ASTM D1434-23
(Test Method)Standard Test Method for Determining Gas Permeability Characteristics of Plastic Film and Sheeting
Standard Test Method for Determining Gas Permeability Characteristics of Plastic Film and Sheeting
SIGNIFICANCE AND USE
5.1 These measurements give semiquantitative estimates for the gas transmission of single pure gases through film and sheeting. Correlation of measured values with any given use, such as packaged contents protection, must be determined by experience. The gas transmission rate is affected by conditions not specifically provided for in these tests, such as moisture content (Note 2), plasticizer content, and nonhomogeneities. These tests do not include any provision for testing seals that may be involved in packaging applications.
Note 2: The tests are run using gas with 0 % moisture changes.
5.2 The historic Interlaboratory testing has revealed that permeances measured by these procedures exhibit a strong dependence on the procedure being used, as well as on the laboratory performing the testing. The historic method relied upon manual calibrations of Hg capillary columns and manual data readings of pressure. The references and use of Hg and capillary columns have been removed from this standard as current D1434 instruments rely upon readily calibrated vacuum gauges and automated recording of data. It is planned that the next revision of this standard includes an updated ILS with modern instrumentation. Additionally, it has been noted that an agreement with other gas transmission rate methods is sometimes poor and may be material-dependent. The materials being tested often affect the between-laboratory precision. The causes of these variations are not precisely known at this time, but is likely due to the fact that this method analyzes ALL gasses from the sample and not just the Test gas. This includes pre-absorbed water vapor within the sample and any free solvents remaining within the specimen. The 48 hr desiccator drying period outlined within the method may not be long enough for all materials. Additionally, other gas transmission rate methods (as those used for oxygen transmission rate, water vapor transmission rate and carbon dioxide transmission rate) often...
SCOPE
1.1 This test method utilizes a manometric method to determine the steady-state rate of transmission of a gas through plastics in the form of film, sheeting, laminates, and plastic-coated papers or fabrics. This test method provides for the determination of (1) gas transmission rate (GTR), (2) permeance, and, in the case of homogeneous materials, (3) permeability.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2023
- Technical Committee
- F02 - Primary Barrier Packaging
- Drafting Committee
- F02.10 - Permeation
Relations
- Effective Date
- 29-Sep-2023
- Effective Date
- 01-Nov-2008
- Effective Date
- 01-Nov-2005
- Effective Date
- 10-Nov-2000
Overview
ASTM D1434-23, published by ASTM International, specifies standardized test methods for determining the gas permeability characteristics of plastic films and sheeting. This comprehensive standard outlines procedures for quantifying the transmission rates of pure gases through a variety of polymeric materials, including films, laminates, and coated papers or fabrics. It provides practical approaches for measuring gas transmission rate (GTR), permeance, and, for homogeneous films, permeability, using both manometric and volumetric techniques.
Gas permeability data generated according to this standard are essential for assessing material suitability in applications where gas barrier properties are critical, such as packaging sensitive food items, pharmaceuticals, and industrial products.
Key Topics
- Gas Transmission Rate (GTR): Defines the quantity of a specified pure gas passing through a plastic film per unit time and area under controlled test conditions. Test conditions, such as temperature and pressure, must be reported with results.
- Permeance: Measures the ratio of GTR to the difference in partial pressure of the gas across the film. Important for evaluating relative barrier performance.
- Permeability: For homogeneous materials, the product of permeance and film thickness provides a property indicative of the material's intrinsic gas barrier characteristics.
- Test Methods:
- Procedure M (Manometric): Measures gas transmission using pressure changes in a sealed test chamber with automated or manual recording.
- Procedure V (Volumetric): Assesses gas flow using volume changes.
- Specimen Conditioning: Prior to testing, materials are typically conditioned at 23 °C and dried to reduce variability from absorbed moisture or solvents.
- Factors Influencing Results: Gas transmission rates are affected by variables such as moisture content, plasticizer presence, and non-uniformities in the sample. Test results reflect only the film or sheeting-not any seals or closures used in application.
Applications
- Packaging Industry: Evaluation of plastic films and laminates to ensure appropriate barrier properties against oxygen, carbon dioxide, and other gases, directly impacting food preservation and shelf life.
- Medical and Pharmaceutical: Selection of reliable film materials for pharmaceutical packaging, where controlled gas transmission is necessary for product stability.
- Material Development and Comparison: Enables manufacturers and researchers to benchmark and improve polymer formulations for enhanced gas barrier performance.
- Quality Assurance: Routine quality checks in manufacturing to confirm that product batches meet specified gas permeability criteria.
Data obtained using ASTM D1434-23 help predict performance in real-world applications, guiding material selection and package design. However, correlation with actual product protection must be confirmed through experience and additional testing as the method does not replicate all real-use conditions.
Related Standards
- ASTM D618: Practice for Conditioning Plastics for Testing-details environmental conditioning prior to testing for consistent, reliable data.
- Packaging Barrier Standards: Standards such as those for oxygen transmission rate (OTR), water vapor transmission rate (WVTR), and carbon dioxide transmission rate methods further specify barrier assessment for individual gases.
- ISO 15105: International standard for gas permeability in plastic films and sheeting, often referenced alongside ASTM D1434.
Practical Value
Implementing ASTM D1434-23 facilitates consistent, comparable measurement of gas permeability in plastic packaging materials, supporting quality control, R&D, and procurement processes. Manufacturers, converters, and product developers rely on this standard to validate material properties and ensure regulatory compliance in key industries where barrier performance is a critical differentiator.
Keywords: ASTM D1434, gas permeability test, plastic film, sheeting, gas transmission rate, permeance, packaging barrier, quality control, polymer testing, manometric method, volumetric method, industry standards.
Buy Documents
ASTM D1434-23 - Standard Test Method for Determining Gas Permeability Characteristics of Plastic Film and Sheeting
REDLINE ASTM D1434-23 - Standard Test Method for Determining Gas Permeability Characteristics of Plastic Film and Sheeting
Get Certified
Connect with accredited certification bodies for this standard

Smithers Quality Assessments
US management systems and product certification.

DIN CERTCO
DIN Group product certification.
Sponsored listings
Frequently Asked Questions
ASTM D1434-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Determining Gas Permeability Characteristics of Plastic Film and Sheeting". This standard covers: SIGNIFICANCE AND USE 5.1 These measurements give semiquantitative estimates for the gas transmission of single pure gases through film and sheeting. Correlation of measured values with any given use, such as packaged contents protection, must be determined by experience. The gas transmission rate is affected by conditions not specifically provided for in these tests, such as moisture content (Note 2), plasticizer content, and nonhomogeneities. These tests do not include any provision for testing seals that may be involved in packaging applications. Note 2: The tests are run using gas with 0 % moisture changes. 5.2 The historic Interlaboratory testing has revealed that permeances measured by these procedures exhibit a strong dependence on the procedure being used, as well as on the laboratory performing the testing. The historic method relied upon manual calibrations of Hg capillary columns and manual data readings of pressure. The references and use of Hg and capillary columns have been removed from this standard as current D1434 instruments rely upon readily calibrated vacuum gauges and automated recording of data. It is planned that the next revision of this standard includes an updated ILS with modern instrumentation. Additionally, it has been noted that an agreement with other gas transmission rate methods is sometimes poor and may be material-dependent. The materials being tested often affect the between-laboratory precision. The causes of these variations are not precisely known at this time, but is likely due to the fact that this method analyzes ALL gasses from the sample and not just the Test gas. This includes pre-absorbed water vapor within the sample and any free solvents remaining within the specimen. The 48 hr desiccator drying period outlined within the method may not be long enough for all materials. Additionally, other gas transmission rate methods (as those used for oxygen transmission rate, water vapor transmission rate and carbon dioxide transmission rate) often... SCOPE 1.1 This test method utilizes a manometric method to determine the steady-state rate of transmission of a gas through plastics in the form of film, sheeting, laminates, and plastic-coated papers or fabrics. This test method provides for the determination of (1) gas transmission rate (GTR), (2) permeance, and, in the case of homogeneous materials, (3) permeability. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 These measurements give semiquantitative estimates for the gas transmission of single pure gases through film and sheeting. Correlation of measured values with any given use, such as packaged contents protection, must be determined by experience. The gas transmission rate is affected by conditions not specifically provided for in these tests, such as moisture content (Note 2), plasticizer content, and nonhomogeneities. These tests do not include any provision for testing seals that may be involved in packaging applications. Note 2: The tests are run using gas with 0 % moisture changes. 5.2 The historic Interlaboratory testing has revealed that permeances measured by these procedures exhibit a strong dependence on the procedure being used, as well as on the laboratory performing the testing. The historic method relied upon manual calibrations of Hg capillary columns and manual data readings of pressure. The references and use of Hg and capillary columns have been removed from this standard as current D1434 instruments rely upon readily calibrated vacuum gauges and automated recording of data. It is planned that the next revision of this standard includes an updated ILS with modern instrumentation. Additionally, it has been noted that an agreement with other gas transmission rate methods is sometimes poor and may be material-dependent. The materials being tested often affect the between-laboratory precision. The causes of these variations are not precisely known at this time, but is likely due to the fact that this method analyzes ALL gasses from the sample and not just the Test gas. This includes pre-absorbed water vapor within the sample and any free solvents remaining within the specimen. The 48 hr desiccator drying period outlined within the method may not be long enough for all materials. Additionally, other gas transmission rate methods (as those used for oxygen transmission rate, water vapor transmission rate and carbon dioxide transmission rate) often... SCOPE 1.1 This test method utilizes a manometric method to determine the steady-state rate of transmission of a gas through plastics in the form of film, sheeting, laminates, and plastic-coated papers or fabrics. This test method provides for the determination of (1) gas transmission rate (GTR), (2) permeance, and, in the case of homogeneous materials, (3) permeability. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D1434-23 is classified under the following ICS (International Classification for Standards) categories: 83.140.10 - Films and sheets. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D1434-23 has the following relationships with other standards: It is inter standard links to ASTM D1898-68(1989), ASTM D618-08, ASTM D618-05, ASTM D618-00. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D1434-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D1434 − 23
Standard Test Method for
Determining Gas Permeability Characteristics of Plastic Film
and Sheeting
This standard is issued under the fixed designation D1434; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 3.1.1 gas transmission rate, GTR, n—the quantity of a given
gas passing through a unit of the parallel surfaces of a plastic
1.1 This test method utilizes a manometric method to
film in unit time under the conditions of test. The SI unit of
determine the steady-state rate of transmission of a gas through
GTR is mol/(m ·s). The test conditions, including temperature
plastics in the form of film, sheeting, laminates, and plastic-
and partial pressure of the gas on both sides of the film, must
coated papers or fabrics. This test method provides for the
be stated. Other factors, such as relative humidity and hydro-
determination of (1) gas transmission rate (GTR), (2)
static pressure, that influence the transport of the gas must also
permeance, and, in the case of homogeneous materials, (3)
be stated. A commonly used unit of GTR is mL (STP)/(m ·d)
permeability.
at a pressure differential of one atmosphere.
1.2 The values stated in SI units are to be regarded as
standard. No other units of measurement are included in this 3.1.2 permeance, P, n—the ratio of the gas transmission rate
standard. to the difference in partial pressure of the gas on the two sides
of the film. The SI unit of permeance is mol/ (m ·s·Pa). The test
1.3 This standard does not purport to address all of the
conditions (see 5.1) must be stated.
safety concerns, if any, associated with its use. It is the
responsibility of the user of this standard to establish appro-
3.1.3 permeability, P’, n—the product of the permeance and
priate safety, health, and environmental practices and deter-
the thickness of a film. The permeability is meaningful only for
mine the applicability of regulatory limitations prior to use.
homogeneous materials, in which it is a property characteristic
1.4 This international standard was developed in accor-
of the bulk material. This quantity should not be used unless
dance with internationally recognized principles on standard-
the constancy of the permeability has been verified using
ization established in the Decision on Principles for the
several different thicknesses of the material. The SI unit of P is
Development of International Standards, Guides and Recom- 2
(mol·m)/(m ·s·Pa) = mol ⁄(m·s·Pa). The test conditions (see 5.1)
mendations issued by the World Trade Organization Technical
must be stated.
Barriers to Trade (TBT) Committee.
NOTE 1—One milliliter (STP) is 44.62 μmol, one atmosphere is 0.1013
2. Referenced Documents
MPa, and one day is 86.4 × 10 s. GTR in SI units is obtained by
−10
multiplying the value in inch-pound units by 5.160 × 10 . Additional
2.1 ASTM Standards:
units and conversions are shown in Appendix X1.
D618 Practice for Conditioning Plastics for Testing
3.1.4 steady state—the state attained when the amount of
D1898 Practice for Sampling of Plastics (Withdrawn 1998)
gas absorbed in the film is in equilibrium with the flux of gas
3. Terminology through the film.
3.1 Definitions of Terms Specific to This Standard:
4. Summary of Test Method
4.1 The sample is mounted in a gas transmission cell so as
This test method is under the jurisdiction of ASTM Committee F02 on Primary
to form a sealed semi-barrier between two chambers. One
Barrier Packaging and is the direct responsibility of Subcommittee F02.10 on
chamber contains the test gas at a specific high pressure, and
Permeation.
Current edition approved May 1, 2023. Published June 2023. Originally the other chamber, at a lower pressure, receives the permeating
ɛ1
approved in 1956. Last previous edition approved in 2015 as D1434 – 82 (2015) .
gas.
DOI: 10.1520/D1434-23.
4.1.1 The lower pressure chamber is initially evacuated.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Following the evacuation, the low-pressure chamber is allowed
Standards volume information, refer to the standard’s Document Summary page on
to accumulate the permeating test gas. The transmission of the
the ASTM website.
gas through the test specimen is determined from monitoring
The last approved version of this historical standard is referenced on
www.astm.org. the slope of the increasing pressure over time.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D1434 − 23
5. Significance and Use 6.3 Mark the side of the material facing the test gas. Note:
It’s most common to orientate the sample as it is utilized in
5.1 These measurements give semiquantitative estimates for
real-life (for example, if measuring oxygen passing from the
the gas transmission of single pure gases through film and
outside of a package to the inside, then the “outside face” of the
sheeting. Correlation of measured values with any given use,
sample should be exposed to the high pressure oxygen and the
such as packaged contents protection, must be determined by
“inside face” should be exposed to the vacuum side of the test
experience. The gas transmission rate is affected by conditions
cell).
not specifically provided for in these tests, such as moisture
6.4 If required for permeability normalization, the thickness
content (Note 2), plasticizer content, and nonhomogeneities.
of the specimen shall be measured to the nearest 2.5 μm with
These tests do not include any provision for testing seals that
a calibrated dial gage (or equivalent) at a minimum of five
may be involved in packaging applications.
points distributed over the entire test area. Maximum,
NOTE 2—The tests are run using gas with 0 % moisture changes.
minimum, and average values should be recorded. An alterna-
tive measure of thickness involving the weighing of a known
5.2 The historic Interlaboratory testing has revealed that
area of specimens having a known density is also suitable for
permeances measured by these procedures exhibit a strong
homogeneous materials.
dependence on the procedure being used, as well as on the
laboratory performing the testing. The historic method relied
7. Conditioning and Test Temperature
upon manual calibrations of Hg capillary columns and manual
7.1 Standard Conditioning—To reduce innate outgassing
data readings of pressure. The references and use of Hg and
concerns of the material (such as absorbed water vapor or
capillary columns have been removed from this standard as
solvents), condition all test specimens at 23 °C 6 2 °C in a
current D1434 instruments rely upon readily calibrated vacuum
desiccator over calcium chloride or other suitable desiccant for
gauges and automated recording of data. It is planned that the
not less than 48 h prior to test in accordance with Practice
next revision of this standard includes an updated ILS with
D618, for those tests where conditioning is required.
modern instrumentation. Additionally, it has been noted that an
agreement with other gas transmission rate methods is some-
7.2 Alternative Conditioning—Alternatives to 7.1 may be
times poor and may be material-dependent. The materials
used for conditioning the specimens provided that these
being tested often affect the between-laboratory precision. The
conditions are described in the report. As an example, an
causes of these variations are not precisely known at this time,
alternative conditioning could be placing the sample into a N2
but is likely due to the fact that this method analyzes ALL
purging box to remove absorbed humidity or solvents.
gasses from the sample and not just the Test gas. This includes
7.3 Test Temperature—For ambient testing, the temperature
pre-absorbed water vapor within the sample and any free
of the cell should be controlled at 23 °C 6 2 °C. For instru-
solvents remaining within the specimen. The 48 hr desiccator
ments that can control the cell temperature a tolerance of 6
drying period outlined within the method may not be long
1 °C shall be utilized.
enough for all materials. Additionally, other gas transmission
rate methods (as those used for oxygen transmission rate, water
8. Sampling
vapor transmission rate and carbon dioxide transmission rate)
8.1 The techniques used in sampling a batch of material to
often incorporate test gas specific sensors and therefore would
be tested by these procedures must depend upon the kind of
minimize influence from other gasses. It is suggested that this
information that is sought. Care should be taken to ensure that
method not be used for referee purposes unless purchaser and
samples represent conditions across the width and along the
seller can both establish that they are measuring the same
length of rolls of film. Practice D1898 provides guidelines for
quantity to a mutually agreed upon level of precision.
deciding what procedures to use in sampling a batch of
5.3 Use of the permeability coefficient (involving conver-
material. Enough specimens must be tested to ensure that the
sion of the gas transmission rate to a unit thickness basis) is not information obtained is representative of the batch or other lot
recommended unless the thickness-to-transmission rate rela-
size being tested.
tionship is known from previous studies. Even in essentially
9. Apparatus
homogeneous structures, variations in morphology (as
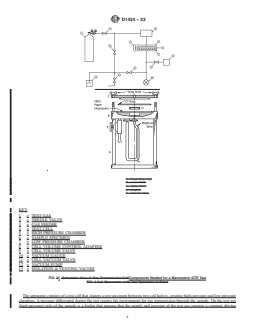
9.1 The apparatus shown in Fig. 1 outlines the components
indicated, for example, by density) and thermal history may
influence permeability. needed for a manometric GTR test.
The apparatus consists of a test cell that clamps a test
specimen between two cell halves, creating high-pressure and
6. Test Specimen
low-pressure chambers. A pressure differential during the test
6.1 The test specimen shall be representative of the material,
creates the environment for gas transmission through the
free of wrinkles, creases, pinholes, and other imperfections,
sample. On the test gas (high-pressure) side of the sample is a
and shall be of uniform thickness and be capable of being
feeder that ensures that the supply and pressure of the test gas
mounted airtight (for example, no edge leakage, which can
remains a constant driving force. On the measurement (low-
occur with papers). The test specimen shall be cut to an
pressure) side of the sample is a pressure sensor (vacuum
appropriate size (generally circular) to fit the test cell.
gauge) that measures the pressure change of the system as gas
6.2 Use three specimens unless otherwise specified or permeates through the specimen. Plumbed beneath the low-
agreed upon among interested parties. pressure chamber is a vacuum pump to initially evacuate the
D1434 − 23
flat as the vacuum is applied and ensures that the test area of
the sample is not occluded and allows for gas transmission rate
flow.
9.1.5 Cell Volume-Control Adapter or Fillers—Optional:
Some instruments incorporate a volume control adapter to aid
with adjusting the range of the instrument. This adapter can be
an additional reservoir plumbed into the system. Alternatively,
using filler blocks within the low-pressure chamber reduces the
chamber volume resulting in faster pressure accumulation,
which is useful for measuring low transmission rate materials.
9.1.6 Vacuum Gage, to register the pressure during evacua-
tion of the system to the nearest 13 Pa.
9.1.7 Cell Vacuum Valve, capable of maintaining a vacuum-
KEY
tight seal.
9.1.8 Vacuum pump, capable of reducing the pressure in the
1 = TEST GAS
system to 26 Pa or less.
2 = NEEDLE VALVE
3 = GAS FEEDER 9.1.9 Additional valving can be utilized to isolate and vent
4 = TEST CELL
the system as needed.
5 = HIGH PRESSURE CHAMBER
6 = SAMPLE SPECIMEN
10. Materials
7 = LOW PRESSURE CHAMBER
10.1 Test Gas—The test gas shall be dry and pure. As a
8 = CELL VOLUME CONTROL ADAPTER
guidance, ensure that the gas pressure remains constant
9 = CELL VOLUME VALVE
throughout the test, the ratio of the volume of gas available for
10 = VACUUM GAUGE
transmission to the volume of gas transmitted at the completion
11 = CELL VACUUM VALVE
12 = VACUUM PUMP of the test shall be at least 100:1.
13 = ISOLATION & VENTING VALVES
10.2 Vacuum Grease—Low volatility/high vacuum grease.
10.3 Filter Paper—Any high-grade, medium retention
FIG. 1 Components Needed for a Manometric GTR Test
qualitative nonashing cellulosic filter paper will be satisfactory
for this purpose. A thickness of 0.2 mm to 0.3 mm is recom-
system for the transmission rate measurement. An optional
mended. As the filter paper supports the specimen, it needs to
volume control adapter plumbed into the low-pressure chamber
be cut to the same size as the gas transmission area of the
can be utilized for extending the range of the GTR test.
low-pressure chamber.
9.1.1 Test Gas—A tank of known gas with enough volume
and pressure to conduct the test.
11. Calibration
9.1.2 Needle Valve, for slowly admitting and adjusting the
11.1 Pressure gauges and temperature sensors within the
pressure of the test gas.
apparatus should be calibrated per manufacturer’s recom-
9.1.3 Gas Feeder—The gas supplied by the Gas Feeder to
mended calibration specifications and schedule.
the upper chamber is a reservoir (usually a tank gas) that has
11.2 Some instruments incorporate a film calibration fea-
enough capacity and constant driving force pressure as to not
impact the GTR measurement. Additionally, the Gas Feeder ture. The GTR calculation r
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
´1
Designation: D1434 − 82 (Reapproved 2015) D1434 − 23
Standard Test Method for
Determining Gas Permeability Characteristics of Plastic Film
and Sheeting
This standard is issued under the fixed designation D1434; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Editorial corrections were made in September 2015.
1. Scope
1.1 This test method covers the estimation of the utilizes a manometric method to determine the steady-state rate of transmission
of a gas through plastics in the form of film, sheeting, laminates, and plastic-coated papers or fabrics. This test method provides
for the determination of (1) gas transmission rate (GTR), (2) permeance, and, in the case of homogeneous materials, (3)
permeability.
1.2 Two procedures are provided:
1.2.1 Procedure M—Manometric.
1.2.2 Procedure V—Volumetric.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and healthsafety, health, and environmental practices and determine
the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D618 Practice for Conditioning Plastics for Testing
D1898 Practice for Sampling of Plastics (Withdrawn 1998)
3. Terminology
3.1 Definitions of Terms Specific to This Standard:
This test method is under the jurisdiction of ASTM Committee F02 on FlexiblePrimary Barrier Packaging and is the direct responsibility of Subcommittee F02.10 on
Permeation.
Current edition approved June 1, 2015May 1, 2023. Published September 2015June 2023. Originally approved in 1956. Last previous edition approved in 20092015 as
ϵ1ɛ1
D1434 – 82 (2009)(2015) . DOI: 10.1520/D1434-82R15E01.10.1520/D1434-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D1434 − 23
3.1.1 gas transmission rate, GTR—GTR, n—the quantity of a given gas passing through a unit of the parallel surfaces of a plastic
film in unit time under the conditions of test. The SI unit of GTR is 1 mol/(m ·s). The test conditions, including temperature and
partial pressure of the gas on both sides of the film, must be stated. Other factors, such as relative humidity and hydrostatic
pressure, that influence the transport of the gas must also be stated. The inch-pound unit of GTR, a A commonly used unit of
GTR,GTR is 1 mL (STP)/(m ·d) at a pressure differential of one atmosphere.
3.1.2 permeance, P—P, n—the ratio of the gas transmission rate to the difference in partial pressure of the gas on the two sides
of the film. The SI unit of permeance is 1 mol/ (m ·s·Pa). The test conditions (see 5.1) must be stated.
3.1.3 permeability, PP’,—n—the product of the permeance and the thickness of a film. The permeability is meaningful only for
homogeneous materials, in which it is a property characteristic of the bulk material. This quantity should not be used unless the
constancy of the permeability has been verified using several different thicknesses of the material. The SI unit of P is
1(mol·m)/(m ·s·Pa) = mol mol/(m·s·Pa). ⁄(m·s·Pa). The test conditions (see 3.15.1) must be stated.
NOTE 1—One millilitremilliliter (STP) is 44.62 μmol, one atmosphere is 0.1013 MPa, and one day is 86.4 × 10 s. GTR in SI units is obtained by
−10
multiplying the value in inch-pound units by 5.160 × 10 . Additional units and conversions are shown in Appendix X1.
3.1.4 steady state—the state attained when the amount of gas absorbed in the film is in equilibrium with the flux of gas through
the film. For Method V, this is obtained when the GTR is constant.
4. Summary of Test Method
4.1 The sample is mounted in a gas transmission cell so as to form a sealed semibarriersemi-barrier between two chambers. One
chamber contains the test gas at a specific high pressure, and the other chamber, at a lower pressure, receives the permeating gas.
Either of the following procedures is used:
4.1.1 Procedure M—In Procedure M the The lower pressure chamber is initially evacuated and the evacuated. Following the
evacuation, the low-pressure chamber is allowed to accumulate the permeating test gas. The transmission of the gas through the
test specimen is indicated by an increase in pressure.determined from monitoring the slope of the increasing pressure over time.
4.1.2 Procedure V—In Procedure V the lower pressure chamber is maintained near atmospheric pressure and the transmission of
the gas through the test specimen is indicated by a change in volume.
5. Significance and Use
5.1 These measurements give semiquantitative estimates for the gas transmission of single pure gases through film and sheeting.
Correlation of measured values with any given use, such as packaged contents protection, must be determined by experience. The
gas transmission rate is affected by conditions not specifically provided for in these tests, such as moisture content (Note 2),
plasticizer content, and nonhomogeneities. These tests do not include any provision for testing seals that may be involved in
packaging applications.
NOTE 2—The tests are run using gas with 0 % moisture changes.
5.2 The historic Interlaboratory testing has revealed that permeances measured by these procedures exhibit a strong dependence
on the procedure being used, as well as on the laboratory performing the testing. Agreement with other The historic method relied
upon manual calibrations of Hg capillary columns and manual data readings of pressure. The references and use of Hg and
capillary columns have been removed from this standard as current D1434 instruments rely upon readily calibrated vacuum gauges
and automated recording of data. It is planned that the next revision of this standard includes an updated ILS with modern
instrumentation. Additionally, it has been noted that an agreement with other gas transmission rate methods is sometimes poor and
may be material-dependent. The materials being tested often affect the between-laboratory precision. The causes of these variations
are not precisely known at this time. time, but is likely due to the fact that this method analyzes ALL gasses from the sample and
not just the Test gas. This includes pre-absorbed water vapor within the sample and any free solvents remaining within the
specimen. The 48 hr desiccator drying period outlined within the method may not be long enough for all materials. Additionally,
other gas transmission rate methods (as those used for oxygen transmission rate, water vapor transmission rate and carbon dioxide
transmission rate) often incorporate test gas specific sensors and therefore would minimize influence from other gasses. It is
D1434 − 23
suggested that this method not be used for referee purposes unless purchaser and seller can both establish that they are measuring
the same quantity to a mutually agreed upon level of precision.
5.3 Use of the permeability coefficient (involving conversion of the gas transmission rate to a unit thickness basis) is not
recommended unless the thickness-to-transmission rate relationship is known from previous studies. Even in essentially
homogeneous structures, variations in morphology (as indicated, for example, by density) and thermal history may influence
permeability.
6. Test Specimen
6.1 The test specimen shall be representative of the material, free of wrinkles, creases, pinholes, and other imperfections, and shall
be of uniform thickness. thickness and be capable of being mounted airtight (for example, no edge leakage, which can occur with
papers). The test specimen shall be cut to an appropriate size (generally circular) to fit the test cell.
6.2 Use three specimens unless otherwise specified or agreed upon among interested parties.
6.3 Mark the side of the material facing the test gas. Note: It’s most common to orientate the sample as it is utilized in real-life
(for example, if measuring oxygen passing from the outside of a package to the inside, then the “outside face” of the sample should
be exposed to the high pressure oxygen and the “inside face” should be exposed to the vacuum side of the test cell).
6.4 The If required for permeability normalization, the thickness of the specimen shall be measured to the nearest 2.5 μm with
a calibrated dial gage (or equivalent) at a minimum of five points distributed over the entire test area. Maximum, minimum, and
average values should be recorded. An alternative measure of thickness involving the weighing of a known area of specimens
having a known density is also suitable for homogeneous materials.
7. Conditioning and Test Temperature
7.1 Standard Conditioning—Condition To reduce innate outgassing concerns of the material (such as absorbed water vapor or
solvents), condition all test specimens at 23 6 2°C23 °C 6 2 °C in a desiccator over calcium chloride or other suitable desiccant
for not less than 48 h prior to test in accordance with Practice D618, for those tests where conditioning is required. In cases of
disagreement, the tolerances shall be 61°C.
7.2 Alternative Conditioning—Alternatives to 7.1 may be used for conditioning the specimens provided that these conditions are
described in the report. As an example, an alternative conditioning could be placing the sample into a N2 purging box to remove
absorbed humidity or solvents.
7.3 Test Temperature—For ambient testing, the temperature of the cell should be controlled at 23 °C 6 2 °C. For instruments that
can control the cell temperature a tolerance of 6 1 °C shall be utilized.
8. Sampling
8.1 The techniques used in sampling a batch of material to be tested by these procedures must depend upon the kind of information
that is sought. Care should be taken to ensure that samples represent conditions across the width and along the length of rolls of
film. Practice D1898 provides guidelines for deciding what procedures to use in sampling a batch of material. Enough specimens
must be tested to ensure that the information obtained is representative of the batch or other lot size being tested.
PROCEDURE M
(Pressure changes in the manometric cell may be determined by either visual or automatic recording.)
MANOMETRIC VISUAL DETERMINATION
9. Apparatus
9.1 The apparatus shown in Fig. 1 andoutlines Fig. 2 consists of the following items:the components needed for a manometric
GTR test.
D1434 − 23
A—Supporting Legs
B—Lower Plate
C—Upper Plate
D—Adapter
E—Vacuum Valve
KEY
1 = TEST GAS
2 = NEEDLE VALVE
3 = GAS FEEDER
4 = TEST CELL
5 = HIGH PRESSURE CHAMBER
6 = SAMPLE SPECIMEN
7 = LOW PRESSURE CHAMBER
8 = CELL VOLUME CONTROL ADAPTER
9 = CELL VOLUME VALVE
10 = VACUUM GAUGE
11 = CELL VACUUM VALVE
12 = VACUUM PUMP
13 = ISOLATION & VENTING VALVES
FIG. 21 Schematic View of Gas Transmission CellComponents Needed for a Manometric GTR Test
FIG. 3 Cell Manometer with Test Specimen in Place
The apparatus consists of a test cell that clamps a test specimen between two cell halves, creating high-pressure and low-pressure
chambers. A pressure differential during the test creates the environment for gas transmission through the sample. On the test gas
(high-pressure) side of the sample is a feeder that ensures that the supply and pressure of the test gas remains a constant driving
D1434 − 23
force. On the measurement (low-pressure) side of the sample is a pressure sensor (vacuum gauge) that measures the pressure
change of the system as gas permeates through the specimen. Plumbed beneath the low-pressure chamber is a vacuum pump to
initially evacuate the system for the transmission rate measurement. An optional volume control adapter plumbed into the
low-pressure chamber can be utilized for extending the range of the GTR test.
9.1.1 Test Gas—A tank of known gas with enough volume and pressure to conduct the test.
9.1.2 Cell Manometer System—Needle Valve, The calibrated cell manometer leg, which indicates for slowly admitting and
adjusting the pressure of transmitted gas, shall consist of precision-bore glass capillary tubing at least 65 mm long with an inside
diameter of 1.5 mm.the test gas.
9.1.3 Cell Reservoir System, Gas Feeder—consisting of a glass reservoir of sufficient size to contain all the mercury required in
the cell.The gas supplied by the Gas Feeder to the upper chamber is a reservoir (usually a tank gas) that has enough capacity and
constant driving force pressure as to not impact the GTR measurement. Additionally, the Gas Feeder incorporates a pressure gauge
with a range from 0 kPa to 333 kPa absolute for measuring the supplied test gas pressure.
9.1.4 Adapters—Transmission Rate Test Cell—Solid and hollow adapters for measurement of widely varying gas transmission
rates. The solid adapter provides a minimum void volume for slow transmission rates. The hollow adapter increases the void
volume by about a factor of eight for faster transmission rates.The transmission test cell shall consist of a high-pressure chamber
and a low-pressure chamber, designed so that the gas transmission area is constant for any specimen mounted in the cell. Note:
typical transmission rate cell area diameters can range from 10 mm to 150 mm.
9.1.4.1 The high-pressure chamber shall have an inlet for the test gas. The high-pressure chamber’s surface that contacts the
specimen shall be smooth and flat and incorporates an O-ring for a leak-free seal.
9.1.4.2 The low-pressure chamber shall be connected to a pressure sensor (vacuum gauge) for the transmission rate pressure
measurement. The surface of the low-pressure chamber that contacts the test specimen shall be smooth and flat for a leak-free seal.
Beneath the sample is a filter paper/support structure. The filter paper/support structure keeps the sample flat as the vacuum is
applied and ensures that the test area of the sample is not occluded and allows for gas transmission rate flow.
9.1.4 Cell Vacuum Valve, capable of maintaining a vacuum-tight seal.
9.1.5 Plate Surfaces, that contact the specimen and filter paper shall be smooth and flat.
9.1.6 O-Ring, for sealing the upper and lower plates.
9.1.7 Pressure Gage, mechanical or electrical type with a range from 0 to 333 kPa absolute. Used for measuring upstream gas
pressure.
9.1.5 Barometer, Cell Volume-Control Adapter or Fillers—suitable for measuring the pressure of the atmosphere to the nearest 133
Pa.Optional: Some instruments incorporate a volume control adapter to aid with adjusting the range of the instrument. This adapter
can be an additional reservoir plumbed into the system. Alternatively, using filler blocks within the low-pressure chamber reduces
the chamber volume resulting in faster pressure accumulation, which is useful for measuring low transmission rate materials.
9.1.6 Vacuum Gage, to register the pressure during evacuation of the system to the nearest 13 Pa.
9.1.7 Cell Vacuum Valve, capable of maintaining a vacuum-tight seal.
9.1.8 Vacuum Pump,pump, capable of reducing the pressure in the system to 26 Pa or less.
9.1.11 Needle Valve, for slowly admitting and adjusting the pressure of the test gas.
9.1.9 Cathetometer, to measure the height of mercury in the cell manometer leg accurately. This instrument should be capable of
measuring changes to the nearest 0.5 mm.Additional valving can be utilized to isolate and vent the system as needed.
9.1.13 Micrometer, to measure specimen thickness, graduated to 2.5 μm (0.1 mil) or better.
9.1.14 Elevated-Temperature Fittings—Special cell fittings are required for high-temperature testing.
D1434 − 23
10. Materials
10.1 Test Gas—The test gas shall be dry and pure. The As a guidance, ensure that the gas pressure remains constant throughout
the test, the ratio of the volume of gas available for transmission to the volume of gas transmitted at the completion of the test shall
be at least 100:1.
10.2 Vacuum Grease—Low volatility/high vacuum grease.
10.3 Mercury—Filter Paper—Mercury used in the cell shall be triple distilled, checked regularly for purity, and replaced with
clean mercury when necessary.Any high-grade, medium retention qualitative nonashing cellulosic filter paper will be satisfactory
for this purpose. A thickness of 0.2 mm to 0.3 mm is recommended. As the filter paper supports the specimen, it needs to be cut
to the same size as the gas transmission area of the low-pressure chamber.
10.2.1 Warning—Very low concentrations of mercury vapor in the air are known to be hazardous. Guidelines for using mercury
in the laboratory have been published by Steere. Be sure to collect all spilled mercury in a closed container. Transfers of mercury
should be made over a large plastic tray. Under normal daily laboratory-use conditions, the cells should be cleaned about every
3 months. Dirty mercury is indicated when the drop of the capillary becomes erratic or when mercury clings to the side of the
capillary, or both. Whenever such discontinuities occur, the mercury should be removed and the cell cleaned as follows:
(1) Wash with toluene (to remove greases and oils).
(2) Wash with acetone (to remove toluene).
(3) Wash with distilled water (to remove acetone).
(4) Wash with a 1 + 1 mixture of nitric acid and distilled water (to remove any mercury salts that may be present). This
operation may be repeated if necessary in order to ensure complete cleaning of glassware.
(5) Wash with distilled water (to remove nitric acid).
(6) Wash with acetone (to remove water).
(7) Dry the cell at room temperature or by blowing a small amount of clean dry air through it.
11. Calibration
11.1 Pressure gauges and temperature sensors within the apparatus should be calibrated per manufacturer’s recommended
calibration specifications and schedule.
11.2 Each cell should be calibrated at the test temperature as follows (Some instruments incorporate a film calibration feature. The
GTR calculation relies upon the following: the change in pressure over time (dp/dt); the high-pressure value of the test gas; the
volume ofFig. 3): the low-pressure side of the system; the test temperature; the transmission rate exposure area of the low-pressure
chamber and the Ideal Gas Constant (R). By using a known GTR film standard, one can determine a calibration coefficient to adjust
for any variances with the calculation inputs. If an instrument does have this feature, follow the manufacture’s guidance for film
calibration.
11.1.1 Determine the void volume of the filter paper from the absolute density of its fiber content (Note 3), the weight of the filter
paper, and its apparent volume (Note 4). Express the void volume determined in this way in microlitres and designate as V .
CD
NOTE 3—Any high-grade, medium-retention qualitative nonashing cellulosic filter paper, 90 mm in diameter will be satisfactory for this purpose. Cellulose
fiber has an approximate density of 1.45 g/mL.
NOTE 4—The apparent volume may be calculated from the thickness and diameter of the filter paper.
11.1.2 Determine the volume of the cell manometer leg from B to C,Fig. 3, by mercury displacement. (Since the void volume of
the adapters is included in this part of the calibration, the volume from B to C should be determined twice, once with the solid
adapter in place, and once with the hollow.) This volume is obtained by dividing the weight of the mercury displaced by its density
(Note 5). Determine this volume to nearest 1 μL and designate as V .
BC
NOTE 5—The density of mercury at 23°C is 13.54 g/mL.
11.1.3 Determine the volume, in microlitres, of the cell manometer leg from A to B,Fig. 3, by mercury displacement. Determine
D1434 − 23
the average cross-sectional area of the capillary by dividing this volume by the length (expressed to the nearest 0.1 mm) from A
to B.Determine this area to the nearest 0.01 mm and designate as a .
c
11.1.4 Determine the area of the filter paper cavity to the nearest 1 mm . Designate this area as A, the area of transmission.
11.1.5 Pour the mercury from the reservoir into the manometer of the cell by carefully tipping the cell. Record the distance from
the datum plane to the upper calibration line B in the capillary leg as h . Record the distance from the datum plane to the top of
B
the mercury meniscus in the reservoir leg as h . Determine h and h to the nearest 0.5 mm.
L B L
11.2 NBS Standard Reference Material 1470 is a polyester film whose permeance to oxygen gas has been certified for a range
of experimental conditions. The calibration steps in 11.1 can be verified by comparing measurements obtained using this method
of test in the user’s laboratory with the values provided on the certificate accompanying the SRM.
12. Procedure
12.1 Transfer all the mercury into the reservoir of the cell manometer system by carefully tipping the cell in such a way that the
mercury pours into the reservoir.If utilized, insert the appropriate volume filler into the low-pressure chamber.
12.2 Insert the appropriate adapter in the cell body.
12.2 Center a filter paper in the lower plate cavity.low-pressure chamber.
12.3 Apply a light thin and uniform coating of vacuum grease on the flat metal that the sealing surface of the specimen will
contact. Avoid excessive grease. low-pressure chamber that contacts the specimen. Avoid excessive grease as it can overflow onto
the filter paper and reduce the transmission rate exposure area.
12.4 PlaceMount the conditioned specimen smoothly on the lower lightly greased plate so that it covers the filter paper and the
entire exposed face of the lower plate.on the low-pressure chamber ensuring no creasing or slackness occurs. Sample creasing can
create leakage from the chamber and a sample mounted with slack will have a larger surface area. Both of these can lead to
inaccurate test results.
12.5 Locate Lightly grease the O-ring on the upper plate; and sealing surface on the high-pressure chamber and then carefully
position this plate it over the specimen and fix the plateclamp into place with uniform pressure to ensure a vacuum-tight seal.
12.7 Connect the line in which the test gas will be subsequently admitted to the upper plate. (The entire cell is now directly
connected to the test gas line.)
12.8 Connect the vacuum source to the nipple attached to the cell vacuum valve. Evacuate the bottom of the cell; then, with the
bottom still being evacuated, evacuate the top of the cell. Close off the vacuum line to the top of the cell; then close the line to
the bottom (Fig. 4).
12.9 Flush the connecting line and the top of the chamber with test gas.
12.10 Reevacuate the system in the same manner as 12.8. The cell manometer system should be evacuated to a pressure of 26 Pa
or less, as indicated on the vacuum gage.
12.6 Pour mercury from the reservoir into the manometer Engage the vacuum pump and evacuate ambient air from the system
of the cell by carefully tipping the cell. The height of the mercury in the capillary leg should be at approximately the same level
as line low-pressure chamber; followed by the high-pressure chamber.B(Fig. 3) and stationary.
NOTE 6—A leak is indicated if the height of the mercury does not remain stationary. If such a leak occurs, discontinue the test and repeat the entire
procedure. (If a leak occurs on a second trial, this may indicate a mechanical failure of the equipment.)
D1434 − 23
12.12 Record the height of the mercury in the capillary leg, h , at the start of each test, that is, immediately before the test gas
o
has been admitted to the top of the cell.
12.7 After a suitable estimated time for attaining steady-state conditions, record the height of the mercury in the capillary
leg,When the air has been evacuated to a level of 26 Pa or less, close the related valving hand , to the nearest 0.5 mm and the
o
elapsed time,stop the vacuum pump. Note: the time needed to evacuate the system will tbe , to the nearest 1 min. dependent upon
o
the system volume and sample outgassing.
12.14 Record the height of the mercury, h, in the capillary leg to the nearest 0.5 mm versus time, t, in hours, to the nearest 1 min.
Take several readings (at least six are recommended) during the test. Calculate the function g (h) for each t as defined in 13.1. Plot
these values versus time, (t−t ), and construct the best straight line through these points. Use any observed values of h and t for
o
h and t , respectively, if these values are within the steady-state region. A nonlinear plot of g(h) versus (t−t ) that does not pass
o o o
through the origin could indicate an improper selection of h and t ; a new selection should then be made by using a larger mercury
o o
depression for the initial conditions.
NOTE 7—If, after all the mercury has been displaced from the capillary, any doubt exists as to the attainment of steady state, perform a check as follows:
(1) Return the mercury to the reservoir.
(2) Reevacuate the bottom of the cell only, leaving the top pressurized with test gas.
(3) Repeat 12.11, 12.13, and 12.14.
12.8 Return the mercury in the capillary leg to the reservoir by tipping the cell upon completion of the test and prior to opening
the cell vacuum valve.If the pressure within the low-pressure chamber immediately rises, this could be an indication of a leak or
a large amount of sample outgassing and the need to repeat the sample mounting or the initial evacuation, or both.
12.9 Remove the specimen from the cell and measure the thickness with a micrometer (Introduce the Test Gas to the high-pressure
chamber. Shut off the gas supply when the pressureNote 8). Record the average of five determinations made uniformly throughout
the specimen to the nearest 2.5 μm. reaches the desired test pressure level (commonly 101 kPa or 1 atm). Record the high-pressure
value. With the test gas addition, an increase in pressure on the low-pressure side confirms gas is transmitting through the sample.
NOTE 8—If there is reason to believe that the specimen will expand or contract during transmission, the thickness should be measured prior to 12.5, as
well as after transmission. If any change in thickness occurs, a note to this effect shall be included with the results.
12.10 Test three specimens with each gas.Collect and plot the pressure reading within the low-pressure chamber versus time.
12.11 If the requirements ofWhen the graph indicates 12.14 are not met in the normal atmospheric pressure test, repeat the
procedure at a higher (up to 304 kPa) or lower (not less than 50 kPa) test pressure.a straight line, this indicates that the material
has reached equilibrium. The slope of the straight-line portion of the graph relates directly to the GTR. Calculate and report the
GTR.
13. CalculationCalculations
13.1 Calculate the permeance,Gas P,Transmission Rate (GTR) in SI units from the following relationship (relationship:Note 9):
P5 g~h!/~t2 t ! (1)
o
where:
g h 5 2 V 1a p 1h 2 h
~ !
@@ ~ !#
f u B L
ART
h 2 h
~ !
o
·1n 12 12a~h 2 h! (2)
H J o G
P 2 h 2 h
~ !
u L o
¯
a =
area of capillary AB, mm ,
c
A = area of transmission, cm ,
h = height of mercury in the capillary leg at the start of the actual transmission run, after steady-state conditions have been
o
attained, mm,
D1434 − 23
h = height of mercury in cell capillary leg at any given time, mm,
h = maximum height of mercury in the cell manometer leg from the datum plane to upper calibration line B, mm,
B
h = height of mercury in cell reservoir leg from datum plane to top of mercury meniscus, mm,
L
P = upstream pressure of gas to be transmitted,
u
R = universal gas constant 8.3143 × 10 L·Pa/(mol·K),
t = time at the start of the actual transmission run, h, after steady
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...