is necessary for drinking water and wastewater matrices. Typically, only a dilution with water
is needed.
5.4 This test method is intended as an alternative to other multi-analyte methods and various wet chemistries for the
determination of inorganic anions in water and wastewater. Compared to other multi-analyte methods the major benefits of CIE
are speed of analysis, simplicity, and reduced reagent consumption and operating costs.
FIG. 8 Selectivity Diagram of Anion Mobility Using Capillary Ion Electrophoresis
D6508 − 15
FIG. 9 Pictorial Diagram of Indirect UV Detection
6. Interferences
6.1 Analyte identification, quantitation, and possible comigration occur when one anion is in significant excess to other anions
in the sample matrix. For two adjacent peaks, reliable quantitation can be achieved when the concentration differential is less than
100:1. As the resolution between two anion peaks increase so does the tolerated concentration differential. In samples containing
1000 mg/L Cl, 1 mg/L SO can be resolved and quantitated, however, the high Cl will interfere with Br and NO quantitation.
4 2
-1
6.2 Dissolved carbonate, detected as HCO , is an anion present in all aqueous samples, especially alkaline samples. Carbonate
concentrations greater than 500 mg/L will interfere with PO quantitation.
6.3 Monovalent organic acids, except for formate, and neutral organics commonly found in wastewater migrate later in the
electropherogram, after carbonate, and do not interfere. Formate, a common organic acid found in environmental samples, migrates
shortly after fluoride but before phosphate. Formate concentrations greater than 5 mg/L will interfere with fluoride identification
and quantitation. Inclusion of 2 mg/L formate into the mixed anion working solution aids in fluoride and formate identification and
quantitation.
6.4 Divalent organic acids usually found in wastewater migrate after phosphate. At high concentrations, greater than 10 mg/L,
they may interfere with phosphate identification and quantitation.
6.5 Chlorate also migrates after phosphate and at concentrations greater than 10 mg/L will interfere with phosphate
identification and quantitation. Inclusion of 5 mg/L chlorate into the mixed anion working solution aids in phosphate and chlorate
identification and quantitation.
6.6 As analyte concentration increases, analyte peak shape becomes asymmetrical. If adjacent analyte peaks are not baseline
resolved, the data system will drop a perpendicular between them to the baseline. This causes a decrease in peak area for both
analyte peaks and a low bias for analyte amounts. For optimal quantitation, insure that adjacent peaks are fully resolved, if they
are not, dilute the sample 1:1 with water.
7. Apparatus
7.1 Capillary Ion Electrophoresis System—theThe system consists of the following components, as shown in Fig. 10 or
equivalent:
7.1.1 High Voltage Power Supply, capable of generating voltage (potential) between 0 and minus 30 kV relative to ground with
the capability working in a constant current mode.
7.1.2 Covered Sample Carousel, to prevent environmental contamination of the samples and electrolytes during a multisample
batch analysis.
7.1.3 Sample Introduction Mechanism, capable of hydrostatic sampling technique, using gravity, positive pressure, or
equivalent.
7.1.4 Capillary Purge Mechanism, to purge the capillary after every analysis with fresh electrolyte to eliminate any interference
from the previous sample matrix, and to clean the capillary with other reagent, such as sodium hydroxide.
7.1.5 UV Detector, having the capability of monitoring 254 nm, or equivalent, with a time constant of 0.3 s.
7.1.6 Fused Silica Capillary—a 75 μm (inner diameter) x× 375 μm (outer diameter) x× 60 cm (length) having a polymer coating
4,5
for flexibility, and noncoated section to act as the cell window for UV detection.
The sole source of supply of the apparatus known to the committee at this time is Waters Corp., 34 Maple St., Milford, MA 01757.
D6508 − 15
FIG. 10 General Hardware Schematic of a Capillary Ion Electrophoresis System
7.1.7 Constant Temperature Compartment—Compartment, to keep the samples, capillary, and electrolytes at constant
temperature.
7.2 Data System—aA computer system that can acquire data at 20 points/s minimum, express migration time in minutes to three
decimal places, use midpoint of the analyte peak width, or center of gravity, to determine the analyte migration time, use
normalized migration times with respect to a reference peak for qualitative identification, use time corrected peak area response
for analyte quantitation, and express results in concentration units.
NOTE 2—It is recommended that integrators or standard chromatographic data processing not be used with this test method.
4,6
7.3 Anion Exchange Cartridges in the Hydroxide Form.
7.4 Plastic Syringe, 20-mL, disposable.
7.5 Vacuum Filtration Apparatus , Apparatus, capable for filtering 100 mL of reagent through a 0.45 μm aqueous filter.0.45-μm
aqueous filter (see 8.14).
8. Reagents and Materials
8.1 Purity of Reagents—Unless otherwise indicated, it is intended that all reagents shall conform to the reagent grade
specification of the Analytical Reagents of the American Chemical Society, where such specifications are available. Other grades
may be used, provided it is first ascertained that the reagent is of sufficient high purity to permit its use without lessening the
performance or accuracy of the determination. Reagent chemicals shall be used for all tests.
NOTE 3—Calibration and detection limits of this test method are biased by the purity of the reagents.
8.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean Type I reagent water
conforming or exceeding specificationSpecification D1193. Freshly drawn water should be used for preparation of all stock and
working standards, electrolytes, and solutions. Performance and detection limits of this test method are limited by the purity of
reagent water, especially TOC. Other reagent water types may be used provided it is first ascertained that the water is of sufficiently
high purity to permit its use without adversely affecting the bias and precision of the test method.
8.3 Reagent Blank—Reagent water, or any other solution, used to preserve or dilute the sample.
8.4 Individual Anion Solution, StockStock:
If you are aware of alternative suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a
meeting of the responsible technical committee, which you may attend.
The sole source of supply of the apparatus known to the committee at this time is Alltech Associates, P/N 30254, 2051 Waukegan Rd., Deerfield, IL, 60015.
Reagent Chemicals, American Chemical Society Specifications, Am. Chem. Soc., Washington, DC. For suggestions on the testing of reagents not listed by the American
Chemical Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S.
Pharmacopoeia Convention, Inc. (USPC), Rockville, Md.
Although the reagent water may exceed Specification D1193, the reagent water needs to be periodically tested for bacterial contamination. Bacteria and their waste
products may adversely affect system performance. As a guide, ASTM Type IA water specifies a total bacteria count of 10 colonies/L. Refer to Test Method F488 for analysis
procedure.
D6508 − 15
NOTE 4—It is suggested that certified individual 1000 mg/L anion standards of appropriate known purity be purchased for use with this test method.
NOTE 5—All weights given are for anhydrous or dried salts. Reagent purity must be accounted for in order to calculate true value concentration. Certify
against NIST traceable standards.
8.4.1 Bromide Solution, Standard (1.0 mL = 1.00 mg Bromide)—Dry approximately 0.5 g of sodium bromide (NaBr) for 6 h
at 150°C and cool in a desiccator. Dissolve 0.128 g of the dry salt in a 100 mL volumetric flask with water, and fill to mark with
water.
8.4.2 Chloride Solution, Standard (1.0 mL = 1.00 mg Chloride)—Dry approximately 0.5 g of sodium chloride (NaCl) for 1 h
at 100°C and cool in a desiccator. Dissolve 0.165 g of the dry salt in a 100 mL a volumetric flask with water, and fill to mark with
water.
8.4.3 Fluoride Solution, Standard (1.0 mL = 1.00 mg Fluoride)—Dry approximately 0.5 g of sodium fluoride (NaF) for 1 h at
100°C and cool in a desiccator. Dissolve 0.221 g of the dry salt in a 100 mL volumetric flask with water, and fill to mark with
water.
8.4.4 Formate Solution, Standard (1.0 mL = 1.00 mg Formate)—Dissolve 0.151 g of sodium formate in a 100-mL volumetric
flask with water, and fill to mark with water.
8.4.5 Nitrate Solution, Standard (1.0 mL = 1.00 mg Nitrate)—Dry approximately 0.5 g of sodium nitrate (NaNO ) for 48 h at
105°C and cool in a desiccator. Dissolve 0.137 g of the dry salt in a 100-mL volumetric flask with water, and fill to mark with
water.
8.4.6 Nitrite Solution, Standard (1.0 mL = 1.00 mg Nitrite)—Dry approximately 0.5 g of sodium nitrite (NaNO ) for 24 h in
a desiccator containing concentrated sulfuric acid. Dissolve 0.150 g of the dry salt in a 100-mL volumetric flask with water, and
fill to mark with water. Store in a sterilized glass bottle. Refrigerate and prepare monthly.
NOTE 6—Nitrite is easily oxidized, especially in the presence of moisture. Use only fresh reagent.
NOTE 7—Prepare sterile bottles for storing nitrite solutions by heating for 1 h at 170°C in an air oven.
8.4.7 Ortho-Phosphate Solution, Standard (1.0 mL = 1.00 mg o-Phosphate)—Dissolve 0.150 g of anhydrous dibasic sodium
phosphate (Na HPO ) in a 100-mL volumetric flask with water, and fill to mark with water.
2 4
8.4.8 Sulfate Solution, Standard (1.0 mL = 1.00 mg Sulfate)—Dry approximately 0.5 g of anhydrous sodium sulfate (Na SO )
2 4
for 1 h at 110°C and cool in a dessicator. Dissolve 0.148 g of the dry salt in a 100-mL volumetric flask with water, and fill to mark
with water.
8.5 Mixed Anion Solution, Working—Prepare at least three different working standard concentrations for the analyte anions of
interest bracketing the desired range of analysis, typically between 0.1 and 50 mg/L, and add 2 mg/L formate to all standards. Add
an appropriate aliquot of Individual anion stock solution (see 8.4) to a prerinsed 100-mL volumetric flask, and dilute to 100 mL
with water.
NOTE 8—Use 100 μL of Individual anion stock solution (see 8.4) per 100 mL for 1 mg/L anion.
NOTE 9—Anions of no interest may be omitted.
NOTE 10—The midrange mixed anion solution, working may be used for the determination of migration times and resolution described in 12.1.
8.6 Calibration Verification Solution (CVS)—A solution formulated by the laboratory of mixed analytes of known concentration
prepared in water. The CVS solution must be prepared from a different source to the calibration standards.
8.7 Performance Evaluation Solution (PES)—A solution formulated by an independent source of mixed analytes of known
concentration prepared in water. Ideally, the PES solution should be purchased from an independent source.
8.8 Quality Control Solution (QCS)—A solution of known analyte concentrations added to a synthetic sample matrix such as
substitute wastewater that sufficiently challenges the test method.
8.9 Buffer Solution (100 mM CHES/1 mM Calcium Gluconate)—Dissolve 20.73 g of CHES (2-[N-Cyclohexylamino]-Ethane
Sulfonic Acid) and 0.43 g of calcium gluconate in a 1-L volumetric flask with water, and dilute to 1 L with water. This concentrate
may be stored in a capped glass or plastic container for up to one year.
8.10 Chromate Concentrate Solution (100 mM Sodium Chromate)—Dissolve 23.41 g of sodium chromate tetrahydrate
(Na CrO ·4 H O) in a 1-L volumetric flask with water, and dilute to 1 L with water. This concentrate may be stored in a capped
2 4 2
glass or plastic container for up to one year.
8.11 OFM Concentrate Solution (100 mM Tetradecyltrimethyl Ammonium Bromide)—Dissolve 33.65 g of Tetradecyltrimethyl
Ammonium Bromide (TTABr) in a 1-L volumetric flask with water, and dilute to 1 L with water. Store this solution in a capped
glass or plastic container for up to one year.
NOTE 11—TTABr needs to be converted to the hydroxide form (TTAOH) for use with this test method. TTAOH is commercially available as 100 mM
TTAOH, which is an equivalent substitute.
8.12 Sodium Hydroxide Solution (500 mM Sodium Hydroxide)—Dissolve 20 g of sodium hydroxide (NaOH) in a 1-L plastic
volumetric flask with water, and dilute to 1 L with water.
D6508 − 15
4,9
8.13 Electrolyte Solution, Working (4.7 mM Chromate/4 mM TTAOH/10 mM CHES/0.1 mM Calcium Gluconate) —Wash the
anion exchange cartridge in the hydroxide form (see 7.3) using the 20-mL plastic syringe (see 7.4) with 10 mL of 500 mM NaOH
(see 8.12) followed by 10 mL of water. Discard the washings. Slowly pass 4-mL of the 100 mM TTABr solution (see 8.11) through
the cartridge into a 100-mL volumetric flask. Rinse the cartridge with 20 mL of water, adding the washing to the volumetric flask.
NOTE 12—The above procedure is used to convert the TTABr to TTAOH, which is used in the electrolyte. If using commercially available 100 mM
TTAOH, the above conversion step is not necessary; substitute 4 mL of 100 mM TTAOH and continue below.
8.13.1 Into the 100-mL volumetric flask add 4.7 mL of chromate concentrate solution (see 8.10) and 10 mL of buffer solution
(see 8.9). Mix and dilute to 100 mL with water. The natural pH of the electrolyte should be 9 6 0.1. Filter and degas using the
vacuum filtration apparatus. Store the any remaining electrolyte in a capped glass or plastic container at ambient temperature. The
electrolyte is stable for one year.
8.14 Filter Paper—Purchase suitable filter paper. Typically the filter papers have a pore size of 0.45-μm membrane. Material
such as fine-textured, acid-washed, ashless paper, or glass fiber paper are acceptable. The user must first ascertain that the filter
paper is of sufficient purity to use without adversely affecting the bias and precision of the test method.
9. Precautions
9.1 Chemicals used in this test method are typical of many useful laboratory chemicals, reagents, and cleaning solutions, which
can be hazardous if not handled properly. Refer to Guide D3856.
9.2 It is the responsibility of the user to prepare, handle, and dispose of chemical solutions in accordance with all applicable
federal, state, and local regulations. (Warning—This capillary electrophoresis method uses high voltage as a means for separating
the analyte anions, and can be hazardous if not used properly. Use only those instruments that have all proper safety features.)
10. Sampling
10.1 Collect samples in accordance with PracticePractices D3370 or Practice D1066.
10.2 Rinse sample containers with sample and discard to eliminate any contamination from the container. Fill to overflowing
and cap to exclude air.
10.3 Analyze samples, as soon as possible, after collection. For nitrite, nitrate, and phosphate refrigerate the sample at 4°C after
collection. Warm to room temperature before dilution and analysis.
10.4 At the laboratory, filter samples containing suspended solids through a prerinsed 0.45 μm 0.45-μm aqueous compatible
membrane filter (8.14) before analysis.
10.5 If sample dilution is required to remain within the scope of this test method, dilute with water only.
11. Preparation of Apparatus
11.1 Set up the CE and data system according to the manufacturer’s instructions.
11.2 Program the CE system to maintain a constant temperature of 25 6 0.5°C, or 5°C above ambient laboratory temperature.
Fill the electrolyte reservoirs with fresh chromate electrolyte working solution (see 8.13), and allow 10 minutes for thermal
equilibration.
11.3 Condition a new capabilitycapillary (see 7.1.6) with 500 mM NaOH solution (see 8.12) for 5 minutes followed by water
for 5 minutes. Purge the capillary with electrolyte (see 8.13) for 3 minutes.
11.4 Apply 15 kV of voltage and test for current. The current should be 14 6 1 μA. If no current is observed, then there is a
bubble, or blockage, or both, in the capillary. Degas the chromate electrolyte working solution and retry. If still no current, replace
the capillary.
11.5 Set the UV detector to 254 nm detection, or equivalent. Zero the detector to 0.000 absorbance. UV offset is less than 0.1
AU.
11.6 Program the CE system for constant current of 14 μA.
11.7 Program the CE system for a hydrostatic sampling of 30 s. Approximately 37 nL of sample is siphoned into the capillary.
Different sampling times may be used provided that the samples and standards are analyzed identically.
11.8 Program the CE system for 1 minute purge with the chromate electrolyte working solution between each analysis. Using
a 15 psi 103 kPa (15 psi) vacuum purge mechanism, one 60-cm capillary volume can be displaced in 30 s.
11.9 Program the data system for an acquisition rate of at least 20 points/s. Program the data system to identify analyte peaks
based upon normalized migration time using Cl as the reference peak, and to quantitate analyte peak response using time corrected
peak area.
The sole source of supply of the apparatus known to the committee at this time is Waters Corp., 34 Maple St., Milford, MA 01757, as IonSelect High MobilityAnion
Electrolyte, P/N 49385.
D6508 − 15
NOTE 13—Under the analysis conditions Cl is always the first peak in the electropherogram, and can be used as migration time reference peak.
12. Calibration
12.1 Determination of Migration Times (Calibrate Daily)—The migration time of an anion is dependent upon the electrolyte
composition, pH, capillary surface and length, applied voltage, the ionic strength of the sample, and temperature. For every fresh
electrolyte determine the analyte migration time, in min to the third decimal place, of the midrange mixed anion standard working
solution (see 8.5), described in Section 11. Use the midpoint of analyte peak width as the determinant of analyte migration time.
NOTE 14—Analyte peak apex may be used as the migration time determinant, but potential analyte misidentification may result with asymmetrical peak
shape at high analyte concentrations.
12.2 Analyze the blank (see 8.3) and at least three working mg/L solutions (see 8.5), using the set-up described in Section 11.
For each anion concentration (X-axis) plot time corrected peak area response (Y-axis). Determine the best linear calibration line
through the data points, or use the linear regression calibration routine (linear through zero) available in the data system.
NOTE 15—Do not use peak height for calibration. Peak area is directly related to migration time, that is, for the same analyte concentration, increasing
migration time give increasing peak area.
2 2
12.2.1 The r (coefficient of determination) values should be greater than 0.995; typical r values obtained from the
interlaboratory collaborative are given in Table A1.2.
12.3 Calibrate daily and with each change in electrolyte, and validate by analyzing the CVS solution (see 8.6) according to
procedure in 16.4.
12.4 After validation of linear multiple point calibration, a single point calibration solution can be used between 0.1 and 50
mg/L for recalibration provided the quality control requirements in 16.4 are met.
13. Procedure
13.1 Dilute the sample, if necessary with water, to remain within the scope (see 1.2 and 1.3) and calibration of this test method.
Refer to A1.5.1.
13.2 Analyze all blanks (see 8.3), standards (see 8.5), and samples as described in Section 11 using the quality control criteria
described in 16.5 – 16.9. Refer to Figs. 1-6 for representative anion standard, detection limit standard, substitute wastewater,
drinking water, and wastewater electropherograms.
13.3 Analyze all blanks, calibration standards, samples, and quality control solutions in singlicate.
13.3.1 Optional—Duplicate analyses are preferred due to short analysis times.
NOTE 16—Collaborative data was acquired, submitted and evaluated as the average of duplicate samplings.
13.4 After 20 sample analyses, or batch, analyze the QCS solution (see 8.8) If necessary, recalibrate using a single mixed anion
standard working solution (see 8.5), and replace analyte migration time.
NOTE 17—A change in analyte migration time of the mixed anion standard working solution by more than +5% +5 % suggests that components in
the previously analyzed sample matrices have contaminated the capillary surface. Continue but wash the capillary with NaOH solution (see 8.12) before
the next change in electrolyte.
14. Calculation
14.1 Relate the time corrected peak area response for each analyte with the calibration curve generated in 12.2 to determine
mg/L concentration of analyte anion. If the sample was diluted prior to analysis, then multiply mg/L anion by the dilution factor
to obtain the original sample concentration, as follows:
Original Sample mg/L Analyte 5 ~A x SF! (1)
where:
A = analyte concentration determined from the calibration curve, in mg/L, and
SF = scale or dilution factor.
15. Report Format
15.1 The sample analysis report should contain the sample name, analyte anion name, migration time reported to three decimal
places, migration time ratio, peak area, time corrected peak area, sample dilution, and original solution analyte concentration.
15.1.1 Optional—Report analysis method parameters, date of sample data acquisition, and date of result processing for
documentation and validation purposes.
16. Quality Control
16.1 Before this test method is applied to the analysis of unknown samples, the analyst should establish control according to
procedures recommended in Practice D5847, and Guide D5810.
D6508 − 15
16.2 The laboratory using this test should perform an initial demonstration of laboratory capability according to procedures
outlines in Practice D5847.
NOTE 18—Certified performance evaluation solutions (PES) and QC solutions (QCS and CVS) are commercially available and recommended.
16.3 Initial Demonstration of Performance—Analyze seven replicates of a performance evaluation solution (PES) (see 8.7). The
analyte concentration mean and standard deviation of the seven replicates should be calculated and compared to the test methods
single operator precision for equivalent concentrations in reagent water given in Section 17.
16.3.1 Repeat the seven replicate analysis protocol before using a freshly prepared CVS solution (see 8.6) and QCS solution
(see 8.8) for the first time. Calculate the standard deviation and compare with previous results using the student t-test. If no
significant difference is noted, then use the combined standard deviation to determine the QC limits, generally the mean 6 three
standard deviations, for the CVS and QCS solutions.
16.4 Calibration Verification—After calibration, verify the calibration linearity and acceptable instrument performance using a
calibration verification solution (see 8.6) treated as an unknown. If the determined CVS concentrations (see 8.6) are not within 6
3 63 standard deviations of the known true values as described in 16.3.1, the calibration solutions may be out of control.
Reanalyze, and if analyte concentration still falls outside the acceptable limits, fresh calibration solutions (see 8.5) are required.
Successful CVS analyte concentration must be confirmed after recalibration before continuing with the test method.
16.5 Analyze a reagent blank (see 8.3) with each laboratory-defined batch to check for contamination introduced by the
laboratory or use of the test method.
16.6 Quality Control Solution—Analyze one QCS (see 8.8) after 20 samples, or batch. The analyte concentrations for the QCS
should fall within 6 3 standard deviations of historical values for the equivalent concentration and matrix. They are determined
as described in 16.3.1.
Analyze one QCS (see 8.8) after 20 samples, or laboratory-defined batch. It is recommended, but not required to use a second
source, if possible and practical for the QCS. The analyte concentrations for the QCS should fall within 6 3 standard deviations
of historical values for the equivalent concentration and matrix. They are determined as described in 16.3.1.
Upper Control Limit = Analyte Mean Value + 3 times the Standard Deviation
Lower Control Limit = Analyte Mean Value – 3 times the Standard Deviation
16.7 Matrix Spike Recovery—One matrix spike (MS) should be analyzed with each batch of samples to test method recovery.
Spike a portion of one sample from each batch with a known concentration of analyte, prepared in accordance with Guide D3856.
The % recovery of the spike should fall within %recovery 6 analyst %RSD for an equivalent spike concentration and matrix given
in Tables 1-7. If it does not, an interference may be present and the data for the set of similar samples matrices must be qualified
with a warning that the data are suspect, or an alternate test method should be used. Refer to Guide D5810.
16.7.1 If the known analyte concentration is between 15 and 50 mg/L, then spike the sample solution to increase analyte
concentration by 50 %.
16.7.2 If the known analyte concentration is less than 15 mg/L, then spike the sample solution to increase analyte concentration
by 100 %, but not less than 2 mg/L.
16.7.3 Calculate the percent recovery of the spike using the following formula:
% Recovery 5 100 A V 1V 2 B V /C V (2)
@ ~ ! #
s s
where:
A = Analyte concentration (mg/L) in spiked sample,
B = Analyte concentration (mg/L) in unspiked sample,
C = Concentration (mg/L) of analyte in spiking solution,
V = Volume (mL) of sample used, and
s
V = Volume (mL) of spiking solution added.
A = analyte concentration (mg/L) in spiked sample,
B = analyte concentration (mg/L) in unspiked sample,
C = concentration (mg/L) of analyte in spiking solution,
V = volume (mL) of sample used, and
s
V = volume (mL) of spiking solution added.
Evaluate the performance according to Practice D5847.
16.8 Method Precision—One unknown sample should be analyzed in triplicate with each batch to test method precision.
Calculate the standard deviation and use the F-test to compare with the single operator precision give in Tables 1-7 for the
equivalent analyte concentration and matrix type. Evaluate performance according to Practice D5847.
16.9 The laboratory may perform additional quality control as desired or appropriate.
D6508 − 15
TABLE 1 Precision, Bias, and Matrix Recovery for Chloride
No. of Mean Bias Versus Recovery Versus Interlab Interlab Single Operator Analyst
Matrix True Value
Values Result True Value True Value Std Dev S(t) %RSD Std Dev. S(o) %RSD
Reagent water 9 0.50 0.55 0.05 110.0 0.11 19.8
10 0.71 0.69 –0.02 97.2 0.08 11.5 0.05 7.5
10 2.00 1.97 –0.03 98.5 0.14 6.8
9 2.98 2.97 –0.01 99.7 0.11 3.8 0.05 2.1
10 14.92 14.76 –0.16 98.9 0.61 4.2
10 19.91 19.81 –0.10 99.5 0.81 4.1 0.48 2.8
10 39.81 38.58 –1.23 96.9 1.43 3.7
10 49.76 48.70 –1.06 97.9 1.94 4.0 1.36 3.1
Substitute wastewater 9 0.50 0.46 –0.04 92.0 0.51 111.1
9 0.71 0.43 –0.28 60.6 0.69 160.7 0.42 93.8
9 2.00 1.52 –0.48 76.0 0.68 45.0
9 2.98 2.58 –0.40 86.6 0.63 24.5 0.50 24.3
9 14.92 14.29 –0.63 95.8 1.02 7.1
9 19.91 18.93 –0.98 95.1 1.24 6.6 0.60 3.6
9 39.81 37.34 –2.47 93.8 5.44 14.6
9 49.76 47.54 –2.22 95.5 3.13 6.6 4.43 10.4
Drinking water 12 0.50 0.63 0.13 126.0 0.67 106.1
12 0.71 0.75 0.04 105.6 0.34 45.5 0.40 57.2
12 2.00 2.15 0.15 107.5 0.51 23.6
12 2.98 2.95 –0.03 99.0 0.39 13.1 0.47 18.5
12 14.92 14.54 –0.38 97.5 0.71 4.9
12 19.91 19.09 –0.82 95.9 1.11 5.8 0.37 2.2
12 39.81 38.38 –1.43 96.4 1.56 4.1
12 49.76 47.97 –1.79 96.4 2.19 4.6 1.26 3.9
9Real9 Wastewater 9 0.50 0.42 –0.08 84.0 0.34 81.0
10 0.71 0.47 –0.24 66.2 0.34 72.6 0.26 59.3
10 2.00 1.56 –0.44 78.0 0.51 32.7
9 2.98 2.78 –0.20 93.3 0.19 6.8 0.37 17.3
10 14.92 14.29 –0.63 95.8 0.63 4.4
10 19.91 18.83 –1.08 94.6 0.78 4.1 0.46 2.8
9 39.81 37.01 –2.80 93.0 2.78 7.5
10 49.76 48.24 –1.52 96.9 3.15 6.5 2.54 6.0
17. Precision and Bias
17.1 The precision and bias data presented in this test method meet the requirements of Practice D2777, and are given in Tables
1-7.
17.2 This test method interlaboratory collaborative was performed by 11 laboratories using one operator each. Four Youden-Pair
spike concentrations for the seven analytes anions yielding eight analyte concentration levels. Test data was submitted for eleven
reagent waters, eleven substitute wastewaters (Practice D5905), 15 drinking waters, and 13 wastewater sample matrices.
17.3 The precision, bias, and matrix recovery of this test method per anion analyte in four tested sample matrices are based upon
the analyte true value, calculated using weight, volume, and purity. True value spiking solution concentrations are given in Table
A1.4.
17.4 The bias and matrix recovery statements for less than 2 mg/L of chloride, sulfate, and nitrate in naturally occurring sample
matrices may be misleading due to spiking of small analyte concentration into a high naturally occurring analyte concentration
observed with the matrix blank. The commonly occurring analyte concentrations observed in the sample matrix blanks for the
naturally occurring tested matrices are given in Table A1.5.
17.5 The high nitrate bias and %recovery noted for the 0.5 mg/L NO spike solution are attributed to the spiking solution
containing 50 mg/L nitrite and 0.5 mg/L nitrate. Refer to Annex Table A1.4, Solution 3. Some of the nitrite converted to nitrate
prior to analysis. Similar NO conversion effect is observed with the 2-mg/L nitrate and 2 mg/L nitrite spike, Solution 7.
x
17.6 All collaborative participants used the premade chromate electrolyte. Ten laboratories used a Waters CIA Analyzer with
Millennium Data Processing Software, and one laboratory used a Agilent CE System with Diode Array Detector that provided
equivalent results.
NOTE 19—Refer to reference B1.16 and Agilent (the former HP company) website for recommended operating conditions.
Supporting data have been filed at ASTM International Headquarters and may be obtained by requesting Research Report RR:D19-1165. Contact ASTM Customer
Service at service@astm.org.
D6508 − 15
TABLE 2 Precision, Bias, and Matrix Recovery for Bromide
No. of Mean Bias Versus Recovery Versus Interlab Interlab Single Operator Analyst
Matrix True Value
Values Result True Value True Value Std Dev S(t) %RSD Std Dev. S(o) %RSD
Reagent water 10 0.51 0.60 0.09 117.6 0.19 31.0
10 0.70 0.83 0.13 118.6 0.23 28.2 0.10 14.6
10 2.00 2.06 0.06 103.0 0.14 6.6
10 3.01 2.88 –0.13 95.7 0.23 7.9 0.15 6.3
10 14.93 15.00 0.07 100.5 0.58 3.9
10 19.91 19.32 –0.59 97.0 0.97 5.0 0.75 4.4
10 39.81 39.66 –0.15 99.6 1.24 3.1
10 49.77 50.04 0.27 100.5 2.94 5.9 1.61 3.6
Substitute wastewater 9 0.51 0.67 0.16 131.4 0.19 28.8
9 0.70 0.96 0.26 137.1 0.21 21.8 0.08 9.3
9 2.00 2.14 0.14 107.0 0.22 10.2
9 3.01 2.72 –0.29 90.4 0.35 12.8 0.17 7.0
9 14.93 14.70 –0.23 98.5 0.58 3.9
9 19.91 18.91 –1.00 95.0 2.62 13.8 1.63 9.7
9 39.81 38.76 –1.05 97.4 1.11 2.9
9 49.77 48.81 –0.96 98.1 1.52 3.1 0.48 1.1
Drinking water 13 0.51 0.58 0.07 113.7 0.25 43.4
13 0.70 0.83 0.13 118.6 0.22 26.5 0.14 19.9
13 2.00 1.98 –0.02 99.0 0.25 12.5
13 3.01 2.56 –0.45 85.0 0.25 9.7 0.15 6.8
13 14.93 14.63 –0.30 98.0 0.50 3.4
13 19.91 19.22 –0.69 96.5 1.10 5.7 0.77 4.6
13 39.81 38.97 –0.84 97.9 1.99 5.1
13 49.77 48.74 –1.03 97.9 1.49 3.1 1.13 2.6
9Real9 Wastewater 11 0.51 0.59 0.08 115.7 0.11 19.3
12 0.70 0.78 0.08 111.4 0.19 24.4 0.10 14.0
11 2.00 2.08 0.08 104.0 0.13 6.3
12 3.01 2.70 –0.31 89.7 0.41 15.1 0.27 11.5
12 14.93 15.16 0.23 101.5 0.90 6.0
11 19.91 19.46 –0.45 97.7 1.63 8.4 1.09 6.3
12 39.81 40.24 0.43 101.1 2.27 5.7
12 49.77 49.97 0.20 100.4 2.52 5.0 0.91 2.0
17.7 Precision and bias for this test method conforms to Practice -98, – 98, which was in place at the time of collaborative
testing. Under the allowances made in 1.4 of Practice D2777-08, – 13, these precision and bias data do meet existing requirements
for interlaboratory studies of Committee D19 test methods.
18. Keywords
18.1 anion; bromide; capillary electrophoresis; chloride; drinking water; fluoride; ion analysis; nitrate; nitrite; orthophosphate;
reagent water; substitute wastewater; sulfate; wastewater
D6508 − 15
TABLE 3 Precision, Bias, and Matrix Recovery for Nitrite
No. of Mean Bias Versus Recovery Versus Interlab Interlab Single Operator Analyst
Matrix True Value
Values Result True Value True Value Std Dev S(t) %RSD Std Dev. S(o) %RSD
Reagent water 9 0.50 0.62 0.12 124.0 0.16 26.1
9 0.70 0.72 0.02 102.9 0.08 10.5 0.05 7.1
10 2.00 1.31 –0.69 65.5 0.25 19.2
10 2.98 3.11 0.13 104.4 0.17 5.4 0.
...

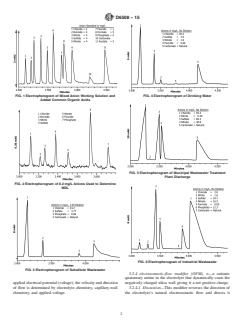



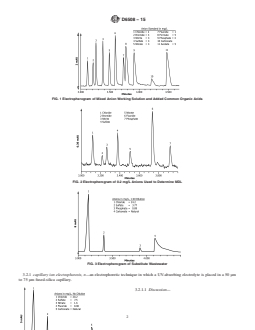
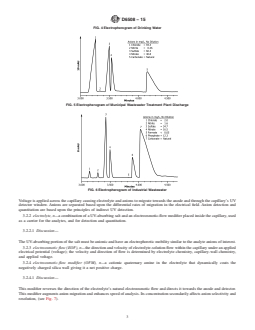

Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...