ASTM D6731-01(2011)
(Test Method)Standard Test Method for Determining the Aerobic, Aquatic Biodegradability of Lubricants or Lubricant Components in a Closed Respirometer
Standard Test Method for Determining the Aerobic, Aquatic Biodegradability of Lubricants or Lubricant Components in a Closed Respirometer
SIGNIFICANCE AND USE
Results from this test method suggest the degree of aerobic, aquatic biodegradation of a lubricant or lubricant component. The rate and extent of oxygen consumption is measured upon exposure of the test material to an inoculum within the confines of a controlled laboratory setting. Test materials which achieve a high degree of biodegradation in this test may be assumed to easily biodegrade in many aerobic aquatic environments.
Because of the stringency of this test method, low results do not necessarily mean that the test material is not biodegradable under environmental conditions, but indicate that further testing is necessary to establish biodegradability.
If the pH value at the end of the test is outside the range from 6 to 8 and if the percentage degradation of the test material is less than 50 %, it is advisable to repeat the test with a lower concentration of the test material or a higher concentration of the buffer solution, or both.
A reference or control material known to biodegrade under the conditions of this test method is necessary in order to verify the activity of the inoculum. The test must be regarded as invalid and shall be repeated using a fresh inoculum if the reference material does not demonstrate biodegradation to the extent of >60 % of the ThO2 within 28 days.
Information on the toxicity of the test material to the inoculum may be useful in the interpretation of low biodegradation results. Toxicity of the test material to the inoculum may be evaluated by testing the test material in combination with the reference material in inhibition control systems. If an inhibition control is included, the test material is assumed to be inhibiting if the degradation percentage of the reference material is lower than 40 % (ISO 8192:1986). In this case, it is advisable to repeat the test with lower concentrations of the test material.
Total oxygen utilization in the blank at the end of the test exceeding 60 mg O2/L invalidates the test.
The wat...
SCOPE
1.1 This test method covers a procedure for determining the degree of biodegradability of lubricants or their components in an aerobic aqueous medium on exposure to an inoculum under controlled laboratory conditions. This test method is an ultimate biodegradation test that measures oxygen demand in a closed respirometer.
1.2 This test method is suitable for evaluating the biodegradation of volatile as well as nonvolatile lubricants or lubricant components.
1.3 This test method is applicable to lubricants and lubricant components which are not toxic and not inhibitory to the test microorganisms at the test concentration.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazards are given in Section 10.
General Information
Relations
Standards Content (Sample)
NOTICE: This standard has either been superseded and replaced by a new version or withdrawn.
Contact ASTM International (www.astm.org) for the latest information
Designation: D6731 − 01 (Reapproved 2011)
Standard Test Method for
Determining the Aerobic, Aquatic Biodegradability of
Lubricants or Lubricant Components in a Closed
Respirometer
This standard is issued under the fixed designation D6731; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope D4447 Guide for Disposal of Laboratory Chemicals and
Samples
1.1 This test method covers a procedure for determining the
D6384 Terminology Relating to Biodegradability and Eco-
degree of biodegradability of lubricants or their components in
toxicity of Lubricants
an aerobic aqueous medium on exposure to an inoculum under
E943 Terminology Relating to Biological Effects and Envi-
controlled laboratory conditions. This test method is an ulti-
ronmental Fate
mate biodegradation test that measures oxygen demand in a
2.2 ISO Standards:
closed respirometer.
ISO 4259:1992(E) Petroleum Products–Determination and
1.2 This test method is suitable for evaluating the biodeg-
Application of Precision Data in Relation to Methods of
radation of volatile as well as nonvolatile lubricants or lubri-
Test
cant components.
ISO 6107-2:1997 Water Quality–Vocabulary–Part 2
1.3 Thistestmethodisapplicabletolubricantsandlubricant ISO 8192:1986 Water Quality–Test for Inhibition of Oxygen
components which are not toxic and not inhibitory to the test
Consumption by Activated Sludge
microorganisms at the test concentration. ISO 9408:1999 Water Quality–Evaluation of UltimateAero-
bic Biodegradability of Organic Compounds in Aqueous
1.4 The values stated in SI units are to be regarded as
Medium by Determination of Oxygen Demand in a
standard. No other units of measurement are included in this
Closed Respirometer
standard.
2.3 OECD Standards:
1.5 This standard does not purport to address all of the
OECD 301F:1992 Ready Biodegradability-Manometric Re-
safety concerns, if any, associated with its use. It is the
spirometry
responsibility of the user of this standard to establish appro-
2.4 APHA Standards:
priate safety and health practices and determine the applica-
2540B Total Solids Dried at 103-105°C
bilityofregulatorylimitationspriortouse.Specifichazardsare
9215 Heterotrophic Plate Count
given in Section 10.
3. Terminology
2. Referenced Documents
2 3.1 Definitions:
2.1 ASTM Standards:
3.1.1 Definitions of terms applicable to this test method
D1129 Terminology Relating to Water
appear in the Compilation of ASTM Standard Definitions and
D1193 Specification for Reagent Water
the following terminology standards: D1129, D4175, D6384,
D1293 Test Methods for pH of Water
E943, and ISO 6107-2:1997.
D4175 Terminology Relating to Petroleum, Petroleum
3.1.2 activated sludge, n—the precipitated solid matter,
Products, and Lubricants
consisting mainly of bacteria and other aquatic
microorganisms, that is produced at a domestic wastewater
This test method is under the jurisdiction of ASTM Committee D02 on
Petroleum Products, Liquid Fuels, and Lubricantsand is the direct responsibility of
Subcommittee D02.12 on Environmental Standards for Lubricants. Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
Current edition approved May 1, 2011. Published May 2011. Originally 4th Floor, New York, NY 10036, http://www.ansi.org.
approved in 2001. Last previous edition approved in 2005 as D6731–01 (2005). Available from Organisation for Economic Cooperation and Development
DOI: 10.1520/D6731-01R11. (OECD), 2 rue André Pascal, F-75775, Paris Cedex 16, France, http://
For referenced ASTM standards, visit the ASTM website, www.astm.org, or www.oecd.org.
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM From Standard Methods for the Examination of Water and Wastewater, latest
Standards volume information, refer to the standard’s Document Summary page on edition. Available from the American Public Health Assoc., 1015 18th St., NW,
the ASTM website. Washington, DC 20036.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6731 − 01 (2011)
treatment plant and is used primarily in secondary sewage defined aquatic medium and measures the biodegradation of
treatmenttomicrobiallyoxidizedissolvedorganicmatterinthe the test material by following the decrease in oxygen in the
effluent. respirometer.
3.1.3 aerobic, adj—(a) taking place in the presence of
4.2 The test material is the sole source of carbon and energy
oxygen; (b) living or active in the presence of oxygen.
inthemedium.Areferencematerialknowntobiodegrade,such
as low erucic acid rapeseed oil (LEAR or canola oil) is run
3.1.4 biochemical oxygen demand (BOD), n—the mass
alongside the test material to confirm that the inoculum is
concentration of dissolved oxygen consumed under specified
viableandcapableofbiodegradingsuitablematerialsunderthe
conditions by the biological oxidation of organic or inorganic
test conditions. The test material or reference material concen-
matter, or both.
tration is normally 50 to 100 mg/L, providing a theoretical
3.1.4.1 Discussion—BOD determination is performed using
oxygen demand of at least 50 mg O /L but no more than 200
empirical tests employing standardized laboratory procedures.
mg O /L. The ThO of the test and reference materials will be
These tests measure oxygen utilization during a specified 2 2
determined from measured elemental compositional analysis
incubation period for the biochemical degradation of organic
and will be calculated as in 13.1.
material (carbonaceous demand) in water.
3.1.5 biodegradation, n—the process of chemical break- 4.3 The inoculated medium is stirred in a closed flask and
down or transformation of a test material caused by microor- the consumption of oxygen is determined either by measuring
ganisms or their enzymes. the amount of oxygen required to maintain a constant gas
3.1.5.1 Discussion—Biodegradation is only one mechanism volume in the respirometer flask, or by measuring the change
by which materials are removed, transformed, or both, in the in volume or pressure (or a combination of the two) in the
environment. apparatus.
3.1.6 lag phase, n—the period of diminished physiological
4.4 EvolvedCO (carbondioxide)isabsorbedinanalkaline
activity and cell division following the addition of microorgan-
trap solution (for example, 10 M NaOH or KOH) or other
isms to a new culture medium.
CO -absorbing system suspended within the test vessel, typi-
cally in the headspace of the test vessel.
3.1.7 log phase, n—the period of growth of microorganisms
during which cells divide at a positive constant rate.
4.5 Biodegradation is followed over a specified period by
3.1.8 pre-adaptation, n—the incubation of an inoculum in determining the consumption of oxygen. The amount of
the presence of the test material which is done prior to the oxygen utilized in oxidation of the test and reference material
initiation of the test and under conditions similar to the test is corrected for oxygen uptake by the inoculum in the blank
conditions. controls and is expressed as a percentage of the theoretical
oxygen demand (ThO ) calculated from the empirical formula
3.1.8.1 Discussion—The aim of pre-adaptation is to im-
prove the precision of the test method by decreasing variability of the material. Evaluation of the biodegradability of the test
material is made on the basis of these data. Normally the test
in the rate of biodegradation produced by the inoculum.
Pre-adaptation may mimic the natural processes which cause duration is 28 days; however, the test may be terminated if
oxygen consumption has plateaued. The test may be extended
changes in the microbial population of the inoculum leading to
more rapid biodegradation of the test material but is not as long as the systems’ integrity is maintained and the
inoculum in the blank systems is viable. The duration of the
expected to change the overall extent of biodegradation of the
test material. testwillbedependentonthelengthoftimerequiredfortherate
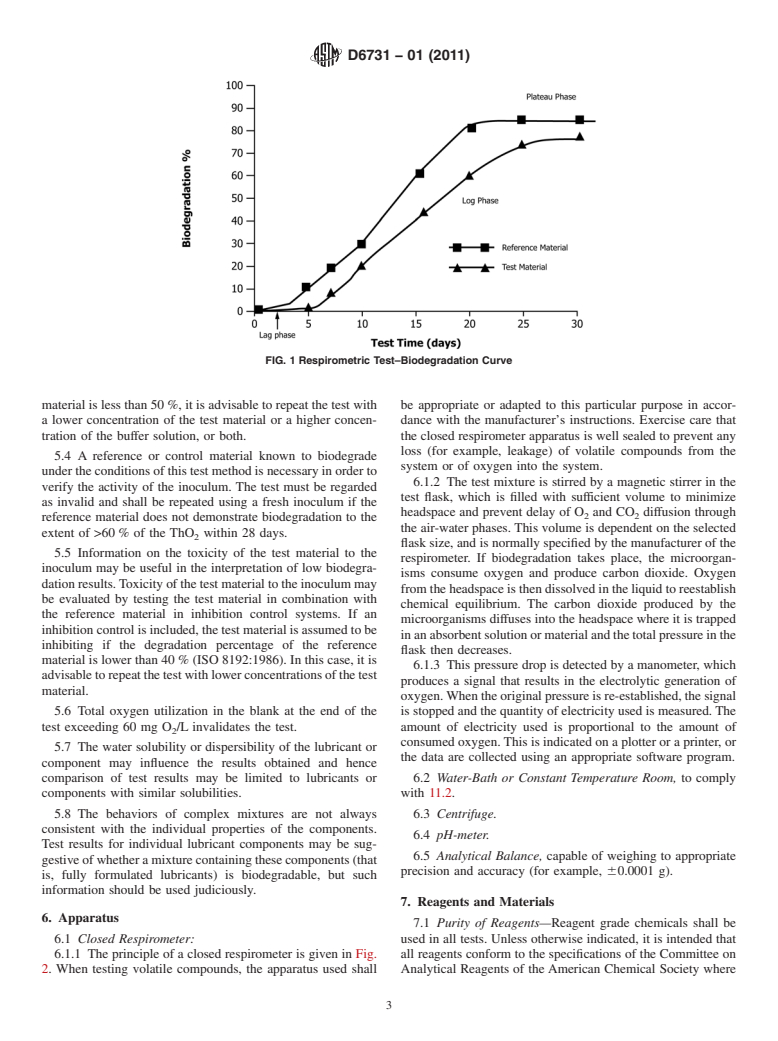
of test material biodegradation to achieve a plateau. A graphi-
3.1.9 pre-condition, n—the pre-incubation of an inoculum
cal illustration of the test results for a biodegradable material is
under the conditions of the test in the absence of the test
presented in Fig. 1.
material.
3.1.10 sludge, n—a water-formed sedimentary deposit.
5. Significance and Use
3.1.11 suspended solids (of an activated sludge or other
5.1 Results from this test method suggest the degree of
inoculum samples), n—solids present in activated sludge or
aerobic, aquatic biodegradation of a lubricant or lubricant
other inoculum samples that are not removed by settling under
component. The rate and extent of oxygen consumption is
specified conditions.
measured upon exposure of the test material to an inoculum
within the confines of a controlled laboratory setting. Test
4. Summary of Test Method
materialswhichachieveahighdegreeofbiodegradationinthis
4.1 Biodegradation of a lubricant or the component(s) of a
test may be assumed to easily biodegrade in many aerobic
lubricant is determined by measuring the oxygen consumed
aquatic environments.
whenthelubricantorcomponentisexposedtomicroorganisms
5.2 Because of the stringency of this test method, low
under controlled aerobic aquatic conditions. This value is then
results do not necessarily mean that the test material is not
compared to the theoretical amount of oxygen (ThO ) which is
biodegradable under environmental conditions, but indicate
required to oxidize all of the elements (that is, carbon,
that further testing is necessary to establish biodegradability.
hydrogen, nitrogen, and so forth) in the test material. This test
method mixes the test material (lubricant or component) with 5.3 If the pH value at the end of the test is outside the range
aerobic microorganisms in a closed respirometer containing a from 6 to 8 and if the percentage degradation of the test
D6731 − 01 (2011)
FIG. 1 Respirometric Test–Biodegradation Curve
material is less than 50 %, it is advisable to repeat the test with be appropriate or adapted to this particular purpose in accor-
a lower concentration of the test material or a higher concen- dance with the manufacturer’s instructions. Exercise care that
tration of the buffer solution, or both. the closed respirometer apparatus is well sealed to prevent any
loss (for example, leakage) of volatile compounds from the
5.4 A reference or control material known to biodegrade
system or of oxygen into the system.
under the conditions of this test method is necessary in order to
6.1.2 The test mixture is stirred by a magnetic stirrer in the
verify the activity of the inoculum. The test must be regarded
test flask, which is filled with sufficient volume to minimize
as invalid and shall be repeated using a fresh inoculum if the
headspace and prevent delay of O and CO diffusion through
2 2
reference material does not demonstrate biodegradation to the
the air-water phases. This volume is dependent on the selected
extent of >60 % of the ThO within 28 days.
flask size, and is normally specified by the manufacturer of the
5.5 Information on the toxicity of the test material to the
respirometer. If biodegradation takes place, the microorgan-
inoculum may be useful in the interpretation of low biodegra-
isms consume oxygen and produce carbon dioxide. Oxygen
dationresults.Toxicityofthetestmaterialtotheinoculummay
fromtheheadspaceisthendissolvedintheliquidtoreestablish
be evaluated by testing the test material in combination with
chemical equilibrium. The carbon dioxide produced by the
the reference material in inhibition control systems. If an
microorganisms diffuses into the headspace where it is trapped
inhibition control is included, the test material is assumed to be
inanabsorbentsolutionormaterialandthetotalpressureinthe
inhibiting if the degradation percentage of the reference
flask then decreases.
material is lower than 40 % (ISO 8192:1986). In this case, it is
6.1.3 This pressure drop is detected by a manometer, which
advisabletorepeatthetestwithlowerconcentrationsofthetest
produces a signal that results in the electrolytic generation of
material.
oxygen.Whentheoriginalpressureisre-established,thesignal
5.6 Total oxygen utilization in the blank at the end of the is stopped and the quantity of electricity used is measured.The
test exceeding 60 mg O /L invalidates the test. amount of electricity used is proportional to the amount of
consumed oxygen.This is indicated on a plotter or a printer, or
5.7 The water solubility or dispersibility of the lubricant or
the data are collected using an appropriate software program.
component may influence the results obtained and hence
comparison of test results may be limited to lubricants or 6.2 Water-Bath or Constant Temperature Room, to comply
components with similar solubilities. with 11.2.
5.8 The behaviors of complex mixtures are not always
6.3 Centrifuge.
consistent with the individual properties of the components.
6.4 pH-meter.
Test results for individual lubricant components may be sug-
6.5 Analytical Balance, capable of weighing to appropriate
gestiveofwhetheramixturecontainingthesecomponents(that
precision and accuracy (for example, 60.0001 g).
is, fully formulated lubricants) is biodegradable, but such
information should be used judiciously.
7. Reagents and Materials
6. Apparatus
7.1 Purity of Reagents—Reagent grade chemicals shall be
6.1 Closed Respirometer: used in all tests. Unless otherwise indicated, it is intended that
6.1.1 The principle of a closed respirometer is given in Fig. all reagents conform to the specifications of the Committee on
2. When testing volatile compounds, the apparatus used shall Analytical Reagents of the American Chemical Society where
D6731 − 01 (2011)
FIG. 2 Principle of a Closed Respirometer
such specifications are available. Other grades may be used, (Na HPO ·2H O), and 0.5 g ammonium chloride (NH Cl) in
2 4 2 4
provided it is first ascertained that the reagent is of sufficiently
water and dilute to 1 L. Alternatively, 50.3 g of disodium
high purity to permit its use without decreasing the accuracy of
hydrogen phosphate, heptahydrate (Na HPO ·7H O) may be
2 4 2
the determination.
used in place of Na HPO ·2H O. The pH of this solution shall
2 4 2
be about 7.4.
7.2 Purity of Water—Unless otherwise indicated, references
to water shall be understood to mean reagent water as defined
8. Ino
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.