ASTM D5391-23
(Test Method)Standard Test Method for Electrical Conductivity and Resistivity of a Flowing High Purity Water Sample
Standard Test Method for Electrical Conductivity and Resistivity of a Flowing High Purity Water Sample
SIGNIFICANCE AND USE
5.1 Conductivity measurements are typically made on samples of moderate to high ionic strength where contamination of open samples in routine laboratory handling is negligible. Under those conditions, standard temperature compensation using coefficients of 1 to 3 % of reading per degree Celsius over wide concentration ranges is appropriate. In contrast, this test method requires special considerations to reduce trace contamination and accommodates the high and variable temperature coefficients of pure water samples that can range as high as 7 % of reading per degree Celsius. In addition, measuring instrument design performance must be proven under high purity conditions.
5.2 This test method is applicable for detecting trace amounts of ionic contaminants in water. It is the primary means of monitoring the performance of demineralization and other high purity water treatment operations. It is also used to detect ionic contamination in boiler waters, microelectronics rinse waters, pharmaceutical process waters, etc., as well as to monitor and control the level of boiler and power plant cycle chemistry treatment chemicals. This test method supplements the basic measurement requirements for Test Methods D1125, D2186, and D4519.
5.3 At very low levels of alkaline contamination, for example, 0–1 μg/L NaOH, conductivity is suppressed, and can actually be slightly below the theoretical value for pure water. (1 and 2)4 Alkaline materials suppress the highly conductive hydrogen ion concentration while replacing it with less conductive sodium and hydroxide ions. This phenomenon is not an interference with conductivity or resistivity measurement itself but could give misleading indications of inferred water purity in this range if it is not recognized.
SCOPE
1.1 This test method covers the determination of electrical conductivity and resistivity of high purity water samples below 10 μS/cm (above 0.1 Mohm-cm). It is applicable to both continuous and periodic measurements but in all cases, the water must be flowing in order to provide representative sampling. Static grab sampling cannot be used for such high purity water. Continuous measurements are made directly in pure water process lines, or in side stream sample lines to enable measurements on high temperature or high pressure samples, or both.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Mar-2023
- Technical Committee
- D19 - Water
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 01-Aug-2018
- Effective Date
- 01-Aug-2018
- Effective Date
- 01-Jun-2016
- Effective Date
- 15-Jun-2012
- Effective Date
- 15-Jun-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-Jan-2008
- Effective Date
- 01-Dec-2007
- Effective Date
- 15-Dec-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 15-Aug-2006
Overview
ASTM D5391-23 is an international standard published by ASTM International, titled "Standard Test Method for Electrical Conductivity and Resistivity of a Flowing High Purity Water Sample." This method outlines procedures for accurately measuring the electrical conductivity and resistivity of high purity water samples below 10 μS/cm (above 0.1 Mohm-cm). These parameters are vital for monitoring minute levels of ionic contaminants in water, crucial to industries where high purity water is essential. The method specifically requires samples to be flowing, as static grab samples are unsuitable due to potential contamination, ensuring representative results for both continuous and periodic measurement scenarios.
Key Topics
- Measurement Scope: Targets high purity water with very low ionic content; applicable to samples below 10 μS/cm.
- Flowing Sample Requirement: Only applies to flowing samples; static or grab samples cannot provide reliable results at these purities.
- Significance: Conductivity and resistivity measurements are the primary indicators for identifying trace levels of ionic contamination.
- Special Considerations:
- High temperature coefficients of pure water can reach up to 7% per °C.
- Extreme care required to prevent trace contamination from sample handling, cell surfaces, and atmospheric exposure.
- Instrument design and temperature compensation must be validated for high purity conditions.
- Interference Factors:
- Atmospheric CO₂ can alter results; closed or in-line measurement is essential.
- Alkaline contamination (e.g., NaOH) at low levels can falsely indicate higher water purity due to ion exchange effects.
- Leadwire capacitance and cell design can introduce errors if not properly managed.
Applications
This standard offers practical value for monitoring high purity water systems in several key sectors, including:
- Power Generation: Essential for boiler feedwater and cycle chemistry control, ensuring the performance of demineralization processes and detection of ionic contamination which can cause corrosion or scaling.
- Microelectronics: Used to monitor rinse and process water, where even trace ions can impact product yield.
- Pharmaceuticals: Utilized for process water purity validation, helping ensure regulatory compliance.
- Industrial Water Treatment: Assesses demineralized water quality in settings requiring ultra-low ionic content.
- Process Control: Enables real-time, online verification of water purity in critical process lines.
- Reference for Compliance and Quality Control: Supplements other ASTM methods for water quality assessment and aligns with international best practices established through the World Trade Organization Technical Barriers to Trade (TBT) guidelines.
Related Standards
Several other ASTM standards, guides, and practices support or complement ASTM D5391-23 and may be referenced in the context of conductivity and resistivity measurement in water:
- ASTM D1125: Test Methods for Electrical Conductivity and Resistivity of Water
- ASTM D2186: Test Methods for Deposit-Forming Impurities in Steam
- ASTM D4519: Test Method for On-Line Determination of Anions and Carbon Dioxide in High Purity Water
- ASTM D1129: Terminology Relating to Water
- ASTM D1193: Specification for Reagent Water
- ASTM D1066, D1192, D2777, D3370, D3864: Additional guidance on sampling, equipment, precision, and on-line analytical systems for water quality assessment
Keywords: high purity water, electrical conductivity, water resistivity, monitoring ionic contamination, ASTM D5391-23, boiler water quality, power plant cycle chemistry, microelectronics water, pharmaceuticals water testing, continuous water sampling, water temperature compensation, water quality standards.
By adhering to this standard, organizations can ensure reliable, interference-free assessment of water purity, maintain stringent operational requirements, and uphold regulatory and industry quality standards.
Buy Documents
ASTM D5391-23 - Standard Test Method for Electrical Conductivity and Resistivity of a Flowing High Purity Water Sample
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D5391-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Electrical Conductivity and Resistivity of a Flowing High Purity Water Sample". This standard covers: SIGNIFICANCE AND USE 5.1 Conductivity measurements are typically made on samples of moderate to high ionic strength where contamination of open samples in routine laboratory handling is negligible. Under those conditions, standard temperature compensation using coefficients of 1 to 3 % of reading per degree Celsius over wide concentration ranges is appropriate. In contrast, this test method requires special considerations to reduce trace contamination and accommodates the high and variable temperature coefficients of pure water samples that can range as high as 7 % of reading per degree Celsius. In addition, measuring instrument design performance must be proven under high purity conditions. 5.2 This test method is applicable for detecting trace amounts of ionic contaminants in water. It is the primary means of monitoring the performance of demineralization and other high purity water treatment operations. It is also used to detect ionic contamination in boiler waters, microelectronics rinse waters, pharmaceutical process waters, etc., as well as to monitor and control the level of boiler and power plant cycle chemistry treatment chemicals. This test method supplements the basic measurement requirements for Test Methods D1125, D2186, and D4519. 5.3 At very low levels of alkaline contamination, for example, 0–1 μg/L NaOH, conductivity is suppressed, and can actually be slightly below the theoretical value for pure water. (1 and 2)4 Alkaline materials suppress the highly conductive hydrogen ion concentration while replacing it with less conductive sodium and hydroxide ions. This phenomenon is not an interference with conductivity or resistivity measurement itself but could give misleading indications of inferred water purity in this range if it is not recognized. SCOPE 1.1 This test method covers the determination of electrical conductivity and resistivity of high purity water samples below 10 μS/cm (above 0.1 Mohm-cm). It is applicable to both continuous and periodic measurements but in all cases, the water must be flowing in order to provide representative sampling. Static grab sampling cannot be used for such high purity water. Continuous measurements are made directly in pure water process lines, or in side stream sample lines to enable measurements on high temperature or high pressure samples, or both. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Conductivity measurements are typically made on samples of moderate to high ionic strength where contamination of open samples in routine laboratory handling is negligible. Under those conditions, standard temperature compensation using coefficients of 1 to 3 % of reading per degree Celsius over wide concentration ranges is appropriate. In contrast, this test method requires special considerations to reduce trace contamination and accommodates the high and variable temperature coefficients of pure water samples that can range as high as 7 % of reading per degree Celsius. In addition, measuring instrument design performance must be proven under high purity conditions. 5.2 This test method is applicable for detecting trace amounts of ionic contaminants in water. It is the primary means of monitoring the performance of demineralization and other high purity water treatment operations. It is also used to detect ionic contamination in boiler waters, microelectronics rinse waters, pharmaceutical process waters, etc., as well as to monitor and control the level of boiler and power plant cycle chemistry treatment chemicals. This test method supplements the basic measurement requirements for Test Methods D1125, D2186, and D4519. 5.3 At very low levels of alkaline contamination, for example, 0–1 μg/L NaOH, conductivity is suppressed, and can actually be slightly below the theoretical value for pure water. (1 and 2)4 Alkaline materials suppress the highly conductive hydrogen ion concentration while replacing it with less conductive sodium and hydroxide ions. This phenomenon is not an interference with conductivity or resistivity measurement itself but could give misleading indications of inferred water purity in this range if it is not recognized. SCOPE 1.1 This test method covers the determination of electrical conductivity and resistivity of high purity water samples below 10 μS/cm (above 0.1 Mohm-cm). It is applicable to both continuous and periodic measurements but in all cases, the water must be flowing in order to provide representative sampling. Static grab sampling cannot be used for such high purity water. Continuous measurements are made directly in pure water process lines, or in side stream sample lines to enable measurements on high temperature or high pressure samples, or both. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D5391-23 is classified under the following ICS (International Classification for Standards) categories: 13.060.60 - Examination of physical properties of water. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D5391-23 has the following relationships with other standards: It is inter standard links to ASTM D1129-13(2020)e2, ASTM D1066-18e1, ASTM D1066-18, ASTM D4519-16, ASTM D2777-12, ASTM D1066-11, ASTM D3370-10, ASTM D1129-10, ASTM D3370-08, ASTM D2777-08, ASTM D3370-07, ASTM D1066-06, ASTM D1129-06a, ASTM D1129-06ae1, ASTM D2777-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D5391-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D5391 − 23
Standard Test Method for
Electrical Conductivity and Resistivity of a Flowing High
Purity Water Sample
This standard is issued under the fixed designation D5391; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope tivity of Water (Withdrawn 2023)
D1129 Terminology Relating to Water
1.1 This test method covers the determination of electrical
D1192 Guide for Equipment for Sampling Water and Steam
conductivity and resistivity of high purity water samples below
in Closed Conduits (Withdrawn 2003)
10 μS/cm (above 0.1 Mohm-cm). It is applicable to both
D1193 Specification for Reagent Water
continuous and periodic measurements but in all cases, the
D2186 Test Methods for Deposit-Forming Impurities in
water must be flowing in order to provide representative
Steam (Withdrawn 2014)
sampling. Static grab sampling cannot be used for such high
D2777 Practice for Determination of Precision and Bias of
purity water. Continuous measurements are made directly in
Applicable Test Methods of Committee D19 on Water
pure water process lines, or in side stream sample lines to
D3370 Practices for Sampling Water from Flowing Process
enable measurements on high temperature or high pressure
Streams
samples, or both.
D3864 Guide for On-Line Monitoring Systems for Water
1.2 The values stated in SI units are to be regarded as
Analysis
standard. No other units of measurement are included in this
D4519 Test Method for On-Line Determination of Anions
standard.
and Carbon Dioxide in High Purity Water by Cation
Exchange and Degassed Cation Conductivity
1.3 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
3. Terminology
responsibility of the user of this standard to establish appro-
3.1 Definitions—For definitions of other terms used in these
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use. test methods, refer to Terminology D1129.
3.1.1 electrical conductivity—refer to Test Methods D1125.
1.4 This international standard was developed in accor-
dance with internationally recognized principles on standard-
3.1.2 electrical resistivity—refer to Test Methods D1125.
ization established in the Decision on Principles for the
3.2 Definitions of Terms Specific to This Standard:
Development of International Standards, Guides and Recom-
3.2.1 cell constant, n—the ratio of the length of the path, L
mendations issued by the World Trade Organization Technical
(cm), and the cross-sectional area of the solution, A (cm ),
Barriers to Trade (TBT) Committee.
between the electrodes of a conductivity/resistivity cell, with
−1
units of cm .
2. Referenced Documents
2 3.2.1.1 Discussion—In high purity water measurements, the
2.1 ASTM Standards:
−1
cell constant is normally between 0.001 and 0.1 cm to
D1066 Practice for Sampling Steam
−1
prevent electrical interference. This is lower than the 1 cm of
D1125 Test Methods for Electrical Conductivity and Resis-
the standard centimetre cube and is taken into account by direct
reading instrument ranges that are matched with specific cell
constants.
This test method is under the jurisdiction of ASTM Committee D19 on Water
and is the direct responsibility of Subcommittee D19.03 on Sampling Water and
4. Summary of Test Method
Water-Formed Deposits, Analysis of Water for Power Generation and Process Use,
On-Line Water Analysis, and Surveillance of Water. 4.1 Conductivity or resistivity is measured with a cell and
Current edition approved April 1, 2023. Published April 2023. Originally
temperature sensor or compensator in a flowing, closed system
approved in 1993. Last previous edition approved in 2014 as D5391 – 14 which
to prevent trace contamination from wetted surfaces and from
was withdrawn January 2023 and reinstated in April 2023. DOI: 10.1520/D5391-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on The last approved version of this historical standard is referenced on
the ASTM website. www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5391 − 23
the atmosphere. Specialized temperature compensation cor- but could give misleading indications of inferred water purity
rects the measurement to 25 °C, taking into account the in this range if it is not recognized.
temperature effects on the ionization of water, the
contaminants, and interactions between the two. In the absence 6. Interferences
of specialized temperature compensation, the sample tempera-
6.1 Exposure of the sample to the atmosphere may cause
ture is controlled to 25 6 0.2 °C.
changes in conductivity/resistivity due to loss or gain of
4.2 To determine the cell constant of a high purity conduc-
dissolved ionizable gases. Carbon dioxide, normally present in
tivity cell with an instrument capable of accurate measurement the air, can reach an equilibrium concentration in water of
over the range of pure water to 150 μS/cm with a single cell
about 1 mg/L and add approximately 1 μS/cm to the conduc-
constant, Test Methods D1125 are used directly. Manufactur- tivity due to formation of carbonic acid. Closed flow-through
ers’ certification of cell constant traceability by this means is an
or sealed in-line cell installation is required for this reason.
acceptable alternative.
6.2 Power plant installations utilizing long sample lines can
4.3 To determine the cell constant of a high purity conduc- experience significant sampling problems. New sample lines
tivity cell with an instrument which does not accurately cover
normally require longterm conditioning. Iron oxides and other
the range from pure water to 150 μS/cm with a single cell deposits accumulate in slow flowing horizontal sample lines
constant, a secondary standard cell is used that has an inter-
and can develop chromatograph-like retention of ionic species,
mediate cell constant with precise value determined by Test resulting in very long delay times. Precautions are described in
Methods D1125. That secondary standard cell is then used in
Section 9.
low conductivity water (not a standard) and readings are
6.3 Cell and flow chamber surfaces will slowly leach trace
compared with those of the low constant cell under test. In this
ionic contaminants, evidenced by increasing conductivity read-
manner, the cell constant of the latter is determined. Manufac-
ings with very low or zero flowrate. There must be sufficient
turers’ certification of cell constant traceability by this means is
flow to keep these contaminants from accumulating to the point
an acceptable alternative.
that they affect the measurement. The high and convoluted
surface area of platinized cells precludes their use for high
5. Significance and Use
purity measurements for this reason.
5.1 Conductivity measurements are typically made on
6.4 Capacitance of the cell and extension leadwire, espe-
samples of moderate to high ionic strength where contamina-
cially in high purity ranges can add significant positive error to
tion of open samples in routine laboratory handling is negli-
conductance readings (negative error to resistance readings).
gible. Under those conditions, standard temperature compen-
The measuring instrument must be designed to accommodate
sation using coefficients of 1 to 3 % of reading per degree
cell and leadwire characteristics in high purity water as
Celsius over wide concentration ranges is appropriate. In
described in 7.1.1 and Annex A1. In addition, the instrument
contrast, this test method requires special considerations to
manufacturers’ recommendations on cell leadwire must be
reduce trace contamination and accommodates the high and
carefully followed.
variable temperature coefficients of pure water samples that
6.5 Conductivity and resistivity measurements are refer-
can range as high as 7 % of reading per degree Celsius. In
addition, measuring instrument design performance must be enced to 25 °C. Either samples must be controlled to 25.0 6
0.2 °C or specialized temperature compensation must be em-
proven under high purity conditions.
ployed that accounts for the characteristics of high purity water
5.2 This test method is applicable for detecting trace
with specific contaminants, as described in 7.1.2.
amounts of ionic contaminants in water. It is the primary means
of monitoring the performance of demineralization and other 6.6 Samples containing dissolved gases must have sufficient
flow through the cell that bubbles cannot accumulate and
high purity water treatment operations. It is also used to detect
ionic contamination in boiler waters, microelectronics rinse occupy sample volume within the cell, causing low conductiv-
ity (high resistivity) readings. This problem is typical in
waters, pharmaceutical process waters, etc., as well as to
monitor and control the level of boiler and power plant cycle makeup water treatment systems where water warms up, drops
chemistry treatment chemicals. This test method supplements in pressure, and is acidified by cation exchange operations.
the basic measurement requirements for Test Methods D1125, This releases dissolved air and converts carbonates to carbon
D2186, and D4519. dioxide gas.
5.3 At very low levels of alkaline contamination, for 6.7 High purity conductivity measurement must not be
example, 0–1 μg/L NaOH, conductivity is suppressed, and can made on a sample downstream of pH sensors since they
actually be slightly below the theoretical value for pure water. invariably contaminate the sample with traces of reference
(1 and 2) Alkaline materials suppress the highly conductive electrolyte salts. Use a dedicated sample line or place the
hydrogen ion concentration while replacing it with less con- conductivity cell upstream from the pH sensors.
ductive sodium and hydroxide ions. This phenomenon is not an
6.8 Conductivity cells mounted downstream from ion ex-
interference with conductivity or resistivity measurement itself
changers are vulnerable to catching resin particles between the
cell electrodes. Resin particles are sufficiently conductive to
short the cell and cause high off-scale conductivity or ex-
The boldface numbers in parentheses refer to a list of references at the end of
this standard. tremely low resistivity readings. Resin retainers must be
D5391 − 23
effective and cells must be accessible for cleaning. Cell designs
with electrode spacing greater than 1.5 mm have been found to
be less likely to trap such particles.
6.9 Conductivity cells, if subjected to demineralizer regen-
eration reagents, would require excessive rinse time to obtain
satisfactory results. Therefore, locate cells where they will be
isolated during regeneration cycles.
7. Apparatus
7.1 Measuring Instrument:
7.1.1 The instrument shall be continuously reading in either
conductivity or resistivity units. It shall be specifically de-
signed to measure in high purity ranges, measuring with
alternating current (ac) of appropriate voltage, frequency, wave
shape, phase correction, and wave sampling technique to
minimize errors due to parallel and series capacitance of cell
and leadwire as well as minimizing electrode polarization
errors and effects of small direct current (dc) potentials. A cell
simulation technique to verify the performance of an unproven
measuring circuit design under high purity conditions is
provided in Annex A1.
7.1.2 The measurement shall include algorithms to tempera-
ture compensate conductivity or resistivity values to 25.0 °C.
The algorithm shall compensate for changes in water ionization
as well as for solute ion mobility for neutral salt contaminants.
The conductivity of pure water has been documented with high
accuracy (3-5).
7.1.3 In the case of samples containing acidic or basic
solutes (such as power plant treatment using ammonia,
morpholine, etc., or acidic cation conductivity samples or
microelectronics acid etch rinse monitoring), special algo-
rithms shall be employed that account for the interaction of
acids and bases with the ionization of water (6-8). The user is
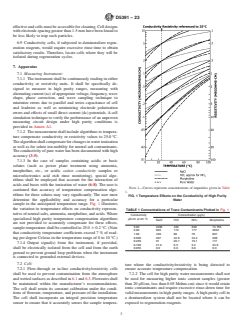
NOTE 1—Curves represent concentrations of impurities given in Table
cautioned that accuracy of temperature compensation algo-
1.
rithms for these solutes may vary significantly. The user must
FIG. 1 Temperature Effects on the Conductivity of High Purity
determine the applicability and accuracy for a particular
Water
sample in the anticipated temperature range. Fig. 1 illustrates
the variation in temperature effects on conductivity represen-
TABLE 1 Concentrations of Trace Contaminants Plotted in Fig. 1
tative of neutral salts, ammonia, morpholine, and acids. Where
Conductivity Concentration (μg/L)
μS/cm at 25 °C
specialized high purity temperature compensation algorithms
NaCl HCl NH Morpholine
are not provided to accurately compensate for these effects,
5.00 2295 430 638 13 755
sample temperature shall be controlled to 25.0 6 0.2 °C. (Note
2.00 903 172 177 2632
that conductivity temperature coefficients exceed 7 % of read-
1.00 439 86 76 837
0.500 207 42.6 34.4 298
ing per degree Celsius in the temperature range of 0 to 10 °C.)
0.250 91 20.7 16.1 117
7.1.4 Output signal(s) from the instrument, if provided,
0.100 21.0 6.5 5.4 34.6
shall be electrically isolated from the cell and from the earth
0.055 0.0 0.0 0.0 0.0
ground to prevent ground loop problems when the instrument
is connected to grounded external devices.
7.2 Cell:
ture where the conductivity/resistivity is being detected to
7.2.1 Flow-through or in-line conductivity/resistivity cells ensure accurate temperature compensation.
shall be used to prevent contamination from the atmosphere 7.2.2 The cell for high purity water measurements shall not
and wetted surfaces as described in 6.1 and 6.3. Flowrates shall be used for measuring higher ionic content samples (greater
be maintained within the manufacturer’s recommendations. than 20 μS/cm, less than 0.05 Mohm-cm) since it would retain
The cell shall retain its constant calibration under the condi- ionic contaminants and require excessive rinse-down time for
tions of flowrate, temperature, and pressure of the installation. valid measurements in high purity ranges. A high purity cell in
The cell shall incorporate an integral precision temperature a demineralizer system shall not be located where it can be
sensor to ensure that it accurately senses the sample tempera- exposed to regeneration reagents.
D5391 − 23
7.2.3 Electrodes of the cell shall not be platinized for pure or dead legs must be avoided to ensure a representative sample
water measurements since the microscopically rough, porous and prevent any bubbles from adhering to cell surfaces.
surface would retain ionic contaminants and produce excessive
9.2 Design and operate side stream sample lines to maintain
downscale response times. Only a trace or flash of platinum
sample integrity.
black is permissible on electrode surfaces. Electrodes of
9.2.1 Do not expose the sample to the atmosphere to prevent
titanium, nickel, monel, stainless steel, or platinum are suitable
absorption or loss of gases, particularly carbon dioxide, that
for high purity measurement. However, extra care must be
affect conductivity.
taken using platinum cells not to exceed manufacturers’
9.2.2 The sample line shall not have pH electrodes upstream
recommended flowrate and not to permit rough handling that
of the conductivity cell as noted in 6.7.
could bend the electrodes and change the cell constant.
9.2.3 In power plant installations where iron oxide and other
7.2.4 If the cell constant as checked does not fall within
solids occur in pure water samples, flowrates must be con-
acceptable limits of its nominal value, it is necessary to clean
trolled to minimize accumulation of deposits that could greatly
or replace the cell. Even in pure water samples, coatings such
delay transport of ionic materials. A sample flow velocity of 2
as iron oxide crud in power plant installations, resin fines, and
m/s in horizontal sample lines has been found optimum.
other solids and films can develop. Insulating coatings over
Additional sample line design criteria have been documented
electrode surfaces can cause negative conductivity errors.
(9-11).
Conductive accumulations between electrodes can short them
9.2.4 The sample shall be continuously flowing at a stable
and cause positive errors. Mechanical cleaning must not be
flowrate to enable sample line wetted surfaces to reach
attempted with platinum electrodes since this would change the
equilibrium with sample conditions. Do not make measure-
cell constant. Cleaning must either follow manufacturer’s
ments following changes to the sample flowrate for the period
recommendations or that given in Test Methods D1125. Ultra-
of time required to recover from transient effects on the
sonic cleaners have also been found effective in some cases.
particular sampling system.
7.3 Extension leadwire type, size, and length shall be
9.2.5 Control the sample temperature within the capabilities
according to manufacturers’ recommendations, primarily to
of the temperature compensation algorithms used in the mea-
prevent parallel leadwire capacitance errors with this high
suring instrument. The sample temperature shall be sufficiently
resistance ac measurement. In particular, the length shall not
stable to maintain reasonable equilibrium with the sample line
exceed the measuring instrument’s ability to minimize capaci-
wetted surfaces.
tance errors.
9.2.6 Do not locate the cell where it would be exposed to
demineralizer reagents as noted in 6.9.
8. Reagents
9.2.7 Additional sampling precautions are provided in Prac-
8.1 Purity of Reagents—Reagent grade chemicals shall be
tices D1066 and D3370 and Guides D1192 and D3864.
used in all tests. Unless otherwise indicated, it is intende
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...