ASTM E3161-21
(Practice)Standard Practice for Preparing a Pseudomonas aeruginosa or Staphylococcus aureus Biofilm using the CDC Biofilm Reactor
Standard Practice for Preparing a <emph type="bdit">Pseudomonas aeruginosa</emph > or <emph type="bdit">Staphylococcus aureus</emph> Biofilm using the CDC Biofilm Reactor
SIGNIFICANCE AND USE
5.1 Bacteria that exist in biofilms are phenotypically different from suspended cells of the same genotype. Research has shown that biofilm bacteria are more difficult to kill than suspended bacteria (4, 5). Laboratory biofilms are engineered in growth reactors designed to produce a specific biofilm type. Altering system parameters will correspondingly result in a change in the biofilm. The purpose of this practice is to direct a user in the growth of a P. aeruginosa or S. aureus biofilm by clearly defining the operational parameters to grow a biofilm that can be assessed for efficacy using the Standard Test Method for Evaluating Disinfectant Efficacy Against Pseudomonas aeruginosa Biofilm Grown in CDC Biofilm Reactor Using Single Tube Method (E2871).
5.2 Operating the CDC Biofilm Reactor at the conditions specified in this method generates biofilm at log densities (log10 CFU per coupon) ranging from 8.0 to 9.5 for P. aeruginosa and 7.5 to 9.0 for S. aureus. These levels of biofilm are anticipated on surfaces conducive to biofilm formation such as the conditions outlined in this method.
5.2.1 To achieve an S. aureus biofilm with a population comparable to that for P. aeruginosa using the bacterial liquid growth medium conditions specified here, the S. aureus biofilm must be grown at 36 °C ±2 °C rather than at room temperature (21 °C ±2 °C).
SCOPE
1.1 This practice specifies the parameters for growing a Pseudomonas aeruginosa (ATCC 15442) or Staphylococcus aureus (ATCC 6538) biofilm that can be used for disinfectant efficacy testing using the Test Method for Evaluating Disinfectant Efficacy Against Pseudomonas aeruginosa Biofilm Grown in CDC Biofilm Reactor Using Single Tube Method (E2871) or in an alternate method capable of accommodating the coupons used in the CDC Biofilm Reactor. The resulting biofilm is representative of generalized situations where biofilm exist on hard, non-porous surfaces under shear rather than being representative of one particular environment. Additional bacteria may be grown using the basic procedure outlined in this document, however, alternative preparation procedures for frozen stock cultures and biofilm generation (for example, medium concentrations, baffle speed, temperature, incubation times, coupon types, etc.) may be necessary.
1.2 This practice uses the CDC Biofilm Reactor created by the Centers for Disease Control and Prevention (1).2 The CDC Biofilm Reactor is a continuously stirred tank reactor (CSTR) with high wall shear. The reactor is versatile and may also be used for growing or characterizing various species of biofilm, or both (2-4) provided appropriate adjustments are made to the growth media and operational parameters of the reactor.
1.3 Basic microbiology training is required to perform this practice.
1.4 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this practice.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Oct-2021
- Technical Committee
- E35 - Pesticides, Antimicrobials, and Alternative Control Agents
- Drafting Committee
- E35.15 - Antimicrobial Agents
Relations
- Effective Date
- 01-Nov-2019
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Oct-2013
- Effective Date
- 01-Apr-2012
- Effective Date
- 01-May-2010
Overview
ASTM E3161-21 is the international standard practice for preparing a Pseudomonas aeruginosa or Staphylococcus aureus biofilm using the CDC Biofilm Reactor. Developed by ASTM International, this guideline specifies the operational parameters for reliably growing laboratory biofilms on hard, non-porous surfaces under shear, using a continuously stirred tank reactor (CSTR) format. The resulting biofilms are suitable for testing disinfectant efficacy following standardized methods such as ASTM E2871 and support broader research into antimicrobial performance by providing reproducible, robust biofilm samples.
Biofilms formed by bacteria like P. aeruginosa and S. aureus are known for their resistance to antimicrobials. The CDC Biofilm Reactor method enables the generation of biofilms at densities comparable to those found in real-world environments, making it highly relevant for product testing, validation, and research.
Key Topics
Biofilm Characteristics
- Biofilms differ significantly from free-floating (planktonic) bacterial cells in phenotype and resistance.
- The standard supports the engineering of high-density biofilms, with P. aeruginosa and S. aureus reaching log10 CFU per coupon from approximately 7.5 to 9.5.
CDC Biofilm Reactor
- Utilizes a high-shear, well-mixed glass vessel with removable coupon carriers for sample collection.
- Operates in two phases: batch phase (initial colonization) and continuous flow phase (steady-state growth).
Operational Parameters
- Details the preparation of bacterial inocula, growth media concentrations, incubation timeframes, temperature controls, and rotational speeds of the baffled stir bar to modulate shear.
- Emphasizes sterility, cleaning, assembly, and calibration of laboratory equipment.
Customization
- While protocols focus on P. aeruginosa (ATCC 15442) and S. aureus (ATCC 6538), the method allows adaptation for additional bacterial species with adjusted media and reactor settings.
Requirements
- Users must have basic microbiological training, observe laboratory safety guidelines, and use SI units throughout testing.
Applications
The ASTM E3161-21 standard is widely applicable in:
Disinfectant Efficacy Testing
- Enables the consistent preparation of biofilm-laden surfaces for evaluating antimicrobial agents, in accordance with ASTM E2871 and related methods.
- Provides a robust basis for regulatory submissions and product validation in healthcare, water treatment, and public health sectors.
Research and Development
- Supports scientific studies on biofilm formation, persistence, and resistance mechanisms.
- Facilitates the comparison of new technologies against established control methods.
Quality Control
- Offers a reproducible protocol for laboratories involved in routine product performance verification.
Training and Standardization
- Enhances skills and methodological consistency among laboratory personnel working with biofilms.
Related Standards
- ASTM E2871 – Test Method for Evaluating Disinfectant Efficacy Against Pseudomonas aeruginosa Biofilm Grown in CDC Biofilm Reactor Using Single Tube Method.
- ASTM E2756 – Terminology Relating to Antimicrobial and Antiviral Agents.
- Other referenced protocols – Procedures for equipment cleaning, sterilization, and calibration as outlined in sector-relevant guidelines.
This standard aligns with international principles set by the World Trade Organization (WTO) for the development of harmonized technical regulations, ensuring global relevance and acceptance.
Keywords: ASTM E3161-21, CDC Biofilm Reactor, biofilm formation, Pseudomonas aeruginosa, Staphylococcus aureus, antimicrobial resistance, disinfectant efficacy, laboratory protocol, biofilm testing, standard practice.
Buy Documents
ASTM E3161-21 - Standard Practice for Preparing a <emph type="bdit">Pseudomonas aeruginosa</emph > or <emph type="bdit">Staphylococcus aureus</emph> Biofilm using the CDC Biofilm Reactor
REDLINE ASTM E3161-21 - Standard Practice for Preparing a <emph type="bdit">Pseudomonas aeruginosa</emph > or <emph type="bdit">Staphylococcus aureus</emph> Biofilm using the CDC Biofilm Reactor
Frequently Asked Questions
ASTM E3161-21 is a standard published by ASTM International. Its full title is "Standard Practice for Preparing a <emph type="bdit">Pseudomonas aeruginosa</emph > or <emph type="bdit">Staphylococcus aureus</emph> Biofilm using the CDC Biofilm Reactor". This standard covers: SIGNIFICANCE AND USE 5.1 Bacteria that exist in biofilms are phenotypically different from suspended cells of the same genotype. Research has shown that biofilm bacteria are more difficult to kill than suspended bacteria (4, 5). Laboratory biofilms are engineered in growth reactors designed to produce a specific biofilm type. Altering system parameters will correspondingly result in a change in the biofilm. The purpose of this practice is to direct a user in the growth of a P. aeruginosa or S. aureus biofilm by clearly defining the operational parameters to grow a biofilm that can be assessed for efficacy using the Standard Test Method for Evaluating Disinfectant Efficacy Against Pseudomonas aeruginosa Biofilm Grown in CDC Biofilm Reactor Using Single Tube Method (E2871). 5.2 Operating the CDC Biofilm Reactor at the conditions specified in this method generates biofilm at log densities (log10 CFU per coupon) ranging from 8.0 to 9.5 for P. aeruginosa and 7.5 to 9.0 for S. aureus. These levels of biofilm are anticipated on surfaces conducive to biofilm formation such as the conditions outlined in this method. 5.2.1 To achieve an S. aureus biofilm with a population comparable to that for P. aeruginosa using the bacterial liquid growth medium conditions specified here, the S. aureus biofilm must be grown at 36 °C ±2 °C rather than at room temperature (21 °C ±2 °C). SCOPE 1.1 This practice specifies the parameters for growing a Pseudomonas aeruginosa (ATCC 15442) or Staphylococcus aureus (ATCC 6538) biofilm that can be used for disinfectant efficacy testing using the Test Method for Evaluating Disinfectant Efficacy Against Pseudomonas aeruginosa Biofilm Grown in CDC Biofilm Reactor Using Single Tube Method (E2871) or in an alternate method capable of accommodating the coupons used in the CDC Biofilm Reactor. The resulting biofilm is representative of generalized situations where biofilm exist on hard, non-porous surfaces under shear rather than being representative of one particular environment. Additional bacteria may be grown using the basic procedure outlined in this document, however, alternative preparation procedures for frozen stock cultures and biofilm generation (for example, medium concentrations, baffle speed, temperature, incubation times, coupon types, etc.) may be necessary. 1.2 This practice uses the CDC Biofilm Reactor created by the Centers for Disease Control and Prevention (1).2 The CDC Biofilm Reactor is a continuously stirred tank reactor (CSTR) with high wall shear. The reactor is versatile and may also be used for growing or characterizing various species of biofilm, or both (2-4) provided appropriate adjustments are made to the growth media and operational parameters of the reactor. 1.3 Basic microbiology training is required to perform this practice. 1.4 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this practice. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Bacteria that exist in biofilms are phenotypically different from suspended cells of the same genotype. Research has shown that biofilm bacteria are more difficult to kill than suspended bacteria (4, 5). Laboratory biofilms are engineered in growth reactors designed to produce a specific biofilm type. Altering system parameters will correspondingly result in a change in the biofilm. The purpose of this practice is to direct a user in the growth of a P. aeruginosa or S. aureus biofilm by clearly defining the operational parameters to grow a biofilm that can be assessed for efficacy using the Standard Test Method for Evaluating Disinfectant Efficacy Against Pseudomonas aeruginosa Biofilm Grown in CDC Biofilm Reactor Using Single Tube Method (E2871). 5.2 Operating the CDC Biofilm Reactor at the conditions specified in this method generates biofilm at log densities (log10 CFU per coupon) ranging from 8.0 to 9.5 for P. aeruginosa and 7.5 to 9.0 for S. aureus. These levels of biofilm are anticipated on surfaces conducive to biofilm formation such as the conditions outlined in this method. 5.2.1 To achieve an S. aureus biofilm with a population comparable to that for P. aeruginosa using the bacterial liquid growth medium conditions specified here, the S. aureus biofilm must be grown at 36 °C ±2 °C rather than at room temperature (21 °C ±2 °C). SCOPE 1.1 This practice specifies the parameters for growing a Pseudomonas aeruginosa (ATCC 15442) or Staphylococcus aureus (ATCC 6538) biofilm that can be used for disinfectant efficacy testing using the Test Method for Evaluating Disinfectant Efficacy Against Pseudomonas aeruginosa Biofilm Grown in CDC Biofilm Reactor Using Single Tube Method (E2871) or in an alternate method capable of accommodating the coupons used in the CDC Biofilm Reactor. The resulting biofilm is representative of generalized situations where biofilm exist on hard, non-porous surfaces under shear rather than being representative of one particular environment. Additional bacteria may be grown using the basic procedure outlined in this document, however, alternative preparation procedures for frozen stock cultures and biofilm generation (for example, medium concentrations, baffle speed, temperature, incubation times, coupon types, etc.) may be necessary. 1.2 This practice uses the CDC Biofilm Reactor created by the Centers for Disease Control and Prevention (1).2 The CDC Biofilm Reactor is a continuously stirred tank reactor (CSTR) with high wall shear. The reactor is versatile and may also be used for growing or characterizing various species of biofilm, or both (2-4) provided appropriate adjustments are made to the growth media and operational parameters of the reactor. 1.3 Basic microbiology training is required to perform this practice. 1.4 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this practice. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E3161-21 is classified under the following ICS (International Classification for Standards) categories: 07.100.01 - Microbiology in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E3161-21 has the following relationships with other standards: It is inter standard links to ASTM E2756-19, ASTM E2756-18, ASTM E2871-13, ASTM E2871-12, ASTM E2756-10. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E3161-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:E3161 −21
Standard Practice for
Preparing a Pseudomonas aeruginosa or Staphylococcus
aureus Biofilm using the CDC Biofilm Reactor
This standard is issued under the fixed designation E3161; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
1.1 This practice specifies the parameters for growing a
1.6 This international standard was developed in accor-
Pseudomonas aeruginosa (ATCC 15442) or Staphylococcus
dance with internationally recognized principles on standard-
aureus (ATCC 6538) biofilm that can be used for disinfectant
ization established in the Decision on Principles for the
efficacy testing using theTest Method for Evaluating Disinfec-
Development of International Standards, Guides and Recom-
tantEfficacyAgainst Pseudomonas aeruginosaBiofilmGrown
mendations issued by the World Trade Organization Technical
inCDCBiofilmReactorUsingSingleTubeMethod(E2871)or
Barriers to Trade (TBT) Committee.
in an alternate method capable of accommodating the coupons
used in the CDC Biofilm Reactor. The resulting biofilm is
2. Referenced Documents
representative of generalized situations where biofilm exist on
2.1 ASTM Standards:
hard, non-porous surfaces under shear rather than being repre-
E2756Terminology Relating toAntimicrobial andAntiviral
sentative of one particular environment. Additional bacteria
Agents
may be grown using the basic procedure outlined in this
E2871Test Method for Determining Disinfectant Efficacy
document, however, alternative preparation procedures for
Against Biofilm Grown in the CDC Biofilm Reactor
frozen stock cultures and biofilm generation (for example,
Using the Single Tube Method
medium concentrations, baffle speed, temperature, incubation
times, coupon types, etc.) may be necessary.
3. Terminology
1.2 This practice uses the CDC Biofilm Reactor created by 3.1 Definitions:
the Centers for Disease Control and Prevention (1). The CDC 3.1.1 For definition of terms used in this method refer to
Biofilm Reactor is a continuously stirred tank reactor (CSTR) Terminology E2756.
with high wall shear. The reactor is versatile and may also be 3.1.2 batch phase, n—establishment of the biofilm by oper-
ating the reactor without the flow of nutrients (batch phase
used for growing or characterizing various species of biofilm,
orboth (2-4)providedappropriateadjustmentsaremadetothe growth medium), but with mixing.
growth media and operational parameters of the reactor.
3.1.3 biofilm, n—microorganisms living in a self-organized
community attached to surfaces, interfaces, or each other,
1.3 Basic microbiology training is required to perform this
embedded in a matrix of extracellular polymeric substances of
practice.
microbial origin, while exhibiting altered phenotypes with
1.4 Units—The values stated in SI units are to be regarded
respect to growth rate and gene transcription.
asstandard.Nootherunitsofmeasurementareincludedinthis
3.1.4 continuously stirred tank reactor (CSTR) phase,
practice.
n—establishment of a steady state biofilm population achieved
1.5 This standard does not purport to address all of the
with the continuous flow of nutrients (continuous flow growth
safety concerns, if any, associated with its use. It is the
medium) in a glass vessel.
responsibility of the user of this standard to establish appro-
3.1.5 coupon, n—biofilm sample surface.
4. Summary of Practice
This practice is under the jurisdiction ofASTM Committee E35 on Pesticides,
4.1 This practice is used for growing a P. aeruginosa or S.
Antimicrobials, and Alternative Control Agents and is the direct responsibility of
aureus biofilm in the CDC Biofilm Reactor. The biofilm is
Subcommittee E35.15 on Antimicrobial Agents.
Current edition approved Nov. 1, 2021. Published January 2022. Originally
approved in 2018. Last previous edition approved in 2018 as E3161–18. DOI: For referenced ASTM standards, visit the ASTM website, www.astm.org, or
10.1520/E3161–21. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Theboldfacenumbersinparenthesesrefertothelistofreferencesattheendof Standards volume information, refer to the standard’s Document Summary page on
this standard. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E3161−21
established by operating the reactor in batch phase (no flow of 6.8 Digital Magnetic Stir Plate—top plate of at least
the nutrients) for 24 h. A steady state population is reached 10.16cm by 10.16 cm that can rotate at a range of 60r⁄min to
while the reactor operates for an additional 24 h with continu- 125 r/min 65 r/min.
ousflowofthenutrients.Theresidencetimeofthenutrientsin
6.9 Silicone Tubing—multiple sizes: size 16 tubing or
thereactorissettoselectforbiofilmgrowth,andisspeciesand
equivalentdesignedforuseinaperistalticpump(usedformost
reactor parameter specific. During the entire 48 h, the biofilm
connections between CSTR growth medium carboy and the
is exposed to continuous fluid shear from the rotation of a
reactor), and size 18 or 25 tubing or equivalent (used for
baffled stir bar. Controlling the rate at which the baffle turns
reactor effluent). All sizes must withstand sterilization (for
determines the intensity of the shear stress to which the
example, platinum cured).
coupons are exposed.At the end of the 48 h, the biofilm-laden
6.10 Norprene Tubing (or equivalent)—size 16 or equiva-
coupons are used for testing.
lent Norprene tubing. Recommended for use in the peristaltic
pump.
5. Significance and Use
6.11 Glass Flow Break—any that will connect with size 16
5.1 Bacteria that exist in biofilms are phenotypically differ-
tubing and withstand sterilization, used to prevent microbial
ent from suspended cells of the same genotype. Research has
contamination of the nutrient reservoir from the biofilm reac-
shown that biofilm bacteria are more difficult to kill than
tor.
suspended bacteria (4, 5). Laboratory biofilms are engineered
6.11.1 Clamp—used to hold flow break, extension clamp
in growth reactors designed to produce a specific biofilm type.
with 0.5 cm minimum grip size.
Altering system parameters will correspondingly result in a
change in the biofilm. The purpose of this practice is to direct 6.11.2 Clamp Stand—heightnolessthan76.2cm,usedwith
a user in the growth of a P. aeruginosa or S. aureus biofilm by clamp to suspend glass flow break vertically and stabilize
clearly defining the operational parameters to grow a biofilm tubing above reactor.
that can be assessed for efficacy using the Standard Test
6.12 Reactor Components.
Method for Evaluating Disinfectant Efficacy Against
6.12.1 Berzelius Borosilicate Glass Tall Beaker—1000 mL
Pseudomonas aeruginosa Biofilm Grown in CDC Biofilm
without pour spout, 9.5cm 60.5 cm diameter. Barbed outlet
Reactor Using Single Tube Method (E2871).
spoutaddedat400mL 650mLmark.Spoutangled30°to45°
5.2 Operating the CDC Biofilm Reactor at the conditions
to ensure drainage. Spout should accommodate size 18 or 25
specified in this method generates biofilm at log densities
flexible silicone tubing.
(log CFU per coupon) ranging from 8.0 to 9.5 for P.
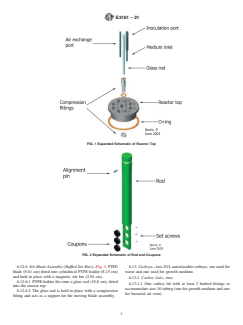
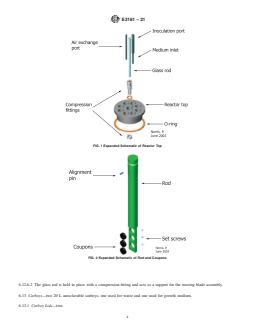
10 6.12.2 Reactor Top—Fig. 1. Ultra-high molecular weight
aeruginosaand7.5to9.0for S. aureus.Theselevelsofbiofilm
(UHMW) polyethylene top (10.1 cm diameter tapering to 8.33
areanticipatedonsurfacesconducivetobiofilmformationsuch
cm) equipped with a minimum of 3 holes accommodating 6 to
as the conditions outlined in this method.
8 cm long pieces of stainless steel or other rigid autoclavable
5.2.1 To achieve an S. aureus biofilm with a population tubing with outside diameter of 5mm to 8 mm for medium
comparable to that for P. aeruginosa using the bacterial liquid inlet, air exchange and inoculation port. Center hole, 1.27 cm
growthmediumconditionsspecifiedhere,the S. aureusbiofilm diameter, to accommodate the glass rod used to support the
mustbegrownat36°C 62°Cratherthanatroomtemperature baffleassembly.Eightrodholes,1.905cmdiameter,notchedto
(21°C 62°C). accommodate stainless steel rod alignment pin (0.236 cm
outside diameter). O-ring, attached to underside of reactor top.
6. Apparatus
6.12.3 Polypropylene Rods—Fig. 2. Eight polypropylene
rods, 21.08 cm long, two types: coupon holder machined to
6.1 Culture Tubes and Culture Tube Closures—any glass or
holdthreecoupons(see6.12.4)attheimmersedend,three316
plastic tube with a volume capacity of at least 15 mL.
stainless steel set screws embedded in side to hold coupons in
6.2 Calibrated Pipetter—continuously adjustable pipetter place; and coupon holder blanks, without coupon recesses.
with volume capability of 1 mL. Rods fit into holes in reactor top and lock into preformed
notches with alignment pin.
6.3 Vortex—any vortex that will ensure proper agitation and
6.12.4 Coupons—twenty-four cylindrical coupons (for
mixing of culture tubes.
example, borosilicate glass) with a diameter of 1.27cm
6.4 Ultrasonic Water Bath—any cavitating sonicating bath
60.013 cm, thickness of approximately 3.0 mm.
that operates at 45 65 kHz and which has a volume large
6.12.5 Small Allen Wrench (1.27 mm, hex)—for adjusting
enough to accommodate 50 mL or 250 mL conical tubes.
set screws.
6.5 Analytical Balance—sensitive to 0.01 g.
6.6 Sterilizer—any steam sterilizer that can produce the
The sole source of supply of the apparatus (CDC Biofilm Reactor) and
conditions of sterilization is acceptable.
associatedcouponsknowntothecommitteeatthistimeisBioSurfaceTechnologies,
Corp. www.biofilms.biz. If you are aware of alternative suppliers, please provide
6.7 Peristaltic Pump—pump head that can hold size 16 or
this information toASTM International Headquarters. Your comments will receive
equivalent peristaltic pump tubing. Use a separate pump for
careful consideration at a meeting of the responsible technical committee, which
each reactor. you may attend. The user may also build the reactor.
E3161−21
FIG. 1Expanded Schematic of Reactor Top
FIG. 2Expanded Schematic of Rod and Coupons
6.12.6 Stir Blade Assembly (Baffled Stir Bar)—Fig.3.PTFE 6.13 Carboys—two20Lautoclavablecarboys,oneusedfor
blade (5.61 cm) fitted into cylindrical PTFE holder (8.13 cm)
waste and one used for growth medium.
and held in place with a magnetic stir bar (2.54 cm).
6.13.1 Carboy Lids—two.
6.12.6.1 PTFE holder fits onto a glass rod (15.8 cm), fitted
6.13.1.1 One carboy lid with at least 2 barbed fittings to
into the reactor top.
accommodate size 16 tubing (one for growth medium and one
6.12.6.2 The glass rod is held in place with a compression
for bacterial air vent).
fitting and acts as a support for the moving blade assembly.
E3161−21
FIG. 3Expanded Schematic of Baffled Stir Bar
6.13.1.2 One carboy lid with at least two 1 cm holes bored 8.1.2 Check each coupon under 20× magnification for
inthesamefashion(oneforeffluentwasteandoneforbacterial scratching, chipping, other damage, or accumulated debris
air vent). before each use. Discard those with visible damage to surface
topography.
NOTE 1—Carboy lids can be purchased with fittings.
8.1.3 For initial use and re-use, sonicate coupons in indi-
6.13.2 Bacterial Air Vent (Filter)—autoclavable0.2µmpore
vidual tubes or welled plates for approximately 5 min in
size,tobesplicedintotubingonwastecarboy,growthmedium
detergent diluted per the manufacturer’s instructions. The
carboy and reactor top, recommended diameter 37 mm.
soapy water must completely cover the coupons.
6.14 Fig. 4 illustrates a schematic of the assembled system.
NOTE 2—Process coupons individually to minimize the risk of damage
6.15 Detergent—laboratory detergent for cleaning coupons
to the coupons.
and reactor parts.
8.1.4 Rinse coupons with reagent grade water and sonicate
for approximately 1 min in reagent grade water.
7. Reagents and Materials
8.1.5 Repeat rinsing and sonication with reagent grade
7.1 Purity of Water—All references to water as diluent or
water until no soap is left on the coupons, as demonstrated by
reagent shall mean de-ionized water or water of equal purity.
alackofvisiblesuds.Oncethecouponsareclean,weargloves
7.2 Culture Media: to prevent oils and other residue from contaminating the
7.2.1 Cryoprotectant—Tryptic Soy Broth (30 g/L) with surface. Store screened and cleaned coupons in a Petri dish.
15% (v/v) glycerol.
NOTE3—Couponsmaybemadeoutofalternativematerials.Adjustthe
7.2.2 Bacterial Liquid Growth Medium—Tryptic Soy Broth
cleaning procedure so that it is appropriate for the coupon material being
(TSB).
used.
7.2.2.1 For P. aeruginosa, use 300 mg/L TSB for the
8.2 Preparation of Reactor Top:
inoculum and batch phase reactor operations, and 100 mg/L
8.2.1 Invert the reactor top and place baffled stir bar onto
TSB for the continuous flow reactor operation.
glass rod positioned in the center of the reactor top.
7.2.2.2 For S. aureus, use 30 g/L TSB for the inoculum, 3
8.2.2 Invertthereactorbeakerandplaceontotheassembled
g/LTSB for the batch phase reactor operation, and 1 g/LTSB
top.Turnthereactoroversothatthereactortopisupright.The
for the continuous flow reactor operation.
baffled stir bar is designed to allow it to rotate freely.
7.2.3 Growth Medium for Stock Culture Generation—
Trypticase Soy Agar (TSA). 8.2.3 Placeacleanedandscreenedcouponintoeachholein
the reactor rods, leaving the top of the coupon flush with the
8. Preparation of Apparatus
inside rod surface. Tighten set screw. If less than 24 coupons
are required for testing, substitute one coupon holder blank for
8.1 Preparation of Borosilicate Glass Coupons:
each polyproplyene rod holding three (3) coupons.
8.1.1 Couponsmaybeusedrepeatedlywithpropercleaning
8.2.4 Place rods into reactor top loosely (not yet fitted into
and screening between each use.After use in the reactor, place
notches).
contaminated coupons in an appropriate vessel, cover with
liquid (e.g., water), and, along with the other parts of the 8.2.5 Connect the bacterial air vent by fitting the vent to a
contaminatedreactorsystem,autoclaveat121°Cfor30minor smallsectionofappropriatelysizedtubingandattachtooneof
using other parameters that ensure sterilization. the rigid tubes on the reactor top.
E3161−21
FIG. 4Schematic of the Completely Assembled Reactor System
8.2.6 Splice the glass flow break into the growth medium reactor,and RT=residencetime.Forexample:iftheoperating
tubing line near the reactor top. volume equals 325 mL and the residence time equals 30 min,
then the pump flow rate should be set to equal 10.8 mL/min.
9. Calibration and Standardization
9.2 Periodic pump calibration: Follow manufacturer’s in-
9.1 Co
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E3161 − 18 E3161 − 21
Standard Practice for
Preparing a Pseudomonas aeruginosa or Staphylococcus
aureus Biofilm using the CDC Biofilm Reactor
This standard is issued under the fixed designation E3161; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Scope*
1.1 This practice specifies the parameters for growing a Pseudomonas aeruginosa (ATCC 15442) or Staphylococcus aureus
(ATCC 6538) biofilm that can be used for disinfectant efficacy testing using the Test Method for Evaluating Disinfectant Efficacy
Against Pseudomonas aeruginosa Biofilm Grown in CDC Biofilm Reactor Using Single Tube Method (E2871) or in an alternate
method capable of accommodating the coupons used in the CDC Biofilm Reactor. The resulting biofilm is representative of
generalized situations where biofilm exist on hard, non-porous surfaces under shear rather than being representative of one
particular environment. Additional bacteria may be grown using the basic procedure outlined in this document, however,
alternative preparation procedures for frozen stock cultures and biofilm generation (for example, medium concentrations, baffle
speed, temperature, incubation times, coupon types, etc.) may be necessary.
1.2 This practice uses the CDC Biofilm Reactor created by the Centers for Disease Control and Prevention (1). The CDC Biofilm
Reactor is a continuously stirred tank reactor (CSTR) with high wall shear. The reactor is versatile and may also be used for
growing or characterizing various species of biofilm, or both (2-4) provided appropriate adjustments are made to the growth media
and operational parameters of the reactor.
1.3 Basic microbiology training is required to perform this practice.
1.4 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this practice.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E2756 Terminology Relating to Antimicrobial and Antiviral Agents
This practice is under the jurisdiction of ASTM Committee E35 on Pesticides, Antimicrobials, and Alternative Control Agents and is the direct responsibility of
Subcommittee E35.15 on Antimicrobial Agents.
Current edition approved April 1, 2018Nov. 1, 2021. Published June 2018January 2022. Originally approved in 2018. Last previous edition approved in 2018 as E3161–18.
DOI: 10.1520/E3161–18.10.1520/E3161–21.
The boldface numbers in parentheses refer to the list of references at the end of this standard.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E3161 − 21
E2871 Test Method for Determining Disinfectant Efficacy Against Biofilm Grown in the CDC Biofilm Reactor Using the Single
Tube Method
3. Terminology
3.1 Definitions:
3.1.1 For definition of terms used in this method refer to Terminology E2756.
3.1.2 batch phase, n—establishment of the biofilm by operating the reactor without the flow of nutrients (batch phase growth
medium), but with mixing.
3.1.3 biofilm, n—microorganisms living in a self-organized community attached to surfaces, interfaces, or each other, embedded
in a matrix of extracellular polymeric substances of microbial origin, while exhibiting altered phenotypes with respect to growth
rate and gene transcription.
3.1.4 continuously stirred tank reactor (CSTR) phase, n—establishment of a steady state biofilm population achieved with the
continuous flow of nutrients (continuous flow growth medium) in a glass vessel.
3.1.5 coupon, n—biofilm sample surface.
4. Summary of Practice
4.1 This practice is used for growing a P. aeruginosa or S. aureus biofilm in the CDC Biofilm Reactor. The biofilm is established
by operating the reactor in batch phase (no flow of the nutrients) for 24 h. A steady state population is reached while the reactor
operates for an additional 24 h with continuous flow of the nutrients. The residence time of the nutrients in the reactor is set to
select for biofilm growth, and is species and reactor parameter specific. During the entire 48 h, the biofilm is exposed to continuous
fluid shear from the rotation of a baffled stir bar. Controlling the rate at which the baffle turns determines the intensity of the shear
stress to which the coupons are exposed. At the end of the 48 h, the biofilm-laden coupons are used for testing.
5. Significance and Use
5.1 Bacteria that exist in biofilms are phenotypically different from suspended cells of the same genotype. Research has shown
that biofilm bacteria are more difficult to kill than suspended bacteria (4, 5). Laboratory biofilms are engineered in growth reactors
designed to produce a specific biofilm type. Altering system parameters will correspondingly result in a change in the biofilm. The
purpose of this practice is to direct a user in the growth of a P. aeruginosa or S. aureus biofilm by clearly defining the operational
parameters to grow a biofilm that can be assessed for efficacy using the Standard Test Method for Evaluating Disinfectant Efficacy
Against Pseudomonas aeruginosa Biofilm Grown in CDC Biofilm Reactor Using Single Tube Method (E2871).
5.2 Operating the CDC Biofilm Reactor at the conditions specified in this method generates biofilm at log densities (log CFU
per coupon) ranging from 8.0 to 9.5 for P. aeruginosa and 7.5 to 9.0 for S. aureus. These levels of biofilm are anticipated on
surfaces conducive to biofilm formation such as the conditions outlined in this method.
5.2.1 To achieve an S. aureus biofilm with a population comparable to that for P. aeruginosa using the bacterial liquid growth
medium conditions specified here, the S. aureus biofilm must be grown at 3636 °C 62 °C rather than at room temperature
(21(21 °C 62 °C).
6. Apparatus
6.1 Culture Tubes and Culture Tube Closures—any glass or plastic tube with a volume capacity of at least 15 mL.
6.2 Calibrated Pipetter—continuously adjustable pipetter with volume capability of 1 mL.
6.3 Vortex—any vortex that will ensure proper agitation and mixing of culture tubes.
6.4 Ultrasonic Water Bath—any cavitating sonicating bath that operates at 45 65 kHz and which has a volume large enough to
accommodate 50 mL or 250 mL conical tubes.
E3161 − 21
6.5 Analytical Balance—sensitive to 0.01 g.
6.6 Sterilizer—any steam sterilizer that can produce the conditions of sterilization is acceptable.
6.7 Peristaltic Pump—pump head that can hold size 16 or equivalent peristaltic pump tubing. Use a separate pump for each
reactor.
6.8 Digital Magnetic Stir Plate—top plate of at least 10.1610.16 cm by 10.16 cm that can rotate at a range of 6060 r ⁄min to 125
r/min 65 r/min.
6.9 Silicone Tubing—multiple sizes: size 16 tubing or equivalent designed for use in a peristaltic pump (used for most connections
between CSTR growth medium carboy and the reactor), and size 18 or 25 tubing or equivalent (used for reactor effluent). All sizes
must withstand sterilization (for example, platinum cured).
6.10 Norprene Tubing (or equivalent)—size 16 or equivalent Norprene tubing. Recommended for use in the peristaltic pump.
6.11 Glass Flow Break—any that will connect with size 16 tubing and withstand sterilization, used to prevent microbial
contamination of the nutrient reservoir from the biofilm reactor.
6.11.1 Clamp—used to hold flow break, extension clamp with 0.5 cm minimum grip size.
6.11.2 Clamp Stand—height no less than 76.2 cm, used with clamp to suspend glass flow break vertically and stabilize tubing
above reactor.
6.12 Reactor Components.
6.12.1 Berzelius Borosilicate Glass Tall Beaker—1000 mL without pour spout, 9.59.5 cm 60.5 cm diameter. Barbed outlet spout
added at 400400 mL 650 mL mark. Spout angled 30° to 45° to ensure drainage. Spout should accommodate size 18 or 25 flexible
silicone tubing.
6.12.2 Reactor Top—Fig. 1. Ultra-high molecular weight (UHMW) polyethylene top (10.1 cm diameter tapering to 8.33 cm)
equipped with a minimum of 3 holes accommodating 6 to 8 cm long pieces of stainless steel or other rigid autoclavable tubing
with outside diameter of 55 mm to 8 mm for medium inlet, air exchange and inoculation port. Center hole, 1.27 cm diameter, to
accommodate the glass rod used to support the baffle assembly. Eight rod holes, 1.905 cm diameter, notched to accommodate
stainless steel rod alignment pin (0.236 cm outside diameter). O-ring, attached to underside of reactor top.
6.12.3 Polypropylene Rods—Fig. 2. Eight polypropylene rods, 21.08 cm long, two types: coupon holder machined to hold three
coupons (see 6.12.4) at the immersed end, three 316 stainless steel set screws embedded in side to hold coupons in place; and
coupon holder blanks, without coupon recesses. Rods fit into holes in reactor top and lock into preformed notches with alignment
pin.
6.12.4 Coupons—twenty-four cylindrical coupons (for example, borosilicate glass) with a diameter of 1.271.27 cm 60.013 cm,
thickness of approximately 3.0 mm.
6.12.5 Small Allen Wrench (1.27 mm, hex)—for adjusting set screws.
6.12.6 Stir Blade Assembly (Baffled Stir Bar)—Fig. 3. PTFE blade (5.61 cm) fitted into cylindrical PTFE holder (8.13 cm) and held
in place with a magnetic stir bar (2.54 cm).
6.12.6.1 PTFE holder fits onto a glass rod (15.8 cm), fitted into the reactor top.
The sole source of supply of the apparatus (CDC Biofilm Reactor) and associated coupons known to the committee at this time is BioSurface Technologies, Corp.
www.biofilms.biz. If you are aware of alternative suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful
consideration at a meeting of the responsible technical committee, which you may attend. The user may also build the reactor.
E3161 − 21
FIG. 1 Expanded Schematic of Reactor Top
FIG. 2 Expanded Schematic of Rod and Coupons
6.12.6.2 The glass rod is held in place with a compression fitting and acts as a support for the moving blade assembly.
6.13 Carboys—two 20 L autoclavable carboys, one used for waste and one used for growth medium.
6.13.1 Carboy Lids—two.
E3161 − 21
FIG. 3 Expanded Schematic of Baffled Stir Bar
6.13.1.1 One carboy lid with at least 2 barbed fittings to accommodate size 16 tubing (one for growth medium and one for bacterial
air vent).
6.13.1.2 One carboy lid with at least two 1 cm holes bored in the same fashion (one for effluent waste and one for bacterial air
vent).
NOTE 1—Carboy lids can be purchased with fittings.
6.13.2 Bacterial Air Vent (Filter)—autoclavable 0.2 μm pore size, to be spliced into tubing on waste carboy, growth medium
carboy and reactor top, recommended diameter 37 mm.
6.14 Fig. 4 illustrates a schematic of the assembled system.
6.15 Detergent—laboratory detergent for cleaning coupons and reactor parts.
7. Reagents and Materials
7.1 Purity of Water—All references to water as diluent or reagent shall mean de-ionized water or water of equal purity.
7.2 Culture Media:
7.2.1 Cryoprotectant—Tryptic Soy Broth (30 g/L) with 15 % (v/v) glycerol.
7.2.2 Bacterial Liquid Growth Medium—Tryptic Soy Broth (TSB).
7.2.2.1 For P. aeruginosa, use 300 mg/L TSB for the inoculum and batch phase reactor operations, and 100 mg/L TSB for the
continuous flow reactor operation.
7.2.2.2 For S. aureus, use 30 g/L TSB for the inoculum, 3 g/L TSB for the batch phase reactor operation, and 1 g/L TSB for the
continuous flow reactor operation.
7.2.3 Growth Medium for Stock Culture Generation—Trypticase Soy Agar (TSA).
8. Preparation of Apparatus
8.1 Preparation of Borosilicate Glass Coupons:
E3161 − 21
FIG. 4 Schematic of the Completely Assembled Reactor System
8.1.1 Coupons may be used repeatedly with proper cleaning and screening between each use. After use in the reactor, place
contaminated coupons in an appropriate vessel, cover with liquid (e.g., water), and, along with the other parts of the contaminated
reactor system, autoclave at 121°C for 30 min or using other parameters that ensure sterilization.
8.1.2 Check each coupon under 20× magnification for scratching, chipping, other damage, or accumulated debris before each use.
Discard those with visible damage to surface topography.
8.1.3 For initial use and re-use, sonicate coupons in individual tubes or welled plates for approximately 5 min in detergent diluted
per the manufacturer’s instructions. The soapy water must completely cover the coupons.
NOTE 2—Process coupons individually to minimize the risk of damage to the coupons.
8.1.4 Rinse coupons with reagent grade water and sonicate for approximately 1 min in reagent grade water.
8.1.5 Repeat rinsing and sonication with reagent grade water until no soap is left on the coupons, as demonstrated by a lack of
visible suds. Once the coupons are clean, wear gloves to prevent oils and other residue from contaminating the surface. Store
screened and cleaned coupons in a Petri dish.
NOTE 3—Coupons may be made out of alternative materials. Adjust the cleaning procedure so that it is appropriate for the coupon material being used.
8.2 Preparation of Reactor Top:
E3161 − 21
8.2.1 Invert the reactor top and place baffled stir bar onto glass rod positioned in the center of the reactor top.
8.2.2 Invert the reactor beaker and place onto the assembled top. Turn the reactor over so that the reactor top is upright. The baffled
stir bar is designed to allow it to rotate freely.
8.2.3 Place a cleaned and screened coupon into each hole in the reactor rods, leaving the top of the coupon flush with the inside
rod surface. Tighten set screw. If less than 24 coupons are required for testing, substitute one coupon holder blank for each
polyproplyene rod holding three (3) coupons.
8.2.4 Place rods into reactor top loosely (not yet fitted into notches).
8.2.5 Connect the bacterial air vent by fitting the vent to a small section of appropriately sized tubing and attach to one of the rigid
tubes on the reactor top.
8.2.6 Splice the glass flow break into the growth medium tubing line near the reactor top.
9. Calibration and Standardization
9.1 Confirm the operating volume of each reactor (that is, new Berzelius beaker with spout) prior to initial use.
9.1.1 Fully assemble the reactor (including rods with coupons and baffle apparatus) and place on a stir plate set to the appropriate
speed (f
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...