ASTM F2914-12(2024)
(Guide)Standard Guide for Identification of Shelf-Life Test Attributes for Endovascular Devices
Standard Guide for Identification of Shelf-Life Test Attributes for Endovascular Devices
SIGNIFICANCE AND USE
3.1 The purpose of this guide is to provide a procedure for determining the appropriate attributes to evaluate in a shelf-life study for an endovascular device.
SCOPE
1.1 This guide addresses the determination of appropriate device attributes for testing as part of a shelf-life study for endovascular devices. Combination and biodegradable devices (for example, drug devices, biologic devices, or drug biologics) may require additional considerations, depending on their nature.
1.2 This guide does not directly provide any test methods for conducting shelf-life testing.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Jan-2024
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.30 - Cardiovascular Standards
Relations
- Effective Date
- 15-Jan-2024
- Effective Date
- 15-Jan-2024
- Effective Date
- 15-Jan-2024
- Effective Date
- 15-Jan-2024
Overview
ASTM F2914-12(2024), Standard Guide for Identification of Shelf-Life Test Attributes for Endovascular Devices, provides a structured approach for determining the appropriate device attributes to evaluate during shelf-life studies of endovascular medical devices. Developed by ASTM International, this guide is essential for manufacturers, regulatory professionals, and quality assurance engineers involved in assessing medical device stability and compliance. The document aids users in identifying the specific attributes of endovascular devices that need verification to ensure performance and patient safety throughout the product’s shelf life.
Key Topics
- Attribute Identification: The guide details a clear procedure for selecting device attributes most likely to be affected by aging, packaging, sterilization, and storage conditions.
- Risk-Based Approach: Emphasis is placed on risk assessment, where only those attributes that could potentially change over time and impact safety or performance require detailed shelf-life evaluation.
- Scientific Justification: The standard encourages use of references from scientific literature, vendor data, or in-house research to substantiate decisions on attribute stability.
- Data-Driven Decisions: Where scientific principles are insufficient, actual data and relevant performance testing are required to support shelf-life determinations.
- Reporting Requirements: ASTM F2914-12(2024) includes a template for documenting which attributes are tested, why certain tests are omitted, and the supporting rationale, enhancing transparency and regulatory compliance.
Common examples of attributes generally impacted by aging include:
- Tensile strength and flexibility of catheter materials
- Balloon rated burst and fatigue resistance
- Stent securement
Attributes typically not impacted by aging (given appropriate materials and storage) include:
- Metal component surface area
- MR compatibility
- Marker radiopacity
- Corrosion resistance
Applications
This standard is widely applicable across the lifecycle management of endovascular device products:
- Research & Development: Supports R&D teams in selecting relevant shelf-life attributes during product design and initial testing phases.
- Quality Assurance: Enables QA teams to establish robust shelf-life protocols aligned with regulatory expectations for device aging, storage, and performance.
- Regulatory Submissions: Provides essential rationale and documentation required for submissions to regulatory bodies such as the FDA or EU Notified Bodies.
- Risk Management: Facilitates informed decisions on when in-depth shelf-life testing is necessary by linking attribute changes to potential patient or product safety risks.
- Product Stewardship: Encourages periodic review, helping device manufacturers ensure continued compliance as materials, packaging, or sterilization processes evolve.
The guide is relevant for a wide range of endovascular devices, including catheters, stents, balloons, and their combination/biodegradable variants, though additional considerations may apply for the latter.
Related Standards
Several standards and guidelines complement ASTM F2914-12(2024) in the evaluation of medical device shelf life and performance:
- ASTM F1980: Guide for Accelerated Aging of Sterile Barrier Systems for Medical Devices
- ISO 11607-1: Packaging for Terminally Sterilized Medical Devices - Requirements for Materials, Sterile Barrier Systems and Packaging Systems
- ISO 14971: Medical Devices - Application of Risk Management to Medical Devices
- ISO 25539-1 & ISO 25539-2: Cardiovascular Implants - Endovascular Devices
- FDA Guidance Documents: For non-clinical engineering tests and labeling recommendations
- AAMI TIR 17: Compatibility of Materials Subject to Sterilization
By applying ASTM F2914-12(2024) in conjunction with these related standards, organizations can achieve comprehensive, compliant shelf-life assessments and facilitate smooth regulatory approvals for endovascular medical devices.
Keywords: endovascular device shelf life, shelf-life test attributes, medical device aging, ASTM F2914, shelf-life study, device stability, risk assessment, regulatory compliance.
Buy Documents
ASTM F2914-12(2024) - Standard Guide for Identification of Shelf-Life Test Attributes for Endovascular Devices
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2914-12(2024) is a guide published by ASTM International. Its full title is "Standard Guide for Identification of Shelf-Life Test Attributes for Endovascular Devices". This standard covers: SIGNIFICANCE AND USE 3.1 The purpose of this guide is to provide a procedure for determining the appropriate attributes to evaluate in a shelf-life study for an endovascular device. SCOPE 1.1 This guide addresses the determination of appropriate device attributes for testing as part of a shelf-life study for endovascular devices. Combination and biodegradable devices (for example, drug devices, biologic devices, or drug biologics) may require additional considerations, depending on their nature. 1.2 This guide does not directly provide any test methods for conducting shelf-life testing. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 3.1 The purpose of this guide is to provide a procedure for determining the appropriate attributes to evaluate in a shelf-life study for an endovascular device. SCOPE 1.1 This guide addresses the determination of appropriate device attributes for testing as part of a shelf-life study for endovascular devices. Combination and biodegradable devices (for example, drug devices, biologic devices, or drug biologics) may require additional considerations, depending on their nature. 1.2 This guide does not directly provide any test methods for conducting shelf-life testing. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2914-12(2024) is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2914-12(2024) has the following relationships with other standards: It is inter standard links to ASTM F2914-12(2018), ASTM F3172-15(2021), ASTM F3089-23, ASTM F3036-21. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2914-12(2024) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2914 − 12 (Reapproved 2024)
Standard Guide for
Identification of Shelf-Life Test Attributes for Endovascular
Devices
This standard is issued under the fixed designation F2914; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 4. Procedure
1.1 This guide addresses the determination of appropriate
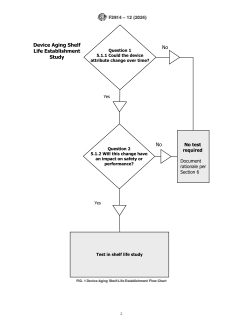
4.1 Shelf-Life Establishment Model Introduction—The deci-
device attributes for testing as part of a shelf-life study for
sion flow chart (Fig. 1) assists study developers in selecting
endovascular devices. Combination and biodegradable devices
and justifying risk-appropriate test protocols for medical de-
(for example, drug devices, biologic devices, or drug biologics)
vices to establish shelf life. The decision flow chart is intended
may require additional considerations, depending on their
to elicit questions and an appropriate rationale for testing or not
nature.
testing a particular attribute during aging. The risk to the
patient as the device ages is one of the primary drivers. It is
1.2 This guide does not directly provide any test methods
for conducting shelf-life testing. recommended that all regulatory requirements and guidances
be considered during development of the shelf-life establish-
1.3 This standard does not purport to address all of the
ment test plan. See Fig. 1.
safety concerns, if any, associated with its use. It is the
responsibility of the user of this standard to establish appro-
4.2 Question 1: Could the device attribute change over
priate safety, health, and environmental practices and deter-
time?
mine the applicability of regulatory limitations prior to use.
4.2.1 Considerations in Evaluating Question 1—This ques-
1.4 This international standard was developed in accor-
tion must be addressed based on the device design character-
dance with internationally recognized principles on standard-
istics (and also in relation to the device being packaged,
ization established in the Decision on Principles for the
sterilized, shipped, and stored).
Development of International Standards, Guides and Recom-
4.2.1.1 Consider attributes such as the following, for ex-
mendations issued by the World Trade Organization Technical
ample:
Barriers to Trade (TBT) Committee.
(1) Material Properties/Characterization—Composition;
2. Terminology
Mechanical Properties; Corrosion Resistance
(2) Dimensional and Functional Properties—Dimensions;
2.1 Definitions:
Surface Area; Foreshortening
2.1.1 endovascular device—device used to treat vascular
(3) Deliverability and Functionality—Balloon Fatigue;
disease from within the vessel.
Balloon Rated Burst; Bond Tensile Strength
2.1.2 product—final packaged and sterilized device with all
4.2.1.2 Various sources may provide sufficient evidence to
included components.
confirm that some specific attributes do not change over time
2.1.3 shelf life—the amount of real time that a fully pack-
for the application or that the change is not a risk to the patient.
aged (and sterilized, if applicable) product can be expected to
(1) Scientific literature.
remain in storage at specified conditions and maintain its
(2) Appropriate vendor publication.
critical performance properties.
(3) In-house research.
3. Significance and Use (4) Assessment of clinically accepted device.
4.2.1.3 When using such data to justify why certain attri-
3.1 The purpose of this guide is to provide a procedure for
butes may not require shelf-life testing, consider all differences
determining the appropriate attributes to evaluate in a shelf-life
between the subject device and the source of those data to
study for an endovascular device.
ensure applicability. For example, vendor literature may not
represent the actual use of the material by the device manu-
This guide is under the jurisdiction of ASTM Committee F04 on Medical and
Surgical Materials and Devices and is the direct responsibility of Subcommittee
facturer. Additionally, further processing (for example, steril-
F04.30 on Cardiovascular Standards.
ization) may change the physical or chemical attribute(s) of the
Current edition approved Jan. 15, 2024. Published January 2024. Originally
material. Finally, consider whether there are interactions
approved in 2012. Last previous edition approved in 2018 as F2914 – 12 (2018).
DOI:10.1520/F2914-12R24. (chemical or physical) that may impact your assessment.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2914 − 12 (2024)
FIG. 1 Device Aging Shelf-Life Establishment Flow Chart
F2914 − 12 (2024)
4.2.1.4 In order for testing to be applicable, the testing must way of stating the question is: “Will a change in the device
be conducted on articles that are representative of the final attribute resulting from aging pose a significant risk to the
device (that is, utilizing the same sterilization method and dose, patient or clinician?” Risk analysis is an appropriate technique
dimensions, material, processing conditions, and packaging). If used to answer this question. However, since risk analysis
test articles are not identical, provide appropriate justification methodologies have yet to be standardized, there is no defini-
for applicability of the testing. tive risk level that can be applied universally for all devices and
4.2.2 Justification Based upon Scientific Principles—When parameters. It will be the responsibility of individual compa-
one considers whether an attribute should be included in a nies to carefully develop the threshold for acceptable risk.
shelf-life study, the first question is whether the attribute 4.3.1 Basis for Risk Assessment—The assessment of risk
changes over time. There are several device attributes that may related to a device attribute may be conducted using clinical
be driven by physical parameters of the device that would not history (in literature or privately held) or the complaint history
change over time and therefore will not require shelf-life of a similar device used in a similar application. Additionally,
testing. The assessment should be conducted using universal a scientific/medical argument might provide adequate informa-
scientific/physical principles. In cases where the assessment is tion to assess the risk.
based on universal scientific/physical principle, appropriate 4.3.2 Risk Assessment Examples—The following examples
references should be provided. In cases where justifications of risk assessment of selected attributes are for illustrative
may be less obvious, data to support the scientific/physical purposes only; this guide cannot claim to address all
rationale shall be generated. Tables 1 and 2 list two groups of circumstances, and thus these examples should not be used to
device attributes with accompanying scientific rationale. overly influence a company’s policies. When not expected to
4.2.3 Justification Based upon Data—Scientific principles impact safety or performance, the scientific justification shall
for some device attributes/requirements are not readily evident. be documented in detail.
In such cases, one may generate data to support a rationale. It
5. Shelf-Life Establishment Report
may be advantageous to conduct testing in a manner that
allows for the data to be applicable to various size devices. In
5.1 The report shall include a complete device description,
this case, it is important to translate the device attribute (such
assumptions for device storage, and the device attributes
as system flexibility) into the underlying size independent
considered for testing in conducting a device aging shelf-life
scientific parameters (such as Young’s modulus). Testing is
establishment study. The decision to conduct testing or not for
then conducted to evaluate the stability of the core scientific
each device attribute shall be reported. The rationale for why
parameter. For each device attribute, more than one scientific
testing of a specific device attribute was determined to not be
parameter may be necessary to demonstrate stability over the
necessary (answered “no” to Question 1 or 2) shall be reported.
aging period. (For simplicity of the examples, only one test
The reported rationale shall provide sufficient detail to con-
parameter is illustrated in Table 3.) Each device attribute
vince a person with adequate engineering/scientific experience.
should be evaluated to determine what scientific parameters
References supporting rationale to not conduct testing should
may be affected by aging, and the appropriate testing to
be provided as appropriate. When testing of a specific device
mitigate each of those risks should then be conducted. The
attribute was determined to be neces
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...