ASTM E2709-23
(Practice)Standard Practice for Demonstrating Capability to Comply with an Acceptance Procedure
Standard Practice for Demonstrating Capability to Comply with an Acceptance Procedure
ABSTRACT
This practice provides a general methodology for evaluating single-stage or multiple-stage acceptance procedures which involve a quality characteristic measured on a numerical scale. This methodology computes, at a prescribed confidence level, a lower bound on the probability of passing an acceptance procedure, using estimates of the parameters of the distribution of test results from a sampled population.
SIGNIFICANCE AND USE
4.1 This practice considers inspection procedures that may involve multiple-stage sampling, where at each stage one can decide to accept or to continue sampling, and the decision to reject is deferred until the last stage.
4.1.1 At each stage there are one or more acceptance criteria on the test results; for example, limits on each individual test result, or limits on statistics based on the sample of test results, such as the average, standard deviation, or coefficient of variation (relative standard deviation).
4.2 The methodology in this practice defines an acceptance region for a set of test results from the sampled population such that, at a prescribed confidence level, the probability that a sample from the population will pass the acceptance procedure is greater than or equal to a prespecified lower bound.
4.2.1 Having test results fall in the acceptance region is not equivalent to passing the acceptance procedure, but provides assurance that a sample would pass the acceptance procedure with a specified probability.
4.2.2 This information can be used for process demonstration, validation of test methods, and qualification of instruments, processes, and materials.
4.2.3 This information can be used for lot release (acceptance), but the lower bound may be conservative in some cases.
4.2.4 If the results are to be applied to future test results from the same process, then it is assumed that the process is stable and predictable. If this is not the case then there can be no guarantee that the probability estimates would be valid predictions of future process performance.
4.3 This methodology was originally developed (1-4)3 for use in two specific quality characteristics of drug products in the pharmaceutical industry but will be applicable for acceptance procedures in all industries.
4.4 Mathematical derivations would be required that are specific to the individual criteria of each test.
SCOPE
1.1 This practice provides a general methodology for evaluating single-stage or multiple-stage acceptance procedures which involve a quality characteristic measured on a numerical scale. This methodology computes, at a prescribed confidence level, a lower bound on the probability of passing an acceptance procedure, using estimates of the parameters of the distribution of test results from a sampled population.
1.2 For a prescribed lower probability bound, the methodology can also generate an acceptance limit table, which defines a set of test method outcomes (for example, sample averages and standard deviations) that would pass the acceptance procedure at a prescribed confidence level.
1.3 This approach may be used for demonstrating compliance with in-process, validation, or lot-release specifications.
1.4 The system of units for this practice is not specified.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Oct-2023
- Technical Committee
- E11 - Quality and Statistics
- Drafting Committee
- E11.20 - Test Method Evaluation and Quality Control
Relations
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Apr-2023
- Effective Date
- 01-Apr-2022
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
Overview
ASTM E2709-23: Standard Practice for Demonstrating Capability to Comply with an Acceptance Procedure is a widely recognized methodology developed by ASTM International for evaluating single-stage or multiple-stage acceptance procedures. This standard is essential for industries that need to verify a quality characteristic measured on a numerical scale. By applying statistical techniques at a prescribed confidence level, ASTM E2709-23 provides a systematic method for calculating a lower bound on the probability that a process, product, or lot will pass an acceptance procedure based on sampled test results.
This standard offers industries a robust framework for process validation, method verification, and lot release, helping to ensure consistent quality and regulatory compliance.
Key Topics
- Acceptance Procedures: Outlines evaluation of both single-stage and multiple-stage acceptance procedures, addressing various decision points and acceptance criteria at each stage.
- Quality Characteristics: Focuses on characteristics measured on a numerical scale, emphasizing parameters such as mean, standard deviation, and coefficient of variation.
- Statistical Confidence: Establishes a methodology to compute lower bounds for the probability of passing acceptance based on a specified confidence level, promoting reliable decision-making.
- Acceptance Region & Limits: Defines the concept of an "acceptance region," where test results indicate a process is capable, and "acceptance limits," which are statistically derived boundaries for decision-making.
- Sampling Methods: Supports both simple random and two-stage sampling plans, making the standard versatile across a broad range of industries and applications.
- Applicability Across Industries: While originally utilized in pharmaceutical quality control, the approach is generalizable to other sectors needing statistical process validation.
Applications
Organizations can leverage ASTM E2709-23 to:
- Process Demonstration: Provide statistical assurance that a manufacturing process or test method consistently produces results meeting predefined specifications.
- Validation and Qualification: Support validation of analytical methods, instrumentation, and manufacturing processes by demonstrating their capability to meet acceptance criteria.
- Lot Release Decisions: Use acceptance limit tables generated under this standard to make informed and statistically sound lot release decisions.
- Regulatory Compliance: Establish objective, quantifiable evidence of process capability required by regulators in pharmaceuticals, biotechnology, medical devices, and other sectors.
- Statistical Sampling Plans: Apply the standard's methodology in planning and confirmation phases to minimize risk and enhance product quality assurance.
Common users of ASTM E2709-23 include quality assurance professionals, statisticians, manufacturing engineers, laboratory personnel, and regulatory affairs teams across diverse industries.
Related Standards
ASTM E2709-23 aligns with and references several key ASTM standards for quality and statistics:
- ASTM E456: Terminology Relating to Quality and Statistics
- ASTM E2282: Guide for Defining the Test Result of a Test Method
- ASTM E2586: Practice for Calculating and Using Basic Statistics
These companion standards further support statistical quality control, method validation, and the implementation of robust testing protocols.
By applying ASTM E2709-23, organizations demonstrate commitment to statistically sound quality management practices, improve process capability, and maintain compliance with international standards and regulatory expectations. For more information, consult ASTM's official documentation and related statistical standards.
Buy Documents
ASTM E2709-23 - Standard Practice for Demonstrating Capability to Comply with an Acceptance Procedure
REDLINE ASTM E2709-23 - Standard Practice for Demonstrating Capability to Comply with an Acceptance Procedure
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

Bureau Veritas
Bureau Veritas is a world leader in laboratory testing, inspection and certification services.

DNV
DNV is an independent assurance and risk management provider.
Sponsored listings
Frequently Asked Questions
ASTM E2709-23 is a standard published by ASTM International. Its full title is "Standard Practice for Demonstrating Capability to Comply with an Acceptance Procedure". This standard covers: ABSTRACT This practice provides a general methodology for evaluating single-stage or multiple-stage acceptance procedures which involve a quality characteristic measured on a numerical scale. This methodology computes, at a prescribed confidence level, a lower bound on the probability of passing an acceptance procedure, using estimates of the parameters of the distribution of test results from a sampled population. SIGNIFICANCE AND USE 4.1 This practice considers inspection procedures that may involve multiple-stage sampling, where at each stage one can decide to accept or to continue sampling, and the decision to reject is deferred until the last stage. 4.1.1 At each stage there are one or more acceptance criteria on the test results; for example, limits on each individual test result, or limits on statistics based on the sample of test results, such as the average, standard deviation, or coefficient of variation (relative standard deviation). 4.2 The methodology in this practice defines an acceptance region for a set of test results from the sampled population such that, at a prescribed confidence level, the probability that a sample from the population will pass the acceptance procedure is greater than or equal to a prespecified lower bound. 4.2.1 Having test results fall in the acceptance region is not equivalent to passing the acceptance procedure, but provides assurance that a sample would pass the acceptance procedure with a specified probability. 4.2.2 This information can be used for process demonstration, validation of test methods, and qualification of instruments, processes, and materials. 4.2.3 This information can be used for lot release (acceptance), but the lower bound may be conservative in some cases. 4.2.4 If the results are to be applied to future test results from the same process, then it is assumed that the process is stable and predictable. If this is not the case then there can be no guarantee that the probability estimates would be valid predictions of future process performance. 4.3 This methodology was originally developed (1-4)3 for use in two specific quality characteristics of drug products in the pharmaceutical industry but will be applicable for acceptance procedures in all industries. 4.4 Mathematical derivations would be required that are specific to the individual criteria of each test. SCOPE 1.1 This practice provides a general methodology for evaluating single-stage or multiple-stage acceptance procedures which involve a quality characteristic measured on a numerical scale. This methodology computes, at a prescribed confidence level, a lower bound on the probability of passing an acceptance procedure, using estimates of the parameters of the distribution of test results from a sampled population. 1.2 For a prescribed lower probability bound, the methodology can also generate an acceptance limit table, which defines a set of test method outcomes (for example, sample averages and standard deviations) that would pass the acceptance procedure at a prescribed confidence level. 1.3 This approach may be used for demonstrating compliance with in-process, validation, or lot-release specifications. 1.4 The system of units for this practice is not specified. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ABSTRACT This practice provides a general methodology for evaluating single-stage or multiple-stage acceptance procedures which involve a quality characteristic measured on a numerical scale. This methodology computes, at a prescribed confidence level, a lower bound on the probability of passing an acceptance procedure, using estimates of the parameters of the distribution of test results from a sampled population. SIGNIFICANCE AND USE 4.1 This practice considers inspection procedures that may involve multiple-stage sampling, where at each stage one can decide to accept or to continue sampling, and the decision to reject is deferred until the last stage. 4.1.1 At each stage there are one or more acceptance criteria on the test results; for example, limits on each individual test result, or limits on statistics based on the sample of test results, such as the average, standard deviation, or coefficient of variation (relative standard deviation). 4.2 The methodology in this practice defines an acceptance region for a set of test results from the sampled population such that, at a prescribed confidence level, the probability that a sample from the population will pass the acceptance procedure is greater than or equal to a prespecified lower bound. 4.2.1 Having test results fall in the acceptance region is not equivalent to passing the acceptance procedure, but provides assurance that a sample would pass the acceptance procedure with a specified probability. 4.2.2 This information can be used for process demonstration, validation of test methods, and qualification of instruments, processes, and materials. 4.2.3 This information can be used for lot release (acceptance), but the lower bound may be conservative in some cases. 4.2.4 If the results are to be applied to future test results from the same process, then it is assumed that the process is stable and predictable. If this is not the case then there can be no guarantee that the probability estimates would be valid predictions of future process performance. 4.3 This methodology was originally developed (1-4)3 for use in two specific quality characteristics of drug products in the pharmaceutical industry but will be applicable for acceptance procedures in all industries. 4.4 Mathematical derivations would be required that are specific to the individual criteria of each test. SCOPE 1.1 This practice provides a general methodology for evaluating single-stage or multiple-stage acceptance procedures which involve a quality characteristic measured on a numerical scale. This methodology computes, at a prescribed confidence level, a lower bound on the probability of passing an acceptance procedure, using estimates of the parameters of the distribution of test results from a sampled population. 1.2 For a prescribed lower probability bound, the methodology can also generate an acceptance limit table, which defines a set of test method outcomes (for example, sample averages and standard deviations) that would pass the acceptance procedure at a prescribed confidence level. 1.3 This approach may be used for demonstrating compliance with in-process, validation, or lot-release specifications. 1.4 The system of units for this practice is not specified. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2709-23 is classified under the following ICS (International Classification for Standards) categories: 03.120.30 - Application of statistical methods. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2709-23 has the following relationships with other standards: It is inter standard links to ASTM E2709-19(2023), ASTM E2282-23, ASTM E2282-14(2023), ASTM E456-13a(2022)e1, ASTM E2810-19, ASTM E456-13a(2022). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2709-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2709 − 23 An American National Standard
Standard Practice for
Demonstrating Capability to Comply with an Acceptance
Procedure
This standard is issued under the fixed designation E2709; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope E2282 Guide for Defining the Test Result of a Test Method
1.1 This practice provides a general methodology for evalu-
3. Terminology
ating single-stage or multiple-stage acceptance procedures
3.1 Definitions—Unless otherwise noted in this standard, all
which involve a quality characteristic measured on a numerical
terms relating to quality and statistics are defined in Terminol-
scale. This methodology computes, at a prescribed confidence
ogy E456.
level, a lower bound on the probability of passing an accep-
3.1.1 characteristic, n—a property of items in a sample or
tance procedure, using estimates of the parameters of the
population which, when measured, counted or otherwise
distribution of test results from a sampled population.
observed, helps to distinguish between the items. E2282
1.2 For a prescribed lower probability bound, the method-
3.1.2 multiple-stage acceptance procedure, n—a procedure
ology can also generate an acceptance limit table, which
that involves more than one stage of sampling and testing a
defines a set of test method outcomes (for example, sample
given quality characteristic and one or more acceptance criteria
averages and standard deviations) that would pass the accep-
per stage.
tance procedure at a prescribed confidence level.
3.1.3 test method, n—a definitive procedure that produces a
1.3 This approach may be used for demonstrating compli-
test result. E2282
ance with in-process, validation, or lot-release specifications.
3.2 Definitions of Terms Specific to This Standard:
1.4 The system of units for this practice is not specified.
3.2.1 acceptable parameter region, n—the set of values of
1.5 This standard does not purport to address all of the
parameters characterizing the distribution of test results for
safety concerns, if any, associated with its use. It is the
which the probability of passing the acceptance procedure is
responsibility of the user of this standard to establish appro-
greater than a prescribed lower bound.
priate safety, health, and environmental practices and deter-
3.2.2 acceptance region, n—the set of values of parameter
mine the applicability of regulatory limitations prior to use.
estimates that will attain a prescribed lower bound on the
1.6 This international standard was developed in accor-
probability of passing an acceptance procedure at a prescribed
dance with internationally recognized principles on standard-
level of confidence.
ization established in the Decision on Principles for the
3.2.3 acceptance limit, n—the boundary of the acceptance
Development of International Standards, Guides and Recom-
region, for example, the maximum sample standard deviation
mendations issued by the World Trade Organization Technical
test results for a given sample mean.
Barriers to Trade (TBT) Committee.
4. Significance and Use
2. Referenced Documents
4.1 This practice considers inspection procedures that may
2.1 ASTM Standards:
involve multiple-stage sampling, where at each stage one can
E456 Terminology Relating to Quality and Statistics
decide to accept or to continue sampling, and the decision to
reject is deferred until the last stage.
4.1.1 At each stage there are one or more acceptance criteria
This practice is under the jurisdiction of ASTM Committee E11 on Quality and
on the test results; for example, limits on each individual test
Statistics and is the direct responsibility of Subcommittee E11.20 on Test Method
Evaluation and Quality Control.
result, or limits on statistics based on the sample of test results,
Current edition approved Nov. 1, 2023. Published November 2023. Originally
such as the average, standard deviation, or coefficient of
approved in 2009. Last previous edition approved in 2023 as E2709 – 19 (2023).
variation (relative standard deviation).
DOI: 10.1520/E2709-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
4.2 The methodology in this practice defines an acceptance
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
region for a set of test results from the sampled population such
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. that, at a prescribed confidence level, the probability that a
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2709 − 23
sample from the population will pass the acceptance procedure 5.3 Determine the contour of the region of parameter values
is greater than or equal to a prespecified lower bound. for which the expression for the probability of passing the
given acceptance procedure is at least equal to the required
4.2.1 Having test results fall in the acceptance region is not
equivalent to passing the acceptance procedure, but provides lower bound (LB) on the probability of acceptance (p). This
defines the acceptable parameter region. Since the acceptance
assurance that a sample would pass the acceptance procedure
with a specified probability. parameter region is a lower bound, it should be compared to
the simulated probability of passing the acceptance procedure.
4.2.2 This information can be used for process
demonstration, validation of test methods, and qualification of
5.4 For each value of a statistic or set of statistics, derive a
instruments, processes, and materials.
joint confidence region for the distribution parameters at
4.2.3 This information can be used for lot release
confidence level, expressed as a percentage, of 100(1-α). The
(acceptance), but the lower bound may be conservative in some
size of sample to be taken, n, and the statistics to be used, must
cases.
be predetermined (see 5.6).
4.2.4 If the results are to be applied to future test results
5.5 Determine the contour of the acceptance region, which
from the same process, then it is assumed that the process is
consists of values of the statistics for which the confidence
stable and predictable. If this is not the case then there can be
region at level 100(1-α) is entirely contained in the acceptable
no guarantee that the probability estimates would be valid
parameter region. The acceptance limits lie on the contour of
predictions of future process performance.
the acceptance region.
4.3 This methodology was originally developed (1-4) for
5.6 To select the size of sample, n, to be taken, the
use in two specific quality characteristics of drug products in
probability that sample statistics will lie within acceptance
the pharmaceutical industry but will be applicable for accep-
limits should be evaluated over a range of values of n, for
tance procedures in all industries.
values of population parameters of practical interest, and for
4.4 Mathematical derivations would be required that are
which probabilities of passing the given acceptance procedure
specific to the individual criteria of each test.
are well above the lower bound. The larger the sample size n
that is chosen, the larger will be the acceptance region and the
5. Methodology
tighter the distribution of the statistics. Choose n so that the
probability of passing acceptance limits is greater than a
5.1 The process for defining the acceptance limits, starting
predetermined value.
from the definition of the acceptance procedure, is outlined in
this section. A computer program is normally required to
5.7 To use the acceptance limit, sample randomly from the
produce the acceptable parameter region and the acceptance
population. Compute statistics for the sample. If statistics fall
limits.
within the acceptance limits, then there is 1-α confidence that
5.1.1 An expression for the exact probability of passing the
the probability of acceptance is at least p.
acceptance procedure might be intractable when the procedure
6. Procedures for Sampling from a Normal Distribution
consists of multiple stages with multiple criteria, hence a lower
bound for the probability may be used.
6.1 An important class of procedures is for the case where
the quality characteristic is normally distributed. Particular
5.2 Express the probability of passing the acceptance pro-
instructions for that case are given in this section, for two
cedure as a function of the parameters characterizing the
sampling methods, simple random and two-stage. In this
distribution of the quality characteristic for items in the
standard, these sampling methods are denoted Sampling Plan 1
sampled population.
and Sampling Plan 2, respectively.
5.2.1 For each stage in the procedure having multiple
acceptance criteria, determine the lower bound on the prob-
6.2 When the characteristic is normally distributed, param-
ability of that stage as a function of the probabilities of passing eters are the mean (μ) and standard deviation (σ) of the
each of the criteria in the stage: population. The acceptable parameter region will be the region
m under a curve in the half-plane where μ is on the horizontal
P S 5 P C and C … and C $ 1 2 1 2 P C (1)
~ ! ~ ! ~ ~ !!
i i1 i2 im (j51 ij
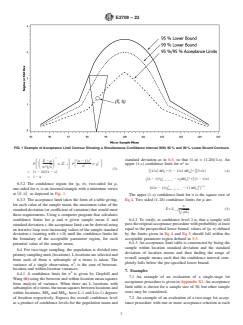
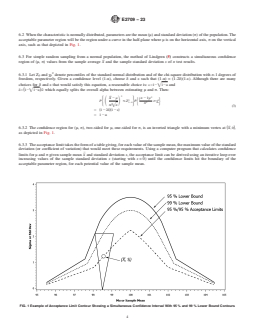
axis, σ on the vertical axis, such as that depicted in Fig. 1.
where:
6.3 For simple random sampling from a normal population,
P(S ) = is the probability of passing stage i,
the method of Lindgren (5) constructs a simultaneous confi-
i
P(C ) = is the probability of passing the j-th criterion of m
¯
ij
dence region of (μ, σ) values from the sample average X and
within the i-th stage.
the sample standard deviation s of n test results.
5.2.2 Determine the lower bound on the probability of
6.3.1 Let Z and χ denote percentiles of the standard
p p
passing a k-stage procedure as a function of probabilities of
normal distribution and of the chi-square distribution with n-1
passing each of the individual stages:
degrees of freedom, respectively. Given a confidence level
(1-α), choose δ and ε such that (1-α) = (1-2δ)(1-ε). Although
P pass k 2 stage procedure $ max P S , P S , … , P S (2)
~ ! $ ~ ! ~ ! ~ !%
1 2 k
there are many choices for δ and ε that would satisfy this
=
equation, a reasonable choice is: ε512 12α and
3 ~ = !
δ5 12 12α /2 which equally splits the overall alpha be-
The boldface numbers in parentheses refer to a list of references at the end of
this standard. tween estimating μ and σ. Then:
E2709 − 23
FIG. 1 Example of Acceptance Limit Contour Showing a Simultaneous Confidence Interval With 95 % and 99 % Lower Bound Contours
¯ standard deviation as in 6.3, so that (1-α) = (1-2δ)(1-ε). An
X 2 μ ~n 2 1!s
2 2
P # Z P $ χ
H J
12δ 2 ε
HS D J upper (1-ε) confidence limit for σ is:
σ
σ/=n
(3)
@~1/n! MS 1~1 2 1/n! MS #1$@~1/n! (4)
L E
5 ~1 2 2δ!~1 2 ε!
5 1 2 α
2 2
L 2 1 /χ 2 1 MS # 1 1 2 1/n
~ ! @~ !
~ !
L21, 12ϵ L
6.3.2 The confidence region for (μ, σ), two-sided for μ,
2 2 1/2
L n 2 1 /χ 2 1 MS # %
~ !
~ L n21 , 12ϵ ! E
one-sided for σ, is an inverted triangle with a minimum vertex ~ !
¯
at ~X, 0!, as depicted in Fig. 1.
The upper (1-ε) confidence limit for σ is the square root of
Eq 4. Two sided (1-2δ) confidence limits for μ are:
6.3.3 The acceptance limit takes the form of a table giving,
for each value of the sample mean, the maximum value of the
σ
¯
X6Z (5)
standard deviation (or coefficient of variation) that would meet 12δ
=~nL!
these requirements. Using a computer program that calculates
¯
6.4.2 To verify, at confidence level 1-α, that a sample will
confidence limits for μ and σ given sample mean X and
pass the original acceptance procedure with probability at least
standard deviation s, the acceptance limit can be derived using
equal to the prespecified lower bound, values of (μ, σ) defined
an iterative loop over increasing values of the sample standard
by the limits given in Eq 4 and Eq 5 should fall within the
deviation s (starting with s = 0) until the confidence limits hit
acceptable parameter region defined in 5.3.
the boundary of the acceptable parameter region, for each
6.4.3 An acceptance limit table is constructed by fixing the
potential value of the sample mean.
sample within location standard deviation and the standard
6.4 For two-stage sampling, the population is divided into
deviation of location means and then finding the range of
primary sampling units (locations). L locations are selected and
overall sample means such that the confidence interval com-
from each of them a subsample of n items is taken. The
pletely falls below the pre-specified lower bound.
variance of a single observation, σ , is the sum of between-
location and within-location variances.
7. Examples
6.4.1 A confidence limit for σ is given by Graybill and
7.1 An example of an evaluation of a single-stage lot
Wang (6) using the between and within location mean squares
acceptance procedure is given in Appendix X1. An acceptance
from analysis of variance. When there are L locations with
limit table is shown for a sample size of 30, but other sample
subsampl
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E2709 − 19 (Reapproved 2023) E2709 − 23 An American National Standard
Standard Practice for
Demonstrating Capability to Comply with an Acceptance
Procedure
This standard is issued under the fixed designation E2709; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This practice provides a general methodology for evaluating single-stage or multiple-stage acceptance procedures which
involve a quality characteristic measured on a numerical scale. This methodology computes, at a prescribed confidence level, a
lower bound on the probability of passing an acceptance procedure, using estimates of the parameters of the distribution of test
results from a sampled population.
1.2 For a prescribed lower probability bound, the methodology can also generate an acceptance limit table, which defines a set
of test method outcomes (for example, sample averages and standard deviations) that would pass the acceptance procedure at a
prescribed confidence level.
1.3 This approach may be used for demonstrating compliance with in-process, validation, or lot-release specifications.
1.4 The system of units for this practice is not specified.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E456 Terminology Relating to Quality and Statistics
E2282 Guide for Defining the Test Result of a Test Method
E2586 Practice for Calculating and Using Basic Statistics
3. Terminology
3.1 Definitions—See Terminology—Unless otherwise E456 for a more extensive listing of terms in ASTM Committee E11
standards.noted in this standard, all terms relating to quality and statistics are defined in Terminology E456.
This practice is under the jurisdiction of ASTM Committee E11 on Quality and Statistics and is the direct responsibility of Subcommittee E11.20 on Test Method
Evaluation and Quality Control.
Current edition approved April 1, 2023Nov. 1, 2023. Published April 2023November 2023. Originally approved in 2009. Last previous edition approved in 20192023 as
E2709 – 19.E2709 – 19 (2023). DOI: 10.1520/E2709-19R23.10.1520/E2709-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2709 − 23
3.1.1 characteristic, n—a property of items in a sample or population which, when measured, counted or otherwise observed,
helps to distinguish between the items. E2282
¯
3.1.2 mean, n—of a population, μ, average or expected value of a characteristic in a population, of a sampleX, sum of the observed
values in a sample divided by the sample size. E2586
3.1.2 multiple-stage acceptance procedure, n—a procedure that involves more than one stage of sampling and testing a given
quality characteristic and one or more acceptance criteria per stage.
3.1.4 standard deviation, n—of a population, σ, the square root of the average or expected value of the squared deviation of a
variable from its mean – of a sample, s, the square root of the sum of the squared deviations of the observed values in the sample
divided by the sample size minus 1. E2586
3.1.3 test method, n—a definitive procedure that produces a test result. E2282
3.2 Definitions of Terms Specific to This Standard:
3.2.1 acceptable parameter region, n—the set of values of parameters characterizing the distribution of test results for which the
probability of passing the acceptance procedure is greater than a prescribed lower bound.
3.2.2 acceptance region, n—the set of values of parameter estimates that will attain a prescribed lower bound on the probability
of passing an acceptance procedure at a prescribed level of confidence.
3.2.3 acceptance limit, n—the boundary of the acceptance region, for example, the maximum sample standard deviation test
results for a given sample mean.
4. Significance and Use
4.1 This practice considers inspection procedures that may involve multiple-stage sampling, where at each stage one can decide
to accept or to continue sampling, and the decision to reject is deferred until the last stage.
4.1.1 At each stage there are one or more acceptance criteria on the test results; for example, limits on each individual test result,
or limits on statistics based on the sample of test results, such as the average, standard deviation, or coefficient of variation (relative
standard deviation).
4.2 The methodology in this practice defines an acceptance region for a set of test results from the sampled population such that,
at a prescribed confidence level, the probability that a sample from the population will pass the acceptance procedure is greater
than or equal to a prespecified lower bound.
4.2.1 Having test results fall in the acceptance region is not equivalent to passing the acceptance procedure, but provides assurance
that a sample would pass the acceptance procedure with a specified probability.
4.2.2 This information can be used for process demonstration, validation of test methods, and qualification of instruments,
processes, and materials.
4.2.3 This information can be used for lot release (acceptance), but the lower bound may be conservative in some cases.
4.2.4 If the results are to be applied to future test results from the same process, then it is assumed that the process is stable and
predictable. If this is not the case then there can be no guarantee that the probability estimates would be valid predictions of future
process performance.
4.3 This methodology was originally developed (1-4) for use in two specific quality characteristics of drug products in the
pharmaceutical industry but will be applicable for acceptance procedures in all industries.
The boldface numbers in parentheses refer to a list of references at the end of this standard.
E2709 − 23
4.4 Mathematical derivations would be required that are specific to the individual criteria of each test.
5. Methodology
5.1 The process for defining the acceptance limits, starting from the definition of the acceptance procedure, is outlined in this
section. A computer program is normally required to produce the acceptable parameter region and the acceptance limits.
5.1.1 An expression for the exact probability of passing the acceptance procedure might be intractable when the procedure consists
of multiple stages with multiple criteria, hence a lower bound for the probability may be used.
5.2 Express the probability of passing the acceptance procedure as a function of the parameters characterizing the distribution of
the quality characteristic for items in the sampled population.
5.2.1 For each stage in the procedure having multiple acceptance criteria, determine the lower bound on the probability of that
stage as a function of the probabilities of passing each of the criteria in the stage:
m
P S 5 P C and C … and C $ 12 12 P C (1)
~ ! ~ ! ~ ~ !!
i i1 i2 im (j51 ij
where:
P(S ) = is the probability of passing stage i,
i
P(C ) = is the probability of passing the j-th criterion of m within the i-th stage.
ij
5.2.2 Determine the lower bound on the probability of passing a k-stage procedure as a function of probabilities of passing each
of the individual stages:
P ~pass k 2 stage procedure!$ max$P~S !, P~S !, … , P~S !% (2)
1 2 k
5.3 Determine the contour of the region of parameter values for which the expression for the probability of passing the given
acceptance procedure is at least equal to the required lower bound (LB) on the probability of acceptance (p). This defines the
acceptable parameter region. Since the acceptance parameter region is a lower bound, it should be compared to the simulated
probability of passing the acceptance procedure.
5.4 For each value of a statistic or set of statistics, derive a joint confidence region for the distribution parameters at confidence
level, expressed as a percentage, of 100(1-α). The size of sample to be taken, n, and the statistics to be used, must be predetermined
(see 5.6).
5.5 Determine the contour of the acceptance region, which consists of values of the statistics for which the confidence region at
level 100(1-α) is entirely contained in the acceptable parameter region. The acceptance limits lie on the contour of the acceptance
region.
5.6 To select the size of sample, n, to be taken, the probability that sample statistics will lie within acceptance limits should be
evaluated over a range of values of n, for values of population parameters of practical interest, and for which probabilities of
passing the given acceptance procedure are well above the lower bound. The larger the sample size n that is chosen, the larger will
be the acceptance region and the tighter the distribution of the statistics. Choose n so that the probability of passing acceptance
limits is greater than a predetermined value.
5.7 To use the acceptance limit, sample randomly from the population. Compute statistics for the sample. If statistics fall within
the acceptance limits, then there is 1-α confidence that the probability of acceptance is at least p.
6. Procedures for Sampling from a Normal Distribution
6.1 An important class of procedures is for the case where the quality characteristic is normally distributed. Particular instructions
for that case are given in this section, for two sampling methods, simple random and two-stage. In this standard, these sampling
methods are denoted Sampling Plan 1 and Sampling Plan 2, respectively.
E2709 − 23
6.2 When the characteristic is normally distributed, parameters are the mean (μ) and standard deviation (σ) of the population. The
acceptable parameter region will be the region under a curve in the half-plane where μ is on the horizontal axis, σ on the vertical
axis, such as that depicted in Fig. 1.
6.3 For simple random sampling from a normal population, the method of Lindgren (5) constructs a simultaneous confidence
¯
region of (μ, σ) values from the sample average X and the sample standard deviation s of n test results.
6.3.1 Let Z and χ denote percentiles of the standard normal distribution and of the chi-square distribution with n-1 degrees of
p p
freedom, respectively. Given a confidence level (1-α), choose δ and ε such that (1-α) = (1-2δ)(1-ε). Although there are many
choices for δ and ε that would satisfy this equation, a reasonable choice is: ε512=12α and
δ5~12=12α!/2 which equally splits the overall alpha between estimating μ and σ. Then:
¯ 2
X 2 μ n 2 1 s
~ !
2 2
P # Z P $χ
H J
HS D 12δJ 2 ε
σ
σ/=n
(3)
5 12 2δ 12ε
~ !~ !
5 12α
¯
6.3.2 The confidence region for (μ, σ), two-sided for μ, one-sided for σ, is an inverted triangle with a minimum vertex at ~X, 0!,
as depicted in Fig. 1.
6.3.3 The acceptance limit takes the form of a table giving, for each value of the sample mean, the maximum value of the standard
deviation (or coefficient of variation) that would meet these requirements. Using a computer program that calculates confidence
¯
limits for μ and σ given sample mean X and standard deviation s, the acceptance limit can be derived using an iterative loop over
increasing values of the sample standard deviation s (starting with s = 0) until the confidence limits hit the boundary of the
acceptable parameter region, for each potential value of the sample mean.
FIG. 1 Example of Acceptance Limit Contour Showing a Simultaneous Confidence Interval With 95 % and 99 % Lower Bound Contours
E2709 − 23
6.4 For two-stage sampling, the population is divided into primary sampling units (locations). L locations are selected and from
each of them a subsample of n items is taken. The variance of a single observation, σ , is the sum of between-location and
within-location variances.
6.4.1 A confidence limit for σ is given by Graybill and Wang (6) using the between and within location mean squares from
analysis of variance. When there are L locations with subsamples of n items, the mean squares between locations and within
locations, MS and MS , have L-1 and L(n-1) degrees of freedom respectively. Express the overall confidence level as a product
L E
of confidence levels for the population mean and standard deviation as in 6.3, so that (1-α) = (1-2δ)(1-ε). An upper (1-ε) confidence
limit for σ is:
@~1/n! MS 1~12 1/n! MS #1$@~1/n! (4)
L E
2 2
~L 2 1!/χ 2 1 MS # 1@~12 1/n!
~ !
L21, 12ϵ L
2 2 1/2
L n 2 1 /χ 2 1 MS # %
~ !
~ !
L~n21!, 12ϵ E
The upper (1-ε) confidence limit for σ is the square root of Eq 4. Two sided (1-2δ) confidence limits for μ are:
σ
¯
X6Z (5)
12δ
=
~nL!
6.4.2 To verify, at confidence level 1-α, that a sample will pass the original acceptance procedure with probability at leas
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...