ASTM F2267-24
(Test Method)Standard Test Method for Measuring Load-Induced Subsidence of Intervertebral Body Fusion Device Under Static Axial Compression
Standard Test Method for Measuring Load-Induced Subsidence of Intervertebral Body Fusion Device Under Static Axial Compression
SIGNIFICANCE AND USE
5.1 Intervertebral body fusion devices are generally simple geometric-shaped devices, which are often porous or hollow in nature. Their function is to support the anterior column of the spine to facilitate arthrodesis of the motion segment.
5.2 This test method is designed to quantify the subsidence characteristics of different designs of intervertebral body fusion devices since this is a potential clinical failure mode. These tests are conducted in vitro in order to simplify the comparison of simulated vertebral body subsidence induced by the intervertebral body fusion devices.
5.3 The static axial compressive loads that will be applied to the intervertebral body fusion devices and test blocks will differ from the complex loading seen in vivo, and therefore, the results from this test method may not be used to directly predict in vivo performance. The results, however, can be used to compare the varying degrees of subsidence between different intervertebral body fusion device designs for a given density of simulated bone.
5.4 The location within the simulated vertebral bodies and position of the intervertebral body fusion device with respect to the loading axis will be dependent upon the design and manufacturer's recommendation for implant placement.
SCOPE
1.1 This test method specifies the materials and methods for the axial compressive subsidence testing of non-biologic intervertebral body fusion devices, spinal implants designed to promote arthrodesis at a given spinal motion segment.
1.2 This test method is intended to provide a basis for the mechanical comparison among past, present, and future non-biologic intervertebral body fusion devices. This test method is intended to enable the user to mechanically compare intervertebral body fusion devices and does not purport to provide performance standards for intervertebral body fusion devices.
1.3 This test method describes a static test method by specifying a load type and a specific method of applying this load. This test method is designed to allow for the comparative evaluation of intervertebral body fusion devices.
1.4 Guidelines are established for measuring test block deformation and determining the subsidence of intervertebral body fusion devices.
1.5 Since some intervertebral body fusion devices require the use of additional implants for stabilization, the testing of these types of implants may not be in accordance with the manufacturer's recommended usage.
1.6 Units—The values stated in SI units are to be regarded as the standard with the exception of angular measurements, which may be reported in terms of either degrees or radians.
1.7 The use of this standard may involve the operation of potentially hazardous equipment. This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Apr-2024
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.25 - Spinal Devices
Relations
- Effective Date
- 15-Apr-2024
- Effective Date
- 15-Apr-2024
- Effective Date
- 15-Apr-2024
- Effective Date
- 15-Apr-2024
Overview
ASTM F2267-24: Standard Test Method for Measuring Load-Induced Subsidence of Intervertebral Body Fusion Device Under Static Axial Compression provides a reliable framework for evaluating the mechanical behavior of spinal implants-specifically, intervertebral body fusion devices-under static axial compressive loads. Developed by ASTM Committee F04 on Medical and Surgical Materials and Devices, this test method supports meaningful comparison of non-biologic intervertebral body fusion devices in a laboratory setting. The primary aim is to quantify the propensity of devices to subside into simulated vertebral bodies, facilitating spinal arthrodesis and supporting improved device design and selection.
Key Topics
- Intervertebral Body Fusion Devices: These implants, often porous or hollow in design, function to support the anterior column of the spine and facilitate spinal fusion.
- Subsidence Testing: The standard establishes procedures to measure subsidence-the phenomenon where an implant sinks into the vertebral body-under controlled, static loading.
- Mechanical Comparison: The method is intended for comparative evaluation, not for establishing minimum performance criteria, allowing assessment of device design trends and material impacts over time.
- Test Apparatus and Protocols: The method specifies the use of a test fixture incorporating polyurethane foam test blocks, mimicking vertebral body stiffness. It details test setup, implant positioning, and load application for reproducible results.
- Data Collection and Reporting: Guidelines are established for recording load-displacement curves, calculating stiffness, determining yield load, and reporting all relevant test parameters and outcomes.
Applications
- Product Development and Benchmarking: Medical device manufacturers use ASTM F2267-24 to evaluate and compare the mechanical integrity and subsidence behavior of new intervertebral body fusion device prototypes against existing products.
- Regulatory Submissions: This standard provides supporting data for regulatory filings by quantifying device performance, especially when seeking approvals or market clearances.
- Quality Assurance: Consistent mechanical comparison aids in maintaining product quality over time, ensuring ongoing conformance to baseline mechanical characteristics.
- Clinical Relevance: While in vitro results do not replicate the complexity of in vivo loading, the test method offers insight into relative device performance and helps identify designs with reduced risk of subsidence- a known clinical concern.
- R&D and Academic Research: Researchers and engineers leverage the method for comparative studies and biomechanical investigations related to spinal fusion device efficacy and design optimization.
Related Standards
ASTM F2267-24 references several related standards essential for accurate subsidence testing:
- ASTM E4: Practices for Force Calibration and Verification of Testing Machines
- ASTM F1582: Terminology Relating to Spinal Implants
- ASTM F1839: Specification for Rigid Polyurethane Foam for Use as a Standard Material in Orthopaedic Device Testing
- ASTM F2077: Test Methods for Intervertebral Body Fusion Devices
- ASTM E177 & ASTM E691: Statistical practices for precision and bias and for interlaboratory studies
Practical Value
ASTM F2267-24 is vital for stakeholders in the spinal implant sector, including engineers, quality managers, and regulatory professionals. By delivering a standardized, reproducible test method for measuring load-induced subsidence, the standard supports improvements in spinal implant safety, longevity, and clinical success. Its comparability fosters innovation, informs evidence-based device selection, and underpins regulatory confidence in preclinical device data.
Keywords: intervertebral body fusion device, spinal implants, subsidence test method, static axial compression, mechanical comparison, polyurethane foam, spinal arthrodesis, ASTM F2267-24.
Buy Documents
ASTM F2267-24 - Standard Test Method for Measuring Load-Induced Subsidence of Intervertebral Body Fusion Device Under Static Axial Compression
REDLINE ASTM F2267-24 - Standard Test Method for Measuring Load-Induced Subsidence of Intervertebral Body Fusion Device Under Static Axial Compression
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2267-24 is a standard published by ASTM International. Its full title is "Standard Test Method for Measuring Load-Induced Subsidence of Intervertebral Body Fusion Device Under Static Axial Compression". This standard covers: SIGNIFICANCE AND USE 5.1 Intervertebral body fusion devices are generally simple geometric-shaped devices, which are often porous or hollow in nature. Their function is to support the anterior column of the spine to facilitate arthrodesis of the motion segment. 5.2 This test method is designed to quantify the subsidence characteristics of different designs of intervertebral body fusion devices since this is a potential clinical failure mode. These tests are conducted in vitro in order to simplify the comparison of simulated vertebral body subsidence induced by the intervertebral body fusion devices. 5.3 The static axial compressive loads that will be applied to the intervertebral body fusion devices and test blocks will differ from the complex loading seen in vivo, and therefore, the results from this test method may not be used to directly predict in vivo performance. The results, however, can be used to compare the varying degrees of subsidence between different intervertebral body fusion device designs for a given density of simulated bone. 5.4 The location within the simulated vertebral bodies and position of the intervertebral body fusion device with respect to the loading axis will be dependent upon the design and manufacturer's recommendation for implant placement. SCOPE 1.1 This test method specifies the materials and methods for the axial compressive subsidence testing of non-biologic intervertebral body fusion devices, spinal implants designed to promote arthrodesis at a given spinal motion segment. 1.2 This test method is intended to provide a basis for the mechanical comparison among past, present, and future non-biologic intervertebral body fusion devices. This test method is intended to enable the user to mechanically compare intervertebral body fusion devices and does not purport to provide performance standards for intervertebral body fusion devices. 1.3 This test method describes a static test method by specifying a load type and a specific method of applying this load. This test method is designed to allow for the comparative evaluation of intervertebral body fusion devices. 1.4 Guidelines are established for measuring test block deformation and determining the subsidence of intervertebral body fusion devices. 1.5 Since some intervertebral body fusion devices require the use of additional implants for stabilization, the testing of these types of implants may not be in accordance with the manufacturer's recommended usage. 1.6 Units—The values stated in SI units are to be regarded as the standard with the exception of angular measurements, which may be reported in terms of either degrees or radians. 1.7 The use of this standard may involve the operation of potentially hazardous equipment. This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Intervertebral body fusion devices are generally simple geometric-shaped devices, which are often porous or hollow in nature. Their function is to support the anterior column of the spine to facilitate arthrodesis of the motion segment. 5.2 This test method is designed to quantify the subsidence characteristics of different designs of intervertebral body fusion devices since this is a potential clinical failure mode. These tests are conducted in vitro in order to simplify the comparison of simulated vertebral body subsidence induced by the intervertebral body fusion devices. 5.3 The static axial compressive loads that will be applied to the intervertebral body fusion devices and test blocks will differ from the complex loading seen in vivo, and therefore, the results from this test method may not be used to directly predict in vivo performance. The results, however, can be used to compare the varying degrees of subsidence between different intervertebral body fusion device designs for a given density of simulated bone. 5.4 The location within the simulated vertebral bodies and position of the intervertebral body fusion device with respect to the loading axis will be dependent upon the design and manufacturer's recommendation for implant placement. SCOPE 1.1 This test method specifies the materials and methods for the axial compressive subsidence testing of non-biologic intervertebral body fusion devices, spinal implants designed to promote arthrodesis at a given spinal motion segment. 1.2 This test method is intended to provide a basis for the mechanical comparison among past, present, and future non-biologic intervertebral body fusion devices. This test method is intended to enable the user to mechanically compare intervertebral body fusion devices and does not purport to provide performance standards for intervertebral body fusion devices. 1.3 This test method describes a static test method by specifying a load type and a specific method of applying this load. This test method is designed to allow for the comparative evaluation of intervertebral body fusion devices. 1.4 Guidelines are established for measuring test block deformation and determining the subsidence of intervertebral body fusion devices. 1.5 Since some intervertebral body fusion devices require the use of additional implants for stabilization, the testing of these types of implants may not be in accordance with the manufacturer's recommended usage. 1.6 Units—The values stated in SI units are to be regarded as the standard with the exception of angular measurements, which may be reported in terms of either degrees or radians. 1.7 The use of this standard may involve the operation of potentially hazardous equipment. This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2267-24 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2267-24 has the following relationships with other standards: It is inter standard links to ASTM F2267-22, ASTM F2789-10(2020), ASTM F3631-24, ASTM F3292-19. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2267-24 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2267 − 24
Standard Test Method for
Measuring Load-Induced Subsidence of Intervertebral Body
Fusion Device Under Static Axial Compression
This standard is issued under the fixed designation F2267; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
1.1 This test method specifies the materials and methods for
mendations issued by the World Trade Organization Technical
the axial compressive subsidence testing of non-biologic in-
Barriers to Trade (TBT) Committee.
tervertebral body fusion devices, spinal implants designed to
promote arthrodesis at a given spinal motion segment.
2. Referenced Documents
1.2 This test method is intended to provide a basis for the
2.1 ASTM Standards:
mechanical comparison among past, present, and future non-
E4 Practices for Force Calibration and Verification of Test-
biologic intervertebral body fusion devices. This test method is
ing Machines
intended to enable the user to mechanically compare interver-
E177 Practice for Use of the Terms Precision and Bias in
tebral body fusion devices and does not purport to provide
ASTM Test Methods
performance standards for intervertebral body fusion devices.
E691 Practice for Conducting an Interlaboratory Study to
Determine the Precision of a Test Method
1.3 This test method describes a static test method by
F1839 Specification for Rigid Polyurethane Foam for Use as
specifying a load type and a specific method of applying this
a Standard Material for Testing Orthopaedic Devices and
load. This test method is designed to allow for the comparative
Instruments
evaluation of intervertebral body fusion devices.
F2077 Test Methods for Intervertebral Body Fusion Devices
1.4 Guidelines are established for measuring test block
deformation and determining the subsidence of intervertebral 3. Terminology
body fusion devices.
3.1 All subsidence testing terminology is consistent with the
1.5 Since some intervertebral body fusion devices require referenced standards above, unless otherwise stated.
the use of additional implants for stabilization, the testing of
3.2 Definitions:
these types of implants may not be in accordance with the
3.2.1 coordinate system/axes—three orthogonal axes are
manufacturer’s recommended usage.
defined following a right-handed Cartesian coordinate system
1.6 Units—The values stated in SI units are to be regarded (Fig. 4). The XY plane bisects the sagittal plane between the
as the standard with the exception of angular measurements, superior and inferior surfaces that are intended to simulate the
which may be reported in terms of either degrees or radians. adjacent vertebral end plates. The positive Z-axis is to be
directed superiorly. Force components parallel to the XY plane
1.7 The use of this standard may involve the operation of
are shear components of loading. The compressive axial force
potentially hazardous equipment. This standard does not pur-
is defined to be the component in the negative Z direction.
port to address all of the safety concerns, if any, associated
Torsional load is defined to be the component of moment about
with its use. It is the responsibility of the user of this standard
the Z-axis.
to establish appropriate safety, health, and environmental
practices and determine the applicability of regulatory limita- 3.2.1.1 origin—the center of the coordinate system is lo-
tions prior to use. cated at the center of rotation of the testing fixture.
1.8 This international standard was developed in accor-
3.2.1.2 X-axis—the positive X-axis is a global fixed axis
dance with internationally recognized principles on standard-
relative to the testing machine’s stationary base and is to be
directed anteriorly relative to the specimen’s initial unloaded
position.
This test method is under the jurisdiction of ASTM Committee F04 on Medical
and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.25 on Spinal Devices. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved April 15, 2024. Published April 2024. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2003. Last previous edition approved in 2022 as F2267 – 22. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/F2267-24. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2267 − 24
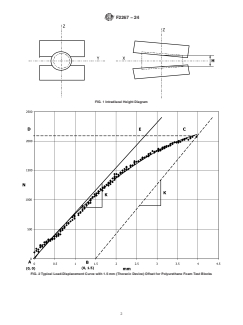
FIG. 1 Intradiscal Height Diagram
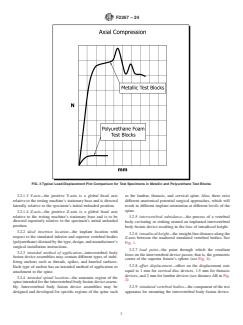
FIG. 2 Typical Load-Displacement Curve with 1.5 mm (Thoracic Device) Offset for Polyurethane Foam Test Blocks
F2267 − 24
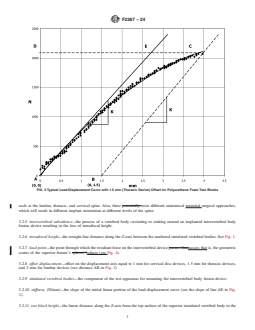
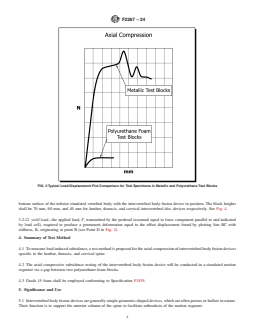
FIG. 3 Typical Load-Displacement Plot Comparison for Test Specimens in Metallic and Polyurethane Test Blocks
3.2.1.3 Y-axis—the positive Y-axis is a global fixed axis as the lumbar, thoracic, and cervical spine. Also, there exist
relative to the testing machine’s stationary base and is directed different anatomical potential surgical approaches, which will
laterally relative to the specimen’s initial unloaded position. result in different implant orientation at different levels of the
spine.
3.2.1.4 Z-axis—the positive Z-axis is a global fixed axis
relative to the testing machine’s stationary base and is to be
3.2.5 intervertebral subsidence—the process of a vertebral
directed superiorly relative to the specimen’s initial unloaded
body cavitating or sinking around an implanted intervertebral
position.
body fusion device resulting in the loss of intradiscal height.
3.2.2 ideal insertion location—the implant location with
3.2.6 intradiscal height—the straight-line distance along the
respect to the simulated inferior and superior vertebral bodies
Z-axis between the unaltered simulated vertebral bodies. See
(polyurethane) dictated by the type, design, and manufacturer’s
Fig. 1.
surgical installation instructions.
3.2.7 load point—the point through which the resultant
3.2.3 intended method of application—intervertebral body
force on the intervertebral device passes; that is, the geometric
fusion device assemblies may contain different types of stabi-
center of the superior fixture’s sphere (see Fig. 4).
lizing anchors such as threads, spikes, and knurled surfaces.
3.2.8 offset displacement—offset on the displacement axis
Each type of anchor has an intended method of application or
equal to 1 mm for cervical disc devices, 1.5 mm for thoracic
attachment to the spine.
devices, and 2 mm for lumbar devices (see distance AB in Fig.
3.2.4 intended spinal location—the anatomic region of the
2)
spine intended for the intervertebral body fusion device assem-
3.2.9 simulated vertebral bodies—the component of the test
bly. Intervertebral body fusion device assemblies may be
designed and developed for specific regions of the spine such apparatus for mounting the intervertebral body fusion device.
F2267 − 24
FIG. 4 Subsidence Test Fixture
3.2.10 stiffness, (N/mm)—the slope of the initial linear nature. Their function is to support the anterior column of the
portion of the load-displacement curve (see the slope of line spine to facilitate arthrodesis of the motion segment.
AE in Fig. 2).
5.2 This test method is designed to quantify the subsidence
3.2.11 test block height—the linear distance along the Z-axis
characteristics of different designs of intervertebral body fusion
from the top surface of the superior simulated vertebral body to
devices since this is a potential clinical failure mode. These
the bottom surface of the inferior simulated vertebral body with
tests are conducted in vitro in order to simplify the comparison
the intervertebral body fusion device in position. The block
of simulated vertebral body subsidence induced by the in-
heights shall be 70 mm, 60 mm, and 40 mm for lumbar,
tervertebral body fusion devices.
thoracic, and cervical intervertebral disc devices respectively.
5.3 The static axial compressive loads that will be applied to
See Fig. 4.
the intervertebral body fusion devices and test blocks will
3.2.12 yield load—the applied load, F, transmitted by the
differ from the complex loading seen in vivo, and therefore, the
pushrod (assumed equal to force component parallel to and
results from this test method may not be used to directly predict
indicated by load cell), required to produce a permanent
in vivo performance. The results, however, can be used to
deformation equal to the offset displacement found by plotting
compare the varying degrees of subsidence between different
line BC with stiffness, K, originating at point B (see Point D in
intervertebral body fusion device designs for a given density of
Fig. 2).
simulated bone.
4. Summary of Test Method
5.4 The location within the simulated vertebral bodies and
position of the intervertebral body fusion device with respect to
4.1 To measure load-induced subsidence, a test method is
the loading axis will be dependent upon the design and
proposed for the axial compression of intervertebral body
manufacturer’s recommendation for implant placement.
fusion devices specific to the lumbar, thoracic, and cervical
spine.
6. Apparatus
4.2 The axial compressive subsidence testing of the in-
6.1 Test machines will conform to the requirements of
tervertebral body fusion device will be conducted in a simu-
Practices E4.
lated motion segment via a gap between two polyurethane
foam blocks.
6.2 The intradiscal height, H (Fig. 1), shall be determined
from vertebral body and disc morphometric data at the in-
4.3 Grade 15 foam shall be employed conforming to Speci-
tended level of application. Suggested heights are as follows:
fication F1839.
10 mm for the lumbar spine, 6 mm for the thoracic spine, and
5. Significance and Use
4 mm for the cervical spine. The user of this test method should
5.1 Intervertebral body fusion devices are generally simple select the intradiscal height that is appropriate for the device
geometric-shaped devices, which are often porous or hollow in being tested.
F2267 − 24
6.3 Axial Compressive Testing Apparatus—An example superior sphere should be of minimal weight so as to be
axial compressive test fixture can be referenced in Figs. 4 and considered a “two force” member. It thus applies to the
5. Two pieces of polyurethane foam or rigid metal are rigidly intervertebral device a resultant force directed along the
mounted inside the test fixture. The actuator of the testing pushrod’s axes and located at the center of the superior
machine is connected to the pushrod by a minimal friction fixture’s sphere joint (the geometric center of the device being
ball-and-socket joint or universal joint (that is, unconstrained tested). The polyurethane blocks are to have surfaces that mate
in bending). The pushrod is connected to the superior fixture by geometrically with the intervertebral device similar to how the
a minimal friction sphere joint (that is, unconstrained in device is intended to mate with vertebral end plates. The test
bending and torsion). The inferior sphere portion firmly holds apparatus will be assembled such that the Z-axis of the
the inferior polyurethane block and is rigidly fixe
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2267 − 22 F2267 − 24
Standard Test Method for
Measuring Load-Induced Subsidence of Intervertebral Body
Fusion Device Under Static Axial Compression
This standard is issued under the fixed designation F2267; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method specifies the materials and methods for the axial compressive subsidence testing of non-biologic
intervertebral body fusion devices, spinal implants designed to promote arthrodesis at a given spinal motion segment.
1.2 This test method is intended to provide a basis for the mechanical comparison among past, present, and future non-biologic
intervertebral body fusion devices. This test method is intended to enable the user to mechanically compare intervertebral body
fusion devices and does not purport to provide performance standards for intervertebral body fusion devices.
1.3 This test method describes a static test method by specifying a load type and a specific method of applying this load. This test
method is designed to allow for the comparative evaluation of intervertebral body fusion devices.
1.4 Guidelines are established for measuring test block deformation and determining the subsidence of intervertebral body fusion
devices.
1.5 Since some intervertebral body fusion devices require the use of additional implants for stabilization, the testing of these types
of implants may not be in accordance with the manufacturer’s recommended usage.
1.6 Units—The values stated in SI units are to be regarded as the standard with the exception of angular measurements, which may
be reported in terms of either degrees or radians.
1.7 The use of this standard may involve the operation of potentially hazardous equipment. This standard does not purport to
address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish
appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
This test method is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.25 on Spinal Devices.
Current edition approved Sept. 1, 2022April 15, 2024. Published September 2022April 2024. Originally approved in 2003. Last previous edition approved in 20182022
as F2267 – 04 (2018).F2267 – 22. DOI: 10.1520/F2267-22.10.1520/F2267-24.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2267 − 24
E4 Practices for Force Calibration and Verification of Testing Machines
E177 Practice for Use of the Terms Precision and Bias in ASTM Test Methods
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
F1582 Terminology Relating to Spinal Implants
F1839 Specification for Rigid Polyurethane Foam for Use as a Standard Material for Testing Orthopaedic Devices and
Instruments
F2077 Test Methods for Intervertebral Body Fusion Devices
3. Terminology
3.1 All subsidence testing terminology is consistent with the referenced standards above, unless otherwise stated.
3.2 Definitions:
3.2.1 coordinate system/axes—three orthogonal axes are defined by Terminologyfollowing a F1582 as seen inright-handed
Cartesian coordinate system (Fig. 4. The center of the coordinate system is located at the geometric center of the intervertebral
body fusion device assembly. ). The XY plane bisects the sagittal plane between the superior and inferior surfaces that are intended
to simulate the adjacent vertebral end plates. The Xpositive-axis is along the longitudinal axis of the implant, with positive Z-axis
is to be directed superiorly. Force components parallel to the XY plane are shear components of loading. The compressive axial
Xforce in the anterior direction,is defined to be the component in the negative Z Ydirection. is lateral, andTorsional load is defined
to Zbe is cephalic.the component of moment about the Z-axis.
3.2.1.1 origin—the center of the coordinate system is located at the center of rotation of the testing fixture.
3.2.1.2 X-axis—the positive X-axis is a global fixed axis relative to the testing machine’s stationary base and is to be directed
anteriorly relative to the specimen’s initial unloaded position.
3.2.1.3 Y-axis—the positive Y-axis is a global fixed axis relative to the testing machine’s stationary base and is directed laterally
relative to the specimen’s initial unloaded position.
3.2.1.4 Z-axis—the positive Z-axis is a global fixed axis relative to the testing machine’s stationary base and is to be directed
superiorly relative to the specimen’s initial unloaded position.
3.2.2 ideal insertion location—the implant location with respect to the simulated inferior and superior vertebral bodies
(polyurethane) dictated by the type, design, and manufacturer’s surgical installation instructions.
3.2.3 intended method of application—intervertebral body fusion devices device assemblies may contain different types of
stabilizing featuresanchors such as threads, spikes, and knurled surfaces. Each type of featureanchor has an intended method of
application or attachment to the spine.
3.2.4 intended spinal location—the anatomic region of the spine intended for the intervertebral body fusion device. device
assembly. Intervertebral body fusion devices device assemblies may be designed and developed for specific regions of the spine
FIG. 1 Intradiscal Height Diagram
F2267 − 24
FIG. 2 Typical Load-Displacement Curve with 1.5 mm (Thoracic Device) Offset for Polyurethane Foam Test Blocks
such as the lumbar, thoracic, and cervical spine. Also, there potentially exist different anatomical potential surgical approaches,
which will result in different implant orientation at different levels of the spine.
3.2.5 intervertebral subsidence—the process of a vertebral body cavitating or sinking around an implanted intervertebral body
fusion device resulting in the loss of intradiscal height.
3.2.6 intradiscal height—the straight-line distance along the Z-axis between the unaltered simulated vertebral bodies. See Fig. 1.
3.2.7 load point—the point through which the resultant force on the intervertebral device passes (thatpasses; that is, the geometric
center of the superior fixture’s sphere) (sphere (see Fig. 4).
3.2.8 offset displacement—offset on the displacement axis equal to 1 mm for cervical disc devices, 1.5 mm for thoracic devices,
and 2 mm for lumbar devices (see distance AB in Fig. 2)
3.2.9 simulated vertebral bodies—the component of the test apparatus for mounting the intervertebral body fusion device.
3.2.10 stiffness, (N/mm)—the slope of the initial linear portion of the load-displacement curve (see the slope of line AE in Fig.
2).
3.2.11 test block height—the linear distance along the Z-axis from the top surface of the superior simulated vertebral body to the
F2267 − 24
FIG. 3 Typical Load-Displacement Plot Comparison for Test Specimens in Metallic and Polyurethane Test Blocks
bottom surface of the inferior simulated vertebral body with the intervertebral body fusion device in position. The block heights
shall be 70 mm, 60 mm, and 40 mm for lumbar, thoracic, and cervical intervertebral disc devices respectively. See Fig. 4.
3.2.12 yield load—the applied load, F, transmitted by the pushrod (assumed equal to force component parallel to and indicated
by load cell), required to produce a permanent deformation equal to the offset displacement found by plotting line BC with
stiffness, K, originating at point B (see Point D in Fig. 2).
4. Summary of Test Method
4.1 To measure load-induced subsidence, a test method is proposed for the axial compression of intervertebral body fusion devices
specific to the lumbar, thoracic, and cervical spine.
4.2 The axial compressive subsidence testing of the intervertebral body fusion device will be conducted in a simulated motion
segment via a gap between two polyurethane foam blocks.
4.3 Grade 15 foam shall be employed conforming to Specification F1839.
5. Significance and Use
5.1 Intervertebral body fusion devices are generally simple geometric-shaped devices, which are often porous or hollow in nature.
Their function is to support the anterior column of the spine to facilitate arthrodesis of the motion segment.
F2267 − 24
FIG. 4 Subsidence Test Fixture
5.2 This test method is designed to quantify the subsidence characteristics of different designs of intervertebral body fusion
devices since this is a potential clinical failure mode. These tests are conducted in vitro in order to simplify the comparison of
simulated vertebral body subsidence induced by the intervertebral body fusion devices.
5.3 The static axial compressive loads that will be applied to the intervertebral body fusion devices and test blocks will differ from
the complex loading seen in vivo, and therefore, the results from this test method may not be used to directly predict in vivo
performance. The results, however, can be used to compare the varying degrees of subsidence between different intervertebral body
fusion device designs for a given density of simulated bone.
5.4 The location within the simulated vertebral bodies and position of the intervertebral body fusion device with respect to the
loading axis will be dependent upon the design and manufacturer’s recommendation for implant placement.
6. Apparatus
6.1 Test machines will conform to the requirements of Practices E4.
6.2 The intradiscal height, H (Fig. 1), shall be determined from vertebral body and disc morphometric data at the intended level
of application. Suggested heights are as follows: 10 mm for the lumbar spine, 6 mm for the thoracic spine, and 4 mm for the
cervical spine. The user of this test method should select the intradiscal height that is appropriate for the device being tested.
6.3 Axial Compressive Testing Apparatus—An example axial compressive test fixture can be referenced in Figs. 4 and 5. Two
pieces of polyurethane foam or rigid metal are rigidly mounted inside the test fixture. The actuator of the testing machine is
connected to the pushrod by a minimal friction ball-and-socket joint or universal joint (that is, unconstrained in bending). The
pushrod is connected to the superior fixture by a minimal friction sphere joint (that is, unconstrained in bending and torsion). The
inferior sphere portion firmly holds the inferior polyurethane block and is rigidly fixed within the base socket so that no rotation
occurs. The hollow pushrod and superior sphere should be of minimal weight so as to be considered a “two force” member. It thus
applies to the intervertebral device a resultant force directed along the pushrod’s axes and located at the cen
...




Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...