ASTM D5134-21
(Test Method)Standard Test Method for Detailed Analysis of Petroleum Naphthas through n-Nonane by Capillary Gas Chromatography
Standard Test Method for Detailed Analysis of Petroleum Naphthas through n-Nonane by Capillary Gas Chromatography
SIGNIFICANCE AND USE
5.1 A knowledge of the hydrocarbon components comprising a petroleum naphtha, reformate, or alkylate is useful in valuation of crude oils, in alkylation and reforming process control, in product quality assessment, and for regulatory purposes. Detailed hydrocarbon composition is also used as input in the mathematical modeling of refinery processes.
5.2 Separation of naphtha components by the procedure described in this test method can result in some peaks that represent coeluting compounds. This test method cannot attribute relative concentrations to the coelutants. In the absence of supporting information, use of the results of this test method for purposes which require such attribution is not recommended.
SCOPE
1.1 This detailed hydrocarbon analysis (DHA) test method covers the determination of hydrocarbon components paraffins, naphthenes, and monoaromatics (PNA) of petroleum naphthas as enumerated in Table 1. Components eluting after n-nonane (bp 150.8 °C) are determined as a single group.
1.2 This test method is applicable to olefin-free (D1319 or D6839. The hydrocarbon mixture must have a 98 % point of 250 °C or less as determined by Test Method D3710 or D7096 or equivalent.
1.3 Components that are present at the 0.05 % by mass level or greater can be determined.
1.4 This test method may not be completely accurate for PNA above carbon number C7; Test Method D5443 or D6839 may be used to verify or complement the results of this test method for carbon numbers >C7.
1.5 Detailed hydrocarbon components in olefin containing samples may be determined by DHA Test Methods D6729, D6730, or D6733.
1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific warning statements are given in Section 8.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Nov-2021
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.04.0L - Gas Chromatography Methods
Relations
- Refers
ASTM D4175-23a - Standard Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants - Effective Date
- 15-Dec-2023
- Refers
ASTM D4175-23e1 - Standard Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants - Effective Date
- 01-Jul-2023
- Effective Date
- 01-Sep-2019
- Effective Date
- 20-Apr-2016
- Effective Date
- 01-Dec-2015
- Effective Date
- 01-Oct-2014
- Effective Date
- 01-Oct-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Nov-2012
- Effective Date
- 01-Nov-2011
- Refers
ASTM D4057-06(2011) - Standard Practice for Manual Sampling of Petroleum and Petroleum Products - Effective Date
- 01-Jun-2011
- Effective Date
- 01-May-2011
- Effective Date
- 15-Feb-2010
- Effective Date
- 15-Apr-2009
- Effective Date
- 15-Oct-2008
Overview
ASTM D5134-21: Standard Test Method for Detailed Analysis of Petroleum Naphthas through n-Nonane by Capillary Gas Chromatography provides a standardized approach for determining the detailed hydrocarbon composition of petroleum naphthas, reformates, and alkylates. Using capillary gas chromatography, this method identifies and quantifies paraffins, naphthenes, and monoaromatics (PNA) up to n-nonane, enabling improved process control, quality assessment, and regulatory compliance in petroleum refining and product valuation.
Key Topics
- Hydrocarbon Component Analysis: The method separates, identifies, and quantifies hydrocarbons in petroleum naphthas, focusing on paraffins, naphthenes, and monoaromatics eluting up to n-nonane. Components after n-nonane are reported as a grouped sum.
- Capillary Gas Chromatography: Utilizes a highly specified capillary column and flame ionization detection to deliver reproducible retention times and resolution, minimizing interpretive errors and ensuring result comparability.
- Method Applicability: Designed for olefin-free liquid hydrocarbon mixtures, including virgin naphthas, reformates, and alkylates with a 98% point of 250°C or less. For samples containing olefins or those with higher carbon numbers, alternative or complementary methods are recommended.
- Quantitative and Qualitative Data: Determines components present at 0.05% mass or higher, providing both mass percent and identity. Coeluting compounds may present as single peaks; this method does not differentiate their individual concentrations.
- Precision and Limitations: Offers clear guidelines on repeatability and reproducibility for selected representative naphtha components. Strict adherence to method conditions is critical for accurate peak identification and quantification.
- Safety and Regulatory Compliance: Includes recommendations for safe handling, analysis, and environmental considerations associated with hydrocarbon volatile materials.
Applications
- Refinery Process Control: Supports process optimization in alkylation and reforming by providing detailed hydrocarbon composition data.
- Product Quality Assessment: Essential for evaluating and certifying gasoline blending streams, ensuring compliance with specifications and performance requirements.
- Valuation of Crude Oils and Feedstocks: Detailed hydrocarbon profiles enable more accurate valuation and selection of feedstocks for refining and petrochemical processing.
- Regulatory Reporting: Facilitates reporting of detailed hydrocarbon data for environmental and regulatory submission, supporting compliance with both national and international standards.
- Refinery Modeling: Supplies key input data for mathematical modeling of refinery processes, improving yield predictions and process efficiency.
Related Standards
Several ASTM standards complement or support ASTM D5134-21:
- ASTM D1319: Hydrocarbon Types in Liquid Petroleum Products by Fluorescent Indicator Adsorption
- ASTM D3710, D7096: Boiling Range Distribution of Gasoline and Gasoline Fractions by Gas Chromatography
- ASTM D5443, D6839: Multi-dimensional Gas Chromatography Methods for Hydrocarbon Analysis
- ASTM D6729, D6730, D6733: Determination of Individual Components in Spark Ignition Engine Fuels by Capillary High-Resolution Gas Chromatography
- ASTM D4057, D3700: Practices for Sampling Petroleum and Petroleum Products
- ASTM E355, E594: Gas Chromatography Terminology, Flame Ionization Detector Testing
Practical Value
ASTM D5134-21 equips laboratories, refineries, and regulatory agencies with a clear, consistent methodology for hydrocarbon analysis in petroleum naphthas, supporting critical activities such as process control, product quality, compliance, and technical reporting. By standardizing column specifications and operating conditions, the method ensures consistent, reliable hydrocarbon data, essential for fuel blending, process optimization, and meeting environmental standards. Its precise, reproducible protocols make it a cornerstone of modern petroleum laboratory analysis.
Buy Documents
ASTM D5134-21 - Standard Test Method for Detailed Analysis of Petroleum Naphthas through n-Nonane by Capillary Gas Chromatography
REDLINE ASTM D5134-21 - Standard Test Method for Detailed Analysis of Petroleum Naphthas through n-Nonane by Capillary Gas Chromatography
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D5134-21 is a standard published by ASTM International. Its full title is "Standard Test Method for Detailed Analysis of Petroleum Naphthas through n-Nonane by Capillary Gas Chromatography". This standard covers: SIGNIFICANCE AND USE 5.1 A knowledge of the hydrocarbon components comprising a petroleum naphtha, reformate, or alkylate is useful in valuation of crude oils, in alkylation and reforming process control, in product quality assessment, and for regulatory purposes. Detailed hydrocarbon composition is also used as input in the mathematical modeling of refinery processes. 5.2 Separation of naphtha components by the procedure described in this test method can result in some peaks that represent coeluting compounds. This test method cannot attribute relative concentrations to the coelutants. In the absence of supporting information, use of the results of this test method for purposes which require such attribution is not recommended. SCOPE 1.1 This detailed hydrocarbon analysis (DHA) test method covers the determination of hydrocarbon components paraffins, naphthenes, and monoaromatics (PNA) of petroleum naphthas as enumerated in Table 1. Components eluting after n-nonane (bp 150.8 °C) are determined as a single group. 1.2 This test method is applicable to olefin-free (D1319 or D6839. The hydrocarbon mixture must have a 98 % point of 250 °C or less as determined by Test Method D3710 or D7096 or equivalent. 1.3 Components that are present at the 0.05 % by mass level or greater can be determined. 1.4 This test method may not be completely accurate for PNA above carbon number C7; Test Method D5443 or D6839 may be used to verify or complement the results of this test method for carbon numbers >C7. 1.5 Detailed hydrocarbon components in olefin containing samples may be determined by DHA Test Methods D6729, D6730, or D6733. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific warning statements are given in Section 8. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 A knowledge of the hydrocarbon components comprising a petroleum naphtha, reformate, or alkylate is useful in valuation of crude oils, in alkylation and reforming process control, in product quality assessment, and for regulatory purposes. Detailed hydrocarbon composition is also used as input in the mathematical modeling of refinery processes. 5.2 Separation of naphtha components by the procedure described in this test method can result in some peaks that represent coeluting compounds. This test method cannot attribute relative concentrations to the coelutants. In the absence of supporting information, use of the results of this test method for purposes which require such attribution is not recommended. SCOPE 1.1 This detailed hydrocarbon analysis (DHA) test method covers the determination of hydrocarbon components paraffins, naphthenes, and monoaromatics (PNA) of petroleum naphthas as enumerated in Table 1. Components eluting after n-nonane (bp 150.8 °C) are determined as a single group. 1.2 This test method is applicable to olefin-free (D1319 or D6839. The hydrocarbon mixture must have a 98 % point of 250 °C or less as determined by Test Method D3710 or D7096 or equivalent. 1.3 Components that are present at the 0.05 % by mass level or greater can be determined. 1.4 This test method may not be completely accurate for PNA above carbon number C7; Test Method D5443 or D6839 may be used to verify or complement the results of this test method for carbon numbers >C7. 1.5 Detailed hydrocarbon components in olefin containing samples may be determined by DHA Test Methods D6729, D6730, or D6733. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific warning statements are given in Section 8. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D5134-21 is classified under the following ICS (International Classification for Standards) categories: 71.080.10 - Aliphatic hydrocarbons. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D5134-21 has the following relationships with other standards: It is inter standard links to ASTM D4175-23a, ASTM D4175-23e1, ASTM E594-96(2019), ASTM D6839-16, ASTM D6839-15, ASTM D1319-14, ASTM D6839-13, ASTM D1319-13, ASTM D3700-12, ASTM E594-96(2011), ASTM D4057-06(2011), ASTM D6730-01(2011), ASTM D7096-10, ASTM D6729-04(2009), ASTM D1319-08. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D5134-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D5134 − 21
Standard Test Method for
Detailed Analysis of Petroleum Naphthas through n-Nonane
by Capillary Gas Chromatography
This standard is issued under the fixed designation D5134; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
INTRODUCTION
Despite the many advances in capillary gas chromatography instrumentation and the remarkable
resolution achievable, it has proven difficult to standardize a test method for the analysis of a mixture
as complex as petroleum naphtha. Because of the proliferation of numerous, similar columns and the
endless choices of phase thickness, column internal diameter, length, etc., as well as instrument
operating parameters, many laboratories use similar but not identical methods for the capillary GC
analysis of petroleum naphthas. Even minute differences in column polarity or column oven
temperature, for example, can change resolution or elution order of components and make their
identification an individual interpretive process rather than the desirable, objective application of
standard retention data. To avoid this, stringent column specifications and temperature and flow
conditions have been adopted in this test method to ensure consistent elution order and resolution and
reproducible retention times. Strict adherence to the specified conditions is essential to the successful
application of this test method.
1. Scope* 1.5 Detailed hydrocarbon components in olefin containing
samples may be determined by DHA Test Methods D6729,
1.1 This detailed hydrocarbon analysis (DHA) test method
D6730,or D6733.
coversthedeterminationofhydrocarboncomponentsparaffins,
1.6 The values stated in SI units are to be regarded as
naphthenes, and monoaromatics (PNA) of petroleum naphthas
standard. No other units of measurement are included in this
as enumerated in Table 1. Components eluting after n-nonane
standard.
(bp 150.8°C) are determined as a single group.
1.7 This standard does not purport to address all of the
1.2 This test method is applicable to olefin-free (<2%
safety concerns, if any, associated with its use. It is the
olefins by liquid volume) liquid hydrocarbon mixtures includ-
responsibility of the user of this standard to establish appro-
ing virgin naphthas, reformates, and alkylates. Olefin content
priate safety, health, and environmental practices and deter-
can be determined by Test Method D1319 or D6839. The
mine the applicability of regulatory limitations prior to use.
hydrocarbon mixture must have a 98% point of 250°C or less
Specific warning statements are given in Section 8.
as determined by Test Method D3710 or D7096 or equivalent.
1.8 This international standard was developed in accor-
dance with internationally recognized principles on standard-
1.3 Componentsthatarepresentatthe0.05%bymasslevel
ization established in the Decision on Principles for the
or greater can be determined.
Development of International Standards, Guides and Recom-
1.4 This test method may not be completely accurate for mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
PNAabove carbon number C7; Test Method D5443 or D6839
may be used to verify or complement the results of this test
2. Referenced Documents
method for carbon numbers >C7.
2.1 ASTM Standards:
D1319Test Method for HydrocarbonTypes in Liquid Petro-
leum Products by Fluorescent Indicator Adsorption
This test method is under the jurisdiction of ASTM Committee D02 on
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of
Subcommittee D02.04.0L on Gas Chromatography Methods. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Dec. 1, 2021. Published December 2021. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1990. Last previous edition approved in 2017 as D5134–13 (2017). Standards volume information, refer to the standard’s Document Summary page on
DOI: 10.1520/D5134-21. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5134 − 21
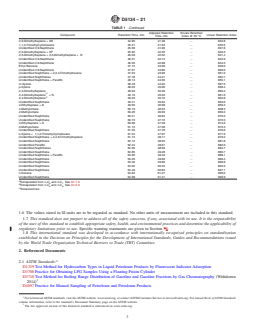
TABLE 1 Typical Retention Characteristics of Naphtha Components
NOTE 1—The abbreviations N and P refer to unidentified naphthenes and paraffins respectively.
Adjusted Retention Kovats Retention
Compound Retention Time, min Linear Retention Index
Time, min Index @ 35 °C
Methane 3.57 0.00 100.0 .
Ethane 3.65 0.08 200.0 .
Propane 3.84 0.27 300.0 .
Isobutane 4.14 0.57 367.3 .
n-Butane 4.39 0.82 400.0 .
2,2-Dimethylpropane 4.53 0.96 415.5 .
Isopentane 5.33 1.76 475.0 .
n-Pentane 5.84 2.27 500.0 .
2,2-Dimethylbutane 6.81 3.24 536.2 .
Cyclopentane 7.83 4.26 564.1 .
2,3-Dimethylbutane 7.89 4.32 565.5 .
2-Methylpentane 8.06 4.49 569.5 .
3-Methylpentane 8.72 5.15 583.4 .
n-Hexane 9.63 6.06 600.0 .
2,2-Dimethylpentane 11.22 7.65 624.2 .
Methylcyclopentane 11.39 7.82 626.5 .
2,4-Dimethylpentane 11.68 8.11 630.3 .
2,2,3-Trimethylbutane 12.09 8.52 635.4 .
Benzene 13.29 9.72 649.1 .
3,3-dimethylpentane 13.84 10.27 654.8 .
Cyclohexane 14.19 10.62 658.3 .
2-Methylhexane 15.20 11.63 667.8 .
2,3-Dimethylpentane 15.35 11.78 669.1 .
1,1-Dimethylcyclopentane 15.61 12.04 671.4 .
3-Methylhexane 16.18 12.61 676.2 .
cis-1,3-Dimethylcyclopentane 16.88 13.31 681.8 .
trans-1,3-Dimethylcyclopentane 17.22 13.65 684.4 .
3-Ethylpentane 17.44 13.87 686.1 .
trans-1,2-Dimethylcyclopentane 17.57 14.00 687.0 .
2,2,4-Trimethylpentane 17.80 14.23 688.7 .
n-Heptane 19.43 15.86 700.0 .
A
Methylcyclohexane + cis-1,2-Dimethylcyclopentane 22.53 18.96 718.6 .
A
1,1,3-Trimethylcyclopentane + 2,2-Dimethylhexane 23.05 19.48 721.4 .
A
Ethylcyclopentane 24.59 21.02 729.3 .
A
2,5-Dimethylhexane + 2,2,3-Trimethylpentane 25.12 21.55 731.9 .
A
2,4-Dimethylhexane 25.47 21.90 733.5 .
A
1,trans-2,cis-4-Trimethylcyclopentane 26.43 22.86 738.0 .
A
3,3-Dimethylhexane 26.79 23.22 739.6 .
A
1,trans-2,cis-3-Trimethylcyclopentane 28.01 24.44 744.9 .
A
2,3,4-Trimethylpentane 28.70 25.13 747.8 .
A B
Toluene + 2,3,3-Trimethylpentane 29.49 25.92 751.1 730.2
B
1,1,2-Trimethylcyclopentane 31.21 27.64 . 741.7
B
2,3-Dimethylhexane 31.49 27.92 . 743.6
A
2-Methyl-3-ethylpentane 31.69 28.12 . 744.9
B
2-Methylheptane 33.06 29.49 . 754.1
B
4-Methylheptane + 3-Methyl-3-ethylpentane 33.34 29.77 . 756.0
B
3,4-Dimethylhexane 33.49 29.92 . 757.0
B
1,cis-2,trans-4-Trimethylcyclopentane + 1,cis-2,cis-4-Trimethylcyclopentane 33.73 30.16 . 758.6
B
cis-1,3-Dimethylcyclohexane 34.45 30.88 . 763.4
B
3-Methylheptane + 1,cis-2,trans-3-Trimethylcyclopentane 34.64 31.07 . 764.7
B
3-Ethylhexane + trans-1,4-Dimethylcyclohexane 34.83 31.26 . 766.0
B
1,1-Dimethylcyclohexane 35.81 32.24 . 772.5
B
2,2,5-Trimethylhexane + trans-1,3-Ethylmethylcyclopentane 36.75 33.18 . 778.8
B
cis-1,3-Ethylmethylcyclopentane 37.14 33.57 . 781.4
B
trans-1,2-Ethylmethylcyclopentane 37.39 33.82 . 783.1
B
2,2,4-Trimethylhexane + 1,1-Ethylmethylcyclopentane 37.68 34.11 . 785.1
B
trans-1,2-Dimethylcylohexane 38.14 34.57 . 788.1
B
1,cis-2,cis-3-Trimethylcyclopentane 39.21 35.64 . 795.3
trans-1,3-Dimethylcyclohexane + cis-1,4-Dimethylcyclohexane 39.54 35.97 . 797.5
n-Octane 39.91 36.34 . 800.0
Isopropylcyclopentane + 2,4,4-Trimethylhexane 40.76 37.19 . 805.7
Unidentified C9-Naphthene 40.88 37.31 . 806.5
Unidentified C8-Naphthene 41.52 37.95 . 810.8
Unidentified C9-Naphthene 41.88 38.31 . 813.2
cis-1,2-Ethylmethylcyclopentane + 2,3,5-Trimethylhexane 42.55 38.98 . 817.7
2,2-Dimethylheptane 43.20 39.63 . 822.0
cis-1,2-Dimethylcyclohexane 43.43 39.86 . 823.6
2,2,3-Trimethylhexane + 9N 43.76 40.19 . 825.8
2,4-Dimethylheptane 43.88 40.31 . 826.6
4,4-Dimethylheptane + 9N 44.09 40.52 . 828.0
Ethylcyclohexane + n-Propylcyclopentane 44.36 40.79 . 829.8
2-Methyl- 4-Ethylhexane 44.74 41.17 . 832.4
D5134 − 21
TABLE 1 Continued
Adjusted Retention Kovats Retention
Compound Retention Time, min Linear Retention Index
Time, min Index @ 35 °C
2,6-Dimethylheptane + 9N 44.95 41.38 . 833.8
1,1,3-Trimethylcyclohexane 45.21 41.64 . 835.5
Unidentified C9-Naphthene 45.56 41.99 . 837.8
2,5-Dimethylheptane + 9P 45.92 42.35 . 840.3
3,5-Dimethylheptane + 3,3-Dimethylheptane + N 46.09 42.52 . 841.4
Unidentified C9-Naphthene 46.31 42.74 . 842.9
Unidentified C9-Naphthene 46.55 42.98 . 844.5
Ethyl Benzene 47.15 43.58 . 848.5
Unidentified C9-Naphthene 47.37 43.80 . 850.0
Unidentified Naphthene + 2,3,4-Trimethylhexane 47.53 43.96 . 851.0
Unidentified Naphthenes 47.78 44.21 . 852.7
Unidentified Naphthene + Paraffin 48.13 44.56 . 855.1
m-Xylene 48.49 44.92 . 857.5
p-Xylene 48.63 45.06 . 858.4
2,3-Dimethylheptane 48.93 45.36 . 860.4
C
3,4-Dimethylheptane + N 49.10 45.53 . 861.6
C
3,4-Dimethylheptane 49.29 45.72 . 862.8
Unidentified Naphthene 49.41 45.84 . 863.6
4-Ethylheptane + N 49.65 46.08 . 865.2
4-Methyloctane 50.10 46.53 . 868.3
2-Methyloctane 50.26 46.69 . 869.3
Unidentified Naphthene 50.41 46.84 . 870.3
Unidentified Naphthene 50.73 47.16 . 872.5
3-Ethylheptane + N 50.96 47.39 . 874.0
3-Methyloctane 51.15 47.58 . 875.3
Unidentified Naphthene 51.35 47.78 . 876.6
o-Xylene + 1,1,2-Trimethylcyclohexane 51.54 47.97 . 877.9
Unidentified Naphthene + 2,4,6-Trimethylheptane 51.74 48.17 . 879.2
Unidentified Naphthene 52.12 48.55 . 881.8
Unidentified Paraffin 52.24 48.67 . 882.6
Unidentified Naphthenes 52.56 48.99 . 884.7
Unidentified Naphthene 52.85 49.28 . 886.7
Unidentified Naphthene + Paraffin 53.06 49.49 . 888.1
Unidentified Naphthene 53.26 49.69 . 889.4
Unidentified Naphthene 53.46 49.89 . 890.8
Unidentified Naphthene 54.02 50.45 . 894.5
Unidentified Naphthene 54.40 50.83 . 897.1
n-Nonane 54.84 51.27 . 900.0
Unidentified Naphthene 54.98 51.41 . 900.9
A
Extrapolated from n-C and n-C . See A1.1.3.
6 7
B
Extrapolated from n-C and n-C . See A1.2.3.
8 9
C
Stereoisomers.
D3700Practice for Obtaining LPG Samples Using a Float- D6730Test Method for Determination of Individual Com-
ing Piston Cylinder ponents in Spark Ignition Engine Fuels by 100-Metre
D3710TestMethodforBoilingRangeDistributionofGaso-
Capillary (with Precolumn) High-Resolution Gas Chro-
line and Gasoline Fractions by Gas Chromatography
matography
(Withdrawn 2014)
D6733Test Method for Determination of Individual Com-
D4057Practice for Manual Sampling of Petroleum and
ponents in Spark Ignition Engine Fuels by 50-Metre
Petroleum Products
Capillary High Resolution Gas Chromatography
D4175Terminology Relating to Petroleum Products, Liquid
D7096Test Method for Determination of the Boiling Range
Fuels, and Lubricants
Distribution of Gasoline by Wide-Bore Capillary Gas
D5443Test Method for Paraffin, Naphthene, and Aromatic
Chromatography
Hydrocarbon Type Analysis in Petroleum Distillates
E355PracticeforGasChromatographyTermsandRelation-
Through 200 °C by Multi-Dimensional Gas Chromatog-
ships
raphy
E594Practice for Testing Flame Ionization Detectors Used
D6839Test Method for Hydrocarbon Types, Oxygenated
in Gas or Supercritical Fluid Chromatography
Compounds, Benzene, and Toluene in Spark Ignition
Engine Fuels by Multidimensional Gas Chromatography
3. Terminology
D6729Test Method for Determination of Individual Com-
ponents in Spark Ignition Engine Fuels by 100 Metre 3.1 Definitions:
Capillary High Resolution Gas Chromatography
3.1.1 This test method makes reference to common gas
chromatographicprocedures,terms,andrelationships.Detailed
3 definitions of these can be found in Practices E355 and E594,
The last approved version of this historical standard is referenced on
www.astm.org. and Terminology D4175.
D5134 − 21
4. Summary of Test Method ionization detector designed for optimum response with capil-
lary columns (with the required gas controls and electronics)
4.1 A representative sample of the naphtha is introduced
must meet or exceed the following specifications:
into a gas chromatograph equipped with a methyl silicone
Operating temperature 100 °C to 300 °C
bondedphasefusedsilicacapillarycolumn.Heliumcarriergas
Sensitivity >0.015 C/g
transports the vaporized sample through the column in which
−12
Minimum detectability 5 × 10 g carbon/second
the components are separated. Components are sensed by a Linearity >10
flame ionization detector as they elute from the column. The
7.2 Sample Introduction System—Manual or automatic liq-
detector signal is processed by an electronic data acquisition
uid syringe sample injection to the splitting injector may be
system or integrating computer. Each eluting peak is identified
employed. Devices capable of 0.2 µL to 1.0 µL injections are
by comparing its retention index to a table of retention indices
suitable. It should be noted that inadequate splitter design or
and by visual matching with a standard chromatogram. The
poor injection technique, or both, can result in sample frac-
table of retention indices has been established by running
tionation. Operating conditions which preclude fractionation
reference compounds under identical conditions or by gas
should be determined in accordance with Section 12.
chromatographic—mass spectrometric (GC/MS) analysis of
7.3 Electronic Data Acquisition System—Any data acquisi-
reference samples under the same conditions, or both.
tion and integration device used for quantitation of these
4.2 The mass concentration of each component is deter-
analyses must meet or exceed these minimum requirements:
mined by area normalization with response factors. Peaks
7.3.1 Capacity for at least 250 peaks/analysis.
eluting after n-nonane are summed and reported as C .
10+ 7.3.2 Normalized area percent calculation with response
factors.
5. Significance and Use
7.3.3 Identification of individual components by retention
5.1 A knowledge of the hydrocarbon components compris-
time.
ing a petroleum naphtha, reformate, or alkylate is useful in
7.3.4 Noise and spike rejection capability.
valuation of crude oils, in alkylation and reforming process
7.3.5 Sampling rates for fast (<1 s) peaks.
control, in product quality assessment, and for regulatory
7.3.6 Positive and negative sloping baseline correction.
purposes. Detailed hydrocarbon composition is also used as
7.3.7 Peakdetectionsensitivityfornarrowandbroadpeaks.
input in the mathematical modeling of refinery processes.
7.3.8 Perpendicular drop and tangent skimming as needed.
5.2 Separation of naphtha components by the procedure
7.4 Capillary Column—This test method utilizes a 50 m
described in this test method can result in some peaks that
(0.2 mm inside diameter) fused silica capillary column with
represent coeluting compounds. This test method cannot attri-
bonded (cross-linked) methyl silicone phase and a film thick-
buterelativeconcentrationstothecoelutants.Intheabsenceof
ness (d) of 0.5 µm. Other columns with these nominal
f
supporting information, use of the results of this test method
dimensions may be suitable. However, all columns must meet
for purposes which require such attribution is not recom-
the criteria set out in Section 11 for efficiency, resolution, and
mended.
polarity.
6. Interferences
8. Reagents and Materials
6.1 Ifpresent,olefinichydrocarbonswithboilingpointsless
8.1 Carrier Gas, helium, mol fraction is 99.99% pure.
than 150°C will be separated and detected along with the
(Warning—Compressed gas under high pressure.)
saturates and aromatics. Some of the olefins will coelute with
8.2 Fuel Gas, hydrogen, mol fraction is 99.9% pure.
saturates or aromatics and give erroneously high concentra-
(Warning—Extremely flammable gas under pressure.)
tions for those components. Some coelutions of PNA compo-
nents above C7 may occur and results may not be completely 8.3 Make-up Gas, helium or nitrogen, 99.99 % pure.
accurate. Test Method D5443 may be used for carbon number
(Warning—Compressed gases under higher pressure.)
distribution above C7 to verify results from this test method.
8.4 n-Heptane, mol fraction is 99+ %. (Warning—
6.2 Alcohols, ethers, and other organic compounds of simi-
Flammable. Harmful if inhaled.)
lar volatility can also interfere by coeluting with saturate or
8.5 Methane—(Warning—Extremely flammable gas.)
aromatic hydrocarbons thereby causing erroneously high val-
8.6 2-Methylheptane, mol fraction is 99+%. (Warning—
ues to be determined.
Flammable. Harmful if inhaled.)
7. Apparatus
8.7 4-Methylheptane, mol fraction is 99+%. (Warning—
Flammable. Harmful if inhaled.)
7.1 Instrumentation—A gas chromatograph capable of col-
umn oven temperature programming from 35°C to 200°C in
8.8 2-Methylpentane, mol fraction is 99+%. (Warning—
1°C⁄min increments is required. A heated flash vaporizing
Extremely flammable. Harmful if inhaled.)
injector designed to provide a linear sample split injection (for
8.9 n-Octane, mol fraction is 99+ %. (Warning—
example, 200:1) is also required for proper sample introduc-
Flammable. Harmful if inhaled.)
tion. The associated carrier gas controls must be of adequate
precision to provide reproducible column flows and split ratios 8.10 Toluene, mol fraction is 99+ %. (Warning—
in order to maintain analytical integrity. A hydrogen flame Flammable. Vapor harmful.)
D5134 − 21
8.11 2,3,3-Trimethylpentane, mol fraction is 99+ %. 8.15 Reference Reformate, actual refinery production re-
(Warning—Extremely flammable. Harmful if inhaled.) former product (‘reformate’) for compound identification as in
Fig. 3.(Warning—Extremely flammable. Harmful if inhaled.)
8.12 Column Evaluation Mixture, a qualitative synthetic
mixture of pure liquid hydrocarbons with the following ap-
NOTE 1—Alkylate, virgin naphtha, and reformer production refinery
proximate composition: 0.5%toluene, 1% n-heptane, 1%
reference samples may be available from several vendors; alternatively,
2,3,3-trimethylpentane, 1%2-methylheptane, 1%4-methyl-
in-house production materials or equivalent that matches closely the
heptane, 1% n-octane in 2-methylpentane solvent. fingerprints in the chromatograms (Figs. 1-3) may be used.
8.13 Reference Alkylate, actual refinery production alky-
9. Sampling
lation product used for compound identification as in Fig. 1.
(Warning—Extremely flammable. Harmful if inhaled.)
9.1 Hydrocarbon liquids (including naphthas) with Reid
vapor pressures of 110 kPa (16 psi) or less may be sampled
8.14 Reference Virgin Naphtha, actual refinery production
either into a floating piston cylinder or into an open container.
stream used for compound identification as in Fig. 2.
(Warning—Extremely flammable. Harmful if inhaled.) Samples taken into piston samplers may be sampled into a GC
FIG. 1 Reference Alkylate Chromatogram
D5134 − 21
FIG. 2 Reference Virgin Naphtha Chromatogram
vialorequivalentprovidedthatuponchillingandtransferdoes 9.1.2 Open Container Sampling—Refer to Practice D4057
not lead to significant losses of light components. for instructions on manual sampling from bulk storage into
open containers. Stopper container immediately after drawing
NOTE2—Althoughpossible,thistestmethodhasnotbeenevaluatedfor
sample.
injectionofpressurizedsamplesthatrequirehighpressureliquidinjection
valves.
9.2 Preserve the sample by cooling to approximately 4°C
and by maintaining that temperature until immediately prior to
9.1.1 Cylinder Sampling—Refer to Test Method D3700 for
analysis.
instructionsontransferringarepresentativesampleofahydro-
carbon fluid from a source into a floating piston cylinder.Add 9.3 Transfer an aliquot of the cooled sample into a pre-
inert gas to the ballast side of the floating piston cylinder to cooled septum vial, then seal appropriately. Obtain the test
achieveapressureof350kPa(45psi)abovethevaporpressure specimen for analysis directly from the sealed septum vial, for
of the sample. either manual or automatic syringe injection.
D5134 − 21
FIG. 3 Reference Reformer Product (Reformate) Chromatogram
10. Preparation of Apparatus 10.2.2 Set the oven temperature to 35°C and allow oven to
equilibrate for at least 15 min, and then observe the tempera-
10.1 Install and condition column as per manufacturer’s or
ture reading.
supplier’s instructions. After conditioning, attach column out-
10.2.3 If the reading of the independent temperature sensor
let to flame ionization detector inlet and check for leaks
is more than 0.5°C different from 35°C, follow manufactur-
throughout the system. If leaks are found, tighten or replace
er’s instructions to adjust calibration of GC oven temperature.
fittings before proceeding.
10.2 Calibrate the gas chromatograph column oven tem-
NOTE 3—Differences of as little as 1°C c
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D5134 − 13 (Reapproved 2017) D5134 − 21
Standard Test Method for
Detailed Analysis of Petroleum Naphthas through n-Nonane
by Capillary Gas Chromatography
This standard is issued under the fixed designation D5134; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
INTRODUCTION
Despite the many advances in capillary gas chromatography instrumentation and the remarkable
resolution achievable, it has proven difficult to standardize a test method for the analysis of a mixture
as complex as petroleum naphtha. Because of the proliferation of numerous, similar columns and the
endless choices of phase thickness, column internal diameter, length, etc., as well as instrument
operating parameters, many laboratories use similar but not identical methods for the capillary GC
analysis of petroleum naphthas. Even minute differences in column polarity or column oven
temperature, for example, can change resolution or elution order of components and make their
identification an individual interpretive process rather than the desirable, objective application of
standard retention data. To avoid this, stringent column specifications and temperature and flow
conditions have been adopted in this test method to ensure consistent elution order and resolution and
reproducible retention times. Strict adherence to the specified conditions is essential to the successful
application of this test method.
1. Scope Scope*
1.1 This detailed hydrocarbon analysis (DHA) test method covers the determination of hydrocarbon components paraffins,
naphthenes, and monoaromatics (PNA) of petroleum naphthas as enumerated in Table 1. Components eluting after n-nonane (bp
150.8 °C) are determined as a single group.
1.2 This test method is applicable to olefin-free (<2 % olefins by liquid volume) liquid hydrocarbon mixtures including virgin
naphthas, reformates, and alkylates. Olefin content can be determined by Test Method D1319 or D6839. The hydrocarbon mixture
must have a 98 % point of 250 °C or less as determined by Test Method D3710 or D7096 or equivalent.
1.3 Components that are present at the 0.05 % by mass level or greater can be determined.
1.4 This test method may not be completely accurate for PNA above carbon number C7; Test Method D5443 or D6839 may be
used to verify or complement the results of this test method for carbon numbers >C7.
1.5 Detailed hydrocarbon components in olefin containing samples may be determined by DHA Test Methods D6729, D6730, or
D6733.
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.04.0L on Gas Chromatography Methods.
Current edition approved Oct. 1, 2017Dec. 1, 2021. Published November 2017December 2021. Originally approved in 1990. Last previous edition approved in 20132017
as D5134 – 13.D5134 – 13 (2017). DOI: 10.1520/D5134-13R17.10.1520/D5134-21.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5134 − 21
TABLE 1 Typical Retention Characteristics of Naphtha Components
NOTE 1—The abbreviations N and P refer to unidentified naphthenes and paraffins respectively.
Adjusted Retention Kovats Retention
Compound Retention Time, min Linear Retention Index
Time, min Index @ 35 °C
Methane 3.57 0.00 100.0 .
Ethane 3.65 0.08 200.0 .
Propane 3.84 0.27 300.0 .
Isobutane 4.14 0.57 367.3 .
n-Butane 4.39 0.82 400.0 .
2,2-Dimethylpropane 4.53 0.96 415.5 .
Isopentane 5.33 1.76 475.0 .
n-Pentane 5.84 2.27 500.0 .
2,2-Dimethylbutane 6.81 3.24 536.2 .
Cyclopentane 7.83 4.26 564.1 .
2,3-Dimethylbutane 7.89 4.32 565.5 .
2-Methylpentane 8.06 4.49 569.5 .
3-Methylpentane 8.72 5.15 583.4 .
n-Hexane 9.63 6.06 600.0 .
2,2-Dimethylpentane 11.22 7.65 624.2 .
Methylcyclopentane 11.39 7.82 626.5 .
2,4-Dimethylpentane 11.68 8.11 630.3 .
2,2,3-Trimethylbutane 12.09 8.52 635.4 .
Benzene 13.29 9.72 649.1 .
3,3-dimethylpentane 13.84 10.27 654.8 .
Cyclohexane 14.19 10.62 658.3 .
2-Methylhexane 15.20 11.63 667.8 .
2,3-Dimethylpentane 15.35 11.78 669.1 .
1,1-Dimethylcyclopentane 15.61 12.04 671.4 .
3-Methylhexane 16.18 12.61 676.2 .
cis-1,3-Dimethylcyclopentane 16.88 13.31 681.8 .
trans-1,3-Dimethylcyclopentane 17.22 13.65 684.4 .
3-Ethylpentane 17.44 13.87 686.1 .
trans-1,2-Dimethylcyclopentane 17.57 14.00 687.0 .
2,2,4-Trimethylpentane 17.80 14.23 688.7 .
n-Heptane 19.43 15.86 700.0 .
A
Methylcyclohexane + cis-1,2-Dimethylcyclopentane 22.53 18.96 718.6 .
A
1,1,3-Trimethylcyclopentane + 2,2-Dimethylhexane 23.05 19.48 721.4 .
A
Ethylcyclopentane 24.59 21.02 729.3 .
A
2,5-Dimethylhexane + 2,2,3-Trimethylpentane 25.12 21.55 731.9 .
A
2,4-Dimethylhexane 25.47 21.90 733.5 .
A
1,trans-2,cis-4-Trimethylcyclopentane 26.43 22.86 738.0 .
A
3,3-Dimethylhexane 26.79 23.22 739.6 .
A
1,trans-2,cis-3-Trimethylcyclopentane 28.01 24.44 744.9 .
A
2,3,4-Trimethylpentane 28.70 25.13 747.8 .
A B
Toluene + 2,3,3-Trimethylpentane 29.49 25.92 751.1 730.2
B
1,1,2-Trimethylcyclopentane 31.21 27.64 . 741.7
B
2,3-Dimethylhexane 31.49 27.92 . 743.6
A
2-Methyl-3-ethylpentane 31.69 28.12 . 744.9
B
2-Methylheptane 33.06 29.49 . 754.1
B
4-Methylheptane + 3-Methyl-3-ethylpentane 33.34 29.77 . 756.0
B
3,4-Dimethylhexane 33.49 29.92 . 757.0
B
1,cis-2,trans-4-Trimethylcyclopentane + 1,cis-2,cis-4-Trimethylcyclopentane 33.73 30.16 . 758.6
B
cis-1,3-Dimethylcyclohexane 34.45 30.88 . 763.4
B
3-Methylheptane + 1,cis-2,trans-3-Trimethylcyclopentane 34.64 31.07 . 764.7
B
3-Ethylhexane + trans-1,4-Dimethylcyclohexane 34.83 31.26 . 766.0
B
1,1-Dimethylcyclohexane 35.81 32.24 . 772.5
B
2,2,5-Trimethylhexane + trans-1,3-Ethylmethylcyclopentane 36.75 33.18 . 778.8
B
cis-1,3-Ethylmethylcyclopentane 37.14 33.57 . 781.4
B
trans-1,2-Ethylmethylcyclopentane 37.39 33.82 . 783.1
B
2,2,4-Trimethylhexane + 1,1-Ethylmethylcyclopentane 37.68 34.11 . 785.1
B
trans-1,2-Dimethylcylohexane 38.14 34.57 . 788.1
B
1,cis-2,cis-3-Trimethylcyclopentane 39.21 35.64 . 795.3
trans-1,3-Dimethylcyclohexane + cis-1,4-Dimethylcyclohexane 39.54 35.97 . 797.5
n-Octane 39.91 36.34 . 800.0
Isopropylcyclopentane + 2,4,4-Trimethylhexane 40.76 37.19 . 805.7
Unidentified C9-Naphthene 40.88 37.31 . 806.5
Unidentified C8-Naphthene 41.52 37.95 . 810.8
Unidentified C9-Naphthene 41.88 38.31 . 813.2
cis-1,2-Ethylmethylcyclopentane + 2,3,5-Trimethylhexane 42.55 38.98 . 817.7
2,2-Dimethylheptane 43.20 39.63 . 822.0
cis-1,2-Dimethylcyclohexane 43.43 39.86 . 823.6
2,2,3-Trimethylhexane + 9N 43.76 40.19 . 825.8
2,4-Dimethylheptane 43.88 40.31 . 826.6
4,4-Dimethylheptane + 9N 44.09 40.52 . 828.0
Ethylcyclohexane + n-Propylcyclopentane 44.36 40.79 . 829.8
2-Methyl- 4-Ethylhexane 44.74 41.17 . 832.4
D5134 − 21
TABLE 1 Continued
Adjusted Retention Kovats Retention
Compound Retention Time, min Linear Retention Index
Time, min Index @ 35 °C
2,6-Dimethylheptane + 9N 44.95 41.38 . 833.8
1,1,3-Trimethylcyclohexane 45.21 41.64 . 835.5
Unidentified C9-Naphthene 45.56 41.99 . 837.8
2,5-Dimethylheptane + 9P 45.92 42.35 . 840.3
3,5-Dimethylheptane + 3,3-Dimethylheptane + N 46.09 42.52 . 841.4
Unidentified C9-Naphthene 46.31 42.74 . 842.9
Unidentified C9-Naphthene 46.55 42.98 . 844.5
Ethyl Benzene 47.15 43.58 . 848.5
Unidentified C9-Naphthene 47.37 43.80 . 850.0
Unidentified Naphthene + 2,3,4-Trimethylhexane 47.53 43.96 . 851.0
Unidentified Naphthenes 47.78 44.21 . 852.7
Unidentified Naphthene + Paraffin 48.13 44.56 . 855.1
m-Xylene 48.49 44.92 . 857.5
p-Xylene 48.63 45.06 . 858.4
2,3-Dimethylheptane 48.93 45.36 . 860.4
C
3,4-Dimethylheptane + N 49.10 45.53 . 861.6
C
3,4-Dimethylheptane 49.29 45.72 . 862.8
Unidentified Naphthene 49.41 45.84 . 863.6
4-Ethylheptane + N 49.65 46.08 . 865.2
4-Methyloctane 50.10 46.53 . 868.3
2-Methyloctane 50.26 46.69 . 869.3
Unidentified Naphthene 50.41 46.84 . 870.3
Unidentified Naphthene 50.73 47.16 . 872.5
3-Ethylheptane + N 50.96 47.39 . 874.0
3-Methyloctane 51.15 47.58 . 875.3
Unidentified Naphthene 51.35 47.78 . 876.6
o-Xylene + 1,1,2-Trimethylcyclohexane 51.54 47.97 . 877.9
Unidentified Naphthene + 2,4,6-Trimethylheptane 51.74 48.17 . 879.2
Unidentified Naphthene 52.12 48.55 . 881.8
Unidentified Paraffin 52.24 48.67 . 882.6
Unidentified Naphthenes 52.56 48.99 . 884.7
Unidentified Naphthene 52.85 49.28 . 886.7
Unidentified Naphthene + Paraffin 53.06 49.49 . 888.1
Unidentified Naphthene 53.26 49.69 . 889.4
Unidentified Naphthene 53.46 49.89 . 890.8
Unidentified Naphthene 54.02 50.45 . 894.5
Unidentified Naphthene 54.40 50.83 . 897.1
n-Nonane 54.84 51.27 . 900.0
Unidentified Naphthene 54.98 51.41 . 900.9
A
Extrapolated from n-C and n-C . See A1.1.3.
6 7
B
Extrapolated from n-C and n-C . See A1.2.3.
8 9
C
Stereoisomers.
1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. Specific warning statements are given in Section 78.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1319 Test Method for Hydrocarbon Types in Liquid Petroleum Products by Fluorescent Indicator Adsorption
D3700 Practice for Obtaining LPG Samples Using a Floating Piston Cylinder
D3710 Test Method for Boiling Range Distribution of Gasoline and Gasoline Fractions by Gas Chromatography (Withdrawn
2014)
D4057 Practice for Manual Sampling of Petroleum and Petroleum Products
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
D5134 − 21
D4175 Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants
D5443 Test Method for Paraffin, Naphthene, and Aromatic Hydrocarbon Type Analysis in Petroleum Distillates Through 200 °C
by Multi-Dimensional Gas Chromatography
D6839 Test Method for Hydrocarbon Types, Oxygenated Compounds, Benzene, and Toluene in Spark Ignition Engine Fuels by
Multidimensional Gas Chromatography
D6729 Test Method for Determination of Individual Components in Spark Ignition Engine Fuels by 100 Metre Capillary High
Resolution Gas Chromatography
D6730 Test Method for Determination of Individual Components in Spark Ignition Engine Fuels by 100-Metre Capillary (with
Precolumn) High-Resolution Gas Chromatography
D6733 Test Method for Determination of Individual Components in Spark Ignition Engine Fuels by 50-Metre Capillary High
Resolution Gas Chromatography
D7096 Test Method for Determination of the Boiling Range Distribution of Gasoline by Wide-Bore Capillary Gas
Chromatography
E355 Practice for Gas Chromatography Terms and Relationships
E594 Practice for Testing Flame Ionization Detectors Used in Gas or Supercritical Fluid Chromatography
3. Terminology
3.1 Definitions:
3.1.1 This test method makes reference to common gas chromatographic procedures, terms, and relationships. Detailed definitions
of these can be found in Practices E355 and E594, and Terminology D4175.
4. Summary of Test Method
4.1 A representative sample of the naphtha is introduced into a gas chromatograph equipped with a methyl silicone bonded phase
fused silica capillary column. Helium carrier gas transports the vaporized sample through the column in which the components are
separated. Components are sensed by a flame ionization detector as they elute from the column. The detector signal is processed
by an electronic data acquisition system or integrating computer. Each eluting peak is identified by comparing its retention index
to a table of retention indices and by visual matching with a standard chromatogram. The table of retention indices has been
established by running reference compounds under identical conditions or by gas chromatographic—mass spectrometric (GC/MS)
analysis of reference samples under the same conditions, or both.
4.2 The mass concentration of each component is determined by area normalization with response factors. Peaks eluting after
n-nonane are summed and reported as C .
10+
5. Significance and Use
5.1 A knowledge of the hydrocarbon components comprising a petroleum naphtha, reformate, or alkylate is useful in valuation
of crude oils, in alkylation and reforming process control, in product quality assessment, and for regulatory purposes. Detailed
hydrocarbon composition is also used as input in the mathematical modeling of refinery processes.
5.2 Separation of naphtha components by the procedure described in this test method can result in some peaks that represent
coeluting compounds. This test method cannot attribute relative concentrations to the coelutants. In the absence of supporting
information, use of the results of this test method for purposes which require such attribution is not recommended.
6. Interferences
6.1 If present, olefinic hydrocarbons with boiling points less than 150 °C will be separated and detected along with the saturates
and aromatics. Some of the olefins will coelute with saturates or aromatics and give erroneously high concentrations for those
components. Some coelutions of PNA components above C7 may occur and results may not be completely accurate. Test Method
D5443 may be used for carbon number distribution above C7 to verify results from this test method.
6.2 Alcohols, ethers, and other organic compounds of similar volatility can also interfere by coeluting with saturate or aromatic
hydrocarbons thereby causing erroneously high values to be determined.
7. Apparatus
7.1 Instrumentation—A gas chromatograph capable of column oven temperature programming from 35 °C to 200 °C in 1 °C ⁄min
D5134 − 21
increments is required. A heated flash vaporizing injector designed to provide a linear sample split injection (for example, 200:1)
is also required for proper sample introduction. The associated carrier gas controls must be of adequate precision to provide
reproducible column flows and split ratios in order to maintain analytical integrity. A hydrogen flame ionization detector designed
for optimum response with capillary columns (with the required gas controls and electronics) must meet or exceed the following
specifications:
Operating temperature 100 °C to 300 °C
Sensitivity >0.015 C/g
−12
Minimum detectability 5 × 10 g carbon/second
Linearity >10
7.2 Sample Introduction System—Manual or automatic liquid syringe sample injection to the splitting injector may be employed.
Devices capable of 0.2 μL to 1.0 μL injections are suitable. It should be noted that inadequate splitter design or poor injection
technique, or both, can result in sample fractionation. Operating conditions which preclude fractionation should be determined in
accordance with Section 1112.
7.3 Electronic Data Acquisition System—Any data acquisition and integration device used for quantitation of these analyses must
meet or exceed these minimum requirements:
7.3.1 Capacity for at least 250 peaks/analysis.
7.3.2 Normalized area percent calculation with response factors.
7.3.3 Identification of individual components by retention time.
7.3.4 Noise and spike rejection capability.
7.3.5 Sampling rates for fast (<1 s) peaks.
7.3.6 Positive and negative sloping baseline correction.
7.3.7 Peak detection sensitivity for narrow and broad peaks.
7.3.8 Perpendicular drop and tangent skimming as needed.
7.4 Capillary Column—This test method utilizes a 50 m (0.2 mm inside diameter) fused silica capillary column with bonded
(cross-linked) methyl silicone phase and a film thickness (d ) of 0.5 μm. Other columns with these nominal dimensions may be
f
suitable. However, all columns must meet the criteria set out in Section 1011 for efficiency, resolution, and polarity.
8. Reagents and Materials
8.1 Carrier Gas, helium, mol fraction is 99.99 % pure. (Warning—Compressed gas under high pressure.)
8.2 Fuel Gas, hydrogen, mol fraction is 99.9 % pure. (Warning—Extremely flammable gas under pressure.)
8.3 Make-up Gas, helium or nitrogen, 99.99 % pure. (Warning—Compressed gases under higher pressure.)
8.4 n-Heptane, mol fraction is 99+ %. (Warning—Flammable. Harmful if inhaled.)
8.5 Methane—(Warning—Extremely flammable gas.)
8.6 2-Methylheptane, mol fraction is 99+ %. (Warning—Flammable. Harmful if inhaled.)
8.7 4-Methylheptane, mol fraction is 99+ %. (Warning—Flammable. Harmful if inhaled.)
D5134 − 21
8.8 2-Methylpentane, mol fraction is 99+ %. (Warning—Extremely flammable. Harmful if inhaled.)
8.9 n-Octane, mol fraction is 99+ %. (Warning—Flammable. Harmful if inhaled.)
8.10 Toluene, mol fraction is 99+ %. (Warning—Flammable. Vapor harmful.)
8.11 2,3,3-Trimethylpentane, mol fraction is 99+ %. (Warning—Extremely flammable. Harmful if inhaled.)
8.12 Column Evaluation Mixture, a qualitative synthetic mixture of pure liquid hydrocarbons with the following approximate
composition: 0.5 % toluene, 1 % n-heptane, 1 % 2,3,3-trimethylpentane, 1 % 2-methylheptane, 1 % 4-methylheptane, 1 % n-
octane in 2-methylpentane solvent.
8.13 Reference Alkylate, actual refinery production alkylation product used for compound identification as in Fig. 1.
(Warning—Extremely flammable. Harmful if inhaled.)
8.14 Reference Virgin Naphtha, actual refinery production stream used for compound identification as in Fig. 2. (Warning—
Extremely flammable. Harmful if inhaled.)
8.15 Reference Reformate, actual refinery production reformer product (‘reformate’) for compound identification as in Fig. 3.
(Warning—Extremely flammable. Harmful if inhaled.)
NOTE 1—Alkylate, virgin naphtha, and reformer production refinery reference samples may be available from several vendors; alternatively, in-house
production materials or equivalent that matches closely the fingerprints in the chromatograms (Figs. 1-3) may be used.
9. Sampling
9.1 Hydrocarbon liquids (including naphthas) with Reid vapor pressures of 110 kPa (16 psi) or less may be sampled either into
a floating piston cylinder or into an open container. Samples taken into piston samplers may be sampled into a GC vial or
equivalent provided that upon chilling and transfer does not lead to significant losses of light components.
NOTE 2—Although possible, this test method has not been evaluated for injection of pressurized samples that require high pressure liquid injection valves.
9.1.1 Cylinder Sampling—Refer to Test Method D3700 for instructions on transferring a representative sample of a hydrocarbon
fluid from a source into a floating piston cylinder. Add inert gas to the ballast side of the floating piston cylinder to achieve a
pressure of 350 kPa (45 psi) above the vapor pressure of the sample.
9.1.2 Open Container Sampling—Refer to Practice D4057 for instructions on manual sampling from bulk storage into open
containers. Stopper container immediately after drawing sample.
9.2 Preserve the sample by cooling to approximately 4 °C and by maintaining that temperature until immediately prior to analysis.
9.3 Transfer an aliquot of the cooled sample into a precooled septum vial, then seal appropriately. Obtain the test specimen for
analysis directly from the sealed septum vial, for either manual or automatic syringe injection.
10. Preparation of Apparatus
10.1 Install and condition column as per manufacturer’s or supplier’s instructions. After conditioning, attach column outlet to
flame ionization detector inlet and check for leaks throughout the system. If leaks are found, tighten or replace fittings before
proceeding.
10.2 Calibrate the gas chromatograph column oven temperature sensors using an independent, electronic temperature measuring
device such as a thermocouple or platinum resistance temperature detector.
D5134 − 21
FIG. 1 Reference Alkylate Chromatogram
10.2.1 Place the independent temperature measuring probe in the oven in the region occupied by the column. Do not allow sensor
to touch the walls of the oven.
10.2.2 Set the oven temperature to 35 °C and allow oven to equilibrate for at least 15 min, and then observe the temperature
reading.
10.2.3 If the reading of the independent temperature sensor is more than 0.5 °C different from 35 °C, follow manufacturer’s
instructions to adjust calibration of GC oven temperature.
NOTE 3—Differences of as lit
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...