ASTM D3352-15
(Test Method)Standard Test Method for Strontium Ion in Brackish Water, Seawater, and Brines
Standard Test Method for Strontium Ion in Brackish Water, Seawater, and Brines
SIGNIFICANCE AND USE
5.1 This test method4 can be used to determine strontium ions in brackish water, seawater, and brines.
SCOPE
1.1 This test method covers the determination of soluble strontium ion in brackish water, seawater, and brines by atomic absorption spectrophotometry.
1.2 Samples containing from 5 to 2100 mg/L of strontium may be analyzed by this test method.
1.3 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 31-Jan-2015
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.05 - Inorganic Constituents in Water
Relations
- Replaces
ASTM D3352-08a - Standard Test Method for Strontium Ion in Brackish Water, Seawater, and Brines - Effective Date
- 01-Feb-2015
- Effective Date
- 01-May-2020
- Effective Date
- 15-Jun-2012
- Effective Date
- 01-May-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-Jan-2008
- Effective Date
- 01-Dec-2007
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 15-Aug-2006
- Effective Date
- 15-Aug-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 15-Feb-2006
Overview
ASTM D3352-15 is the international standard test method for determining strontium ion concentrations in brackish water, seawater, and brines using atomic absorption spectrophotometry. Developed by ASTM International, this standard provides guidance for accurate analysis of soluble strontium ions in challenging aquatic matrices common to environmental monitoring, water quality assessment, and industrial processes. The method is applicable for samples containing between 5 to 2100 mg/L of strontium.
This standard supports sound laboratory practices in line with global trade and regulatory expectations by adopting internationally recognized principles for standardization.
Key Topics
- Sample Applicability: Suitable for brackish water, seawater, and brines with strontium ion concentrations in the 5-2100 mg/L range.
- Analytical Method: Utilizes atomic absorption spectrophotometry, ensuring reliable quantification in highly saline or complex matrices.
- Quality Assurance: Emphasizes stringent quality control procedures, including calibration with working standards, method blanks, and regular verification using laboratory control samples and spike recovery tests.
- Interference Management: Addresses potential interference from matrix components such as silicon, aluminum, and phosphate by introducing lanthanum to suppress adverse chemical and ionization effects.
- Precision and Bias: Provides statistical guidance for acceptable performance, including interlaboratory precision and accuracy expectations.
- Reporting Units: Results are to be expressed in SI units (mg/L), supporting international data exchange and regulatory compliance.
- Safety Considerations: Reminds users to establish and observe all relevant safety and health protocols during test execution.
Applications
ASTM D3352-15 is widely adopted across industries where precise measurement of strontium ions is required, including:
- Environmental Monitoring: Tracking strontium levels in marine and estuarine ecosystems, ensuring compliance with environmental protection standards.
- Industrial Brine Management: Supporting oil and gas operations, desalination, and other processes where control of strontium concentrations in brines is critical for operational efficiency and regulatory compliance.
- Water Quality Assessment: Used by laboratories, utilities, and regulatory agencies to evaluate the suitability of water resources and prevent contamination.
- Geochemical Research: Assists in characterizing groundwater and formation brines for resource extraction, reservoir management, and geochemical studies.
Laboratories benefit from the method's clarity on sample handling, preparation, and instrumentation, helping to minimize analytical errors and improve confidence in measurement results. The quality control requirements facilitate traceable and defensible data, essential for regulatory reporting and decision-making.
Related Standards
Several ASTM standards are referenced within ASTM D3352-15 to ensure comprehensive water analysis and quality assurance:
- ASTM D1129: Terminology Relating to Water - Provides standard definitions relevant to water analysis.
- ASTM D1193: Specification for Reagent Water - Outlines requirements for purity of water used in analytical procedures.
- ASTM D2777: Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water - Supports statistical evaluation of test method performance.
- ASTM D3370: Practices for Sampling Water from Closed Conduits - Ensures representative sampling for laboratory analysis.
- ASTM D5810: Guide for Spiking into Aqueous Samples - Provides instructions for matrix spike and recovery procedures.
- ASTM D5847: Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis - Enhances analytical rigor through comprehensive QC protocols.
By integrating ASTM D3352-15 into laboratory workflows, organizations ensure reliable assessment of strontium in saline waters, achieving regulatory compliance and supporting environmental stewardship initiatives.
Keywords: ASTM D3352-15, strontium ion, brackish water, seawater, brines, atomic absorption spectrophotometry, water analysis, environmental monitoring, ASTM standard.
Buy Documents
ASTM D3352-15 - Standard Test Method for Strontium Ion in Brackish Water, Seawater, and Brines
REDLINE ASTM D3352-15 - Standard Test Method for Strontium Ion in Brackish Water, Seawater, and Brines
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D3352-15 is a standard published by ASTM International. Its full title is "Standard Test Method for Strontium Ion in Brackish Water, Seawater, and Brines". This standard covers: SIGNIFICANCE AND USE 5.1 This test method4 can be used to determine strontium ions in brackish water, seawater, and brines. SCOPE 1.1 This test method covers the determination of soluble strontium ion in brackish water, seawater, and brines by atomic absorption spectrophotometry. 1.2 Samples containing from 5 to 2100 mg/L of strontium may be analyzed by this test method. 1.3 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 5.1 This test method4 can be used to determine strontium ions in brackish water, seawater, and brines. SCOPE 1.1 This test method covers the determination of soluble strontium ion in brackish water, seawater, and brines by atomic absorption spectrophotometry. 1.2 Samples containing from 5 to 2100 mg/L of strontium may be analyzed by this test method. 1.3 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM D3352-15 is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D3352-15 has the following relationships with other standards: It is inter standard links to ASTM D3352-08a, ASTM D1129-13(2020)e2, ASTM D2777-12, ASTM D5810-96(2011), ASTM D3370-10, ASTM D1129-10, ASTM D3370-08, ASTM D2777-08, ASTM D3370-07, ASTM D1129-06a, ASTM D1129-06ae1, ASTM D5810-96(2006), ASTM D2777-06, ASTM D1193-06, ASTM D1129-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D3352-15 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:D3352 −15
Standard Test Method for
Strontium Ion in Brackish Water, Seawater, and Brines
This standard is issued under the fixed designation D3352; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* wavelength they emit when excited. When radiation from a
given excited element is passed through a flame containing
1.1 This test method covers the determination of soluble
ground state atoms of that element, the intensity of the
strontium ion in brackish water, seawater, and brines by atomic
transmitted radiation will decrease in proportion to the amount
absorption spectrophotometry.
of the ground state element in the flame. A hollow cathode
1.2 Samples containing from 5 to 2100 mg/L of strontium
lamp whose cathode is made of the element to be determined
may be analyzed by this test method. 3
provides the radiation. The metal atoms to be measured are
placed in the beam of radiation by aspirating the specimen into
1.3 The values stated in SI units are to be regarded as
standard. The values given in parentheses are mathematical an oxidant-fuel flame. A monochromator isolates the charac-
teristic radiation from the hollow cathode lamp and a photo-
conversions to inch-pound units that are provided for informa-
tion only and are not considered standard. sensitive device measures the attenuated transmitted radiation.
1.4 This standard does not purport to address all of the 4.2 Sincethevariableandsometimeshighconcentrationsof
safety concerns, if any, associated with its use. It is the
matrix materials in the waters and brines affect absorption
responsibility of the user of this standard to establish appro- differently,itisdifficulttopreparestandardssufficientlysimilar
priate safety and health practices and determine the applica-
to the waters and brines. To overcome this difficulty, the
bility of regulatory limitations prior to use. method of additions is used in which three identical samples
are prepared and varying amounts of a standard added to two
2. Referenced Documents
of them. The three samples are then aspirated, the concentra-
2.1 ASTM Standards:
tion readings recorded, and the original sample concentration
D1129 Terminology Relating to Water
calculated.
D1193 Specification for Reagent Water
5. Significance and Use
D2777 Practice for Determination of Precision and Bias of
Applicable Test Methods of Committee D19 on Water
5.1 This test method can be used to determine strontium
D3370 Practices for Sampling Water from Closed Conduits
ions in brackish water, seawater, and brines.
D5810 Guide for Spiking into Aqueous Samples
6. Interferences
D5847 Practice for Writing Quality Control Specifications
for Standard Test Methods for Water Analysis
6.1 The chemical suppression caused by silicon, aluminum,
and phosphate is controlled by adding lanthanum. The lantha-
3. Terminology
num also controls ionization interference.
3.1 Definitions—For definitions of terms used in this test
7. Apparatus
method, refer to Terminology D1129.
7.1 Atomic Absorption Spectrophotometer—The instrument
4. Summary of Test Method
shall consist of atomizer and burner, suitable pressure-
4.1 This test method is dependent on the fact that metallic
regulating devices capable of maintaining constant oxidant and
elements, in the ground state, will absorb light of the same
For additional information on atomic absorption, see the following references:
This test method is under the jurisdiction of ASTM Committee D19 on Water Angino, E. E., and Billings, G. K., Atomic Absorption Spectrophotometry in
and is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents Geology, Elsevier Publishing Co., NewYork, N.Y., 1967; Dean, J.A., and Rains, T.
in Water. C., Editors, Flame Emission and Atomic Absorption Spectrometry Vol 1−Theory,
Current edition approved Feb. 1, 2015. Published April 2015. Originally Marcel Dekker, New York, NY, 1969.
approved in 1974. Last previous edition approved in 2008 as D3352 – 08a. DOI: Additional information is contained in the following references: Fletcher, G. F.,
10.1520/D3352-15. and Collins, A. G., “Atomic Absorption Methods of Analysis of Oilfield Brines:
For referenced ASTM standards, visit the ASTM website, www.astm.org, or Barium,Calcium,Copper,Iron,Lead,Lithium,Magnesium,Manganese,Potassium,
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Sodium,Strontium,andZinc,”U.S.BureauofMines,ReportofInvestigations7861,
Standards volume information, refer to the standard’s Document Summary page on 1974, 14 pp.; Collins, A. G., Geochemistry of Oilfield Waters, Elsevier Publishing
the ASTM website. Co., Amsterdam, Netherlands, 1975.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D3352−15
fuelpressureforthedurationofthetest,ahollowcathodelamp 8.5 Strontium Solution, Standard (1 mL = 1 mg Sr)—
for each metal to be tested, an optical system capable of Dissolve 2.415 g of strontium nitrate [Sr(NO )]in10mLof
3 2
isolating the desired line of radiation, an adjustable slit, a concentrated hydrochloric acid (sp gr 1.19) and about 700 mL
photomultiplier tube or other photosensitive device as a light ofwater.Dilutesolutionto1Lwithwater.Onemillilitreofthis
measuring and amplifying device, and a read-out mechanism solution contains 1 mg of strontium. A purchased strontium
for indicating the amount of absorbed radiation. stock solution of appropriate known purity is also acceptable.
7.1.1 Multi-Element Hollow Cathode Lamps are available
8.6 Oxidant, for Atomic Absorption Spectrophotometer:
and have been found satisfactory.
8.6.1 Air, which has been cleaned and dried through a
7.2 Pressure-Reducing Valves—The supplies of fuel and suitable filter to remove oil, water, and other foreign
oxidant shall be maintained at pressures somewhat higher than substances, is the usual oxidant.
the controlled operating pressure of the instrument by suitable 8.6.2 Nitrous Oxide may be required as an oxidant for
valves. refractory-type metals.
8.7 Fuel, for Atomic Absorption Spectrophotometer:
8. Reagents and Materials
8.7.1 Acetylene—Standard, commercially available acety-
8.1 Purity of Reagents—Reagent grade chemicals shall be
lene is the usual fuel. Acetone, always present in acetylene
used in all tests. Unless otherwise indicated, it is intended that
cylinders, can be prevented from entering and damaging the
all reagents shall conform to the specifications of the Commit-
burner head by replacing a cylinder which only has 689 kPa
teeonAnalyticalReagentsoftheAmericanChemicalSociety,
(100 psi) of acetylene remaining.
where such specifications are available. Other grades may be
used, provided it is first ascertained that the reagent is of 9. Sampling
sufficiently high purity to permit its use without lessening the
9.1 Collect the sample in accordance with Practices D3370.
accuracy of the determination.
10. Procedure
8.2 Purity of Water—Unless otherwise indicated, reference
towatershallbeunderstoodtomeanreagentwaterconforming
10.1 Strontium is determined at the 460.7-nm wavelength
to Specification D1193, Type I. Other reagent water types may
with an air-acetylene flame.
be used provided it is first ascertained that the water is of
10.2 Preliminary Calibration—Using micropipets prepare
sufficiently high purity to permit its use without adversely
standard strontium solutions containing 1 to 10 mg/L of
affecting the precision and bias of the test method. Type III
strontium using the standard strontium solution (8.5) and
water was specified at the time of round robin testing of this
50-mL volumetric flasks. Before making up to volume, add to
test method.
each of these and to a blank, 5 mL of the lanthanum solution
8.3 Filter Paper—Purchase suitable filter paper. Typically
(8.4). Analyze at least three working standards containing
the filter papers have a pore size of 0.45-µm membrane.
concentrations of strontium that bracket the expected sample
Material such as fine-textured, acid-washed, ashless paper, or
concentration, prior to analysis of samples, to calibrate the
glass fiber paper are acceptable. The user must first ascertain
instrument. Aspirate these standards and the blank (for back-
that the filter paper is of sufficient purity to use without
ground setting) and adjust the curvature controls, if necessary,
adversely affecting the bias and precision of the test method.
to obtain a linear relationship between absorbance and the
actual concentration of the standards.
8.4 Lanthanum Solution (5 % La)—Wet 58.65 g of lantha-
num oxide (La O ) with water. Add 250 mL of concentrated
10.3 Transfer an aliquot of water or brine (previously
2 3
hydrochloric acid (sp gr 1.19) very slowly until the material is
filtered (8.3) through a 0.45-µm filter) to a 50-mL volumetric
dissolved. Dilute solution to 1 litre with water.
flask. The specific gravity of the water or brine can be used to
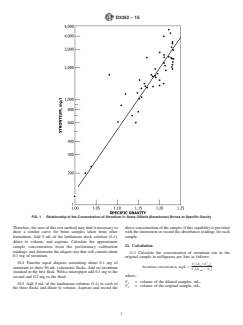
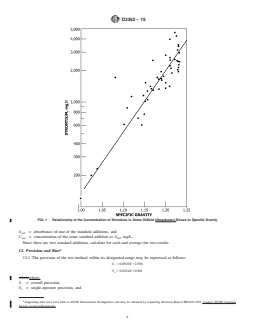
estimatethestrontiumcontentofthesampleand,thereby,serve
as a basis for selecting the aliquot size that will contain about
Reagent Chemicals, American Chemical Society Specifications, American
Chemical Society, Washington, DC. For Suggestions on the testing of reagents not 0.1 mg of strontium. Fig. 1 shows the relationship between
listed by the American Chemical Society, see Annual Standards for Laboratory
strontium concentration and specific gravity for some oilfield
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
brines from the Smackover formation. The concentrations of
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
strontium in the Smackover brines will not necessarily corre-
MD.
late with the concentrations found in other formations.
TABLE 1 Compositions of Artificial Brine Samples
TABLE 2 Determination of Precision and Bias of Strontium Ion
g/L
Sample No.
1 234 Statistically
Amount Amount Significant
Sr 0.060 0.100 1.600 2.100
Added, Found, S S ± Bias (95 %
O T
NaCl 24.0 170.0 80.0 200.0
mg/L mg/L Confidence
KCl 0.5 2.0 1.5 3.0
Level)
KBr 1.0 2.0 2.0 2.0
KI 0.1 0.5 0.5 1.0 60 63.48 2.96 8.49 + 5.8 yes
CaCl 1.5 3.0 2.0 5.0 100 99.5 4.12 11.84 −0.5 no
MgCl 4.5 5.0 2.0 1.0 1600 1665.6 54.87 157.3 + 4.1 no
BaCl 0.05 1.0 0.5 0.5 2100 2167.2 71.12 203.9 + 3.2 no
D3352−15
FIG. 1 Relationship of the Concentration of Strontium in Some Oilfield (Smackover) Brines to Specific Gravity
Therefore, the user of this test method may find it necessary to directconcentrationofthesample,ifthiscapabilityisprovided
draw a simi
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D3352 − 08a D3352 − 15
Standard Test Method for
Strontium Ion in Brackish Water, Seawater, and Brines
This standard is issued under the fixed designation D3352; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Scope*
1.1 This test method covers the determination of soluble strontium ion in brackish water, seawater, and brines by atomic
absorption spectrophotometry.
1.2 Samples containing from 5 to 2100 mg/L of strontium may be analyzed by this test method.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this The values
given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not
considered standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
2. Referenced Documents
2.1 ASTM Standards:
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D3370 Practices for Sampling Water from Closed Conduits
D5810 Guide for Spiking into Aqueous Samples
D5847 Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
3. Terminology
3.1 Definitions—For definitions of terms used in this test method, refer to Terminology D1129.
4. Summary of Test Method
4.1 This test method is dependent on the fact that metallic elements, in the ground state, will absorb light of the same wavelength
they emit when excited. When radiation from a given excited element is passed through a flame containing ground state atoms of
that element, the intensity of the transmitted radiation will decrease in proportion to the amount of the ground state element in the
flame. A hollow cathode lamp whose cathode is made of the element to be determined provides the radiation. The metal atoms
to be measured are placed in the beam of radiation by aspirating the specimen into an oxidant-fuel flame. A monochromator isolates
the characteristic radiation from the hollow cathode lamp and a photosensitive device measures the attenuated transmitted
radiation.
4.2 Since the variable and sometimes high concentrations of matrix materials in the waters and brines affect absorption
differently, it is difficult to prepare standards sufficiently similar to the waters and brines. To overcome this difficulty, the method
This test method is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents in Water.
Current edition approved Nov. 15, 2008Feb. 1, 2015. Published November 2008April 2015. Originally approved in 1974. Last previous edition approved in 2008 as
D3352 – 08.D3352 – 08a. DOI: 10.1520/D3352-08A.10.1520/D3352-15.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
For additional information on atomic absorption, see the following references: Angino, E. E., and Billings, G. K., Atomic Absorption Spectrophotometry in Geology,
Elsevier Publishing Co., New York, N.Y., 1967. Dean, J. A., and Rains, T. C., Editors, Flame Emission and Atomic Absorption Spectrometry Vol 1 − Theory, Marcel Dekker,
New York, NY, 1969.For additional information on atomic absorption, see the following references: Angino, E. E., and Billings, G. K., Atomic Absorption Spectrophotometry
in Geology, Elsevier Publishing Co., New York, N.Y., 1967; Dean, J. A., and Rains, T. C., Editors, Flame Emission and Atomic Absorption Spectrometry Vol 1 − Theory,
Marcel Dekker, New York, NY, 1969.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D3352 − 15
of additions is used in which three identical samples are prepared and varying amounts of a standard added to two of them. The
three samples are then aspirated, the concentration readings recorded, and the original sample concentration calculated.
5. Significance and Use
5.1 This test method can be used to determine strontium ions in brackish water, seawater, and brines.
6. Interferences
6.1 The chemical suppression caused by silicon, aluminum, and phosphate is controlled by adding lanthanum. The lanthanum
also controls ionization interference.
7. Apparatus
7.1 Atomic Absorption Spectrophotometer —Spectrophotometer—The instrument shall consist of atomizer and burner, suitable
pressure-regulating devices capable of maintaining constant oxidant and fuel pressure for the duration of the test, a hollow cathode
lamp for each metal to be tested, an optical system capable of isolating the desired line of radiation, an adjustable slit, a
photomultiplier tube or other photosensitive device as a light measuring and amplifying device, and a read-out mechanism for
indicating the amount of absorbed radiation.
7.1.1 Multi-Element Hollow Cathode Lamps are available and have been found satisfactory.
7.2 Pressure-Reducing Valves—The supplies of fuel and oxidant shall be maintained at pressures somewhat higher than the
controlled operating pressure of the instrument by suitable valves.
8. Reagents and Materials
8.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where
such specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high
purity to permit its use without lessening the accuracy of the determination.
8.2 Purity of Water—Unless otherwise indicated, reference to water shall be understood to mean reagent water conforming to
Specification D1193, Type I. Other reagent water types may be used provided it is first ascertained that the water is of sufficiently
high purity to permit its use without adversely affecting the precision and bias of the test method. Type III water was specified at
the time of round robin testing of this test method.
8.3 Filter Paper—Purchase suitable filter paper. Typically the filter papers have a pore size of 0.45-μm membrane. Material such
as fine-textured, acid-washed, ashless paper, or glass fiber paper are acceptable. The user must first ascertain that the filter paper
is of sufficient purity to use without adversely affecting the bias and precision of the test method.
8.4 Lanthanum Solution (5 % La)—Wet 58.65 g of lanthanum oxide (La O ) with water. Add 250 mL of concentrated
2 3
hydrochloric acid (sp gr 1.19) very slowly until the material is dissolved. Dilute solution to 1 litre with water.
Additional information is contained in the following references: Fletcher, G. F., and Collins, A. G., “Atomic Absorption Methods of Analysis of Oilfield Brines: Barium,
Calcium, Copper, Iron, Lead, Lithium, Magnesium, Manganese, Potassium, Sodium, Strontium, and Zinc,” U.S. Bureau of Mines, Report of Investigations 7861, 1974, 14
pp. Collins, A. G., Geochemistry of Oilfield Waters, Elsevier Publishing Co., Amsterdam. The Netherlands, 1975.Additional information is contained in the following
references: Fletcher, G. F., and Collins, A. G., “Atomic Absorption Methods of Analysis of Oilfield Brines: Barium, Calcium, Copper, Iron, Lead, Lithium, Magnesium,
Manganese, Potassium, Sodium, Strontium, and Zinc,” U.S. Bureau of Mines, Report of Investigations 7861, 1974, 14 pp.; Collins, A. G., Geochemistry of Oilfield Waters,
Elsevier Publishing Co., Amsterdam, Netherlands, 1975.
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For Suggestions on the testing of reagents not listed by
the American Chemical Society, see Annual Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
TABLE 1 Compositions of Artificial Brine Samples
g/L
Sample No.
1 2 3 4
Sr 0.060 0.100 1.600 2.100
NaCl 24.0 170.0 80.0 200.0
KCl 0.5 2.0 1.5 3.0
KBr 1.0 2.0 2.0 2.0
KI 0.1 0.5 0.5 1.0
CaCl 1.5 3.0 2.0 5.0
MgCl 4.5 5.0 2.0 1.0
BaCl 0.05 1.0 0.5 0.5
D3352 − 15
8.5 Strontium Solution, Standard (1 mL = 1 mg Sr)—Dissolve 2.415 g of strontium nitrate [Sr(NO ) ] in 10 mL of concentrated
3 2
hydrochloric acid (sp gr 1.19) and about 700 mL of water. Dilute solution to 1 L with water. One millilitre of this solution contains
1 mg of strontium. Alternatively, certified strontium stock solutions are commercially available through chemical supply vendors
and may be used.A purchased strontium stock solution of appropriate known purity is also acceptable.
8.6 Oxidant, for Atomic Absorption Spectrophotometer:
8.6.1 Air, which has been cleaned and dried through a suitable filter to remove oil, water, and other foreign substances, is the
usual oxidant.
8.6.2 Nitrous Oxide may be required as an oxidant for refractory-type metals.
8.7 Fuel, for Atomic Absorption Spectrophotometer:
8.7.1 Acetylene—Standard, commercially available acetylene is the usual fuel. Acetone, always present in acetylene cylinders,
can be prevented from entering and damaging the burner head by replacing a cylinder which only has 100 psig 689 kPa (100 psi)
of acetylene remaining.
9. Sampling
9.1 Collect the sample in accordance with Practices D3370.
10. Procedure
10.1 Strontium is determined at the 460.7-nm wavelength with an air-acetylene flame.
10.2 Preliminary Calibration—Using micropipets prepare standard strontium solutions containing 1 to 10 mg/L of strontium
using the standard strontium solution (8.5) and 50-mL volumetric flasks. Before making up to volume, add to each of these and
to a blank, 5 mL of the lanthanum solution. solution (8.4). Analyze at least three working standards containing concentrations of
strontium that bracket the expected sample concentration, prior to analysis of samples, to calibrate the instrument. Aspirate these
standards and the blank (for background setting) and adjust the curvature controls, if necessary, to obtain a linear relationship
between absorbance and the actual concentration of the standards.
10.3 Transfer an aliquot of water or brine (previously filtered (8.3) through a 0.45-μm filter) to a 50-mL volumetric flask. The
specific gravity of the water or brine can be used to estimate the strontium content of the sample and, thereby, serve as a basis for
selecting the aliquot size that will contain about 0.1 mg of strontium. Fig. 1 shows the relationship between strontium concentration
and specific gravity for some oilfield brines from the Smackover formation. The concentrations of strontium in the Smackover
brines will not necessarily correlate with the concentrations found in other formations. Therefore, the user of this test method may
find it necessary to draw a similar curve for brine samples taken from other formations. Add 5 mL of the lanthanum stock solution,
sol
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...