ASTM D4328-18
(Practice)Standard Practice for Calculation of Supersaturation of Barium Sulfate, Strontium Sulfate, and Calcium Sulfate Dihydrate (Gypsum) in Brackish Water, Seawater, and Brines
Standard Practice for Calculation of Supersaturation of Barium Sulfate, Strontium Sulfate, and Calcium Sulfate Dihydrate (Gypsum) in Brackish Water, Seawater, and Brines
SIGNIFICANCE AND USE
4.1 This practice covers the mathematical calculation of the supersaturation of three principal sulfate scaling compounds found in industrial operations. Application of this standard practice to the prediction of scale formation in a given system, however, requires experience. The calculations tell the user if a water, or mixture of waters, is in a scaling mode. Whether or not scale will in fact form, how quickly it will form, where it will form, in what quantities, and what composition are subject to factors beyond the scope of this practice. However, based on how supersaturated a given water or mixture of waters is, an objective evaluation of the relative likelihood of scale formation can be made.
Note 1: There are several personal computer (PC) type programs that are both available commercially and publicly that will perform these calculations.
SCOPE
1.1 This practice covers the calculation of supersaturation of barium sulfate, strontium sulfate, and calcium sulfate dihydrate (gypsum) in brackish water, seawater, and brines in which barium, strontium, and calcium ions either coexist or exist individually in solution in the presence of sulfate ions.
1.2 This practice is not applicable for calculating calcium sulfate dihydrate supersaturation if the temperatures of saline waters under investigation exceed 95°C. At temperatures above 95°C, hemianhydrate and anhydrite would be major insoluble forms.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2018
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.05 - Inorganic Constituents in Water
Relations
- Effective Date
- 01-May-2018
- Effective Date
- 01-May-2020
- Effective Date
- 01-Jun-2017
- Refers
ASTM D3352-15 - Standard Test Method for Strontium Ion in Brackish Water, Seawater, and Brines - Effective Date
- 01-Feb-2015
- Effective Date
- 06-Feb-2012
- Effective Date
- 01-Sep-2011
- Effective Date
- 01-Sep-2011
- Effective Date
- 01-Sep-2011
- Effective Date
- 01-Sep-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 15-Sep-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-May-2009
- Refers
ASTM D3352-08a - Standard Test Method for Strontium Ion in Brackish Water, Seawater, and Brines - Effective Date
- 15-Nov-2008
- Effective Date
- 01-Oct-2008
Overview
ASTM D4328-18 is the internationally recognized standard practice for the calculation of supersaturation of barium sulfate, strontium sulfate, and calcium sulfate dihydrate (gypsum) in brackish water, seawater, and brines. Published by ASTM International, this document seeks to provide a mathematical approach to assess the scale-forming potential of these key sulfates in natural and industrial water systems, an essential factor in water management for sectors where mineral scaling can impact operational efficiency.
Supersaturation calculations help determine whether a particular water source is likely to precipitate scale-forming compounds, providing a basis for anticipating and managing scale deposition. While this standard provides calculation methods, it does not predict the exact rate, location, or composition of scale development due to system-specific variables.
Key Topics
- Supersaturation Calculation: Outlines step-by-step procedures for calculating the supersaturation of barium sulfate, strontium sulfate, and calcium sulfate dihydrate.
- Scope Limitations: Not suitable for calcium sulfate dihydrate supersaturation calculations at temperatures >95°C, as other mineral forms predominate above this threshold.
- Analytical Prerequisites: Requires chemical analysis of major inorganic ions (Ba, Sr, Ca, SO₄, Na, K, Cl, Mg, carbonate species) and physical parameters (temperature, pressure).
- SI Units: All results are reported using the International System of Units (SI).
- Safety Notice: Users are responsible for observing appropriate health, safety, and environmental protocols.
Applications
- Industrial Water Treatment: Essential for predicting and controlling scale in power generation, oil and gas production, desalination, and chemical processing industries.
- Seawater and Brine Operations: Enables accurate prediction of scale risk in high-salinity environments, informing preventative maintenance strategies.
- Environmental Monitoring: Supports the management of mineral scaling in natural and engineered aquatic systems.
- Process Optimization: By predicting the supersaturation of sulfate salts, operators can adjust water chemistry or treatment regimes to reduce unplanned shutdowns and increase equipment lifespan.
- Software Integration: Calculation methods from the standard are implemented in many commercially and publicly available PC software programs for automated supersaturation and scaling risk assessments.
Related Standards
Several ASTM standards complement or support the use of ASTM D4328-18 for comprehensive water analysis in scale assessment:
- ASTM D511: Test Methods for Calcium and Magnesium in Water
- ASTM D512: Test Methods for Chloride Ion in Water
- ASTM D513: Test Methods for Total and Dissolved Carbon Dioxide in Water
- ASTM D516: Test Method for Sulfate Ion in Water
- ASTM D1129: Terminology Relating to Water
- ASTM D3352: Test Method for Strontium Ion in Brackish Water, Seawater, and Brines
- ASTM D3370: Practices for Sampling Water from Closed Conduits
- ASTM D3561: Test Method for Lithium, Potassium, and Sodium Ions in Brackish Water, Seawater, and Brines by Atomic Absorption Spectrophotometry
- ASTM D3651 and D3986: Test Methods for Barium in Brines, Seawater, and Brackish Water
Practical Value
Adhering to ASTM D4328-18 ensures standardization, reliability, and comparability in the assessment of scale formation risks across industries. The ability to mathematically determine the supersaturation of sulfate salts empowers engineers and water treatment professionals to make informed decisions, optimize operational processes, and comply with industry regulations for sustainable water usage and system integrity. Use of this standard also supports international best practices in water management and aligns with the requirements set by the World Trade Organization for technical trade compliance.
Keywords: sulfate scale, barium sulfate, strontium sulfate, calcium sulfate dihydrate, gypsum, brackish water, seawater, brines, ASTM D4328-18, scaling prediction, water analysis, supersaturation calculation, industrial water treatment.
Buy Documents
ASTM D4328-18 - Standard Practice for Calculation of Supersaturation of Barium Sulfate, Strontium Sulfate, and Calcium Sulfate Dihydrate (Gypsum) in Brackish Water, Seawater, and Brines
REDLINE ASTM D4328-18 - Standard Practice for Calculation of Supersaturation of Barium Sulfate, Strontium Sulfate, and Calcium Sulfate Dihydrate (Gypsum) in Brackish Water, Seawater, and Brines
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D4328-18 is a standard published by ASTM International. Its full title is "Standard Practice for Calculation of Supersaturation of Barium Sulfate, Strontium Sulfate, and Calcium Sulfate Dihydrate (Gypsum) in Brackish Water, Seawater, and Brines". This standard covers: SIGNIFICANCE AND USE 4.1 This practice covers the mathematical calculation of the supersaturation of three principal sulfate scaling compounds found in industrial operations. Application of this standard practice to the prediction of scale formation in a given system, however, requires experience. The calculations tell the user if a water, or mixture of waters, is in a scaling mode. Whether or not scale will in fact form, how quickly it will form, where it will form, in what quantities, and what composition are subject to factors beyond the scope of this practice. However, based on how supersaturated a given water or mixture of waters is, an objective evaluation of the relative likelihood of scale formation can be made. Note 1: There are several personal computer (PC) type programs that are both available commercially and publicly that will perform these calculations. SCOPE 1.1 This practice covers the calculation of supersaturation of barium sulfate, strontium sulfate, and calcium sulfate dihydrate (gypsum) in brackish water, seawater, and brines in which barium, strontium, and calcium ions either coexist or exist individually in solution in the presence of sulfate ions. 1.2 This practice is not applicable for calculating calcium sulfate dihydrate supersaturation if the temperatures of saline waters under investigation exceed 95°C. At temperatures above 95°C, hemianhydrate and anhydrite would be major insoluble forms. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This practice covers the mathematical calculation of the supersaturation of three principal sulfate scaling compounds found in industrial operations. Application of this standard practice to the prediction of scale formation in a given system, however, requires experience. The calculations tell the user if a water, or mixture of waters, is in a scaling mode. Whether or not scale will in fact form, how quickly it will form, where it will form, in what quantities, and what composition are subject to factors beyond the scope of this practice. However, based on how supersaturated a given water or mixture of waters is, an objective evaluation of the relative likelihood of scale formation can be made. Note 1: There are several personal computer (PC) type programs that are both available commercially and publicly that will perform these calculations. SCOPE 1.1 This practice covers the calculation of supersaturation of barium sulfate, strontium sulfate, and calcium sulfate dihydrate (gypsum) in brackish water, seawater, and brines in which barium, strontium, and calcium ions either coexist or exist individually in solution in the presence of sulfate ions. 1.2 This practice is not applicable for calculating calcium sulfate dihydrate supersaturation if the temperatures of saline waters under investigation exceed 95°C. At temperatures above 95°C, hemianhydrate and anhydrite would be major insoluble forms. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D4328-18 is classified under the following ICS (International Classification for Standards) categories: 13.060.30 - Sewage water. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D4328-18 has the following relationships with other standards: It is inter standard links to ASTM D4328-08(2013), ASTM D1129-13(2020)e2, ASTM D3986-17, ASTM D3352-15, ASTM D513-11e1, ASTM D516-11, ASTM D3651-11, ASTM D3561-11, ASTM D3986-11, ASTM D3370-10, ASTM D512-10, ASTM D1129-10, ASTM D511-09, ASTM D3352-08a, ASTM D3370-08. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D4328-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D4328 − 18
Standard Practice for
Calculation of Supersaturation of Barium Sulfate, Strontium
Sulfate, and Calcium Sulfate Dihydrate (Gypsum) in
Brackish Water, Seawater, and Brines
This standard is issued under the fixed designation D4328; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* D513Test Methods forTotal and Dissolved Carbon Dioxide
in Water
1.1 Thispracticecoversthecalculationofsupersaturationof
D516Test Method for Sulfate Ion in Water
bariumsulfate,strontiumsulfate,andcalciumsulfatedihydrate
D1129Terminology Relating to Water
(gypsum) in brackish water, seawater, and brines in which
D3352Test Method for Strontium Ion in Brackish Water,
barium, strontium, and calcium ions either coexist or exist
Seawater, and Brines
individually in solution in the presence of sulfate ions.
D3370Practices for Sampling Water from Closed Conduits
1.2 This practice is not applicable for calculating calcium
D3561Test Method for Lithium, Potassium, and Sodium
sulfate dihydrate supersaturation if the temperatures of saline
Ions in Brackish Water, Seawater, and Brines by Atomic
watersunderinvestigationexceed95°C.Attemperaturesabove
Absorption Spectrophotometry
95°C, hemianhydrate and anhydrite would be major insoluble
D3651TestMethodforBariuminBrackishWater,Seawater,
forms.
and Brines
1.3 The values stated in SI units are to be regarded as D3986Test Method for Barium in Brines, Seawater, and
Brackish Water by Direct-Current Argon Plasma Atomic
standard. No other units of measurement are included in this
standard. Emission Spectroscopy
1.4 This standard does not purport to address all of the
3. Terminology
safety concerns, if any, associated with its use. It is the
3.1 Definitions:
responsibility of the user of this standard to establish appro-
3.1.1 For definitions of terms used in this standard, refer to
priate safety, health, and environmental practices and deter-
Terminology D1129.
mine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accor-
4. Significance and Use
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the 4.1 This practice covers the mathematical calculation of the
Development of International Standards, Guides and Recom-
supersaturation of three principal sulfate scaling compounds
mendations issued by the World Trade Organization Technical found in industrial operations. Application of this standard
Barriers to Trade (TBT) Committee.
practice to the prediction of scale formation in a given system,
however, requires experience. The calculations tell the user if
2. Referenced Documents
a water, or mixture of waters, is in a scaling mode.Whether or
not scale will in fact form, how quickly it will form, where it
2.1 ASTM Standards:
willform,inwhatquantities,andwhatcompositionaresubject
D511Test Methods for Calcium and Magnesium In Water
tofactorsbeyondthescopeofthispractice.However,basedon
D512Test Methods for Chloride Ion In Water
how supersaturated a given water or mixture of waters is, an
objective evaluation of the relative likelihood of scale forma-
tion can be made.
This practice is under the jurisdiction ofASTM Committee D19 on Water and
is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents in
NOTE 1—There are several personal computer (PC) type programs that
Water.
are both available commercially and publicly that will perform these
Current edition approved May 1, 2018. Published May 2018. Originally
calculations.
approved in 1984. Last previous edition approved in 2013 as D4328–08 (2013).
DOI: 10.1520/D4328-18.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or 5. Procedure
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
5.1 Collect water samples for compositional analysis in
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. accordance with Practices D3370.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4328 − 18
NOTE 2—Supersaturation may also be calculated directly from the
5.2 Determine the calcium and magnesium concentrations
equation (1).
in accordance with Test Methods D511.
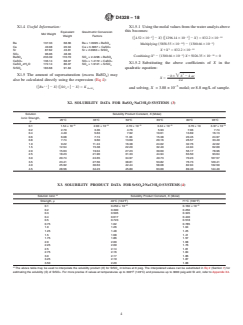
@Ba # 2 y @SO 5# 2 y 5 K (4)
~ !~ !
5.3 Determine the barium concentration in accordance with
Test Methods D3651 or D3986.
where:
2+
5.4 Determine the strontium concentration in accordance Ba = concentration of barium, molal,
2–
SO = concentration of sulfate, molal,
with Test Method D3352. 4
y = excess (supersaturation) of BaSO , molal, and
5.5 Determine sodium and potassium concentrations in
K = solubility product constant (molal) of BaSO at test
accordance with Test Method D3561.
conditions.
5.6 Determine sulfate ion concentration in accordance with
The value X may then be determined from the quadratic
Test Method D516.
equation (see Appendix X1):
5.7 Determine chloride ion concentration in accordance
=
2B6 B 2 4 AC
with Test Methods D512.
X 5 (5)
2A
5.8 Determinecarbonateandbicarbonateionconcentrations
Report BaSO supersaturation in molal terms of the weight
in accordance with Test Methods D513.
of BaSO per volume of water, mg/L.
5.9 Determine the concentrations of all other major inor-
BaSO supersaturation,mg/L (6)
ganic constituents that may be present in the water under
investigation in accordance with appropriate test methods in
1000 3D
2 3
Annual Book of ASTM Standards, Vols 11.01 and 11.02. 5BaSO , ~molal ! 310 3233 3 11000
TDS
S D
5.10 Determine temperature and pressure of the water 1000
system under investigation.
where:
6. Calculation of Ionic Strength
D = sample density.
6.1 Calculate the ionic strength of the water under investi-
8. Calculation of Strontium Sulfate Supersaturation
gation as follows:
(Refer to Appendix X1)
8.1 Calculate strontium sulfate solubility using the same
µ 5 C Z (1)
i i
(
steps described for BaSO (Section 7), but substituting the
appropriate values for SrSO in Eq 2 (refer to Appendix X3 or
where:
Appendix X4).
µ = ionic strength,
C = molal concentration of each ion in solution, and NOTE 3—If barium sulfate supersaturation exists, the amount of sulfate
i
available for strontium sulfate will be less by the amount of sulfate
Z = charge number of ion, i.
i
equivalent to the calculated BaSO supersaturation.
NOTE 4—If carbonate ions are present, strontium carbonate may
7. Calculation of Barium Sulfate Supersaturation (Refer
precipitate. The amount of strontium may then be corrected by that
to Appendix X1)
required for strontium carbonate precipitation prior to the calculation of
7.1 Calculate barium sulfate solubility in the water under SrSO solubility (2). Practically speaking, however, due to the extremely
low solubility of SrCO , this correction may usually be omitted.
investigation, using the equation as follows:
8.2 Calculate the amount of strontium sulfate moles per
S 5 ~=X 14K 2 X!/2 (2)
kilogram water in the sample based on the lesser of the
strontium or remaining sulfate ion concentration.
where:
S = solubility, moles of solute per kilogram of water 8.3 If the amount of SrSO in the sample (8.2) is less than
corrected for the common ion effect,
its calculated solubility (8.1), the water in question is under-
K = solubilityproductconstant(molal)attheionicstrength,
saturatedwithrespecttoSrSO .IftheamountofSrSO present
4 4
temperature and pressure of the water under investiga-
is greater than its solubility, the water is supersaturated with
tion. For BaSO refer to Appendix X2, and
respect to SrSO . Calculate the amount of supersaturation,
X = molal excess of soluble common ion.
moles per kilogram water by difference (Eq 3), or by substi-
tuting appropriate data in Eq 4 (Note 2).
7.2 Calculate the amount of barium sulfate, moles per
8.3.1 ReportSrSO supersaturationintermsoftheweightof
kilogram of water, in the sample based on the lesser of the 4
SrSO per volume of water as follows:
barium or sulfate ion concentration. 4
SrSO supersaturationmg⁄L (7)
7.3 If the amount of BaSO in the sample (7.2) is less than
its calculated solubility (7.1), the water in question is under-
1000 3D
saturated with respect to BaSO . If the amount of BaSO
5SrSO , ~molal! 310 3184 3
4 4
TDS
S D
present is greater than its solubility, the water is supersaturated
withrespecttoBaSO .Calculatetheamountofsupersaturation
as the difference between the two values:
The boldfaced numbers in parentheses refer to a list of references at the end of
supersaturation 5 concentration 2 solubility (3) this standard.
D4328 − 18
9. Calculation of Calcium Sulfate Supersaturation (Refer withrespecttoCaSO .Calculatetheamountofsupersaturation
to Appendix X1) moles per kilogram by difference (Eq 3) or by substituting
appropriate data in Eq 4 (Not
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D4328 − 08 (Reapproved 2013) D4328 − 18
Standard Practice for
Calculation of Supersaturation of Barium Sulfate, Strontium
Sulfate, and Calcium Sulfate Dihydrate (Gypsum) in
Brackish Water, Seawater, and Brines
This standard is issued under the fixed designation D4328; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Scope*
1.1 This practice covers the calculation of supersaturation of barium sulfate, strontium sulfate, and calcium sulfate dihydrate
(gypsum) in brackish water, seawater, and brines in which barium, strontium, and calcium ions either coexist or exist individually
in solution in the presence of sulfate ions.
1.2 This practice is not applicable for calculating calcium sulfate dihydrate supersaturation if the temperatures of saline waters
under investigation exceed 95°C. At temperatures above 95°C, hemianhydrate and anhydrite would be major insoluble forms.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D511 Test Methods for Calcium and Magnesium In Water
D512 Test Methods for Chloride Ion In Water
D513 Test Methods for Total and Dissolved Carbon Dioxide in Water
D516 Test Method for Sulfate Ion in Water
D1129 Terminology Relating to Water
D3352 Test Method for Strontium Ion in Brackish Water, Seawater, and Brines
D3370 Practices for Sampling Water from Closed Conduits
D3561 Test Method for Lithium, Potassium, and Sodium Ions in Brackish Water, Seawater, and Brines by Atomic Absorption
Spectrophotometry
D3651 Test Method for Barium in Brackish Water, Seawater, and Brines
D3986 Test Method for Barium in Brines, Seawater, and Brackish Water by Direct-Current Argon Plasma Atomic Emission
Spectroscopy
3. Terminology
3.1 Definitions—Definitions: For definitions of terms used in this practice, refer to Terminology D1129.
3.1.1 For definitions of terms used in this standard, refer to Terminology D1129.
4. Significance and Use
4.1 This practice covers the mathematical calculation of the supersaturation of three principal sulfate scaling compounds found
in industrial operations. Application of this standard practice to the prediction of scale formation in a given system, however,
This practice is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents in Water.
Current edition approved June 1, 2013May 1, 2018. Published July 2013May 2018. Originally approved in 1984. Last previous edition approved in 20082013 as
D4328 – 08.D4328 – 08 (2013). DOI: 10.1520/D4328-08R13.10.1520/D4328-18.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4328 − 18
requires experience. The calculations tell the user if a water, or mixture of waters, is in a scaling mode. Whether or not scale will
in fact form, how quickly it will form, where it will form, in what quantities, and what composition are subject to factors beyond
the scope of this practice. However, based on how supersaturated a given water or mixture of waters is, an objective evaluation
of the relative likelihood of scale formation can be made.
NOTE 1—There are several personal computer (PC) type programs that are both available commercially and publicly that will perform these
calculations.
5. Procedure
5.1 Collect water samples for compositional analysis in accordance with Practices D3370.
5.2 Determine the calcium and magnesium concentrations in accordance with Test Methods D511.
5.3 Determine the barium concentration in accordance with Test Methods D3651 or D3986.
5.4 Determine the strontium concentration in accordance with Test Method D3352.
5.5 Determine sodium and potassium concentrations in accordance with Test Method D3561.
5.6 Determine sulfate ion concentration in accordance with Test Method D516.
5.7 Determine chloride ion concentration in accordance with Test Methods D512.
5.8 Determine carbonate and bicarbonate ion concentrations in accordance with Test Methods D513.
5.9 Determine the concentrations of all other major inorganic constituents that may be present in the water under investigation
in accordance with appropriate test methods in Annual Book of ASTM Standards, Vols 11.01 and 11.02.
5.10 Determine temperature and pressure of the water system under investigation.
6. Calculation of Ionic Strength
6.1 Calculate the ionic strength of the water under investigation as follows:
μ 5 C Z (1)
( i i
where:
μ = ionic strength,
C = molal concentration of each ion in solution, and
i
Z = charge number of ion, i.
i
7. Calculation of Barium Sulfate Supersaturation (Refer to Appendix X1)
7.1 Calculate barium sulfate solubility in the water under investigation, using the equation as follows:
S 5~=X 14K 2 X!/2 (2)
where:
S = solubility, moles of solute per kilogram of water corrected for the common ion effect,
K = solubility product constant (molal) at the ionic strength, temperature and pressure of the water under investigation. For
BaSO refer to Appendix X2, and
X = molal excess of soluble common ion.
7.2 Calculate the amount of barium sulfate, moles per kilogram of water, in the sample based on the lesser of the barium or
sulfate ion concentration.
7.3 If the amount of BaSO in the sample (7.2) is less than its calculated solubility (7.1), the water in question is undersaturated
with respect to BaSO . If the amount of BaSO present is greater than its solubility, the water is supersaturated with respect to
4 4
BaSO . Calculate the amount of supersaturation as the difference between the two values:
supersaturation 5 concentration 2 solubility (3)
NOTE 2—Supersaturation may also be calculated directly from the equation (1).
@Ba # 2 y @SO 5# 2 y 5 K (4)
~ !~ !
where:
2+
Ba = concentration of barium, molal,
2–
SO = concentration of sulfate, molal,
The boldfaced numbers in parentheses refer to a list of references at the end of this standard.
D4328 − 18
y = excess (supersaturation) of BaSO , molal, and
K = solubility product constant (molal) of BaSO at test conditions.
The value X may then be determined from the quadratic equation (see Appendix X1):
2B6=B 2 4 AC
X 5 (5)
2A
Report BaSO supersaturation in molal terms of the weight of BaSO per volume of water, mg/L.
4 4
BaSO supersaturation,mg/L
1000 3D
2 3
5BaSO , molal 310 3233 3 11000
~ !
TDS
S D
BaSO supersaturation,mg/L (6)
1000 3D
2 3
5BaSO , ~molal !310 3233 3 11000
TDS
S D
where:where:
D = sample density.
8. Calculation of Strontium Sulfate Supersaturation (Refer to Appendix X1)
8.1 Calculate strontium sulfate solubility using the same steps described for BaSO (Section 7), but substituting the appropriate
values for SrSO in Eq 2 (refer to Appendix X3 or Appendix X4).
NOTE 3—If barium sulfate supersaturation exists, the amount of sulfate available for strontium sulfate will be less by the amount of sulfate equivalent
to the calculated BaSO supersaturation.
NOTE 4—If carbonate ions are present, strontium carbonate may precipitate. The amount of strontium may then be corrected by that required for
strontium carbonate precipitation prior to the calculation of SrSO solubility (2). Practically speaking, however, due to the extremely low solubility of
SrCO , this correction may usually be omitted.
8.2 Calculate the amount of strontium sulfate moles per kilogram water in the sample based on the lesser of the strontium or
remaining sulfate ion concentration.
8.3 If the amount of SrSO in the sample (8.2) is less than its calculated solubility (8.1), the water in question is undersaturated
with respect to SrSO . If the amount of SrSO present is greater than its solubility, the water is supersaturated with respect to
4 4
SrSO . Calculate the amount of supersaturation, moles per kilogram water by difference (Eq 3), or by substituting appropriate data
in Eq 4 (Note 2).
8.3.1 Report SrSO supersaturation in terms of the weight of SrSO per volume of water as follows:
4 4
SrSO supersaturation mg⁄L
1000 3D
5SrSO , ~molal!310 3184 3
TDS
S D
SrSO supersaturation mg⁄L (7)
1000 3D
5SrSO , ~molal!310 3184 3
TDS
S D
9. Calculation of Calcium Sulfate Supersaturation (Refer to Appendix X1)
9.1 Calculate calcium sulfate solubility using the same steps described for BaSO (Section 7), but substituting the appropriate
values for CaSO in Eq 2 (refer to Appendix X5).
9.2 Calculate the amount of calcium sulfate moles per
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...