ASTM F3169-16
(Test Method)Standard Test Method for Leak Detection in Blister Packaging by Vacuum Deflection Method by Laser Measurement
Standard Test Method for Leak Detection in Blister Packaging by Vacuum Deflection Method by Laser Measurement
SIGNIFICANCE AND USE
5.1 Leaks in blister packs may affect product quality and such defects can arise from imperfections in the packaging material or bond between the sealed surfaces.
5.2 This method of leak testing is a useful tool as it allows non-destructive and non-subjective leak testing of blister packs. It allows the operator to evaluate how different packaging materials and packaging machine conditions affect the integrity of the packaging. It can also provide indication of unwanted changes in the packaging conditions.
5.3 This type of testing is typically used in pharmaceutical packaging production, during stability trials and for package research and development operations because of its non-destructive nature, cleanliness, and speed.
SCOPE
1.1 Test Packages—This test method can be applied to non-porous blister packs sealed with flexible films such as those used in pharmaceutical packaging. Such blister packs typically consist of thermoformed polymer or cold formed aluminum trays that contain a number of individual blister pockets into which tablets or capsules are placed. The trays are then sealed with a polymer, paper-backed or foil-based flexible laminate lidding material.
1.2 Leaks Detected—This test method detects leaks in blister packs by measuring the deflection of the blister pack surface in response to an applied vacuum. This deflection of the blister pack surface results from the difference in pressure between the gas inside the blister pack and the applied vacuum. Air loss from within a blister pocket as a result of a leak will alter this pressure differential causing a measureable variation in blister pocket deflection. This test method requires that the blister packs are held in appropriate tooling inside a suitable test chamber.
1.3 Test Results—Test results are reported qualitatively (pass/fail). Appropriate acceptance criteria for deflection, height, and collapse values are established by comparing non-leaking packs with those containing defects of a known size. Suitably sized defects in the laminate, tray material, and seal can be detected using this test method. The sensitivity of this test method depends upon a range of factors including blister pocket headspace, blister pocket size, lidding material type, lidding material thickness, lidding material tension, printing, surface texture, test conditions, and the values selected for the pass/fail acceptance criteria. The ability of the test to detect 15 µm, 50 µm, and catastrophic sized holes in four blister pack designs was demonstrated in a study.
1.4 The values stated in SI units are to be regarded as standard and no other units of measurement are included in this test method.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 30-Nov-2016
- Technical Committee
- F02 - Primary Barrier Packaging
- Drafting Committee
- F02.40 - Package Integrity
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 01-Oct-2018
- Effective Date
- 15-Aug-2018
- Effective Date
- 01-Jun-2017
- Effective Date
- 01-Aug-2013
- Effective Date
- 15-Apr-2013
- Effective Date
- 01-Nov-2012
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Aug-2008
- Effective Date
- 01-Sep-2007
- Effective Date
- 01-May-2007
- Effective Date
- 01-Dec-2006
- Effective Date
- 01-Apr-2004
- Effective Date
- 10-Oct-2002
- Effective Date
- 10-Apr-2002
Overview
ASTM F3169-16 is the internationally recognized standard test method for leak detection in blister packaging by the vacuum deflection method using laser measurement. Developed by ASTM International, this standard provides a non-destructive and objective technique for identifying leaks in non-porous blister packs sealed with flexible or foil-based lidding materials. Blister packaging is widely used in the pharmaceutical industry for tablets and capsules, and maintaining the integrity of these packages is crucial to ensure product quality and regulatory compliance.
This standard details how to measure blister pack surface deflection under vacuum to detect leaks. By monitoring changes in the surface profile, especially in the presence of minute holes or defective seals, manufacturers can quickly verify the integrity of each pack without damaging the product.
Key Topics
Leak Detection in Blister Packs: The method involves applying a vacuum to a test chamber containing the blister packs and measuring the deflection of each blister pocket using precise laser equipment. Leaks are identified by a measurable variation in blister deflection compared to non-leaking packs.
Non-Destructive Testing: Unlike some other leak detection techniques, this method does not alter or spoil the packaging, allowing further testing or use of the tested samples.
Quantitative and Qualitative Results: Results are generally reported as pass/fail based on set acceptance criteria for deflection, height, and collapse in the blister pockets. Criteria are tailored according to the packaging design, materials, and known defect sizes.
Factors Affecting Sensitivity: The sensitivity of the test is influenced by blister pocket size, available headspace, lidding material, thickness, material tension, and surface texture, as well as the vacuum parameters selected for the tests.
Equipment and Calibration: The test setup typically includes a vacuum chamber, precise measurement devices (laser sensor and actuator), and tooling specific to the blister pack format. Proper calibration ensures accuracy and repeatability.
Applications
The vacuum deflection leak test method is primarily applied in:
- Pharmaceutical Packaging Production: For routine testing during manufacturing to confirm package integrity and compliance with industry standards.
- Stability Trials: During product development and shelf-life studies to monitor packaging performance over time.
- Research and Development: To evaluate new materials or package designs and verify the performance of packaging machinery.
- Quality Assurance: To detect small defects that could impact product efficacy or patient safety, including micro-holes, seal failures, and gross tears.
- Regulatory Compliance: Satisfying quality control and integrity verification requirements in pharmaceutical packaging processes.
The method's non-destructive nature, cleanliness, and speed are ideal for sensitive products and high-throughput environments.
Related Standards
Several related ASTM standards support or complement ASTM F3169-16 in the field of packaging integrity and leak testing:
- ASTM D996: Terminology of Packaging and Distribution Environments
- ASTM F17: Terminology Relating to Flexible Barrier Packaging
Other standards may be referenced for calibration, packaging materials, and detailed testing protocols for different packaging types or barrier properties. Users should refer to the latest versions and additional guidance where needed for harmonized quality management in pharmaceutical packaging.
Keywords: blister packaging leak detection, vacuum deflection method, laser measurement, pharmaceutical packaging integrity, non-destructive leak testing, ASTM F3169-16, flexible packaging, package quality control, vacuum leak test, pass/fail criteria
Buy Documents
ASTM F3169-16 - Standard Test Method for Leak Detection in Blister Packaging by Vacuum Deflection Method by Laser Measurement
Get Certified
Connect with accredited certification bodies for this standard

BRCGS (Brand Reputation Compliance Global Standards)
Global food safety and quality standards owner.
Sponsored listings
Frequently Asked Questions
ASTM F3169-16 is a standard published by ASTM International. Its full title is "Standard Test Method for Leak Detection in Blister Packaging by Vacuum Deflection Method by Laser Measurement". This standard covers: SIGNIFICANCE AND USE 5.1 Leaks in blister packs may affect product quality and such defects can arise from imperfections in the packaging material or bond between the sealed surfaces. 5.2 This method of leak testing is a useful tool as it allows non-destructive and non-subjective leak testing of blister packs. It allows the operator to evaluate how different packaging materials and packaging machine conditions affect the integrity of the packaging. It can also provide indication of unwanted changes in the packaging conditions. 5.3 This type of testing is typically used in pharmaceutical packaging production, during stability trials and for package research and development operations because of its non-destructive nature, cleanliness, and speed. SCOPE 1.1 Test Packages—This test method can be applied to non-porous blister packs sealed with flexible films such as those used in pharmaceutical packaging. Such blister packs typically consist of thermoformed polymer or cold formed aluminum trays that contain a number of individual blister pockets into which tablets or capsules are placed. The trays are then sealed with a polymer, paper-backed or foil-based flexible laminate lidding material. 1.2 Leaks Detected—This test method detects leaks in blister packs by measuring the deflection of the blister pack surface in response to an applied vacuum. This deflection of the blister pack surface results from the difference in pressure between the gas inside the blister pack and the applied vacuum. Air loss from within a blister pocket as a result of a leak will alter this pressure differential causing a measureable variation in blister pocket deflection. This test method requires that the blister packs are held in appropriate tooling inside a suitable test chamber. 1.3 Test Results—Test results are reported qualitatively (pass/fail). Appropriate acceptance criteria for deflection, height, and collapse values are established by comparing non-leaking packs with those containing defects of a known size. Suitably sized defects in the laminate, tray material, and seal can be detected using this test method. The sensitivity of this test method depends upon a range of factors including blister pocket headspace, blister pocket size, lidding material type, lidding material thickness, lidding material tension, printing, surface texture, test conditions, and the values selected for the pass/fail acceptance criteria. The ability of the test to detect 15 µm, 50 µm, and catastrophic sized holes in four blister pack designs was demonstrated in a study. 1.4 The values stated in SI units are to be regarded as standard and no other units of measurement are included in this test method. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 5.1 Leaks in blister packs may affect product quality and such defects can arise from imperfections in the packaging material or bond between the sealed surfaces. 5.2 This method of leak testing is a useful tool as it allows non-destructive and non-subjective leak testing of blister packs. It allows the operator to evaluate how different packaging materials and packaging machine conditions affect the integrity of the packaging. It can also provide indication of unwanted changes in the packaging conditions. 5.3 This type of testing is typically used in pharmaceutical packaging production, during stability trials and for package research and development operations because of its non-destructive nature, cleanliness, and speed. SCOPE 1.1 Test Packages—This test method can be applied to non-porous blister packs sealed with flexible films such as those used in pharmaceutical packaging. Such blister packs typically consist of thermoformed polymer or cold formed aluminum trays that contain a number of individual blister pockets into which tablets or capsules are placed. The trays are then sealed with a polymer, paper-backed or foil-based flexible laminate lidding material. 1.2 Leaks Detected—This test method detects leaks in blister packs by measuring the deflection of the blister pack surface in response to an applied vacuum. This deflection of the blister pack surface results from the difference in pressure between the gas inside the blister pack and the applied vacuum. Air loss from within a blister pocket as a result of a leak will alter this pressure differential causing a measureable variation in blister pocket deflection. This test method requires that the blister packs are held in appropriate tooling inside a suitable test chamber. 1.3 Test Results—Test results are reported qualitatively (pass/fail). Appropriate acceptance criteria for deflection, height, and collapse values are established by comparing non-leaking packs with those containing defects of a known size. Suitably sized defects in the laminate, tray material, and seal can be detected using this test method. The sensitivity of this test method depends upon a range of factors including blister pocket headspace, blister pocket size, lidding material type, lidding material thickness, lidding material tension, printing, surface texture, test conditions, and the values selected for the pass/fail acceptance criteria. The ability of the test to detect 15 µm, 50 µm, and catastrophic sized holes in four blister pack designs was demonstrated in a study. 1.4 The values stated in SI units are to be regarded as standard and no other units of measurement are included in this test method. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM F3169-16 is classified under the following ICS (International Classification for Standards) categories: 55.040 - Packaging materials and accessories. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3169-16 has the following relationships with other standards: It is inter standard links to ASTM F17-20, ASTM F17-18a, ASTM F17-18, ASTM F17-17, ASTM F17-13a, ASTM F17-13, ASTM F17-12, ASTM D996-10a, ASTM F17-08, ASTM F17-07a, ASTM F17-07, ASTM F17-06, ASTM D996-04, ASTM F17-02, ASTM D996-02. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F3169-16 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:F3169 −16
Standard Test Method for
Leak Detection in Blister Packaging by Vacuum Deflection
Method by Laser Measurement
This standard is issued under the fixed designation F3169; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 1.5 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
1.1 Test Packages—This test method can be applied to
responsibility of the user of this standard to establish appro-
non-porous blister packs sealed with flexible films such as
priate safety and health practices and determine the applica-
those used in pharmaceutical packaging. Such blister packs
bility of regulatory limitations prior to use.
typically consist of thermoformed polymer or cold formed
aluminum trays that contain a number of individual blister
2. Referenced Documents
pockets into which tablets or capsules are placed. The trays are
2.1 ASTM Standards:
then sealed with a polymer, paper-backed or foil-based flexible
D996 Terminology of Packaging and Distribution Environ-
laminate lidding material.
ments
1.2 Leaks Detected—This test method detects leaks in
F17 Terminology Relating to Flexible Barrier Packaging
blister packs by measuring the deflection of the blister pack
surfaceinresponsetoanappliedvacuum.Thisdeflectionofthe
3. Terminology
blister pack surface results from the difference in pressure
3.1 For definitions used in this test method, see Terminolo-
betweenthegasinsidetheblisterpackandtheappliedvacuum.
gies D996 and F17.
Air loss from within a blister pocket as a result of a leak will
3.2 Definitions of Terms Specific to This Standard:
alter this pressure differential causing a measureable variation
3.2.1 blister pack, n—forming material, encapsulated
in blister pocket deflection. This test method requires that the
product, and sealed lidding material.
blister packs are held in appropriate tooling inside a suitable
test chamber.
3.2.2 blister pocket, n—sealed cavity in the forming mate-
rial that contains product.
1.3 Test Results—Test results are reported qualitatively
(pass/fail). Appropriate acceptance criteria for deflection, 3.2.3 collapse, n—difference in height of the blister pocket
height, and collapse values are established by comparing
profile in one plane before and after the reduced vacuum has
non-leaking packs with those containing defects of a known been applied.
size. Suitably sized defects in the laminate, tray material, and
3.2.4 deflection, n—difference in height of the blister pocket
seal can be detected using this test method. The sensitivity of
profileinoneplanebeforeandaftertheinitialvacuumhasbeen
this test method depends upon a range of factors including
applied.
blister pocket headspace, blister pocket size, lidding material
3.2.5 height, n—difference in height of the blister pocket
type, lidding material thickness, lidding material tension,
profile in one plane after initial vacuum when compared
printing, surface texture, test conditions, and the values se-
against blister pack surface height.
lected for the pass/fail acceptance criteria. The ability of the
3.2.6 profile, n—surface of the lidding material measured at
testtodetect15µm,50µm,andcatastrophicsizedholesinfour
regular intervals across the blister pocket.
blister pack designs was demonstrated in a study.
1.4 The values stated in SI units are to be regarded as
4. Summary of Test Method
standard and no other units of measurement are included in this
4.1 The test blister packs are located in the test chamber in
test method.
appropriate tooling to prevent them from moving under
vacuum. The chamber is then sealed and the profile of every
This test method is under the jurisdiction ofASTM Committee F02 on Primary
Barrier Packaging and is the direct responsibility of Subcommittee F02.40 on For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Package Integrity. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Current edition approved Dec. 1, 2016. Published January 2017. DOI: 10.1520/ Standards volume information, refer to the standard’s Document Summary page on
F3169-16 the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3169−16
blister pocket is measured using an appropriate measurement by comparing the deflection and collapse values of good blister
device such as a laser and actuator (see Fig. 1). packs and those containing defects of the required size. Such
defectscanbeproducedbyusingawire,bylaserdrilling,orby
4.2 A vacuum is then applied to the test chamber and each
applying self-adhesive predrilled holes.
blister pocket measured again. Sealed blister pockets can be
identified by the change in measurement between the first and 4.6 Where blister pockets appear pre-swollen the flat sealed
surface of the blister pack is used to project a datum line across
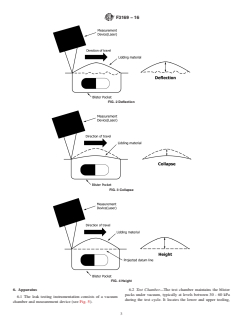
second profiles. This is known as deflection and is caused by
the pressure difference between the air inside the blister pocket the blister pocket from which a change in height under vacuum
can be observed. If this height value is small or negative it can
and the air in the chamber caused by the application of vacuum
(see Fig. 2). be understood that the blister pocket has a gross hole, as the
blister pocket has not deflected due to vacuum or positive
4.3 The deflection of a blister pocket with a gross hole will
pressure before testing (see Fig. 4).
be minimal as the pressure difference between the air inside
4.7 Any blister pocket that falls outside the acceptance
and outside of the blister pocket will equalize through the hole.
criteria for deflection, collapse,or height is recorded as a fail.
4.4 The sensitivity of the test can be increased by holding
The ability of the test to detect 15 and 50 µm sized laser drilled
the test samples under vacuum for a period of time and then
holes was verified using four blister pack designs. This method
reducing the vacuum level. After reducing the vacuum, the
was also used to find large catastrophic defects demonstrated
surface profile of the blister pockets can be measured again and
witha5mm slit in the lidding material.
compared with the profiles recorded after the initial vacuum
4.8 Background noise may occur due to movement of the
was applied. Reducing the vacuum level causes blister pockets
blister pack under vacuum. Such noise can be minimized by
with small holes to have a collapse measurement greater than
selection of an appropriate measurement device, proper reten-
blister pockets with no defects. This is due to air leaking from
tion of the blister pack, and careful selection of pressures used
any small defects (see Fig. 3).
for testing.
4.5 The sensitivity of the test is a function of the test
parameters chosen, namely vacuum levels, hold time, and the
5. Significance and Use
pass/fail criteria selected. Blister pack design including head-
5.1 Leaks in blister packs may affect product quality and
space and blister pocket size, lidding material, lidding material
such defects can arise from imperfections in the packaging
thickness, lidding material tension, printing, and surface tex-
material or bond between the sealed surfaces.
ture can affect test sensitivity. The vacuum levels used are
5.2 This method of leak testing is a useful tool as it allows
typically in the range of 30 - 60 KPa and are chosen based on
non-destructive and non-subjective leak testing of blister
setup tests conducted using good blister packs and those
packs. It allows the operator to evaluate how different pack-
containing defects of known size. The required vacuum level
aging materials and packaging machine conditions affect the
depends upon the blister pack design and materials. For
integrity of the packaging. It can also provide indication of
example, detecting sub-15 µm defects in small blister pockets
unwanted changes in the packaging conditions.
sealed with a stiff lidding material typically requires vacuum
levels towards the higher end of the standard range. The 5.3 This type of testing is typically used in pharmaceutical
vacuum level is then reduced and the packs are rescanned after
packaging production, during stability trials and for package
a holding period (typically 10 - 45 s) at this reduced vacuum. research and development operations because of its non-
The test parameters, including the pass/fail criteria, are chosen
destructive nature, cleanliness, and speed.
FIG. 1Profile
F3169−16
FIG. 2Deflection
FIG. 3Collapse
FIG. 4Height
6. Apparatus 6.2 Test Chamber—The test chamber maintains the blister
packs under vacuum, typically at levels between 30 - 60 kPa
6.1 The leak testing instrumentation consists of a vacuum
during the test cycle. It locates the lower and upper tooling,
chamber and measurement device (see Fig. 5).
F3169−16
FIG. 5Leak Testing Instrumentation
allowing the measurement device to accurately pinpoint the 9. Calibr
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...