ASTM G102-23
(Practice)Standard Practice for Calculation of Corrosion Rates and Related Information from Electrochemical Measurements
Standard Practice for Calculation of Corrosion Rates and Related Information from Electrochemical Measurements
SIGNIFICANCE AND USE
3.1 Electrochemical corrosion rate measurements often provide results in terms of electrical current. Although the conversion of these current values into mass loss rates or penetration rates is based on Faraday’s Law, the calculations can be complicated for alloys and metals with elements having multiple valence values. This practice is intended to provide guidance in calculating mass loss and penetration rates for such alloys. Some typical values of equivalent weights for a variety of metals and alloys are provided.
3.2 Electrochemical corrosion rate measurements may provide results in terms of electrical resistance. The conversion of these results to either mass loss or penetration rates requires additional electrochemical information. Some approaches for estimating this information are given.
3.3 Use of this practice will aid in producing more consistent corrosion rate data from electrochemical results. This will make results from different studies more comparable and minimize calculation errors that may occur in transforming electrochemical results to corrosion rate values.
SCOPE
1.1 This practice covers the providing of guidance in converting the results of electrochemical measurements to rates of uniform corrosion. Calculation methods for converting corrosion current density values to either mass loss rates or average penetration rates are given for most engineering alloys. In addition, some guidelines for converting polarization resistance values to corrosion rates are provided.
1.2 The values stated in SI units are to be regarded as standard. Other units of measurement are included in this standard because of their usage.
1.3 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Feb-2023
- Technical Committee
- G01 - Corrosion of Metals
- Drafting Committee
- G01.11 - Electrochemical Measurements in Corrosion Testing
Relations
- Effective Date
- 01-Nov-2014
- Effective Date
- 01-Feb-2013
- Effective Date
- 01-Feb-2013
- Effective Date
- 01-Feb-2013
- Effective Date
- 15-Nov-2012
- Effective Date
- 01-Dec-2011
- Effective Date
- 15-Nov-2011
- Effective Date
- 01-Nov-2004
- Refers
ASTM G1-03 - Standard Practice for Preparing, Cleaning, and Evaluating Corrosion Test Specimens - Effective Date
- 01-Oct-2003
- Effective Date
- 01-Jan-1999
- Effective Date
- 15-Mar-1994
Overview
ASTM G102-23 - Standard Practice for Calculation of Corrosion Rates and Related Information from Electrochemical Measurements offers a comprehensive methodology for converting electrochemical test results into meaningful corrosion rates for metals and alloys. Developed by ASTM International, this practice is widely used to ensure the consistent calculation of corrosion rates based on parameters such as corrosion current density or polarization resistance derived from electrochemical measurements.
For industries where uniform corrosion assessment is essential, accurate conversion from electrochemical data to practical units like mass loss or penetration rates is critical. This standard aids in minimizing errors and ensuring studies are comparable across different laboratories and test conditions.
Key Topics
- Electrochemical Corrosion Measurement: Guidance on calculations when corrosion rates are reported as electrical current or resistance, converting these to mass loss or penetration rates.
- Faraday’s Law: Principles for converting electrical current to material loss, including complexities for metals with multiple valence states.
- Equivalent Weight Calculation: Methods for calculating the equivalent weight of pure metals and alloys, including how to handle multiple valence scenarios and alloy composition.
- Polarization Resistance: Guidelines for determining corrosion rates using polarization resistance measurements, including estimation of the Stern-Geary constant and consideration of Tafel slopes.
- Units of Measurement: Standardizes the use of SI units, while also including commonly used units such as mpy (mils per year), mm/yr, and g/m²·d.
- Error Sources and Limitations: Discussion of potential errors stemming from incorrect valence assignments, non-uniform corrosion, solution resistivity, and additional electrochemical phenomena that may impact results.
Applications
The ASTM G102-23 standard is broadly applicable across industries where the durability and longevity of metals are of concern:
- Materials Research: Provides a reliable foundation for translating electrochemical data into standardized corrosion rates, essential for comparing new alloys and coatings.
- Quality Control: Assists quality assurance teams in confirming that manufactured metal parts meet corrosion resistance specifications.
- Asset Integrity Management: Used in sectors such as oil & gas, power generation, chemical processing, and marine engineering to estimate the service life of metallic structures, piping, and vessels.
- Failure Analysis: Supports engineers in determining if corrosion was a contributing factor in material failures.
- Electrochemical Testing Laboratories: Standardizes procedures for reporting results, ensuring data comparability between different analysts and equipment setups.
Related Standards
ASTM G102-23 references and complements several other key ASTM corrosion testing standards:
- ASTM G1 - Practice for Preparing, Cleaning, and Evaluating Corrosion Test Specimens
- ASTM G5 - Reference Test Method for Making Potentiodynamic Anodic Polarization Measurements
- ASTM G59 - Test Method for Conducting Potentiodynamic Polarization Resistance Measurements
Together, these standards provide a robust procedural framework for electrochemical corrosion testing, data interpretation, and the conversion of results into actionable corrosion rates.
Keywords: corrosion rate, electrochemical measurements, Faraday’s Law, equivalent weight, polarization resistance, Tafel slopes, ASTM G102, uniform corrosion, corrosion testing, material loss calculation.
Buy Documents
ASTM G102-23 - Standard Practice for Calculation of Corrosion Rates and Related Information from Electrochemical Measurements
REDLINE ASTM G102-23 - Standard Practice for Calculation of Corrosion Rates and Related Information from Electrochemical Measurements
Get Certified
Connect with accredited certification bodies for this standard

Element Materials Technology
Materials testing and product certification.

Inštitut za kovinske materiale in tehnologije
Institute of Metals and Technology. Materials testing, metallurgical analysis, NDT.
Sponsored listings
Frequently Asked Questions
ASTM G102-23 is a standard published by ASTM International. Its full title is "Standard Practice for Calculation of Corrosion Rates and Related Information from Electrochemical Measurements". This standard covers: SIGNIFICANCE AND USE 3.1 Electrochemical corrosion rate measurements often provide results in terms of electrical current. Although the conversion of these current values into mass loss rates or penetration rates is based on Faraday’s Law, the calculations can be complicated for alloys and metals with elements having multiple valence values. This practice is intended to provide guidance in calculating mass loss and penetration rates for such alloys. Some typical values of equivalent weights for a variety of metals and alloys are provided. 3.2 Electrochemical corrosion rate measurements may provide results in terms of electrical resistance. The conversion of these results to either mass loss or penetration rates requires additional electrochemical information. Some approaches for estimating this information are given. 3.3 Use of this practice will aid in producing more consistent corrosion rate data from electrochemical results. This will make results from different studies more comparable and minimize calculation errors that may occur in transforming electrochemical results to corrosion rate values. SCOPE 1.1 This practice covers the providing of guidance in converting the results of electrochemical measurements to rates of uniform corrosion. Calculation methods for converting corrosion current density values to either mass loss rates or average penetration rates are given for most engineering alloys. In addition, some guidelines for converting polarization resistance values to corrosion rates are provided. 1.2 The values stated in SI units are to be regarded as standard. Other units of measurement are included in this standard because of their usage. 1.3 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 3.1 Electrochemical corrosion rate measurements often provide results in terms of electrical current. Although the conversion of these current values into mass loss rates or penetration rates is based on Faraday’s Law, the calculations can be complicated for alloys and metals with elements having multiple valence values. This practice is intended to provide guidance in calculating mass loss and penetration rates for such alloys. Some typical values of equivalent weights for a variety of metals and alloys are provided. 3.2 Electrochemical corrosion rate measurements may provide results in terms of electrical resistance. The conversion of these results to either mass loss or penetration rates requires additional electrochemical information. Some approaches for estimating this information are given. 3.3 Use of this practice will aid in producing more consistent corrosion rate data from electrochemical results. This will make results from different studies more comparable and minimize calculation errors that may occur in transforming electrochemical results to corrosion rate values. SCOPE 1.1 This practice covers the providing of guidance in converting the results of electrochemical measurements to rates of uniform corrosion. Calculation methods for converting corrosion current density values to either mass loss rates or average penetration rates are given for most engineering alloys. In addition, some guidelines for converting polarization resistance values to corrosion rates are provided. 1.2 The values stated in SI units are to be regarded as standard. Other units of measurement are included in this standard because of their usage. 1.3 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM G102-23 is classified under the following ICS (International Classification for Standards) categories: 77.060 - Corrosion of metals. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM G102-23 has the following relationships with other standards: It is inter standard links to ASTM G5-14, ASTM G5-13e2, ASTM G5-13, ASTM G5-13e1, ASTM G5-12, ASTM G1-03(2011), ASTM G5-94(2011)e1, ASTM G5-94(2004), ASTM G1-03, ASTM G1-90(1999)e1, ASTM G5-94(1999)e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM G102-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: G102 − 23
Standard Practice for
Calculation of Corrosion Rates and Related Information
from Electrochemical Measurements
This standard is issued under the fixed designation G102; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 3. Significance and Use
3.1 Electrochemical corrosion rate measurements often pro-
1.1 This practice covers the providing of guidance in
vide results in terms of electrical current. Although the con-
converting the results of electrochemical measurements to rates
version of these current values into mass loss rates or penetra-
of uniform corrosion. Calculation methods for converting
tion rates is based on Faraday’s Law, the calculations can be
corrosion current density values to either mass loss rates or
complicated for alloys and metals with elements having
average penetration rates are given for most engineering alloys.
multiple valence values. This practice is intended to provide
In addition, some guidelines for converting polarization resis-
guidance in calculating mass loss and penetration rates for such
tance values to corrosion rates are provided.
alloys. Some typical values of equivalent weights for a variety
1.2 The values stated in SI units are to be regarded as
of metals and alloys are provided.
standard. Other units of measurement are included in this
3.2 Electrochemical corrosion rate measurements may pro-
standard because of their usage.
vide results in terms of electrical resistance. The conversion of
1.3 This international standard was developed in accor-
these results to either mass loss or penetration rates requires
dance with internationally recognized principles on standard-
additional electrochemical information. Some approaches for
ization established in the Decision on Principles for the
estimating this information are given.
Development of International Standards, Guides and Recom-
3.3 Use of this practice will aid in producing more consis-
mendations issued by the World Trade Organization Technical
tent corrosion rate data from electrochemical results. This will
Barriers to Trade (TBT) Committee.
make results from different studies more comparable and
minimize calculation errors that may occur in transforming
2. Referenced Documents
electrochemical results to corrosion rate values.
2.1 ASTM Standards:
D2776 Methods of Test for Corrosivity of Water in the
4. Corrosion Current Density
Absence of Heat Transfer (Electrical Methods) (With-
3 4.1 Corrosion current values may be obtained from galvanic
drawn 1991)
cells and polarization measurements, including Tafel extrapo-
G1 Practice for Preparing, Cleaning, and Evaluating Corro-
lations or polarization resistance measurements. (See Refer-
sion Test Specimens
ence Test Method G5 and Test Method G59 for examples.) The
G5 Reference Test Method for Making Potentiodynamic
first step is to convert the measured or estimated current value
Anodic Polarization Measurements
to current density. This is accomplished by dividing the total
G59 Test Method for Conducting Potentiodynamic Polariza-
current by the geometric area of the electrode exposed to the
tion Resistance Measurements
solution. The surface roughness is generally not taken into
account when calculating the current density. It is assumed that
the current distributes uniformly across the area used in this
This practice is under the jurisdiction of ASTM Committee G01 on Corrosion
calculation. In the case of galvanic couples, the exposed area of
of Metals and is the direct responsibility of Subcommittee G01.11 on Electrochemi-
the anodic specimen should be used. This calculation may be
cal Measurements in Corrosion Testing.
expressed as follows:
Current edition approved Feb. 15, 2023. Published February 2023. Originally
approved in 1989. Last previous edition approved in 2015 as G102–89 (2015)ɛ .
I
cor
DOI: 10.1520/G0102-23.
i 5 (1)
cor
A
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
where:
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website.
i = corrosion current density, μA/cm ,
cor
The last approved version of this historical standard is referenced on www.ast-
I = total anodic current, μA, and
cor
m.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
G102 − 23
4.5 Valence assignments for elements that exhibit multiple
A = exposed specimen area, cm .
valences can create uncertainty. It is best if an independent
Other units may be used in this calculation. In some
technique can be used to establish the proper valence for each
computerized polarization equipment, this calculation is made
alloying element. Sometimes it is possible to analyze the
automatically after the specimen area is programmed into the
corrosion products and use those results to establish the proper
computer. A sample calculation is given in Appendix X1.
valence. Another approach is to measure or estimate the
4.2 Equivalent Weight—Equivalent weight, EW, may be
electrode potential of the corroding surface. Equilibrium dia-
thought of as the mass of metal in grams that will be oxidized
grams showing regions of stability of various phases as a
by the passage of one Faraday (96 489 6 2 A·s) of electric
function of potential and pH may be created from thermody-
charge.
namic data. These diagrams are known as Potential-pH (Pour-
baix) diagrams and have been published by several authors (2,
NOTE 1—The value of EW is not dependent on the unit system chosen
and so may be considered dimensionless.
3). The appropriate diagrams for the various alloying elements
NOTE 2—The unit of charge A·s has been named Coulomb, abbreviated
can be consulted to estimate the stable valence of each element
C. However, the units A and s are primary SI units, and A·s will be used
at the temperature, potential, and pH of the contacting electro-
exclusively for unit charge below.
lyte that existed during the test.
For pure elements, the equivalent weight is given by:
NOTE 3—Some of the older publications used inaccurate thermody-
W
EW 5 (2)
namic data to construct the diagrams and consequently they are in error.
n
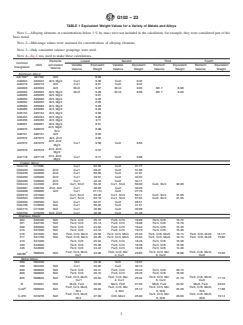
4.6 Some typical values of EW for a variety of metals and
where:
alloys are given in Table 1.
W = the atomic weight of the element, and
n = the number of electrons required to oxidize an atom of
4.7 Calculation of Corrosion Rate—Faraday’s Law can be
the element in the corrosion process, that is, the valence
used to calculate the corrosion rate, either in terms of penetra-
of the element.
tion rate (CR) or mass loss rate (MR) (4):
4.3 For alloys, the equivalent weight is more complex. It is
i
cor
CR 5 K EW (5)
usually assumed that the process of oxidation is uniform and
ρ
does not occur selectively to any component of the alloy. If this
MR 5 K i EW (6)
2 cor
is not true, then the calculation approach will need to be
adjusted to reflect the observed mechanism. In addition, some
where:
rationale must be adopted for assigning values of n to the
CR is given in mm/yr, i in μA/cm ,
cor
elements in the alloy because many elements exhibit more than
one valence value.
−3
K = 3.27 × 10 , mm s g/μA cm yr (Note 4),
4.4 To calculate the alloy equivalent weight, the following
ρ = density in g/cm , (see Practice G1 for density values
approach may be used. Consider 1 g of an alloy oxidized. The
for many metals and alloys used in corrosion testing),
electrical charge equivalent for this mass oxidized, Q, is then:
MR = g/m d, and
−3 2 2
K = 8.954 × 10 , g s cm /μA m d (Note 4).
nifi
Q 5 (3)
(
Wi NOTE 4—EW is considered dimensionless in these calculations.
Other values for K and K for different unit systems are
where:
1 2
th
given in Table 2.
fi = the mass fraction of the i element in the alloy,
th
Wi = the atomic weight of the i element in the alloy, and
4.8 Errors that may arise from this procedure are discussed
th
ni = the valence of the i element of the alloy.
below.
Therefore, the alloy equivalent weight, EW, is the reciprocal
4.8.1 Assignment of incorrect valence values may cause
of this quantity:
serious errors (5).
4.8.2 The calculation of penetration or mass loss from
EW 5 (4)
nifi
electrochemical measurements, as described in this standard,
(
Wi
assumes that uniform corrosion is occurring. In cases where
non-uniform corrosion processes are occurring, the use of these
Normally only elements above 1 mass percent in the alloy
methods may result in a substantial underestimation of the true
are included in the calculation. In cases where the actual
values.
analysis of an alloy is not available, it is conventional to use the
4.8.3 Alloys that include large quantities of metalloids or
mid-range of the composition specification for each element,
oxidized materials may not be able to be treated by the above
unless a better basis is available. A sample calculation is given
procedure.
in Appendix X2 (1).
4.8.4 Corrosion rates calculated by the method above where
abrasion or erosion is a significant contributor to the metal loss
4 process may yield significant underestimation of the metal loss
The boldface numbers in parentheses refer to the list of references at the end of
this standard. rate.
G102 − 23
TABLE 1 Equivalent Weight Values for a Variety of Metals and Alloys
NOTE 1—Alloying elements at concentrations below 1 % by mass were not included in the calculation, for example, they were considered part of the
basis metal.
NOTE 2—Mid-range values were assumed for concentrations of alloying elements.
NOTE 3—Only consistent valence groupings were used.
NOTE 4—Eq 4 was used to make these calculations.
Lowest Second Third Fourth
Elements
Common
UNS w/Constant
Variable Equivalent Variable Equivalent Element/ Equivalent Element/ Equivalent
Designation
Valence
Valence Weight Valence Weight Valence Weight Valence Weight
Aluminum Alloys:
A
AA1100 A91100 Al/3 8.99
AA2024 A92024 Al/3, Mg/2 Cu/1 9.38 Cu/2 9.32
AA2219 A92219 Al/3 Cu/1 9.51 Cu/2 9.42
AA3003 A93003 Al/3 Mn/2 9.07 Mn/4 9.03 Mn 7 8.98
AA3004 A93004 Al/3, Mg/2 Mn/2 9.09 Mn/4 9.06 Mn 7 9.00
AA5005 A95005 Al/3, Mg/2 9.01
AA5050 A95050 Al/3, Mg/2 9.03
AA5052 A95052 Al/3, Mg/2 9.05
AA5083 A95083 Al/3, Mg/2 9.09
AA5086 A95086 Al/3, Mg/2 9.09
AA5154 A95154 Al/3, Mg/2 9.08
AA5454 A95454 Al/3, Mg/2 9.06
AA5456 A95456 Al/3, Mg/2 9.11
AA6061 A96061 Al/3, Mg/2 9.01
Al/3, Mg/2,
AA6070 A96070 8.98
Si/4
AA6101 A96161 Al/3 8.99
AA7072 A97072 Al/3, Zn/2 9.06
Al/3, Zn/2,
AA7075 A97075 Cu/1 9.58 Cu/2 9.55
Mg/2
Al/3, Zn/2,
AA7079 A97079 9.37
Mg/2
Al/3, Zn/2,
AA7178 A97178 Cu/1 9.71 Cu/2 9.68
Mg/2
Copper Alloys:
CDA110 C11000 Cu/1 63.55 Cu/2 31.77
CDA220 C22000 Zn/2 Cu/1 58.07 Cu/2 31.86
CDA230 C23000 Zn/2 Cu/1 55.65 Cu/2 31.91
CDA260 C26000 Zn/2 Cu/1 49.51 Cu/2 32.04
CDA280 C28000 Zn/2 Cu/1 46.44 Cu/2 32.11
CDA444 C44300 Zn/2 Cu/1, Sn/2 50.42 Cu/1, Sn/4 50.00 Cu/2, Sn/4 32.00
CDA687 C68700 Zn/2, Al/3 Cu/1 48.03 Cu/2 30.29
CDA608 C60800 Al/3 Cu/1 47.114 Cu/2 27.76
CDA510 C51000 Cu/1, Sn/2 63.32 Cu/1, Sn/4 60.11 Cu/2, Sn/4 31.66
CDA524 C52400 Cu/1, Sn/2 63.10 Cu/1, Sn/4 57.04 Cu/2, Sn/4 31.55
CDA655 C65500 Si/4 Cu/1 50.21 Cu/2 28.51
CDA706 C70600 Ni/2 Cu/1 56.92 Cu/2 31.51
CDA715 C71500 Ni/2 Cu/1 46.69 Cu/2 30.98
CDA752 C75200 Ni/2, Zn/2 Cu/1 46.38 Cu/2 31.46
Stainless Steels:
304 S30400 Ni/2 Fe/2, Cr/3 25.12 Fe/3, Cr/3 18.99 Fe/3, Cr/6 15.72
321 S32100 Ni/2 Fe/2, Cr/3 25.13 Fe/3, Cr/3 19.08 Fe/3, Cr/6 15.78
309 S30900 Ni/2 Fe/2, Cr/3 24.62 Fe/3, Cr/3 19.24 Fe/3, Cr/6 15.33
310 S31000 Ni/2 Fe/2, Cr/3 24.44 Fe/3, Cr/3 19.73 Fe/3, Cr/6 15.36
316 S31600 Ni/2 Fe/2, Cr/3, Mo/3 25.50 Fe/2, Cr/3, Mo/4 25.33 Fe/3, Cr/6, Mo/6 19.14 Fe/3, Cr/6, Mo/6 16.111
317 S31700 Ni/2 Fe/2, Cr/3, Mo/3 25.26 Fe/2, Cr/3, Mo/4 25.03 Fe/3, Cr/3, Mo/6 19.15 Fe/3, Cr/6, Mo/6 15.82
410 S41000 Fe/2, Cr/3 25.94 Fe/3, Cr/3 18.45 Fe/3, Cr/6 16.28
430 S43000 Fe/2, Cr/3 25.30 Fe/3, Cr/3 18.38 Fe/3, Cr/6 15.58
446 S44600 Fe/2, Cr/3 24.22 Fe/3, Cr/3 18.28 Fe/3, Cr/6 14.46
Fe/2, Cr/3, Mo/3, Fe/2, Cr/3, Mo/ Fe/3, Cr/3, Mo/ Fe/3, Cr/6, Mo/6,
A
20CB3 N08020 Ni/2 23.98 23.83 18.88 15.50
Cu/1 4, Cu/1 6, Cu/2 Cu/2
Nickel Alloys:
200 N02200 NI/2 29.36 Ni/3 19.57
400 N04400 Ni/2 Cu/1 35.82 Cu/2 30.12
600 N06600 Ni/2 Fe/2, Cr/3 26.41 Fe/3, Cr/3 25.44 Fe/3, Cr/6 20.73
800 N08800 Ni/2 Fe/2, Cr/3 25.10 Fe/3, Cr/3 20.76 Fe/3, Cr/6 16.59
Fe/2, Cr/3, Mo/3, Fe/2, Cr/3, Mo/ Fe/3, Cr/3, Mo/ Fe/3, Cr/6, Mo/6,
825 N08825 Ni/2 25.52 25.32 21.70 17.10
Cu/1 4, Cu/1 6, Cu/2 Cu/2
B N10001 Ni/2 Mo/3, Fe/2 30.05 Mo/4, Fe/2 27.50 Mo/6, Fe/2 23.52 Mo/6, Fe/3 23.23
Fe/2, Cr/3, Mo/3, Fe/2, Cr/3, Mo/ Fe/2, Cr/3, Mo/ Fe/3, Cr/6, Mo/6,
B
C-22 N06022 Ni/2 26.04 25.12 23.28 17.88
W/4 4, W/4 6, W/6 W/6
Fe/2, Cr/3, Mo/3, Fe/2, Cr/3, Mo/ Fe/3, Cr/6, Mo/6,
C-276 N10276 Ni/2 27.09 Cr/3, Mo/4 25.90 23.63 19.14
W/4 6, W/6 W/6
G102 − 23
TABLE 1 Continued
Lowest Second Third Fourth
Elements
Common
UNS w/Constant
Variable Equivalent Variable Equivalent Element/ Equivalent Element/ Equivalent
Designation
Valence
Valence Weight Valence Weight Valence Weight Valence Weight
G N06007 Ni/2 (1) 25.46 (2) 22.22 (3) 22.04 (4) 17.03
Carbon Steel: Fe/2 27.92 Fe/3 18.62
(1) = Fe ⁄2, Cr/3, Mo/3, Cu/1, Nb/4,
(3) = Fe ⁄3, Cr ⁄3, Mo/6, Cu/2, Nb/5, Mn/2
Mn/2
(2) = Fe ⁄2, Cr/3, Mo/4, Cu/2, Nb/5,
(4) = Fe ⁄3, Cr/6, Mo/6, Cu/2, Nb/5, Mn/4
Mn/2
Other Metals:
Mg M14142 Mg/2 12.15
Mo R03600 Mo/3 31.98 Mo/4 23.98 Mo/6 15.99
Ag P07016 Ag/1 107.87 Ag/2 53.93
Ta R05210 Ta/5 36.19
Sn L13002 Sn/2 59.34 Sn/4 29.67
Ti R50400 Ti/2 23.95 Ti/3 15.97 Ti/4 11.98
Zn Z19001 Zn/2 32.68
Zr R60701 Zr/4 22.80
Pb L50045 Pb/2 103.59 Pb/4 51.80
A
Registered trademark Carpenter Technology.
B
Registered trademark Haynes International.
TABLE 2 Values of Constants for Use in Faraday’s Equation Rate
5.3.1 Calculate Stern-Geary constants from known Tafel
A slopes where both cathodic and anodic reactions are activation
Penetration
A
controlled, that is, there are distinct linear regions near the
I Unit ρ Unit K Units of K
cor 1 1
Rate Unit (CR)
2 3 corrosion potential on an Elog i plot:
mpy μA/cm g/cm 0.1288 mpy s g/μA cm
B 2B 3B
mm/yr A/m kg/m 327.2 mm s kg/A m y
ba bc
B 2 3 −3
mm/yr μA/cm g/cm 3.27 × 10 mm s g/μA cm y
B 5 (7)
2.303 ~ba1bc!
B
Mass Loss Rate
A
I Unit K Units of K where:
cor 2 2
Unit
2 B 2B
g/m d A/m 0.8953 g s/Ad ba = slope of the anodic Tafel reaction, when plotted on base
2 2 2 2
mg/dm d (mdd) μA/cm 0.0895 mg s cm /μA dm d
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
´1
Designation: G102 − 89 (Reapproved 2015) G102 − 23
Standard Practice for
Calculation of Corrosion Rates and Related Information
from Electrochemical Measurements
This standard is issued under the fixed designation G102; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Editorially corrected the legend below Eq 1 in 4.1 in November 2015.
1. Scope
1.1 This practice covers the providing of guidance in converting the results of electrochemical measurements to rates of uniform
corrosion. Calculation methods for converting corrosion current density values to either mass loss rates or average penetration rates
are given for most engineering alloys. In addition, some guidelines for converting polarization resistance values to corrosion rates
are provided.
1.2 The values stated in SI units are to be regarded as standard. No other Other units of measurement are included in this
standard.standard because of their usage.
1.3 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D2776 Methods of Test for Corrosivity of Water in the Absence of Heat Transfer (Electrical Methods) (Withdrawn 1991)
G1 Practice for Preparing, Cleaning, and Evaluating Corrosion Test Specimens
G5 Reference Test Method for Making Potentiodynamic Anodic Polarization Measurements
G59 Test Method for Conducting Potentiodynamic Polarization Resistance Measurements
3. Significance and Use
3.1 Electrochemical corrosion rate measurements often provide results in terms of electrical current. Although the conversion of
these current values into mass loss rates or penetration rates is based on Faraday’sFaraday’s Law, the calculations can be
complicated for alloys and metals with elements having multiple valence values. This practice is intended to provide guidance in
calculating mass loss and penetration rates for such alloys. Some typical values of equivalent weights for a variety of metals and
alloys are provided.
This practice is under the jurisdiction of ASTM Committee G01 on Corrosion of Metals and is the direct responsibility of Subcommittee G01.11 on Electrochemical
Measurements in Corrosion Testing.
Current edition approved Nov. 1, 2015Feb. 15, 2023. Published December 2015February 2023. Originally approved in 1989. Last previous edition approved in 20102015
as G102–89 (2010).(2015)ɛ . DOI: 10.1520/G0102-89R15E01. 10.1520/G0102-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’sstandard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
G102 − 23
3.2 Electrochemical corrosion rate measurements may provide results in terms of electrical resistance. The conversion of these
results to either mass loss or penetration rates requires additional electrochemical information. Some approaches for estimating this
information are given.
3.3 Use of this practice will aid in producing more consistent corrosion rate data from electrochemical results. This will make
results from different studies more comparable and minimize calculation errors that may occur in transforming electrochemical
results to corrosion rate values.
4. Corrosion Current Density
4.1 Corrosion current values may be obtained from galvanic cells and polarization measurements, including Tafel extrapolations
or polarization resistance measurements. (See Reference Test Method G5 and Practice Test Method G59 for examples.) The first
step is to convert the measured or estimated current value to current density. This is accomplished by dividing the total current
by the geometric area of the electrode exposed to the solution. The surface roughness is generally not taken into account when
calculating the current density. It is assumed that the current distributes uniformly across the area used in this calculation. In the
case of galvanic couples, the exposed area of the anodic specimen should be used. This calculation may be expressed as follows:
I
cor
i 5 (1)
cor
A
where:
where:
i = corrosion current density, μA/cm ,
cor
I = total anodic current, μA, and
cor
A = exposed specimen area, cm .
Other units may be used in this calculation. In some computerized polarization equipment, this calculation is made automatically
after the specimen area is programmed into the computer. A sample calculation is given in Appendix X1.
4.2 Equivalent Weight—Equivalent weight, EW, may be thought of as the mass of metal in grams that will be oxidized by the
passage of one Faraday (96 489 6 2 C (amp-sec)) A·s) of electric charge.
NOTE 1—The value of EW is not dependent on the unit system chosen and so may be considered dimensionless.
NOTE 2—The unit of charge A·s has been named Coulomb, abbreviated C. However, the units A and s are primary SI units, and A·s will be used exclusively
for unit charge below.
For pure elements, the equivalent weight is given by:
W
EW 5 (2)
n
where:
where:
W = the atomic weight of the element, and
n = the number of electrons required to oxidize an atom of the element in the corrosion process, that is, the valence of the
element.
4.3 For alloys, the equivalent weight is more complex. It is usually assumed that the process of oxidation is uniform and does not
occur selectively to any component of the alloy. If this is not true, then the calculation approach will need to be adjusted to reflect
the observed mechanism. In addition, some rationale must be adopted for assigning values of n to the elements in the alloy because
many elements exhibit more than one valence value.
4.4 To calculate the alloy equivalent weight, the following approach may be used. Consider a unit mass of 1 g of an alloy oxidized.
The electron equivalent for 1 g of an alloy, electrical charge equivalent for this mass oxidized, Q, is then:
nifi
Q 5 (3)
(
Wi
G102 − 23
where:
where:
th
fi = the mass fraction of the i element in the alloy,
th
Wi = the atomic weight of the i element in the alloy, and
th
ni = the valence of the i element of the alloy.
Therefore, the alloy equivalent weight, EW, is the reciprocal of this quantity:
EW 5 (4)
nifi
(
Wi
Normally only elements above 1 mass percent in the alloy are included in the calculation. In cases where the actual analysis of
an alloy is not available, it is conventional to use the mid-range of the composition specification for each element, unless a better
basis is available. A sample calculation is given in Appendix X2 (1).
4.5 Valence assignments for elements that exhibit multiple valences can create uncertainty. It is best if an independent technique
can be used to establish the proper valence for each alloying element. Sometimes it is possible to analyze the corrosion products
and use those results to establish the proper valence. Another approach is to measure or estimate the electrode potential of the
corroding surface. Equilibrium diagrams showing regions of stability of various phases as a function of potential and pH may be
created from thermodynamic data. These diagrams are known as Potential-pH (Pourbaix) diagrams and have been published by
several authors (2, 3). The appropriate diagrams for the various alloying elements can be consulted to estimate the stable valence
of each element at the temperature, potential, and pH of the contacting electrolyte that existed during the test.
NOTE 3—Some of the older publications used inaccurate thermodynamic data to construct the diagrams and consequently they are in error.
4.6 Some typical values of EW for a variety of metals and alloys are given in Table 1.
4.7 Calculation of Corrosion Rate—Faraday’s Law can be used to calculate the corrosion rate, either in terms of penetration rate
(CR) or mass loss rate (MR) (4):
i
cor
CR 5 K EW (5)
ρ
MR 5 K i EW (6)
2 cor
where:
CR is given in mm/yr, i in μA/cm ,
cor
−3
K = 3.27 × 10 , mm g/μA cm yr (Note 3),
−3
K = 3.27 × 10 , mm s g/μA cm yr (Note 4),
ρ = density in g/cm , (see Practice G1 for density values for many metals and alloys used in corrosion testing),
MR = g/m d, and
−3 2 2
K = 8.954 × 10 , g cm /μA m d (Note 3).
−3 2 2
K = 8.954 × 10 , g s cm /μA m d (Note 4).
NOTE 4—EW is considered dimensionless in these calculations.
Other values for K and K for different unit systems are given in Table 2.
1 2
4.8 Errors that may arise from this procedure are discussed below.
4.8.1 Assignment of incorrect valence values may cause serious errors (5).
4.8.2 The calculation of penetration or mass loss from electrochemical measurements, as described in this standard, assumes that
uniform corrosion is occurring. In cases where non-uniform corrosion processes are occurring, the use of these methods may result
in a substantial underestimation of the true values.
The boldface numbers in parentheses refer to the list of references at the end of this standard.
G102 − 23
TABLE 1 Equivalent Weight Values for a Variety of Metals and Alloys
NOTE 1—Alloying elements at concentrations below 1 % by mass were not included in the calculation, for example, they were considered part of the
basis metal.
NOTE 2—Mid-range values were assumed for concentrations of alloying elements.
NOTE 3—Only consistent valence groupings were used.
NOTE 4—Eq 4 was used to make these calculations.
Lowest Second Third Fourth
Elements
Common
UNS w/Constant
Variable Equivalent Variable Equivalent Element/ Equivalent Element/ Equivalent
Designation
Valence
Valence Weight Valence Weight Valence Weight Valence Weight
Aluminum Alloys:
A
AA1100 A91100 Al/3 8.99
AA2024 A92024 Al/3, Mg/2 Cu/1 9.38 Cu/2 9.32
AA2219 A92219 Al/3 Cu/1 9.51 Cu/2 9.42
AA3003 A93003 Al/3 Mn/2 9.07 Mn/4 9.03 Mn 7 8.98
AA3004 A93004 Al/3, Mg/2 Mn/2 9.09 Mn/4 9.06 Mn 7 9.00
AA5005 A95005 Al/3, Mg/2 9.01
AA5050 A95050 Al/3, Mg/2 9.03
AA5052 A95052 Al/3, Mg/2 9.05
AA5083 A95083 Al/3, Mg/2 9.09
AA5086 A95086 Al/3, Mg/2 9.09
AA5154 A95154 Al/3, Mg/2 9.08
AA5454 A95454 Al/3, Mg/2 9.06
AA5456 A95456 Al/3, Mg/2 9.11
AA6061 A96061 Al/3, Mg/2 9.01
Al/3, Mg/2,
AA6070 A96070 8.98
Si/4
AA6101 A96161 Al/3 8.99
AA7072 A97072 Al/3, Zn/2 9.06
Al/3, Zn/2,
AA7075 A97075 Cu/1 9.58 Cu/2 9.55
Mg/2
Al/3, Zn/2,
AA7079 A97079 9.37
Mg/2
Al/3, Zn/2,
AA7178 A97178 Cu/1 9.71 Cu/2 9.68
Mg/2
Copper Alloys:
CDA110 C11000 Cu/1 63.55 Cu/2 31.77
CDA220 C22000 Zn/2 Cu/1 58.07 Cu/2 31.86
CDA230 C23000 Zn/2 Cu/1 55.65 Cu/2 31.91
CDA260 C26000 Zn/2 Cu/1 49.51 Cu/2 32.04
CDA280 C28000 Zn/2 Cu/1 46.44 Cu/2 32.11
CDA444 C44300 Zn/2 Cu/1, Sn/2 50.42 Cu/1, Sn/4 50.00 Cu/2, Sn/4 32.00
CDA687 C68700 Zn/2, Al/3 Cu/1 48.03 Cu/2 30.29
CDA608 C60800 Al/3 Cu/1 47.114 Cu/2 27.76
CDA510 C51000 Cu/1, Sn/2 63.32 Cu/1, Sn/4 60.11 Cu/2, Sn/4 31.66
CDA524 C52400 Cu/1, Sn/2 63.10 Cu/1, Sn/4 57.04 Cu/2, Sn/4 31.55
CDA655 C65500 Si/4 Cu/1 50.21 Cu/2 28.51
CDA706 C70600 Ni/2 Cu/1 56.92 Cu/2 31.51
CDA715 C71500 Ni/2 Cu/1 46.69 Cu/2 30.98
CDA752 C75200 Ni/2, Zn/2 Cu/1 46.38 Cu/2 31.46
Stainless Steels:
304 S30400 Ni/2 Fe/2, Cr/3 25.12 Fe/3, Cr/3 18.99 Fe/3, Cr/6 15.72
321 S32100 Ni/2 Fe/2, Cr/3 25.13 Fe/3, Cr/3 19.08 Fe/3, Cr/6 15.78
309 S30900 Ni/2 Fe/2, Cr/3 24.62 Fe/3, Cr/3 19.24 Fe/3, Cr/6 15.33
310 S31000 Ni/2 Fe/2, Cr/3 24.44 Fe/3, Cr/3 19.73 Fe/3, Cr/6 15.36
316 S31600 Ni/2 Fe/2, Cr/3, Mo/3 25.50 Fe/2, Cr/3, Mo/4 25.33 Fe/3, Cr/6, Mo/6 19.14 Fe/3, Cr/6, Mo/6 16.111
317 S31700 Ni/2 Fe/2, Cr/3, Mo/3 25.26 Fe/2, Cr/3, Mo/4 25.03 Fe/3, Cr/3, Mo/6 19.15 Fe/3, Cr/6, Mo/6 15.82
410 S41000 Fe/2, Cr/3 25.94 Fe/3, Cr/3 18.45 Fe/3, Cr/6 16.28
430 S43000 Fe/2, Cr/3 25.30 Fe/3, Cr/3 18.38 Fe/3, Cr/6 15.58
446 S44600 Fe/2, Cr/3 24.22 Fe/3, Cr/3 18.28 Fe/3, Cr/6 14.46
Fe/2, Cr/3, Mo/3, Fe/2, Cr/3, Mo/ Fe/3, Cr/3, Mo/ Fe/3, Cr/6, Mo/6,
A
20CB3 N08020 Ni/2 23.98 23.83 18.88 15.50
Cu/1 4, Cu/1 6, Cu/2 Cu/2
Nickel Alloys:
200 N02200 NI/2 29.36 Ni/3 19.57
400 N04400 Ni/2 Cu/1 35.82 Cu/2 30.12
600 N06600 Ni/2 Fe/2, Cr/3 26.41 Fe/3, Cr/3 25.44 Fe/3, Cr/6 20.73
800 N08800 Ni/2 Fe/2, Cr/3 25.10 Fe/3, Cr/3 20.76 Fe/3, Cr/6 16.59
Fe/2, Cr/3, Mo/3, Fe/2, Cr/3, Mo/ Fe/3, Cr/3, Mo/ Fe/3, Cr/6, Mo/6,
825 N08825 Ni/2 25.52 25.32 21.70 17.10
Cu/1 4, Cu/1 6, Cu/2 Cu/2
B N10001 Ni/2 Mo/3, Fe/2 30.05 Mo/4, Fe/2 27.50 Mo/6, Fe/2 23.52 Mo/6, Fe/3 23.23
Fe/2, Cr/3, Mo/3, Fe/2, Cr/3, Mo/ Fe/2, Cr/3, Mo/ Fe/3, Cr/6, Mo/6,
B
C-22 N06022 Ni/2 26.04 25.12 23.28 17.88
W/4 4, W/4 6, W/6 W/6
Fe/2, Cr/3, Mo/3, Fe/2, Cr/3, Mo/ Fe/3, Cr/6, Mo/6,
C-276 N10276 Ni/2 27.09 Cr/3, Mo/4 25.90 23.63 19.14
W/4 6, W/6 W/6
G102 − 23
TABLE 1 Continued
Lowest Second Third Fourth
Elements
Common
UNS w/Constant
Variable Equivalent Variable Equivalent Element/ Equivalent Element/ Equivalent
Designation
Valence
Valence Weight Valence Weight Valence Weight Valence Weight
G N06007 Ni/2 (1) 25.46 (2) 22.22 (3) 22.04 (4) 17.03
Carbon Steel: Fe/2 27.92 Fe/3 18.62
(1) = Fe ⁄2, Cr/3, Mo/3, Cu/1, Nb/4,
(3) = Fe ⁄3, Cr/3, Mo/6, Cu/2, Nb/5, Mn/2
Mn/2
(1) = Fe ⁄2, Cr/3, Mo/3, Cu/1, Nb/4,
(3) = Fe ⁄3, Cr ⁄3, Mo/6, Cu/2, Nb/5, Mn/2
Mn/2
(2) = Fe ⁄2, Cr/3, Mo/4, Cu/2, Nb/5,
(4) = Fe ⁄3, Cr/6, Mo/6, Cu/2, Nb/5, Mn/4
Mn/2
(2) = Fe ⁄2, Cr/3, Mo/4, Cu/2, Nb/5,
(4) = Fe ⁄3, Cr/6, Mo/6, Cu/2, Nb/5, Mn/4
Mn/2
Other Metals:
Mg M14142 Mg/2 12.15
Mo R03600 Mo/3 31.98 Mo/4 23.98 Mo/6 15.99
Ag P07016 Ag/1 107.87 Ag/2 53.93
Ta R05210 Ta/5 36.19
Sn L13002 Sn/2 59.34 Sn/4 29.67
Ti R50400 Ti/2 23.95 Ti/3 15.97 Ti/4 11.98
Zn Z19001 Zn/2 32.68
Zr R60701 Zr/4 22.80
Pb L50045 Pb/2 103.59 Pb/4 51.80
A
Registered trademark Carpenter Technology.
B
Registered trademark Haynes International.
TABLE 2 Values of Constants for Use in Faraday’s Equation Rate
A
Penetration
A
I Unit ρ Unit K Units of K
cor 1 1
Rate Unit (CR)
2 3
mpy μA/cm g/cm 0.1288 mpy g/μA cm
B 2B 3B
mm/yr A/m kg/m 327.2 mm kg/A m y
B 2 3 −3
mm/yr μA/cm g/cm 3.27 × 10 mm g/μA cm y
B
Mass Loss Rate
A
I Unit K Units of K
cor 2 2
Unit
2 B 2B
g/m d A/m 0.8953 g/Ad
2 2 2 2
mg/dm d (mdd) μA/cm 0.0895 mg cm /μA dm d
2 2B −3 2 2
mg/dm d (mdd) A/m 8.953 × 10 mg m /A dm d
TABLE 2 Values of Constants for Use in Faraday’s Equation Rate
A
Penetration
A
I Unit ρ Unit K Units of K
cor 1 1
Rate Unit (CR)
2 3
mpy μA/cm g/cm 0.1288 mpy s g/μA cm
B 2B 3B
mm/yr A/m kg/m 327.2 mm s kg/A m y
B 2 3 −3
mm/yr μA/cm g/cm 3.27 × 10 mm s g/μA cm y
B
Mass Loss Rate
A
I Unit K Units of K
cor 2 2
Unit
2 B 2B
g/m d A/m 0.8953 g s/Ad
2 2 2 2
mg/dm d (mdd) μA/cm 0.0895 mg s cm /μA dm d
2 2B 2 2
mg/dm d (mdd) A/m 8.953 mg s m /A dm d
A
EW is assumed to be dimensionless.
B
SI unit.
4.8.3 Alloys that include large quantities of metalloids or oxidized materials may not be able to be treated by the above procedure.
4.8.4 Corrosion rates calculated by the method above where abrasion or erosion is a significant contributor to the metal loss
process may yield significant underestimation of the metal loss rate.
5. Polarization Resistance
5.1 Polarization resistance values may be approximated from either potentiodynamic measurements near the corro
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...