ASTM D5790-18
(Test Method)Standard Test Method for Measurement of Purgeable Organic Compounds in Water by Capillary Column Gas Chromatography/Mass Spectrometry
Standard Test Method for Measurement of Purgeable Organic Compounds in Water by Capillary Column Gas Chromatography/Mass Spectrometry
SIGNIFICANCE AND USE
5.1 Purgeable organic compounds have been identified as contaminants in treated drinking water, wastewater, ground water, and Toxicity Characteristic Leaching Procedure (TCLP) leachate. These contaminants may be harmful to the environment and to people. Purge and trap sampling is a generally applicable procedure for concentrating these components prior to gas chromatographic analysis.
SCOPE
1.1 This test method covers the identification and simultaneous measurement of purgeable volatile organic compounds. It has been validated for treated drinking water, wastewater, and ground water. This test method is not limited to these particular aqueous matrices; however, the applicability of this test method to other aqueous matrices must be demonstrated.
1.2 This test method is applicable to a wide range of organic compounds that have sufficiently high volatility and low water solubility to be efficiently removed from water samples using purge and trap procedures. Table 1 lists the compounds that have been validated for this test method. This test method is not limited to the compounds listed in Table 1; however, the applicability of the test method to other compounds must be demonstrated.
1.3 Analyte concentrations up to approximately 200 μg/L may be determined without dilution of the sample. Analytes that are inefficiently purged from water will not be detected when present at low concentrations, but they can be measured with acceptable accuracy and precision when present in sufficient amounts.
1.4 Analytes that are not separated chromatographically, but that have different mass spectra and non-interfering quantitation ions, can be identified and measured in the same calibration mixture or water sample. Analytes that have very similar mass spectra cannot be individually identified and measured in the same calibration mixture or water sample unless they have different retention times. Coeluting compounds with very similar mass spectra, such as structural isomers, must be reported as an isomeric group or pair. Two of the three isomeric xylenes are examples of structural isomers that may not be resolved on the capillary column, and if not, must be reported as an isomeric pair.
1.5 It is the responsibility of the user to ensure the validity of this test method for untested matrices.
1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Dec-2018
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.06 - Methods for Analysis for Organic Substances in Water
Relations
- Effective Date
- 15-Dec-2018
- Effective Date
- 01-Apr-2024
- Effective Date
- 01-Apr-2024
- Effective Date
- 01-May-2020
- Effective Date
- 15-Dec-2017
- Effective Date
- 15-Dec-2017
- Effective Date
- 15-Jun-2012
- Effective Date
- 01-May-2011
- Effective Date
- 01-May-2011
- Effective Date
- 01-Mar-2010
- Effective Date
- 15-Jan-2008
- Effective Date
- 01-Mar-2007
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 15-Aug-2006
Overview
ASTM D5790-18 is a standard test method developed by ASTM International for the measurement of purgeable organic compounds in water using capillary column gas chromatography/mass spectrometry (GC/MS). This method is integral for analyzing volatile organic contaminants that may pose risks to public health and the environment. It has been validated for use with treated drinking water, wastewater, and groundwater, though its applicability can be demonstrated for other aqueous matrices.
The test method provides a robust framework for identifying and quantifying a wide range of purgeable volatile organic compounds (VOCs) that are efficiently extracted from water samples using purge and trap techniques. These compounds, often found as contaminants, can be harmful if present in significant concentrations in water sources.
Key Topics
- Purge and Trap Sampling: The method utilizes purge and trap sampling to concentrate volatile organic compounds from aqueous samples, enhancing the sensitivity and reliability of subsequent GC/MS analysis.
- Wide Applicability: Although validated for specific water matrices, the method can be extended to other types, provided its applicability is verified.
- Analyte Detection Range: The method can accurately measure analytes with concentrations up to approximately 200 μg/L without sample dilution.
- Chromatographic Separation: VOCs are separated using capillary columns, with identification and quantitation based on retention times and mass spectral data.
- Reporting Structural Isomers: Structural isomers that coelute but possess similar mass spectra are reported as isomeric groups or pairs, a practice critical to maintaining data integrity.
- Quality and Safety Requirements: The standard prescribes strict reagent purity, apparatus preparation, sample storage guidelines, and emphasizes compliance with safety and regulatory practices.
- Data Integrity: Use of calibration standards, internal standards, surrogate analytes, blank samples, and fortified samples ensures the accuracy and reliability of measurement results.
Applications
The ASTM D5790-18 standard is widely used across sectors where the assessment and control of volatile organic compound contamination in water is critical:
- Environmental Monitoring: Analyzing surface water, groundwater, and effluent to identify contamination sources and compliance with environmental regulations.
- Drinking Water Testing: Ensuring public safety by monitoring treated drinking water for harmful purgeable organic contaminants.
- Industrial Wastewater Management: Supporting wastewater treatment facilities and industrial dischargers in meeting regulatory discharge standards.
- Remediation Projects: Evaluating the effectiveness of site clean-up operations addressing volatile organic pollutants.
- Toxicity Characteristic Leaching Procedure (TCLP) Leachate Testing: Assessing landfill leachate as required by waste management regulations.
- Quality Assurance Laboratories: Routine use in labs supporting municipal, state, and private sector clients with regulatory compliance and risk assessment.
Related Standards
ASTM D5790-18 references and aligns with a range of supporting standards and guidelines to ensure methodological consistency and comprehensive VOC analysis:
- ASTM D1129 - Terminology Relating to Water
- ASTM D2777 - Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
- ASTM D3871 - Test Method for Purgeable Organic Compounds in Water Using Headspace Sampling
- ASTM D3973 - Test Method for Low-Molecular Weight Halogenated Hydrocarbons in Water
- ASTM E355 - Practice for Gas Chromatography Terms and Relationships
- Code of Federal Regulations 40 CFR Part 261 - Resource Conservation and Recovery Act (RCRA) requirements
ASTM D5790-18 is indispensable for laboratories and organizations focused on water quality, providing the technical rigor required for reliable detection of purgeable VOCs. Its implementation supports efforts to protect public health and the environment by ensuring the accurate identification and quantification of hazardous volatile organic contaminants in water sources.
Buy Documents
ASTM D5790-18 - Standard Test Method for Measurement of Purgeable Organic Compounds in Water by Capillary Column Gas Chromatography/Mass Spectrometry
REDLINE ASTM D5790-18 - Standard Test Method for Measurement of Purgeable Organic Compounds in Water by Capillary Column Gas Chromatography/Mass Spectrometry
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D5790-18 is a standard published by ASTM International. Its full title is "Standard Test Method for Measurement of Purgeable Organic Compounds in Water by Capillary Column Gas Chromatography/Mass Spectrometry". This standard covers: SIGNIFICANCE AND USE 5.1 Purgeable organic compounds have been identified as contaminants in treated drinking water, wastewater, ground water, and Toxicity Characteristic Leaching Procedure (TCLP) leachate. These contaminants may be harmful to the environment and to people. Purge and trap sampling is a generally applicable procedure for concentrating these components prior to gas chromatographic analysis. SCOPE 1.1 This test method covers the identification and simultaneous measurement of purgeable volatile organic compounds. It has been validated for treated drinking water, wastewater, and ground water. This test method is not limited to these particular aqueous matrices; however, the applicability of this test method to other aqueous matrices must be demonstrated. 1.2 This test method is applicable to a wide range of organic compounds that have sufficiently high volatility and low water solubility to be efficiently removed from water samples using purge and trap procedures. Table 1 lists the compounds that have been validated for this test method. This test method is not limited to the compounds listed in Table 1; however, the applicability of the test method to other compounds must be demonstrated. 1.3 Analyte concentrations up to approximately 200 μg/L may be determined without dilution of the sample. Analytes that are inefficiently purged from water will not be detected when present at low concentrations, but they can be measured with acceptable accuracy and precision when present in sufficient amounts. 1.4 Analytes that are not separated chromatographically, but that have different mass spectra and non-interfering quantitation ions, can be identified and measured in the same calibration mixture or water sample. Analytes that have very similar mass spectra cannot be individually identified and measured in the same calibration mixture or water sample unless they have different retention times. Coeluting compounds with very similar mass spectra, such as structural isomers, must be reported as an isomeric group or pair. Two of the three isomeric xylenes are examples of structural isomers that may not be resolved on the capillary column, and if not, must be reported as an isomeric pair. 1.5 It is the responsibility of the user to ensure the validity of this test method for untested matrices. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Purgeable organic compounds have been identified as contaminants in treated drinking water, wastewater, ground water, and Toxicity Characteristic Leaching Procedure (TCLP) leachate. These contaminants may be harmful to the environment and to people. Purge and trap sampling is a generally applicable procedure for concentrating these components prior to gas chromatographic analysis. SCOPE 1.1 This test method covers the identification and simultaneous measurement of purgeable volatile organic compounds. It has been validated for treated drinking water, wastewater, and ground water. This test method is not limited to these particular aqueous matrices; however, the applicability of this test method to other aqueous matrices must be demonstrated. 1.2 This test method is applicable to a wide range of organic compounds that have sufficiently high volatility and low water solubility to be efficiently removed from water samples using purge and trap procedures. Table 1 lists the compounds that have been validated for this test method. This test method is not limited to the compounds listed in Table 1; however, the applicability of the test method to other compounds must be demonstrated. 1.3 Analyte concentrations up to approximately 200 μg/L may be determined without dilution of the sample. Analytes that are inefficiently purged from water will not be detected when present at low concentrations, but they can be measured with acceptable accuracy and precision when present in sufficient amounts. 1.4 Analytes that are not separated chromatographically, but that have different mass spectra and non-interfering quantitation ions, can be identified and measured in the same calibration mixture or water sample. Analytes that have very similar mass spectra cannot be individually identified and measured in the same calibration mixture or water sample unless they have different retention times. Coeluting compounds with very similar mass spectra, such as structural isomers, must be reported as an isomeric group or pair. Two of the three isomeric xylenes are examples of structural isomers that may not be resolved on the capillary column, and if not, must be reported as an isomeric pair. 1.5 It is the responsibility of the user to ensure the validity of this test method for untested matrices. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D5790-18 is classified under the following ICS (International Classification for Standards) categories: 71.080.01 - Organic chemicals in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D5790-18 has the following relationships with other standards: It is inter standard links to ASTM D5790-95(2012), ASTM D3973-85(2024), ASTM D3871-84(2024), ASTM D1129-13(2020)e2, ASTM D3871-84(2017), ASTM D3973-85(2017), ASTM D2777-12, ASTM D3871-84(2011), ASTM D3973-85(2011), ASTM D1129-10, ASTM D2777-08, ASTM E355-96(2007), ASTM D1129-06a, ASTM D1129-06ae1, ASTM D2777-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D5790-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D5790 − 18
Standard Test Method for
Measurement of Purgeable Organic Compounds in Water by
Capillary Column Gas Chromatography/Mass Spectrometry
This standard is issued under the fixed designation D5790; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 1.5 It is the responsibility of the user to ensure the validity
of this test method for untested matrices.
1.1 This test method covers the identification and simulta-
1.6 The values stated in SI units are to be regarded as
neous measurement of purgeable volatile organic compounds.
standard. No other units of measurement are included in this
It has been validated for treated drinking water, wastewater,
standard.
and ground water. This test method is not limited to these
particular aqueous matrices; however, the applicability of this
1.7 This standard does not purport to address all of the
test method to other aqueous matrices must be demonstrated.
safety concerns, if any, associated with its use. It is the
responsibility of the user of this standard to establish appro-
1.2 Thistestmethodisapplicabletoawiderangeoforganic
priate safety, health, and environmental practices and deter-
compounds that have sufficiently high volatility and low water
mine the applicability of regulatory limitations prior to use.
solubility to be efficiently removed from water samples using
1.8 This international standard was developed in accor-
purge and trap procedures. Table 1 lists the compounds that
dance with internationally recognized principles on standard-
havebeenvalidatedforthistestmethod.Thistestmethodisnot
ization established in the Decision on Principles for the
limited to the compounds listed in Table 1; however, the
Development of International Standards, Guides and Recom-
applicability of the test method to other compounds must be
mendations issued by the World Trade Organization Technical
demonstrated.
Barriers to Trade (TBT) Committee.
1.3 Analyte concentrations up to approximately 200 µg/L
2. Referenced Documents
may be determined without dilution of the sample. Analytes
that are inefficiently purged from water will not be detected
2.1 ASTM Standards:
when present at low concentrations, but they can be measured
D1129Terminology Relating to Water
with acceptable accuracy and precision when present in suffi-
D2777Practice for Determination of Precision and Bias of
cient amounts.
Applicable Test Methods of Committee D19 on Water
D3871Test Method for Purgeable Organic Compounds in
1.4 Analytesthatarenotseparatedchromatographically,but
Water Using Headspace Sampling
that have different mass spectra and non-interfering quantita-
D3973TestMethodforLow-MolecularWeightHalogenated
tion ions, can be identified and measured in the same calibra-
Hydrocarbons in Water
tion mixture or water sample. Analytes that have very similar
E355PracticeforGasChromatographyTermsandRelation-
massspectracannotbeindividuallyidentifiedandmeasuredin
ships
the same calibration mixture or water sample unless they have
2.2 Other Documents:
different retention times. Coeluting compounds with very
Code of Federal Regulations40 CFR Part 261
similar mass spectra, such as structural isomers, must be
reportedasanisomericgrouporpair.Twoofthethreeisomeric
3. Terminology
xylenes are examples of structural isomers that may not be
3.1 Definitions:
resolved on the capillary column, and if not, must be reported
3.1.1 For definitions of terms used in this standard, refer to
as an isomeric pair.
Terminology D1129 and Practice E355.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
This test method is under the jurisdiction ofASTM Committee D19 on Water
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
andisthedirectresponsibilityofSubcommitteeD19.06onMethodsforAnalysisfor Standards volume information, refer to the standard’s Document Summary page on
Organic Substances in Water. the ASTM website.
Current edition approved Dec. 15, 2018. Published January 2019. Originally Available from U.S. Government Printing Office, Superintendent of
approved in 1995. Last previous edition approved in 2012 as D5790–95 (2012). Documents, 732 N. Capitol St., NW, Washington, DC 20401-0001, http://
DOI: 10.1520/D5790-18. www.access.gpo.gov.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5790 − 18
3.2 Definitions of Terms Specific to This Standard: standard, or other test compounds) used to evaluate the
3.2.1 calibration standard, n—a solution prepared from the performanceoftheinstrumentsystemwithrespecttoadefined
primarydilutionstandardsolutionandstockstandardsolutions set of test method criteria.
of the internal standards and surrogate analytes.
3.2.9 laboratory reagent blank, n—an aliquot of reagent
3.2.1.1 Discussion—The calibration standards are used to
water that is treated exactly as a sample including exposure to
calibrate the instrument response with respect to analyte
allglassware,equipment,solvents,reagents,internalstandards,
concentration.
and surrogates that are used with other samples.
3.2.2 field duplicates, n—two separate samples collected at 3.2.9.1 Discussion—The laboratory reagent blank is used to
the same time and place under identical circumstances and determine if test method analytes or other interferences are
treated exactly the same throughout field and laboratory present in the laboratory environment, the reagents, or the
procedures. apparatus.
3.2.2.1 Discussion—Analysis of field duplicates gives an
3.2.10 primary dilution standard solution, n—a solution of
indication of the precision associated with sample collection,
severalanalytespreparedinthelaboratoryfromstockstandard
preservation, and storage, as well as with laboratory proce-
solutionsanddilutedasneededtopreparecalibrationsolutions
dures.
and other needed analyte solutions.
3.2.3 field reagent blank, n—reagent water placed in a
3.2.11 purgeable organic, n—any organic material that is
samplecontainer,takentothefieldalongwiththesamples,and
removed from aqueous solution under the purging conditions
treated as a sample in all respects, including exposure to
described in this test method.
sampling site conditions, storage, preservation, and all analyti-
3.2.12 quality control sample, n—a sample matrix contain-
cal procedures.
ing test method analytes or a solution of method analytes in a
3.2.3.1 Discussion—The purpose of the field reagent blank
water-miscible solvent that is used to fortify reagent water or
istodetermineiftestmethodanalytesorotherinterferencesare
environmental samples.
present in the field environment.
3.2.12.1 Discussion—Thequalitycontrolsampleisobtained
3.2.4 internal standard, n—a pure analyte, that is not a
from a source external to the laboratory and is used to check
sample component, added to a solution in a known amount,
laboratoryperformancewithexternallypreparedtestmaterials.
that is used to measure the relative responses of other test
3.2.13 stock standard solution, n—a concentrated solution
method analytes and surrogates that are components of the
containing a single certified standard that is a test method
same solution.
analyte prepared in the laboratory with an assayed reference
3.2.5 laboratory duplicates, n—two sample aliquots taken
compound.
in the analytical laboratory and analyzed separately with
3.2.13.1 Discussion—Stock standard solutions are used to
identical procedures.
prepare primary dilution standards. Commercially available
3.2.5.1 Discussion—Analysis of laboratory duplicates gives
stock standard solutions may be used.
an indication of the precision associated with laboratory
3.2.14 surrogateanalyte,n—apureanalytethatisextremely
procedures, but not with sample collection, preservation, or
unlikely to be found in any sample, that is added to a sample
storage procedures.
aliquot in a known amount, and is measured with the same
3.2.6 laboratory-fortified blank, n—an aliquot of reagent
procedures used to measure other components.
watertowhichknownquantitiesofthetestmethodanalytesare
3.2.14.1 Discussion—The purpose of a surrogate analyte is
added in the laboratory.
to monitor test method performance with each sample.
3.2.6.1 Discussion—The laboratory-fortified blank is ana-
lyzed exactly like a sample, and its purpose is to determine
4. Summary of Test Method
whether the methodology is in control and whether the
4.1 Volatile organic compounds with low water-solubility
laboratory is capable of making accurate and precise measure-
are purged from the sample matrix by bubbling an inert gas
ments at the required detection limit.
through the aqueous sample. Purged sample components are
3.2.7 laboratory-fortifiedsamplematrix,n—analiquotofan
trapped in a tube containing suitable sorbent materials. When
environmental sample to which known quantities of the test
purgingiscomplete,thesorbenttubeisheatedandbackflushed
method analytes are added in the laboratory.
with inert gas to desorb the trapped sample components into a
3.2.7.1 Discussion—The laboratory-fortified sample matrix
capillary gas chromatography (GC) column interfaced to a
is analyzed exactly like a sample, and its purpose is to
mass spectrometer (MS). The GC column is temperature
determine whether or not the sample matrix or the addition of
programmed to separate the test method analytes which are
preservatives or dechlorinating agents to the sample contrib-
then detected with the MS. Compounds eluting from the GC
utes bias to the analytical results. The background concentra-
column are identified by comparing their measured mass
tions of the analytes in the sample matrix must be determined
spectra and retention times to reference spectra and retention
in a separate aliquot, and the measured values in the
times in a database. Reference spectra and retention times for
laboratory-fortified sample matrix must be corrected for back-
analytes are obtained by the measurement of calibration
ground concentrations.
standards under the same conditions used for the samples.The
3.2.8 laboratory performance check solution, n—a solution concentration of each identified component is measured by
of one or more compounds (analytes, surrogates, internal relating the MS response of the quantitation ion produced by
D5790 − 18
that compound to the MS response of the quantitation ion analyzed immediately after a sample containing higher con-
produced by a compound that is used as an internal standard. centrations of volatile organic compounds. Experience gained
Surrogate analytes, whose concentrations are known in every
from the test method validation has shown that there is a
sample, are measured with the same internal standard calibra-
carryover of approximately 2% of the concentration of each
tion procedure.
analyte from one sample to the next. The effect was observed
when samples containing 1 µg/L of analyte were analyzed
5. Significance and Use
immediately after samples containing 20 µg/L of analyte. For
5.1 Purgeable organic compounds have been identified as
that reason, when low concentrations of analytes are measured
contaminants in treated drinking water, wastewater, ground
in a sample, it is very important to examine the results of the
water, andToxicity Characteristic Leaching Procedure (TCLP)
preceding samples and interpret the low-concentration results
leachate. These contaminants may be harmful to the environ-
accordingly.Onepreventivetechniqueisbetween-samplerins-
ment and to people. Purge and trap sampling is a generally
ing of the purging apparatus and sample syringes with two
applicable procedure for concentrating these components prior
portionsofreagentwater.Afteranalysisofasamplecontaining
to gas chromatographic analysis.
high concentrations of volatile organic compounds, one or
6. Interferences
morelaboratoryreagentblanksshouldbeanalyzedtocheckfor
cross contamination. After analyzing a highly contaminated
6.1 During analysis, major contaminant sources are volatile
sample, it may be necessary to use methanol to clean the
materials in the laboratory and impurities in the inert purging
sample chamber, followed by heating in an oven at 105°C.
gas and in the sorbent trap. Avoid the use of plastic tubing or
thread sealants other than PTFE, and avoid the use of flow
6.3 Samples can be contaminated by diffusion of volatile
controllers with rubber components in the purging device.
organics through the septum seal into the sample during
These materials out-gas organic compounds that will be
shipment and storage. The analytical and sample storage area
concentrated in the trap during the purge operation. Analyses
should be isolated from all atmospheric sources of volatile
of laboratory reagent blanks provide information about the
organic compounds, otherwise random background levels may
presenceofcontaminants.Whenpotentialinterferingpeaksare
result. Since methylene chloride will permeate through PTFE
noted in laboratory reagent blanks, the analyst should change
tubing, all gas chromatography carrier gas lines and purge gas
the purge gas source and regenerate the molecular sieve purge
plumbing should be PTFE free. Personnel who have been
gas filter. Reagents should also be checked for the presence of
working directly with solvents such as those used in liquid/
contaminants. Subtracting blank values from sample results is
liquid extraction procedures should not be allowed into the
not permitted.
analytical area until they have washed and changed their
6.2 Interfering contamination may occur when a sample
clothing.
containinglowconcentrationsofvolatileorganiccompoundsis
TABLE 1 Compounds Validated for This Test Method
A
Compound CAS Registry Number Primary Quantitation Ion Secondary Quantitation Ion Approximate Elution Order
Benzene 71-43-2 78 77 20
Bromobenzene 108-86-1 156 77, 158 44
Bromochloromethane 74-97-5 128 49, 130 16
Bromodichloromethane 75-27-4 83 85, 127 25
Bromoform 75-25-2 173 175, 252 41
Bromomethane 74-83-9 94 96 4
n-butylbenzene 104-51-8 91 134 57
sec-butylbenzene 135-98-8 105 134 53
tert-butylbenzene 98-06-6 119 91 52
Carbon disulfide 75-15-0 76 78 8
Carbon tetrachloride 56-23-5 117 119 19
Chlorobenzene 108-90-7 112 77, 114 35
Chloroethane 75-00-3 64 66 5
Chloroform 67-66-3 83 85 15
Chloromethane 74-87-3 50 52 2
2-chlorotoluene 95-49-8 91 126 47
4-chlorotoluene 106-43-4 91 126 50
Dibromochloromethane 124-48-1 129 127 33
1,2-dibromo-3-chloropropane 96-12-8 75 155, 157 60
1,2-dibromoethane 106-93-4 107 109, 188 34
Dibromomethane 74-95-3 93 95, 174 26
1,2-dichlorobenzene 95-50-1 146 111, 148 58
1,3-dichlorobenzene 541-73-1 146 111, 148 54
1,4-dichlorobenzene 106-46-7 146 111, 148 56
trans-1,4-dichloro-2-butene 110-57-6 75 53, 89 48
Dichlorodifluoromethane 75-71-8 85 87 1
1,1-dichloroethane 75-34-3 63 65, 83 11
1,2-dichloroethane 107-06-2 62 98 21
1,1-dichloroethene 75-35-4 96 61, 63 7
cis-1,2-dichloroethene 156-59-4 96 61, 98 13
trans-1,2-dichloroethene 156-60-5 96 61, 98 10
D5790 − 18
TABLE 1 Continued
A
Compound CAS Registry Number Primary Quantitation Ion Secondary Quantitation Ion Approximate Elution Order
1,2-dichloropropane 78-87-5 63 112 24
1,3-dichloropropane 142-28-9 76 78 32
2,2-dichloropropane 590-20-7 77 97 12
1,1-dichloropropene 563-58-6 75 110, 77 18
cis-1,3-dichloropropene 10061-01-5 75 110 27
trans-1,3-dichloropropene 10061-02-6 75 110 29
Ethylbenzene 100-41-4 91 106 36
Hexachlorobutadiene 87-68-3 225 260 62
Hexachloroethane 67-72-1 117 201 59
Isopropylbenzene 98-82-8 105 120 42
p-isopropyltoluene 99-87-6 119 134, 91 55
Methylene chloride 75-09-2 84 86, 49 9
Methyl-tert-butylether 1634-04-4 73 57 14
Methyl-isobutylketone 108-10-1 43 58, 100 23
Naphthalene 91-20-3 128 63
n-propylbenzene 103-65-1 91 120 46
Styrene 100-42-5 104 78 40
1,2,3,4-tetrachlorobenzene 634-66-2 216 108 66
1,2,4,5-tetrachlorobenzene 95-94-3 216 108 65
1,1,1,2-tetrachloroethane 630-20-6 131 133, 119 37
1,1,2,2-tetrachloroethane 79-34-5 83 131, 85 43
Tetrachloroethene 127-18-4 166 168, 129 31
Toluene 108-88-3 92 91 28
1,2,3-trichlorobenzene 87-61-6 180 182 64
1,2,4-trichlorobenzene 120-82-1 180 182 61
1,1,1-trichloroethane 71-55-6 97 99, 61 17
1,1,2-trichloroethane 79-00-5 83 97, 85 30
Trichloroethene 79-01-6 95 130, 132 22
Trichlorofluoromethane 75-69-4 101 103 6
1,2,3-trichloropropane 96-18-4 75 77 45
1,2,4-trimethylbenzene 95-63-6 105 120 51
1,3,5-trimethylbenzene 108-67-8 105 120 49
Vinyl chloride 75-01-4 62 64 3
o-xylene 95-47-6 106 91 39
m-xylene 108-38-3 106 91 38
p-xylene 106-42-3 106 91 38
B
Suggested Internal Standards
Chlorobenzene-d5 3114-55-4 117 82, 119
1,2-dichlorobenzene-d4 2199-69-1 152 115, 150
Fluorobenzene 462-06-6 96 70, 77
Suggested Surrogates
4-bromofluorobenzene 460-00-4 95 174, 176
1,2-dichloroethane-d4 17060-07-0 65 102
Toluene-d8 2037-26-5 98 70, 100
A
Chemical Abstracts Service (CAS).
B
Appendix X2 is a table of the interlaboratory collaborative study analytes and surrogates with internal standards assignments.
7. Apparatus should be installed at the base of the sample chamber so that
the purge gas passes through the water column as finely
7.1 SampleContainers—40to120-mLscrew-capglassvials
dividedbubbleswithadiameterof<3mmattheorigin.Needle
equipped with a PTFE-faced silicone septum. The vials must
spargers may be used, however, the purge gas must be
contain at least twice the volume of water required for the
introduced at a point about 5 mm from the base of the water
analysis. Prior to use, wash vials with detergent and rinse with
column.
tap and reagent water. Allow the vials and septa to air dry at
7.2.2 Trap:
room temperature, place in an oven at 105°C for 1 h, then
7.2.2.1 The trap shall be at least 25 cm long and have an
remove and allow to cool in an area known to be free of
insidediameterofatleast0.267cm.Startingfromtheinlet,the
organics. Purchased, pre-cleaned glass vials may also be used.
trap should contain 1.0 cm of methyl silicone coated packing
and the following amounts of adsorbents: ⁄3 of 2,6-
7.2 Purge and Trap System—The purge and trap system
1 1
diphenyleneoxidepolymer(Tenax ), ⁄3ofsilicagel,and ⁄3of
consists of three basic components: purging device, trap, and
coconut charcoal. If it is not necessary to determine
desorber. Systems are commercially available from several
dichlorodifluoromethane, the charcoal can be eliminated and
sources that meet all of the following specifications.
7.2.1 The all-glass purging device should be designed to
Thesolesourceofsupplyoftheapparatusknowntothecommitteeatthistime
accept eithera5ora 25-mL sample volume. Equipment
is Enka Research Institute-Arnhem, College Station, TX. If you are aware of
designed for either single- or multiple-purging devices is
alternative suppliers, please provide this information to ASTM International
acceptable.Gaseousvolumesabovethesamplemustbekeptto
Headquarters.Your comments will receive careful consideration at a meeting of the
a minimum to eliminate dead volume effects. A glass frit responsible technical committee, which you may attend.
D5790 − 18
the polymer increased to fill two thirds of the trap. Before 7.3.2.1 Column 1—60 m by 0.75 mm inside diameter
initial use, the trap should be conditioned overnight at 225°C VOCOL glass wide-bore capillary with a 1.5-µm film thick-
by backflushing with an inert gas flow of at least 20 mL/min. ness.
Vent the trap effluent to the room rather than to the analytical 7.3.2.2 Column 2—30 m by 0.53 mm inside diameter
DB-624 fused silica capillary with a 3-µm film thickness.
column. Prior to daily use, the trap should be conditioned for
10 min at 225°C with backflushing.The trap may be vented to 7.3.2.3 Column 3—75 m by 0.53 mm inside diameter
DB-624 fused silica capillary with a 3-µm film thickness.
the analytical column during daily conditioning, provided that
the column is run through the temperature program prior to 7.3.2.4 Column 4—30mby0.32mminsidediameterDB-5
5,6
fused silica capillary with a 1-µm film thickness.
analysis of samples.
7.3.2.5 Column 5—105 m by 0.53 mm inside diameter
7.2.2.2 The use of the methyl silicone coated packing is
Rt -502.2 fused silica capillary with a 3-µm film thickness.
x
recommended, but not mandatory. The packing serves the
7.3.2.6 For further discussion of columns and inserts see
purposeofprotectingtheTenaxadsorbantfromaerosols.Since
Refs (7) and (8).
it may adsorb higher boiling compounds, it must be fully
7.3.3 Interfaces Between the GC and MS—The interface
enclosed within the heated zone of the trap. Silanized glass
used depends on the column selected and the gas flow rate.
wool may be used as a spacer at the trap inlet to eliminate
7.3.3.1 The wide-bore Columns 1, 2, 3, and 5 have the
potential cold spots.
capacity to accept the standard gas flows from the trap during
7.2.2.3 The presence of charcoal in the trap may interfere
thermal desorption, and chromatography can begin with the
with the analysis of ketones. When analyzing for ketones, the
onset of thermal desorption. Depending on the pumping
charcoalshouldbeeliminatedandthepolymerincreasedtofill
capacity of the MS, an additional interface between the end of
two thirds of the trap, if dichlorodifluoromethane is not being
the column and the MS may be required. An open split
analyzed.
interface, an all-glass jet separator, or a cryogenic device are
7.2.2.4 Other traps are commercially available which may
acceptable interfaces. Any interface can be used if the perfor-
besuitableforuse.Theequivalencyoftheirperformancemust
mance specifications described in this test method can be
be demonstrated prior to use.
achieved.The end of the transfer line after the interface, or the
7.2.3 The equipment must be capable of rapidly heating the
end of the analytical column if no interface is used, should be
trap to the trap desorb temperature either prior to or at the
placed within a few millimetres of the MS ion source.
beginning of the flow of desorption gas. If the trap has a
7.3.3.2 NarrowboreColumn4maynotbeabletoacceptthe
polymer section, the polymer section of the trap should not be
thermal desorption gas flow, therefore, a cryogenic interface
heatedhigherthan225°C,orthelifeexpectancyofthetrapwill
would be required. This interface condenses the desorbed
decrease. Trap failure is characterized by a pressure drop in
sample components at liquid nitrogen temperature and allows
excess of 20.684 kPa across the trap during purging, by poor
the helium gas to pass through to an exit. The condensed
bromoform sensitivities, or by increased water background.
components are frozen in a narrow band on an uncoated fused
7.2.4 The transfer line between the desorber and the GC silicaprecolumn (9).Whenallcomponentshavebeendesorbed
from the trap, the interface is rapidly heated under a stream of
must be heated within the range of 100 to 150°C.
carrier gas to transfer the analytes to the analytical column.
7.3 Gas Chromatography/Mass Spectrometer/Data System
Alternatively, a subambient oven may be used instead of a
(GC/MS/DS):
cryogenic interface.
7.3.1 TheGCmustbecapableoftemperatureprogramming
7.3.4 The mass spectrometer must be capable of electron
andshouldbeequippedwithvariable-constantdifferentialflow
ionization at a nominal electron energy of 70 eV. The spec-
controllers so that the column flow rate will remain near
trometermustbecapableofscanningfrom48to260amuwith
constant throughout desorption and temperature program op-
a complete scan cycle time (including scan overhead) of2sor
eration. For several of the chromatographic columns listed as
less (scan cycle time=total MS data acquisition time in
below, the column oven must be cooled to 10°C; therefore, a
secondsdividedbynumberofscansinthechromatogram).The
sub-ambient oven controller may be required. One of the
spectrometer must produce a mass spectrum that meets all
columns listed as follows does not require subambient condi-
tions. If syringe injections of 4-bromofluorobenzene (BFB)
will be done, a high efficiency injection port is required. 7
Thesolesourceofsupplyoftheapparatusknowntothecommitteeatthistime
is Supelco, Inc., Bellafonte, PA. If you are aware of alternative suppliers, please
7.3.2 Capillary Gas Chromatography Columns—Any gas
provide this information toASTM International Headquarters.Your comments will
chromatography column that meets the performance specifica-
receive careful consideration at a meeting of the responsible technical committee,
tions of this test method may be used. Separations of the
which you may attend.
Thesolesourceofsupplyoftheapparatusknowntothecommitteeatthistime
calibration mixture must be equivalent or better than those
isJ&WScientific,Inc.,Folsom,CA.Ifyouareawareofalternativesuppliers,please
described in this test method. As examples, the following
provide this information toASTM International Headquarters.Your comments will
columns have been found to be suitable: 1
receive careful consideration at a meeting of the responsible technical committee,
which you may attend.
Thesolesourceofsupplyoftheapparatusknowntothecommitteeatthistime
is Restek Corp., Bellefonte, PA. If you are aware of alternative suppliers, please
5 6
For further discussion on Tenax traps see the Refs (1-6). provide this information toASTM International Headquarters.Your comments will
6 1
The boldface numbers given in parentheses refer to a list of references at the receive careful consideration at a meeting of the responsible technical committee,
end of the text. which you may attend.
D5790 − 18
criteriainTable2when25ngorlessof4-bromofluorobenzene all reagents shall conform to the specifications of the Commit-
(BFB) is introduced into the GC/MS. An average spectrum tee on Analytical Reagents of the American Chemical Soci-
across the BFB GC peak may be used to test instrument ety.
performance.
8.2 Purity of Water—Unless otherwise indicated, references
to water shall be understood to mean reagent water demon-
NOTE 1—If this test method is used for analytes with mass fragments
below48amu(forexample,manyketonesexhibitacharacteristic43amu
strated to be free of the analytes of interest.
massfragment),themassrangemaybemodified.Allcalibrationstandards
8.2.1 Reagent water may be generated by passing tap water
must be analyzed using the same mass range as the samples.
through a carbon filter bed containing about 453 g of activated
NOTE2—ThecriteriainTable2forBFBwereusedforthistestmethod
carbon.
validation. Other criteria, such as those provided in the United States
Environmental Protection Agency 1990 Contract Laboratory Program 8.2.2 A water purification system may be used to generate
Statement of Work, are available. If other mass spectrometer tuning
reagent water.
criteria are used, the precision and bias results presented in Section 15 of
8.2.3 Reagent water may be prepared by boiling distilled
this test method may not apply. Therefore, the applicability of other BFB
water for 15 min. Subsequently, while maintaining the tem-
criteria to the test method must be demonstrated by the user.
perature at 90°C, bubble a contaminant-free inert gas through
7.3.5 Aninterfaceddatasystemisrequiredtoacquire,store,
the water for 1 h. While still hot, transfer the water to a
reduce, and output mass spectral data. The computer software
narrow-mouth screw-cap bottle and seal with a PTFE-lined
shouldhavethecapabilityofprocessingstoredGC/MSdataby
septum and cap.
recognizing a GC peak within any given retention time
8.3 Trap Packing Materials:
window, comparing the mass spectra from the GC peak with
8.3.1 2,6-Diphenylene Oxide Polymer, 60/80 mesh, chro-
spectraldatainauser-createddatabase,andgeneratingalistof
matographic grade, or equivalent.
tentativelyidentifiedcompoundswiththeirretentiontimesand
8.3.2 Methyl Silicone Packing (Optional), OV-1 (3%) on
scan numbers. The software must allow integration of the ion
Chromosorb W, 60/80 mesh, or equivalent.
abundance of any specific ion between specified time or scan
8.3.3 Silica Gel, 35/60 mesh.
number limits. The software should also allow calculation of
8.3.4 Coconut Charcoal, 20/40 mesh.
response factors as defined in 11.2.6 or construction of a
8.3.5 Alternatetrapsmaybeused,sotrappackingmaterials
second or third order regression calibration curve, calculation
may change. However, if using an alternative trap, the QC
of response factor statistics, and calculation of concentrations
criteria of the method must be met or exceeded.
of analytes using either the calibration curve or the equation in
12.1.1.
8.4 Methanol,purgeandtrapgrade,demonstratedtobefree
of analytes.
7.4 Syringe and Syringe Valves:
7.4.1 Glass Hypodermic Syringes, two, 5 to 25 mL, with
8.5 Hydrochloric Acid (1+1)—Carefully add measured vol-
Luer-Lok tip, depending on the sample volume used.
umeofconcentratedHCl(spgr1.19)toequalvolumeofwater.
7.4.2 Two-Way Syringe Valves, three, with Luer ends.
8.6 Vinyl Chloride—Certified mixtures of vinyl chloride in
7.4.3 25 µL Microsyringe, one, witha5cmby 0.15 mm
nitrogen and pure vinyl chloride are commercially available.
inside diameter, 22° bevel needle.
8.7 Ascorbic Acid, granular.
7.4.4 Microsyringes, 10 and 100 µL.
7.4.5 Syringes, 0.5, 1.0, and 5 mL, gas-tight with shut-off
8.8 pH Test Paper, capable of measuring pH 2 with a
valve.
sensitivity of at least 0.5 pH unit.
7.5 Bottles:
8.9 Standard Solutions, Stock—These solutions may be
7.5.1 Standard Solution Storage Containers, 15-mL glass
purchasedascertifiedsolutionsorpreparedfrompurestandard
bottles with PTFE-lined screw caps.
materials using the following procedures. One of these solu-
tions is required for every analyte of concern, every surrogate,
8. Reagents and Materials
and the internal standard. A useful working concentration is
about 1 to 5 µg/µL.
8.1 Purity of Reagents—Reagent grade chemicals shall be
used in all tests. Unless otherwise indicated, it is intended that
Reagent Chemicals, American Chemical Society Specifications, American
Chemical Society, Washington, DC. For suggestions on the testing of reagents not
listed by the American Chemical Society, see Analar Standards for Laboratory
TABLE 2 Ion Abundance Criteria for 4-Bromofluorobenzene
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
(BFB)
and National Formulary,U.S.PharmaceuticalConvention,Inc.(USPC),Rockville,
MD.
Mass Relative Abundance Criteria
Thesolesourceofsupplyoftheapparatusknowntothecommitteeatthistime
50 15 to 40 % of mass 95
is Ohio Valley Specialty Chemical Co., Cincinnati, OH. If you are aware of
75 30 to 60 % of mass 95
alternative suppliers, please provide this information to ASTM International
95 base peak, 100 % relative abundance
Headquarters.Your comments will receive careful consideration at a meeting of the
96 5 to 9 % of mass 95
responsible technical committee, which you may attend.
173 less than 2 % of mass 174
Thesolesourceofsupplyoftheapparatusknowntothecommitteeatthistime
174 greater than 50 % of mass 95
is Johns-Manville Products Corp., Edison, NJ. If you are aware of alternative
175 5 to 9 % of mass 174
suppliers, please provide this information to ASTM International Headquarters.
176 greater than 95 % but less than 101 % of mass 174
177 5 to 9 % of mass 176 Your comments will receive careful consideration at a meeting of the responsible
technical committee, which you may attend.
D5790 − 18
8.9.1 Placeabout9.8mLofmethanolintoa10-mLground- 8.11.2 A solution of the internal standard alone is required
glass stoppered volumetric flask. Allow the flask to stand, topreparecalibrationstandardsandlaboratory-fortifiedblanks.
unstoppered, for about 10 min or until the alcohol-wetted The internal standard should be in methanol at a concentration
surfaces inside the neck of the flask have dried, and weigh to of 5 µg/mL.
the nearest 0.1 mg.
8.12 Laboratory Reagent Blank—Fill a 5-mL (or 25-mL)
8.9.2 If the analyte is a liquid at room temperature, use a
syringe with reagent water and adjust to the mark with no air
100-µLsyringeandimmediatelyaddtwoormoredropsofpure
bubbles.Inject10µLofthefortificationsolutioncontainingthe
standard material to the flask. Be sure that the reference
internal standard and surrogates through the Luer Lok valve
standard falls directly into the alcohol without contacting the
into the reagent water.Transfer the laboratory reagent blank to
neckoftheflask.Iftheanalyteisagasatroomtemperature,fill
the purging device as described in 12.1.3.
a 5-mL valved gas-tight syringe with the standard to the
8.13 Laboratory-Fortified Blank—Prepare this exactly like
5.0-mL mark, lower the needle to 5 mm above the methanol
a calibration standard (see 8.14.2). This is a calibration
meniscus, and slowly inject the standard into the neck area of
standard that is treated as a sample.
the flask. The gas will rapidly dissolve in the methanol.
8.9.3 Reweigh, dilute to volume, stopper, then mix by
8.14 Calibration Standards:
invertingtheflaskseveraltimes.Calculatetheconcentrationin 8.14.1 The number of calibration standards needed depends
micrograms per microlitre from the net gain in weight. When
on the calibration range desired. A minimum of three calibra-
compound purity is certified at 96% or greater, the weight can tion standards is required to calibrate a range of a factor of 20
beusedwithoutcorrectiontocalculatetheconcentrationofthe
inconcentration.Forafactorof50,useatleastfourstandards,
stock standard. and for a factor of 100 at least five standards. The calibration
8.9.4 Store stock standard solutions in 15-mL bottles standards should contain each analyte of concern and each
equipped with PTFE-lined screw caps. Methanol solutions surrogate at concentrations that define the range of the test
prepared from liquid analytes are stable for at least 4 weeks method. Every calibration standard contains the internal stan-
whenstoredat4°C.Methanolsolutionspreparedfromgaseous dard at the same concentration (5 µg/L suggested for a 5-mL
analytes are not stable for more than one week when stored at sample, 1 µg/L suggested for a 25-mL sample).
<0°C; at room temperature, they must be discarded after one 8.14.2 To prepare a calibration standard, add an appropriate
day.
volumeofaprimarydilutionstandard(containinganalytesand
surrogates)toanaliquotofreagentwaterinavolumetricflask.
8.10 Primary Dilution Standards—Use stock standard solu-
Use a microsyringe and rapidly inject the methanol solutions
tionstoprepareprimarydilutionstandardsolutionsthatcontain
into the expanded area of the filled volumetric flask. Remove
all the analytes of concern and the surrogates (but not the
the needle as quickly as possible after injection. Mix by
internal standard) in methanol. The primary dilution standards
inverting the flask three times only. Discard the contents
should be prepared at concentrations that can be easily diluted
contained in the neck of the flask using a disposable pipet.
to prepare aqueous calibration solutions that will bracket the
Aqueous standards are not stable in a volumetric flask and
working concentration range. Store the primary dilution stan-
should be discarded after 1 h unless transferred to a sample
dard solutions with minimal headspace and check frequently
bottle and sealed immediately. Alternatively, the calibration
for signs of deterioration or evaporation, especially just before
standard may be prepared ina5or 25-mL syringe.
preparing calibration solutions. Storage times described for
stock standard solutions in 8.9.4 also apply to primary dilution
9. Hazards
standard solutions.
9.1 Thetoxicityorcarcinogenicityofchemicalsusedinthis
8.11 Solutions for Internal Standard and Surrogates:
test method has not been precisely defined; each chemical
8.11.1 A solution containing the internal standards and the
should be treated as a potential health hazard, and exposure to
surrogates is required to prepare laboratory reagent blanks and
these chemicals should be minimized. Each laboratory is
to fortify each sample. It is also used as a laboratory perfor-
responsible for maintaining awareness of OSHA regulations
mance check solution. Prepare a solution containing the
regarding safe handling of chemicals used in this test method.
desired internal standards and surrogates in methanol. A
number of appropriate internal standards and surrogates are 9.2 The following test method analytes have been tenta-
listed in Table 1. Appendix X2 contains a list of analytes and tively classified as known or suspected human or mammalian
surrogates with assigned internal standards as were used in the carcinogens: benzene, carbon tetrachloride, 1,4-
collaborative study. The concentration of this solution should dichlorobenzene,1,2-dichloroethane, hexachlorobutadiene,
be made as appropriate for the desired calibration range and hexachloroethane, 1,1,2,2-tetrachloroethane, 1,1,2-
expected sample concentration, in order to minimize the trichloroethane, chloroform, 1,2-dibromoethane,
amount of methanol added to the sample. For example, if the tetrachloroethene, trichloroethene, and vinyl chloride. Pure
fortification solution is prepared at a concentration of 5 µg/mL standard materials and stock standard solutions of these com-
of each species, a 5-µL aliquot of this solution added to a poundsshouldbehandledinawell-ventilatedhood.ANational
25-mLwater sample volume gives concentrations of 1 µg/Lof Institute for Occupational Safety and Health/Mine Safety and
each species and a 5-µL aliquot of this solution added to a Health Administration (NIOSH/MSHA) approved toxic gas
5-mLwater sample volume gives a concentration of 5 µg/Lof respirator should be worn when the analyst handles high
each. concentrations of these toxic compounds.
D5790 − 18
10. Sample Collection, Preservation, and Storage bottles. Wherever a set of samples is shipped and stored, it is
accompanied by appropriate blanks.
10.1 Sample Collection, Dechlorination, and Preservation:
10.3.2 Use the same procedures used for samples to add
10.1.1 InordertodeterminetheappropriatequantityofHCl
ascorbic acid and HCl to the field reagent blanks.
(1+1) to be added to the samples, collect a pre-sample of
known volume.
11. Calibration and Standardization
10.1.1.1 Add one drop of HCl (1+1) for every 10 mL of
11.1 Demonstrationanddocumentationofacceptableinitial
pre-sample volume. Check the pH using pH test paper. If the
calibration for compounds of interest is required before any
pH is not less than 2, add more HCl (1+1) to determine the
samples are analyzed and is required intermittently throughout
amount required to get the pH below 2.
sample analysis as dictated by results of continuing calibration
10.1.1.2 If the pre-sample foams upon addition of HCl
checks. After initial calibration is successful, a continuing
(1+1), do not use the HCl (1+1) preservative. Instead, refrig-
calibration check is required at the beginning of each 12-h
erate the sample as described in 10.1.5 and analyze as soon as
period during which analyses are performed. Additional peri-
possible within the holding time as described in 10.2.2.
odic calibration checks are good laboratory practice. The
10.1.2 Samples should be collected in duplicate. It may be
criteria in this section were used for the method validation.
desirable to collect additional samples for screening or other
Other criteria may be more appropriate in a given situation
purposes.Fillthesamplebottlestooverflowing,takingcarenot
depending on the data quality objectives.
to flush out the preservatives. No air bubbles should pass
through the sample as the bottle is filled or be trapped in the 11.2 Initial Calibration:
sample when the bottle is sealed. Seal the sample bottles, 11.2.1 Calibrate the mass and abundance scales of the MS
PTFE-face down, and shake vigorously for 1 min. with calibration compounds and procedures prescribed by the
manufacturer with any modifications necessary to meet the
10.1.2.1 In order to preserve the sample against biological
requirements in 11.2.2.
degradation, add the appropriate quantity of HCl (1+1) as
11.2.2 Introduce 25 ng of BFB into the GC, either by
determined in 10.1.1.1 to the sample bottle before filling.
purging a laboratory reagent blank or making a syringe
10.1.2.2 If the samples are suspected to contain residual
injection, and acquire mass spectra for m/z 48–260 at 70 eV
chlorine, and if measurements of the concentrations of disin-
(see Note 1). Use the purging procedure or GC conditions
fection by-products (for example, trihalomethanes, etc.) are
provided in Section 12, or both. If the spectrum does not meet
desired, add about 25 mg of ascorbic acid to the sample bottle
all criteria in Table 2, retune the MS and adjust to meet all
before filling.
criteria before proceeding with calibration (see Note 2). Use a
10.1.3 When sampling from a water tap, open the tap and
representative spectrum across the GC peak to evaluate the
allow the system to flush until the water temperature has
performance of the system.
stabilized (usually about 10 min).Adjust the flow to about 500
11.2.3 Purge a medium calibration solution, for example 10
mL/minandcollectduplicatesamplesfromtheflowingstream.
to 20 µg/L, using the procedure given in Section 12.
10.1.4 When sampling from an open body of water, fill a
11.2.4 Performance Criteria for the Medium Calibration:
960-mL wide-mouth bottle or 1-L beaker with sample from a
11.2.4.1 GC Performance—Good column performance will
representative area, and carefully fill duplicate sample bottles
produce symmetrical peaks with minimum tailing for most
from the container.
compounds.Ifpeaksarebroad,orsensitivityispoor,see11.3.6
10.1.5 The samples must be chilled to 4°C on the day of
for some possible remedial actions.
collection and must be maintained at that temperature until
11.2.4.2 MS Sensitivity—The GC/MS/DS peak identifica-
analysis. Field samples that will not be received at the
tion software should be able to recognize a GC peak in the
laboratory on the day of collection must be packaged for
appropriate retention time window for each of the compounds
shipment with sufficient ice to ensure that they will be at 4°C
in the calibration solution, and make correct tentative identifi-
on arrival at the laboratory.
cations. If fewer than 99% of the compounds are recognized,
10.2 Sample Storage:
system maintenance is required.
10.2.1 Store samples at 4°C until analysis. The sample
11.2.5 If all performance criteria are met, purge an aliquot
storage area must be free of organic solvent vapors. of each of the other calibration standards using the same
GC/MS conditions.
NOTE 3—If analyzing for light-sensitive analytes, such as some
11.2.6 Calculate a relative response factor (RRF) for each
halogenated compounds, the samples should be stored in the dark or in
analyte and surrogate for each calibration standard. Use a
amber glass bottles.
minimum of one internal standard. A number of appropriate
10.2.2 Analyze all samples within 14 days of collection.
internal standards are listed in Table 1. In complex matrices,
10.3 Field Reagent Blanks:
such as wastewater, more than one internal standard may be
10.3.1 Duplicatefieldreagentblanksmustbehandledalong desirable. Table 1 contains suggested quantitation ions for all
with each sample set, which is composed of the samples compounds. If there is significant interference with a primary
collected from the same general sample site at approximately ion, then a secondary or alternative ion should be selected for
the same time.At the laboratory, fill field blank sample bottles quantitation. Experience gained from the method validation
withwater,seal,andshiptothesamplingsitealongwithempty has shown that the use of these suggested ions and the
sample bottles and back to the laboratory with filled sample suggested internal standards listed in Appendix Appendix X2
D5790 − 18
minimizes method interferences. The calculation of RRF is requirecleaningoftheMSionsource,orothermaintenanceas
supported in acceptable GC/MS data system software. The indicated in 11.3.6, and recalibration. Control charts are useful
RRF is a unitless number, but units used to express quantities aids in documenting sys
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D5790 − 95 (Reapproved 2012) D5790 − 18
Standard Test Method for
Measurement of Purgeable Organic Compounds in Water by
Capillary Column Gas Chromatography/Mass Spectrometry
This standard is issued under the fixed designation D5790; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method covers the identification and simultaneous measurement of purgeable volatile organic compounds. It has
been validated for treated drinking water, wastewater, and ground water. This test method is not limited to these particular aqueous
matrices; however, the applicability of this test method to other aqueous matrices must be demonstrated.
1.2 This test method is applicable to a wide range of organic compounds that have sufficiently high volatility and low water
solubility to be efficiently removed from water samples using purge and trap procedures. Table 1 lists the compounds that have
been validated for this test method. This test method is not limited to the compounds listed in Table 1; however, the applicability
of the test method to other compounds must be demonstrated.
1.3 Analyte concentrations up to approximately 200 μg/L may be determined without dilution of the sample. Analytes that are
inefficiently purged from water will not be detected when present at low concentrations, but they can be measured with acceptable
accuracy and precision when present in sufficient amounts.
1.4 Analytes that are not separated chromatographically, but that have different mass spectra and noninterferingnon-interfering
quantitation ions, can be identified and measured in the same calibration mixture or water sample. Analytes that have very similar
mass spectra cannot be individually identified and measured in the same calibration mixture or water sample unless they have
different retention times. Coeluting compounds with very similar mass spectra, such as structural isomers, must be reported as an
isomeric group or pair. Two of the three isomeric xylenes are examples of structural isomers that may not be resolved on the
capillary column, and if not, must be reported as an isomeric pair.
1.5 It is the responsibility of the user to ensure the validity of this test method for untested matrices.
1.6 The values stated in SI units are to be regarded as the standard. The values given in parentheses are for information only.
standard. No other units of measurement are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1129 Terminology Relating to Water
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D3871 Test Method for Purgeable Organic Compounds in Water Using Headspace Sampling
D3973 Test Method for Low-Molecular Weight Halogenated Hydrocarbons in Water
D4210 Practice for Intralaboratory Quality Control Procedures and a Discussion on Reporting Low-Level Data (Withdrawn
2002)
This test method is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.06 on Methods for Analysis for
Organic Substances in Water.
Current edition approved June 15, 2012Dec. 15, 2018. Published June 2012January 2019. Originally approved in 1995. Last previous edition approved in 20062012 as
D5790 – 95 (2012). (2006). DOI: 10.1520/D5790-95R12.10.1520/D5790-18.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5790 − 18
E355 Practice for Gas Chromatography Terms and Relationships
2.2 Other Document:Documents:
Code of Federal Regulations 40 CFR Part 261
3. Terminology
3.1 Definitions—Definitions: For definitions of terms used in this test method, refer to Definitions D1129 and Practice E355.
3.1.1 For definitions of terms used in this standard, refer to Terminology D1129 and Practice E355.
Available from the Superintendent of Documents, U.S. Government Printing Office, Washington, DC 20402.Superintendent of Documents, 732 N. Capitol St., NW,
Washington, DC 20401-0001, http://www.access.gpo.gov.
D5790 − 18
3.2 Definitions of Terms Specific to This Standard:
3.2.1 calibration standard—standard, n—a solution prepared from the primary dilution standard solution and stock standard
solutions of the internal standards and surrogate analytes. The calibration standards are used to calibrate the instrument response
with respect to analyte concentration.
3.2.1.1 Discussion—
The calibration standards are used to calibrate the instrument response with respect to analyte concentration.
3.2.2 field duplicates, —n—two separate samples collected at the same time and place under identical circumstances and treated
exactly the same throughout field and laboratory procedures. Analysis of field duplicates gives an indication of the precision
associated with sample collection, preservation, and storage, as well as with laboratory procedures.
3.2.2.1 Discussion—
Analysis of field duplicates gives an indication of the precision associated with sample collection, preservation, and storage, as well
as with laboratory procedures.
3.2.3 field reagent blank—blank, n—reagent water placed in a sample container, taken to the field along with the samples, and
treated as a sample in all respects, including exposure to sampling site conditions, storage, preservation, and all analytical
procedures. The purpose of the field reagent blank is to determine if test method analytes or other interferences are present in the
field environment.
3.2.3.1 Discussion—
The purpose of the field reagent blank is to determine if test method analytes or other interferences are present in the field
environment.
3.2.4 internal standard—standard, n—a pure analyte analyte, that is not a sample component, added to a solution in a known
amount, that is used to measure the relative responses of other test method analytes and surrogates that are components of the same
solution. The internal standard must be an analyte that is not a sample component.
3.2.5 laboratory duplicates—duplicates, n—two sample aliquots taken in the analytical laboratory and analyzed separately with
identical procedures. Analysis of laboratory duplicates gives an indication of the precision associated with laboratory procedures,
but not with sample collection, preservation, or storage procedures.
3.2.5.1 Discussion—
Analysis of laboratory duplicates gives an indication of the precision associated with laboratory procedures, but not with sample
collection, preservation, or storage procedures.
3.2.6 laboratory-fortified blank—blank, n—an aliquot of reagent water to which known quantities of the test method analytes
are added in the laboratory. The laboratory-fortified blank is analyzed exactly like a sample, and its purpose is to determine whether
the methodology is in control and whether the laboratory is capable of making accurate and precise measurements at the required
detection limit.
3.2.6.1 Discussion—
The laboratory-fortified blank is analyzed exactly like a sample, and its purpose is to determine whether the methodology is in
control and whether the laboratory is capable of making accurate and precise measurements at the required detection limit.
3.2.7 laboratory-fortified sample matrix—matrix, n—an aliquot of an environmental sample to which known quantities of the
test method analytes are added in the laboratory. The laboratoryfortified sample matrix is analyzed exactly like a sample, and its
purpose is to determine whether or not the sample matrix or the addition of preservatives or dechlorinating agents to the sample
contributes bias to the analytical results. The background concentrations of the analytes in the sample matrix must be determined
in a separate aliquot, and the measured values in the laboratory-fortified sample matrix must be corrected for background
concentrations.
3.2.7.1 Discussion—
The laboratory-fortified sample matrix is analyzed exactly like a sample, and its purpose is to determine whether or not the sample
D5790 − 18
matrix or the addition of preservatives or dechlorinating agents to the sample contributes bias to the analytical results. The
background concentrations of the analytes in the sample matrix must be determined in a separate aliquot, and the measured values
in the laboratory-fortified sample matrix must be corrected for background concentrations.
3.2.8 laboratory performance check solution—solution, n—a solution of one or more compounds (analytes, surrogates, internal
standard, or other test compounds) used to evaluate the performance of the instrument system with respect to a defined set of test
method criteria.
3.2.9 laboratory reagent blank—blank, n—an aliquot of reagent water that is treated exactly as a sample including exposure to
all glassware, equipment, solvents, reagents, internal standards, and surrogates that are used with other samples. The laboratory
reagent blank is used to determine if test method analytes or other interferences are present in the laboratory environment, the
reagents, or the apparatus.
3.2.9.1 Discussion—
The laboratory reagent blank is used to determine if test method analytes or other interferences are present in the laboratory
environment, the reagents, or the apparatus.
3.2.10 primary dilution standard solution—solution, n—a solution of several analytes prepared in the laboratory from stock
standard solutions and diluted as needed to prepare calibration solutions and other needed analyte solutions.
3.2.11 purgeable organic—organic, n—any organic material that is removed from aqueous solution under the purging
conditions described in this test method.
3.2.12 quality control sample—sample, n—a sample matrix containing test method analytes or a solution of method analytes in
a water-miscible solvent that is used to fortify reagent water or environmental samples. The quality control sample is obtained from
a source external to the laboratory and is used to check laboratory performance with externally prepared test materials.
3.2.12.1 Discussion—
The quality control sample is obtained from a source external to the laboratory and is used to check laboratory performance with
externally prepared test materials.
3.2.13 stock standard solution—solution, n—a concentrated solution containing a single certified standard that is a test method
analyte prepared in the laboratory with an assayed reference compound. Stock standard solutions are used to prepare primary
dilution standards. Commercially available stock standard solutions may be used.
3.2.13.1 Discussion—
Stock standard solutions are used to prepare primary dilution standards. Commercially available stock standard solutions may be
used.
3.2.14 surrogate analyte—analyte, n—a pure analyte that is extremely unlikely to be found in any sample, that is added to a
sample aliquot in a known amount, and is measured with the same procedures used to measure other components. The purpose
of a surrogate analyte is to monitor test method performance with each sample.
3.2.14.1 Discussion—
The purpose of a surrogate analyte is to monitor test method performance with each sample.
4. Summary of Test Method
4.1 Volatile organic compounds with low water-solubility are purged from the sample matrix by bubbling an inert gas through
the aqueous sample. Purged sample components are trapped in a tube containing suitable sorbent materials. When purging is
complete, the sorbent tube is heated and backflushed with inert gas to desorb the trapped sample components into a capillary gas
chromatography (GC) column interfaced to a mass spectrometer (MS). The GC column is temperature programmed to separate
the test method analytes which are then detected with the MS. Compounds eluting from the GC column are identified by comparing
their measured mass spectra and retention times to reference spectra and retention times in a database. Reference spectra and
retention times for analytes are obtained by the measurement of calibration standards under the same conditions used for the
samples. The concentration of each identified component is measured by relating the MS response of the quantitation ion produced
by that compound to the MS response of the quantitation ion produced by a compound that is used as an internal standard.
Surrogate analytes, whose concentrations are known in every sample, are measured with the same internal standard calibration
procedure.
D5790 − 18
5. Significance and Use
5.1 Purgeable organic compounds have been identified as contaminants in treated drinking water, wastewater, ground water, and
Toxicity Characteristic Leaching Procedure (TCLP) leachate. These contaminants may be harmful to the environment and to
people. Purge and trap sampling is a generally applicable procedure for concentrating these components prior to gas
chromatographic analysis.
6. Interferences
6.1 During analysis, major contaminant sources are volatile materials in the laboratory and impurities in the inert purging gas
and in the sorbent trap. Avoid the use of plastic tubing or thread sealants other than PTFE, and avoid the use of flow controllers
with rubber components in the purging device. These materials out-gas organic compounds that will be concentrated in the trap
during the purge operation. Analyses of laboratory reagent blanks provide information about the presence of contaminants. When
potential interfering peaks are noted in laboratory reagent blanks, the analyst should change the purge gas source and regenerate
the molecular sieve purge gas filter. Reagents should also be checked for the presence of contaminants. Subtracting blank values
from sample results is not permitted.
6.2 Interfering contamination may occur when a sample containing low concentrations of volatile organic compounds is
analyzed immediately after a sample containing higher concentrations of volatile organic compounds. Experience gained from the
test method validation has shown that there is a carryover of approximately 2 % of the concentration of each analyte from one
sample to the next. The effect was observed when samples containing 1 μg/L of analyte were analyzed immediately after samples
containing 20 μg/L of analyte. For that reason, when low concentrations of analytes are measured in a sample, it is very important
to examine the results of the preceding samples and interpret the low-concentration results accordingly. One preventive technique
is between-sample rinsing of the purging apparatus and sample syringes with two portions of reagent water. After analysis of a
sample containing high concentrations of volatile organic compounds, one or more laboratory reagent blanks should be analyzed
to check for cross contamination. After analyzing a highly contaminated sample, it may be necessary to use methanol to clean the
sample chamber, followed by heating in an oven at 105°C.
6.3 Samples can be contaminated by diffusion of volatile organics through the septum seal into the sample during shipment and
storage. The analytical and sample storage area should be isolated from all atmospheric sources of volatile organic compounds,
otherwise random background levels may result. Since methylene chloride will permeate through PTFE tubing, all gas
chromatography carrier gas lines and purge gas plumbing should be constructed of stainless steel or copper tubing. PTFE free.
Personnel who have been working directly with solvents such as those used in liquid/liquid extraction procedures should not be
allowed into the analytical area until they have washed and changed their clothing.
TABLE 1 Compounds Validated for This Test Method
A
Compound CAS Registry Number Primary Quantitation Ion Secondary Quantitation Ion Approximate Elution Order
Benzene 71-43-2 78 77 20
Bromobenzene 108-86-1 156 77, 158 44
Bromochloromethane 74-97-5 128 49, 130 16
Bromodichloromethane 75-27-4 83 85, 127 25
Bromoform 75-25-2 173 175, 252 41
Bromomethane 74-83-9 94 96 4
n-butylbenzene 104-51-8 91 134 57
sec-butylbenzene 135-98-8 105 134 53
tert-butylbenzene 98-06-6 119 91 52
Carbon disulfide 75-15-0 76 78 8
Carbon tetrachloride 56-23-5 117 119 19
Chlorobenzene 108-90-7 112 77, 114 35
Chloroethane 75-00-3 64 66 5
Chloroform 67-66-3 83 85 15
Chloromethane 74-87-3 50 52 2
2-chlorotoluene 95-49-8 91 126 47
4-chlorotoluene 106-43-4 91 126 50
Dibromochloromethane 124-48-1 129 127 33
1,2-dibromo-3-chloropropane 96-12-8 75 155, 157 60
1,2-dibromoethane 106-93-4 107 109, 188 34
Dibromomethane 74-95-3 93 95, 174 26
1,2-dichlorobenzene 95-50-1 146 111, 148 58
1,3-dichlorobenzene 541-73-1 146 111, 148 54
1,4-dichlorobenzene 106-46-7 146 111, 148 56
trans-1,4-dichloro-2-butene 110-57-6 75 53, 89 48
Dichlorodifluoromethane 75-71-8 85 87 1
1,1-dichloroethane 75-34-3 63 65, 83 11
1,2-dichloroethane 107-06-2 62 98 21
1,1-dichloroethene 75-35-4 96 61, 63 7
cis-1,2-dichloroethene 156-59-4 96 61, 98 13
trans-1,2-dichloroethene 156-60-5 96 61, 98 10
D5790 − 18
TABLE 1 Continued
A
Compound CAS Registry Number Primary Quantitation Ion Secondary Quantitation Ion Approximate Elution Order
1,2-dichloropropane 78-87-5 63 112 24
1,3-dichloropropane 142-28-9 76 78 32
2,2-dichloropropane 590-20-7 77 97 12
1,1-dichloropropene 563-58-6 75 110, 77 18
cis-1,3-dichloropropene 10061-01-5 75 110 27
trans-1,3-dichloropropene 10061-02-6 75 110 29
Ethylbenzene 100-41-4 91 106 36
Hexachlorobutadiene 87-68-3 225 260 62
Hexachloroethane 67-72-1 117 201 59
Isopropylbenzene 98-82-8 105 120 42
p-isopropyltoluene 99-87-6 119 134, 91 55
Methylene chloride 75-09-2 84 86, 49 9
Methyl-tert-butylether 1634-04-4 73 57 14
Methyl-isobutylketone 108-10-1 43 58, 100 23
Naphthalene 91-20-3 128 63
n-propylbenzene 103-65-1 91 120 46
Styrene 100-42-5 104 78 40
1,2,3,4-tetrachlorobenzene 634-66-2 216 108 66
1,2,4,5-tetrachlorobenzene 95-94-3 216 108 65
1,1,1,2-tetrachloroethane 630-20-6 131 133, 119 37
1,1,2,2-tetrachloroethane 79-34-5 83 131, 85 43
Tetrachloroethene 127-18-4 166 168, 129 31
Toluene 108-88-3 92 91 28
1,2,3-trichlorobenzene 87-61-6 180 182 64
1,2,4-trichlorobenzene 120-82-1 180 182 61
1,1,1-trichloroethane 71-55-6 97 99, 61 17
1,1,2-trichloroethane 79-00-5 83 97, 85 30
Trichloroethene 79-01-6 95 130, 132 22
Trichlorofluoromethane 75-69-4 101 103 6
1,2,3-trichloropropane 96-18-4 75 77 45
1,2,4-trimethylbenzene 95-63-6 105 120 51
1,3,5-trimethylbenzene 108-67-8 105 120 49
Vinyl chloride 75-01-4 62 64 3
o-xylene 95-47-6 106 91 39
m-xylene 108-38-3 106 91 38
p-xylene 106-42-3 106 91 38
A
Suggested Internal Standards
B
Suggested Internal Standards
Chlorobenzene-d5 3114-55-4 117 82, 119
1,2-dichlorobenzene-d4 2199-69-1 152 115, 150
Fluorobenzene 462-06-6 96 70, 77
Suggested Surrogates
4-bromofluorobenzene 460-00-4 95 174, 176
1,2-dichloroethane-d4 17060-07-0 65 102
Toluene-d8 2037-26-5 98 70, 100
A
Chemical Abstracts Service (CAS).
B
Appendix X2 is a table of the interlaboratory collaborative study analytes and surrogates with internal standards assignments.
7. Apparatus
7.1 Sample Containers—40 to 120 mL 120-mL screw-cap glass vials equipped with a PTFE-faced silicone septum. The vials
must contain at least twice the volume of water required for the analysis. Prior to use, wash vials with detergent and rinse with
tap and reagent water. Allow the vials and septa to air dry at room temperature, place in an oven at 105°C for 1 h, then remove
and allow to cool in an area known to be free of organics. Purchased, pre-cleaned glass vials may also be used.
7.2 Purge and Trap System—The purge and trap system consists of three basic components: purging device, trap, and desorber.
Systems are commercially available from several sources that meet all of the following specifications.
7.2.1 The all-glass purging device should be designed to accept either a 5 or a 25 mL 25-mL sample volume. Equipment
designed for either single- or multiple-purging devices is acceptable. Gaseous volumes above the sample must be kept to a
minimum to eliminate dead volume effects. A glass frit should be installed at the base of the sample chamber so that the purge
gas passes through the water column as finely divided bubbles with a diameter of <3 mm at the origin. Needle spargers may be
used, however, the purge gas must be introduced at a point about 5 mm from the base of the water column.
7.2.2 Trap:
D5790 − 18
7.2.2.1 The trap shall be at least 25 cm long and have an inside diameter of at least 0.267 cm. Starting from the inlet, the trap
should contain 1.0 cm of methyl silicone coated packing and the following amounts of adsorbents: ⁄3 of 2,6-diphenylene oxide
1 1
polymer (Tenax (Tenax ), ⁄3 of silica gel, and ⁄3 of coconut charcoal. If it is not necessary to determine dichlorodifluoromethane,
the charcoal can be eliminated and the polymer increased to fill two thirds of the trap. Before initial use, the trap should be
conditioned overnight at 225°C by backflushing with an inert gas flow of at least 20 mL/min. Vent the trap effluent to the room
rather than to the analytical column. Prior to daily use, the trap should be conditioned for 10 min at 225°C with backflushing. The
trap may be vented to the analytical column during daily conditioning, provided that the column is run through the temperature
5,6
program prior to analysis of samples.
7.2.2.2 The use of the methyl silicone coated packing is recommended, but not mandatory. The packing serves the purpose of
protecting the Tenax Tenax adsorbant from aerosols. Since it may adsorb higher boiling compounds, it must be fully enclosed
within the heated zone of the trap. Silanized glass wool may be used as a spacer at the trap inlet to eliminate potential cold spots.
7.2.2.3 The presence of charcoal in the trap may interfere with the analysis of ketones. When analyzing for ketones, the charcoal
should be eliminated and the polymer increased to fill two thirds of the trap, if dichlorodifluoromethane is not being analyzed.
7.2.2.4 Other traps are commercially available which may be suitable for use. The equivalency of their performance must be
demonstrated prior to use.
7.2.3 The equipment must be capable of rapidly heating the trap to 225°C the trap desorb temperature either prior to or at the
beginning of the flow of desorption gas. The polymer If the trap has a polymer section, the polymer section of the trap should not
be heated higher than 225°C, or the life expectancy of the trap will decrease. Trap failure is characterized by a pressure drop in
excess of 3 lb/in.20.684 kPa across the trap during purging, by poor bromoform sensitivities, or by increased water background.
7.2.4 The transfer line between the desorber and the GC must be heated within the range of 100 to 150°C.
7.3 Gas Chromatography/Mass Spectrometer/Data System (GC/MS/DS):
7.3.1 The GC must be capable of temperature programming and should be equipped with variable-constant differential flow
controllers so that the column flow rate will remain near constant throughout desorption and temperature program operation. For
several of the chromatographic columns listed as below, the column oven must be cooled to 10°C; therefore, a sub-ambient oven
controller is may be required. One of the columns listed as follows does not require subambient conditions. If syringe injections
of 4-bromofluorobenzene (BFB) will be done, a high efficiency injection port is required.
7.3.2 Capillary Gas Chromatography Columns—Any gas chromatography column that meets the performance specifications of
this test method may be used. Separations of the calibration mixture must be equivalent or better than those described in this test
method. As examples, the following columns have been found to be suitable:
,7
7.3.2.1 Column 1—60 m by 0.75 mm inside diameter VOCOL VOCOL glass wide-bore capillary with a 1.5 μm 1.5-μm film
thickness.
,8
7.3.2.2 Column 2—30 m by 0.53 mm inside diameter DB-624 DB-624 fused silica capillary with a 3 μm 3-μm film thickness.
8,
7.3.2.3 Column 3—75 m by 0.53 mm inside diameter DB-624 DB-624 fused silica capillary with a 3 μm 3-μm film thickness.
8,
7.3.2.4 Column 4—30 m by 0.32 mm inside diameter DB-5 DB-5 fused silica capillary with a 1 μm 1-μm film thickness.
,9
7.3.2.5 Column 5—105 m by 0.53 mm inside diameter Rt -502.2 -502.2 fused silica capillary with a 3 μm 3-μm film thickness.
x
7.3.2.6 For further discussion of columns and inserts see Refs (37) and (48).
7.3.3 Interfaces Between the GC and MS—The interface used depends on the column selected and the gas flow rate.
7.3.3.1 The wide-bore Columns 1, 2, 3, and 5 have the capacity to accept the standard gas flows from the trap during thermal
desorption, and chromatography can begin with the onset of thermal desorption. Depending on the pumping capacity of the MS,
an additional interface between the end of the column and the MS may be required. An open split interface, an all-glass jet
separator, or a cryogenic device are acceptable interfaces. Any interface can be used if the performance specifications described
in this test method can be achieved. The end of the transfer line after the interface, or the end of the analytical column if no interface
is used, should be placed within a few millimetres of the MS ion source.
7.3.3.2 Narrow bore Column 4 cannot may not be able to accept the thermal desorption gas flow, therefore, a cryogenic interface
is would be required. This interface condenses the desorbed sample components at liquid nitrogen temperature and allows the
The sole source of supply of the apparatus known to the committee at this time is Enka Research Institute-Arnhem, 151 Graham Rd., College Station, TX 77845. TX.
If you are aware of alternative suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting
of the responsible technical committee, which you may attend.
6 6
For further discussion on Tenax Tenax traps see the Refs (11-6–2).
The boldface numbers given in parentheses refer to a list of references at the end of the text.
The sole source of supply of the apparatus known to the committee at this time is Supelco, Inc., Supelco Park, Bellafonte, PA 16823-0048.Bellafonte, PA. If you are
aware of alternative suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting of the
responsible technical committee, which you may attend.
The sole source of supply of the apparatus known to the committee at this time is J&W Scientific, Inc., 91 Blue Ravine Rd., Folsom, CA 95630.Folsom, CA. If you are
aware of alternative suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting of the
responsible technical committee, which you may attend.
The sole source of supply of the apparatus known to the committee at this time is Restek Corp., 110 Benner Circle, Bellefonte, PA 16823-8812. Bellefonte, PA. If you
are aware of alternative suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting of
the responsible technical committee, which you may attend.
D5790 − 18
helium gas to pass through to an exit. The condensed components are frozen in a narrow band on an uncoated fused silica
precolumn (59). When all components have been desorbed from the trap, the interface is rapidly heated under a stream of carrier
gas to transfer the analytes to the analytical column. Alternatively, a subambient oven may be used instead of a cryogenic interface.
7.3.4 The mass spectrometer must be capable of electron ionization at a nominal electron energy of 70 eV. The spectrometer
must be capable of scanning from 48 to 260 amu with a complete scan cycle time (including scan overhead) of 2 s or less (scan
cycle time = total MS data acquisition time in seconds divided by number of scans in the chromatogram). The spectrometer must
produce a mass spectrum that meets all criteria in Table 2 when 25 ng or less of 4-bromofluorobenzene (BFB) is introduced into
the GC/MS. An average spectrum across the BFB GC peak may be used to test instrument performance.
NOTE 1—If this test method is used for analytes with mass fragments below 48 amu (for example, many ketones exhibit a characteristic 43 amu mass
fragment), the mass range may be modified. All calibration standards must be analyzed using the same mass range as the samples.
NOTE 2—The criteria in Table 2 for BFB were used for this test method validation. Other criteria, such as those provided in the United States
Environmental Protection Agency 1990 Contract Laboratory Program Statement of Work, are available. If other mass spectrometer tuning criteria are
used, the precision and bias results presented in Section 1515 of this test method may not apply. Therefore, the applicability of other BFB criteria to the
test method must be demonstrated by the user.
7.3.5 An interfaced data system is required to acquire, store, reduce, and output mass spectral data. The computer software
should have the capability of processing stored GC/MS data by recognizing a GC peak within any given retention time window,
comparing the mass spectra from the GC peak with spectral data in a user-created database, and generating a list of tentatively
identified compounds with their retention times and scan numbers. The software must allow integration of the ion abundance of
any specific ion between specified time or scan number limits. The software should also allow calculation of response factors as
defined in 10.2.611.2.6 or construction of a second or third order regression calibration curve, calculation of response factor
statistics, and calculation of concentrations of analytes using either the calibration curve or the equation in 12.1.1.
7.4 Syringe and Syringe Valves:
7.4.1 Glass Hypodermic Syringes, two, 5 to 25 mL, with Luer-Lok tip, depending on the sample volume used.
7.4.2 Two-Way Syringe Valves, three, with Luer ends.
7.4.3 25 μL Microsyringe, one, with a 5 cm by 0.15 mm inside diameter, 22° bevel needle.
7.4.4 Microsyringes, 10 and 100 μL.
7.4.5 Syringes, 0.5, 1.0, and 5 mL, gas-tight with shut-off valve.
7.5 Bottles:
7.5.1 Standard Solution Storage Containers, 15-mL glass bottles with PTFE-lined screw caps.
8. Reagents and Materials
8.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society.
8.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean reagent water demonstrated
to be free of the analytes of interest.
8.2.1 Reagent water may be generated by passing tap water through a carbon filter bed containing about 453 g of activated
carbon.
8.2.2 A water purification system may be used to generate reagent water.
8.2.3 Reagent water may be prepared by boiling distilled water for 15 min. Subsequently, while maintaining the temperature
at 90°C, bubble a contaminant-free inert gas through the water for 1 h. While still hot, transfer the water to a narrow-mouth
screw-cap bottle and seal with a PTFE-lined septum and cap.
TABLE 2 Ion Abundance Criteria for 4-Bromofluorobenzene
(BFB)
Mass Relative Abundance Criteria
50 15 to 40 % of mass 95
75 30 to 60 % of mass 95
95 base peak, 100 % relative abundance
96 5 to 9 % of mass 95
173 less than 2 % of mass 174
174 greater than 50 % of mass 95
175 5 to 9 % of mass 174
176 greater than 95 % but less than 101 % of mass 174
177 5 to 9 % of mass 176
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by
the American Chemical Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmaceutical Convention, Inc. (USPC), Rockville, MD.
D5790 − 18
8.3 Trap Packing Materials:
8.3.1 2,6-Diphenylene Oxide Polymer, 60/80 mesh, chromatographic grade, or equivalent.
, 11 , 12
8.3.2 Methyl Silicone Packing (Optional), OV-1 (3 %) (3 %) on Chromosorb W, W, 60/80 mesh, or equivalent.
8.3.3 Silica Gel, 35/60 mesh.
8.3.4 Coconut Charcoal, 20/40 mesh.
8.3.5 Alternate traps may be used, so trap packing materials may change. However, if using an alternative trap, the QC criteria
of the method must be met or exceeded.
8.4 Methanol, purge and trap grade, demonstrated to be free of analytes.
8.5 Hydrochloric Acid (1+1)—Carefully add measured volume of concentrated HCl (sp gr 1.19) to equal volume of water.
8.6 Vinyl Chloride—Certified mixtures of vinyl chloride in nitrogen and pure vinyl chloride are commercially available.
8.7 Ascorbic Acid, granular.
8.8 pH Test Paper, capable of measuring pH 2 with a sensitivity of at least 0.5 pH unit.
8.9 Standard Solutions, Stock—These solutions may be purchased as certified solutions or prepared from pure standard materials
using the following procedures. One of these solutions is required for every analyte of concern, every surrogate, and the internal
standard. A useful working concentration is about 1 to 5 μg/μL.
8.9.1 Place about 9.8 mL of methanol into a 10-mL ground-glass stoppered volumetric flask. Allow the flask to stand,
unstoppered, for about 10 min or until the alcohol-wetted surfaces inside the neck of the flask have dried, and weigh to the nearest
0.1 mg.
8.9.2 If the analyte is a liquid at room temperature, use a 100-μL syringe and immediately add two or more drops of pure
standard material to the flask. Be sure that the reference standard falls directly into the alcohol without contacting the neck of the
flask. If the analyte is a gas at room temperature, fill a 5-mL valved gas-tight syringe with the standard to the 5.0-mL mark, lower
the needle to 5 mm above the methanol meniscus, and slowly inject the standard into the neck area of the flask. The gas will rapidly
dissolve in the methanol.
8.9.3 Reweigh, dilute to volume, stopper, then mix by inverting the flask several times. Calculate the concentration in
micrograms per microlitre from the net gain in weight. When compound purity is certified at 96 % or greater, the weight can be
used without correction to calculate the concentration of the stock standard.
8.9.4 Store stock standard solutions in 15-mL bottles equipped with PTFE-lined screw caps. Methanol solutions prepared from
liquid analytes are stable for at least 4 weeks when stored at 4°C. Methanol solutions prepared from gaseous analytes are not stable
for more than one week when stored at <0°C; at room temperature, they must be discarded after one day.
8.10 Primary Dilution Standards—Use stock standard solutions to prepare primary dilution standard solutions that contain all
the analytes of concern and the surrogates (but not the internal standard) in methanol. The primary dilution standards should be
prepared at concentrations that can be easily diluted to prepare aqueous calibration solutions that will bracket the working
concentration range. Store the primary dilution standard solutions with minimal headspace and check frequently for signs of
deterioration or evaporation, especially just before preparing calibration solutions. Storage times described for stock standard
solutions in 8.9.4 also apply to primary dilution standard solutions.
8.11 Solutions for Internal Standard and Surrogates:
8.11.1 A solution containing the internal standards and the surrogates is required to prepare laboratory reagent blanks and to
fortify each sample. It is also used as a laboratory performance check solution. Prepare a solution containing the desired internal
standards and surrogates in methanol. A number of appropriate internal standards and surrogates are listed in Table 1. Appendix
X2 contains a list of analytes and surrogates with assigned internal standards as were used in the collaborative study. The
concentration of this solution should be made as appropriate for the desired calibration range and expected sample concentration,
in order to minimize the amount of methanol added to the sample. For example, if the fortification solution is prepared at a
concentration of 5 μg/mL of each species, a 5-μL aliquot of this solution added to a 25-mL water sample volume gives
concentrations of 1 μg/L of each species and a 5-μL aliquot of this solution added to a 5-mL water sample volume gives a
concentration of 5 μg/L of each.
8.11.2 A solution of the internal standard alone is required to prepare calibration standards and laboratory-fortified blanks. The
internal standard should be in methanol at a concentration of 5 μg/mL.
The sole source of supply of the apparatus known to the committee at this time is Ohio Valley Specialty Chemical Co., 432 Walnut St., Cincinnati, OH 45202.Cincinnati,
OH. If you are aware of alternative suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a
meeting of the responsible technical committee, which you may attend.
The sole source of supply of the apparatus known to the committee at this time is Johns-Manville Products Corp., Liddle Ave., Edison, NJ 08837.Edison, NJ. If you
are aware of alternative suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting of
the responsible technical committee, which you may attend.
D5790 − 18
8.12 Laboratory Reagent Blank—Fill a 5-mL (or 25-mL) syringe with reagent water and adjust to the mark with no air bubbles.
Inject 10 μL of the fortification solution containing the internal standard and surrogates through the Luer Lok valve into the reagent
water. Transfer the laboratory reagent blank to the purging device as described in 12.1.3.
8.13 Laboratory-Fortified Blank—Prepare this exactly like a calibration standard (see 8.14.2). This is a calibration standard that
is treated as a sample.
8.14 Calibration Standards:
8.14.1 The number of calibration standards needed depends on the calibration range desired. A minimum of three calibration
standards is required to calibrate a range of a factor of 20 in concentration. For a factor of 50, use at least four standards, and for
a factor of 100 at least five standards. The calibration standards should contain each analyte of concern and each surrogate at
concentrations that define the range of the test method. Every calibration standard contains the internal standard at the same
concentration (5 μg/L suggested for a 5-mL sample, 1 μg/L suggested for a 25-mL sample).
8.14.2 To prepare a calibration standard, add an appropriate volume of a primary dilution standard (containing analytes and
surrogates) to an aliquot of reagent water in a volumetric flask. Use a microsyringe and rapidly inject the methanol solutions into
the expanded area of the filled volumetric flask. Remove the needle as quickly as possible after injection. Mix by inverting the flask
three times only. Discard the contents contained in the neck of the flask using a disposable pipet. Aqueous standards are not stable
in a volumetric flask and should be discarded after 1 h unless transferred to a sample bottle and sealed immediately. Alternatively,
the calibration standard may be prepared in a 5 or 25-mL syringe.
9. Hazards
9.1 The toxicity or carcinogenicity of chemicals used in this test method has not been precisely defined; each chemical should
be treated as a potential health hazard, and exposure to these chemicals should be minimized. Each laboratory is responsible for
maintaining awareness of OSHA regulations regarding safe handling of chemicals used in this test method.
9.2 The following test method analytes have been tentatively classified as known or suspected human or mammalian
carcinogens: benzene, carbon tetrachloride, 1,4-dichlorobenzene,1,2-dichloroethane, hexachlorobutadiene, hexachloroethane,
1,1,2,2-tetrachloroethane, 1,1,2-trichloroethane, chloroform, 1,2-dibromoethane, tetrachloroethene, trichloroethene, and vinyl
chloride. Pure standard materials and stock standard solutions of these compounds should be handled in a well-ventilated hood.
A National Institute for Occupational Safety and Health/Mine Safety and Health Administration (NIOSH/MSHA) approved toxic
gas respirator should be worn when the analyst handles high concentrations of these toxic compounds.
10. Sample Collection, Preservation, and Storage
10.1 Sample Collection, Dechlorination, and Preservation:
10.1.1 In order to determine the appropriate quantity of HCl (1+1) to be added to the samples, collect a pre-sample of known
volume.
10.1.1.1 Add one drop of HCl (1+1) for every 10 mL of pre-sample volume. Check the pH using pH test paper. If the pH is
not less than 2, add more HCl (1+1) to determine the amount required to get the pH below 2.
10.1.1.2 If the pre-sample foams upon addition of HCl (1+1), do not use the HCl (1+1) preservative. Instead, refrigerate the
sample as described in 10.1.5 and analyze as soon as possible within the holding time as described in 10.2.2.
10.1.2 Samples should be collected in duplicate. It may be desirable to collect additional samples for screening or other
purposes. Fill the sample bottles to overflowing, taking care not to flush out the preservatives. No air bubbles should pass through
the sample as the bottle is filled or be trapped in the sample when the bottle is sealed. Seal the sample bottles, PTFE-face down,
and shake vigorously for 1 min.
10.1.2.1 In order to preserve the sample against biological degradation, add the appropriate quantity of HCl (1 + 1) as
determined in 10.1.1.1 to the sample bottle before filling.
10.1.2.2 If the samples are suspected to contain residual chlorine, and if measurements of the concentrations of disinfection
by-products (for example, trihalomethanes, etc.) are desired, add about 25 mg of ascorbic acid to the sample bottle before filling.
10.1.3 When sampling from a water tap, open the tap and allow the system to flush until the water temperature has stabilized
(usually about 10 min). Adjust the flow to about 500 mL/min and collect duplicate samples from the flowing stream.
10.1.4 When sampling from an open body of water, fill a 1-qt960-mL wide-mouth bottle or 1-L beaker with sample from a
representative area, and carefully fill duplicate sample bottles from the container.
10.1.5 The samples must be chilled to 4°C on the day of collection and must be maintained at that temperature until analysis.
Field samples that will not be received at the laboratory on the day of collection must be packaged for shipment with sufficient
ice to ensure that they will be at 4°C on arrival at the laboratory.
10.2 Sample Storage:
10.2.1 Store samples at 4°C until analysis. The sample storage area must be free of organic solvent vapors.
NOTE 3—If analyzing for light-sensitive analytes, such as some halogenated compounds, the samples should be stored in the dark or in amber
...


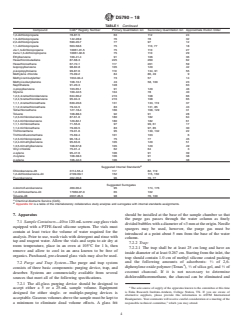




Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...