ASTM F3212-16(2023)
(Test Method)Standard Test Method for Coring Testing of Huber Needles
Standard Test Method for Coring Testing of Huber Needles
SIGNIFICANCE AND USE
5.1 This test method determines whether Huber needles are designed and manufactured such that they will not produce a core during simulated implantable port access.
5.2 If a needle produces a core during actual use, leaking of the implantable port may occur. Also, the core may be flushed into the port’s reservoir and subsequently into the patient’s body.
SCOPE
1.1 This test method covers the qualitative measurement of Huber-type needles’ potential to remove septum material during implantable port access (1).2
1.2 This test method does not address other issues that may include, but are not limited to, force measurement during the perforation/withdrawal, septum integrity, and any safety issues.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Jan-2023
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.33 - Medical/Surgical Instruments
Overview
ASTM F3212-16(2023), Standard Test Method for Coring Testing of Huber Needles, establishes a consistent and qualitative method for evaluating the potential of Huber-type needles to remove (core) septum material when accessing implantable ports. This international standard, developed by ASTM International, is designed to ensure Huber needles are manufactured to a standard that reduces the risk of producing septum cores, which can compromise device integrity and patient safety. Coring, if it occurs, may lead to leakage in the implantable port or migration of septum fragments into the patient's system.
Key Topics
- Huber Needle Coring: Focuses on whether Huber needles remove septum material during simulated access of implantable ports, assessing their design as non-coring needles.
- Qualitative Assessment: The test method is primarily a pass/fail evaluation based on the presence or absence of a core in the needle's cannula after perforation of a septum surrogate.
- Test Apparatus and Procedure:
- Utilizes a clamping fixture to securely hold a molded silicone septum disk.
- Requires observation of the needle's lumen with an optical microscope after simulated access.
- Incorporates additional tools like stylets to confirm the presence or absence of septum cores.
- Limitations: The standard does not address quantitative force measurements, septum durability, or broader safety concerns. It is the user’s responsibility to implement safe practices and compliance with applicable regulations.
Applications
This coring testing method is vital in the following contexts:
- Medical Device Evaluation: Used by manufacturers to ensure Huber needles meet non-coring requirements before being distributed for clinical use.
- Design Verification: Integral for research and development teams to validate the design of new or improved Huber-type needle models.
- Quality Assurance: Applied during production to maintain high standards of device safety and usability, supporting consistent quality in batch manufacturing.
- Regulatory Compliance: Assists in documentation and compliance with international quality and safety management frameworks for medical devices, particularly for products intended for use with implantable ports.
- Clinical Risk Reduction: Reduces the risk of leaked medications and foreign body embolization by ensuring port access devices do not cause coring, safeguarding patient health.
Related Standards
To enhance quality control and harmonize testing protocols, users of ASTM F3212-16(2023) should consider the following related standards:
- ASTM D2240: Standard Test Method for Rubber Property-Durometer Hardness, which is referenced for the characterization of the silicone septum material’s hardness.
- ISO 8536 Series: Standards for infusion equipment and needles, offering additional guidance on safety and performance.
- FDA Quality System Regulation (21 CFR Part 820): U.S. requirements for medical device manufacturers regarding risk management and product safety.
- Other ASTM Medical Device Standards: Especially those developed under the ASTM Committee F04 on Medical and Surgical Materials and Devices.
Practical Value
Implementing ASTM F3212-16(2023) ensures:
- Increased Patient Safety: Minimizes risks of port leakage and embolism.
- Standardized Manufacturing: Facilitates reproducible and reliable production of non-coring Huber needles.
- Regulatory Readiness: Supports compliance for market access in regions upholding international standards.
By integrating this standard into product development and quality assurance procedures, medical device manufacturers uphold the highest standards for injectable device safety and clinical performance.
Buy Documents
ASTM F3212-16(2023) - Standard Test Method for Coring Testing of Huber Needles
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F3212-16(2023) is a standard published by ASTM International. Its full title is "Standard Test Method for Coring Testing of Huber Needles". This standard covers: SIGNIFICANCE AND USE 5.1 This test method determines whether Huber needles are designed and manufactured such that they will not produce a core during simulated implantable port access. 5.2 If a needle produces a core during actual use, leaking of the implantable port may occur. Also, the core may be flushed into the port’s reservoir and subsequently into the patient’s body. SCOPE 1.1 This test method covers the qualitative measurement of Huber-type needles’ potential to remove septum material during implantable port access (1).2 1.2 This test method does not address other issues that may include, but are not limited to, force measurement during the perforation/withdrawal, septum integrity, and any safety issues. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method determines whether Huber needles are designed and manufactured such that they will not produce a core during simulated implantable port access. 5.2 If a needle produces a core during actual use, leaking of the implantable port may occur. Also, the core may be flushed into the port’s reservoir and subsequently into the patient’s body. SCOPE 1.1 This test method covers the qualitative measurement of Huber-type needles’ potential to remove septum material during implantable port access (1).2 1.2 This test method does not address other issues that may include, but are not limited to, force measurement during the perforation/withdrawal, septum integrity, and any safety issues. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F3212-16(2023) is classified under the following ICS (International Classification for Standards) categories: 11.040.30 - Surgical instruments and materials. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3212-16(2023) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F3212 − 16 (Reapproved 2023)
Standard Test Method for
Coring Testing of Huber Needles
This standard is issued under the fixed designation F3212; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
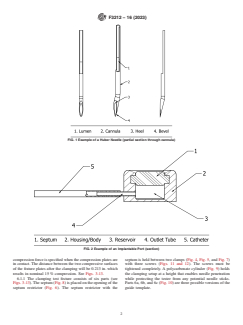
1. Scope 3.1.4 heel, n—the rear cutting edge of the needle bevel.
3.1.5 Huber needle, n—a needle whose tip is angled such
1.1 This test method covers the qualitative measurement of
that the bevel opening is parallel to the main axis of the
Huber-type needles’ potential to remove septum material
cannula. Its special shape slices rather than perforates the
during implantable port access (1).
septum, reducing the chance of leakage due to coring. It is also
1.2 This test method does not address other issues that may
known as a non-coring needed. See Fig. 1.
include, but are not limited to, force measurement during the
3.1.6 implantable port, n—a reservoir placed under the skin
perforation/withdrawal, septum integrity, and any safety issues.
(and usually attached to a catheter) that is made to receive a
1.3 This standard does not purport to address all of the
needle through a septum; it is often used to deliver medication.
safety concerns, if any, associated with its use. It is the
See Fig. 2.
responsibility of the user of this standard to establish appro-
3.1.7 lumen, n—the inside surface of the cannula.
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
3.1.8 septum, n—a feature of an implantable port that allows
1.4 This international standard was developed in accor-
repeated access by a port-access needle, generally composed of
dance with internationally recognized principles on standard-
an elastomeric material. See Fig. 2, Item 1.
ization established in the Decision on Principles for the
3.1.9 stylet, n—a device, preferably metallic, inserted into
Development of International Standards, Guides and Recom-
the lumen to remove a core.
mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
4. Summary of Test Method
4.1 A silicone elastomeric disk (surrogate septum or just
2. Referenced Documents
septum thereafter) is clamped into a specifically designed
2.1 ASTM Standards:
septum holder. The test operator accesses the septum with a
D2240 Test Method for Rubber Property—Durometer Hard-
Huber needle in accordance with the needle manufacturer’s
ness
instructions for use, as if the septum was an implantable port.
The lumen at the bevel is examined for the existence of a core,
3. Terminology
preferably before the needle is withdrawn. This is categorized
3.1 Definitions of Terms Specific to This Standard:
as a pass/fail test. Existence of a core in the needle’s cannula is
3.1.1 bevel, n—the slanted part of a needle that creates a
a failed result.
sharp pointed tip.
3.1.2 cannula, n—the tubular part of a needle through which
5. Significance and Use
fluids pass.
5.1 This test method determines whether Huber needles are
3.1.3 core, n—a sliver of septum material that may be
designed and manufactured such that they will not produce a
produced when a needle perforates a septum.
core during simulated implantable port access.
5.2 If a needle produces a core during actual use, leaking of
This test method is under the jurisdiction of ASTM Committee F04 on Medical
the implantable port may occur. Also, the core may be flushed
and Surgical Materials and Devices and is the direct responsibility of Subcommittee
into the port’s reservoir and subsequently into the patient’s
F04.33 on Medical/Surgical Instruments.
Current edition approved Feb. 1, 2023. Published February 2023. Originally body.
approved in 2016. Last previous edition approved in 2016 as F3212 – 16. DOI:
10.1520/F3212-16R23.
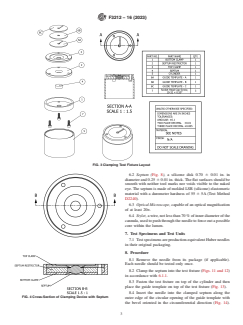
6. Apparatus
The boldface numbers in parentheses refer to a list of references at the end of
this standard.
6.1 Clamping Test Fixture, a clamping device which can
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
hold a septum with nominal dimensions of 0.70 in. in diameter
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
and 0.25 in. thick. The clamping device is such that it restrains
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. radial expansion of the septum under axial compression. The
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3212 − 16 (2023)
FIG. 1 Example of a Huber Needle (partial section through cannula)
FIG. 2 Example of an Implantable Port (section)
compression force is specified when the compression plates are septum is held between two clamps (Fig. 4, Fig. 5, and Fig. 7)
in contact. The distance between the two compressive surfaces with three screws (Figs. 11 and 12). The screws must be
of the fixture plates after the clamping will be 0.213 in. which tightened completely. A polycarbonate cylinder (Fig. 9) holds
results in nominal 15 % compression. See Figs. 3-13. the clamping setup at a height that enables needle penetration
6.1.1 The clamping test fixture consists of six parts (see while protecting the tester from any potential needle sticks.
Figs. 3-13). The septum (Fig. 8) is placed on the opening of the Parts 6a, 6b, and 6c (Fig. 10) are three possible versions of the
septum restrictor (Fig. 6). The septum restrictor with the guide template.
F3212 − 16 (2023)
FIG. 3 Clamping Test Fixture Layout
6.2 Septum (Fig. 8), a silicone disk 0.70 6 0.01 in. in
diameter and 0.25 6 0.01 in. thick. The flat surfaces should be
smooth with neither tool marks nor voids visible to the naked
eye. The septum is made of molded LSR (silicone) elastomeric
material with a durometer hardness of 55 6 5A (Test Method
D2240).
6.3 Optical Microscope, capable of an optical magnification
of at least 20×.
6.4 Stylet, a wire, not less than 70 % of inner diameter of the
cannula, used to push through the needle to force out a possible
core within the lumen.
7. Test Specimens and Test Units
7.1 Test specimens are production-equivalent Huber needles
in their original packaging.
8. Procedure
8.1 Remove the needle from its package (if applicable).
Each needle should be tested only once.
8.2 Clamp the septum into the test fixture (Figs. 11 and 12)
in accordance with 6.1.1.
8.3 Fasten the test fixture on top of the cylinder and then
place the guide template on top of the test fixture (Fig. 13).
8.4 Insert the needle into the clamped septum along the
FIG. 4 Cross-Section of Clamping Device with Septum
outer edge of the circular opening of the guide template with
the
...


Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...