ASTM F1717-21
(Test Method)Standard Test Methods for Spinal Implant Constructs in a Vertebrectomy Model
Standard Test Methods for Spinal Implant Constructs in a Vertebrectomy Model
SIGNIFICANCE AND USE
5.1 Spinal implants are generally composed of several components which, when connected together, form a spinal implant assembly. Spinal implant assemblies are designed to provide some stability to the spine while arthrodesis takes place. These test methods outline standard materials and methods for the evaluation of different spinal implant assemblies so that comparison between different designs may be facilitated.
5.2 These test methods are used to quantify the static and dynamic mechanical characteristics of different designs of spinal implant assemblies. The mechanical tests are conducted in vitro using simplified load schemes and do not attempt to mimic the complex loads of the spine.
5.3 The loads applied to the spinal implant assemblies in vivo will, in general, differ from the loading configurations used in these test methods. The results obtained here cannot be used directly to predict in-vivo performance. The results can be used to compare different component designs in terms of the relative mechanical parameters.
5.4 Fatigue testing in a simulated body fluid or saline may cause fretting, corrosion, or lubricate the interconnections and thereby affect the relative performance of tested devices. This test should be initially performed dry (ambient room conditions) for consistency. The effect of environment may be significant. Repeating all or part of these test methods in simulated body fluid, saline (9 g NaCl per 1000 mL water), a saline drip, water, or a lubricant should be considered. The maximum recommended frequency for this type of cyclic testing should be 5 Hz.
5.5 The location of the longitudinal elements is determined by where the anchors are clinically placed against bony structures. The perpendicular distance to the load direction (block moment arm) between the axis of a hinge pin and the anchor’s attachment points to a UHMWPE block is independent of anchor type. The distance between the anchor’s attachment point to the UHMWPE block an...
SCOPE
1.1 These test methods cover the materials and methods for the static and fatigue testing of spinal implant assemblies in a vertebrectomy model. The test materials for most combinations of spinal implant components can be specific, depending on the intended spinal location and intended method of application to the spine.
1.2 These test methods are intended to provide a basis for the mechanical comparison among past, present, and future spinal implant assemblies. They allow comparison of spinal implant constructs with different intended spinal locations and methods of application to the spine. These test methods are not intended to define levels of performance, since sufficient knowledge is not available to predict the consequences of the use of a particular device.
1.3 These test methods set out guidelines for load types and methods of applying loads. Methods for three static load types and one fatigue test are defined for the comparative evaluation of spinal implant assemblies.
1.4 These test methods establish guidelines for measuring displacements, determining the yield load, and evaluating the stiffness and strength of the spinal implant assembly.
1.5 Some spinal constructs may not be testable in all test configurations.
1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World T...
General Information
- Status
- Published
- Publication Date
- 31-Mar-2021
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.25 - Spinal Devices
Relations
- Effective Date
- 01-Oct-2017
- Effective Date
- 01-Oct-2016
- Effective Date
- 01-Oct-2014
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-May-2014
- Effective Date
- 01-May-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Dec-2011
- Effective Date
- 01-Nov-2011
- Effective Date
- 15-Jul-2011
- Effective Date
- 01-Nov-2010
- Effective Date
- 01-Oct-2010
- Effective Date
- 01-Jun-2010
Overview
ASTM F1717-21 is a widely referenced international standard developed by ASTM International for testing spinal implant constructs in a vertebrectomy model. These test methods provide guidelines for the mechanical evaluation of spinal implant assemblies using static and fatigue testing procedures. The objective of these methods is to facilitate the comparative analysis of different spinal implant designs, assessing their relative mechanical properties in a controlled, in vitro environment. This standard plays a crucial role in the global medical device industry, providing manufacturers, researchers, and regulatory bodies with a consistent methodology for mechanical testing of spinal devices.
Key Topics
Mechanical Testing of Spinal Implant Assemblies
ASTM F1717-21 addresses both static (compression bending, tensile bending, torsion) and dynamic (fatigue) loading scenarios for spinal implant assemblies. These tests simulate loading conditions by using simplified, repeatable setups rather than attempting to replicate the complex in vivo environment of the human spine.Standardized Test Model
The vertebrectomy model employed utilizes Ultra High Molecular Weight Polyethylene (UHMWPE) test blocks, ensuring consistency and reproducibility across evaluations by eliminating the variability of natural bone structure.Quantitative Assessment
The standard outlines detailed metrics for evaluating stiffness, yield load, ultimate load, displacement, and fatigue life of spinal constructs, providing critical data for comparative device analysis.Design Independence
The test methods are intended to be agnostic to the specific spinal location or device design, enabling meaningful comparison between anterior and posterior constructs as well as various anchoring methods (screws, hooks, wires, rods, plates, etc.).Environmental Factors
While initial tests are conducted in dry (ambient) conditions for consistency, ASTM F1717-21 highlights the significance of environmental variables such as simulated body fluids, which may affect device performance over time.
Applications
ASTM F1717-21 is crucial for:
Medical Device Development
Engineers and designers use these mechanical test methods during the development, validation, and verification of new spinal implants, ensuring product safety and performance.Regulatory Submissions
Documentation and results generated using ASTM F1717-21 are frequently required in submissions to regulatory bodies (such as the FDA or international authorities) for market approval of spinal implant systems.Quality Control and Benchmarking
Manufacturers rely on these standardized tests for ongoing quality assurance, post-market surveillance, and competitive benchmarking against existing or emerging spinal fixation technologies.Academic and Clinical Research
Researchers utilize the established test methods to compare the biomechanical performance of new constructs and to publish reproducible, peer-reviewed data.
Related Standards
For a comprehensive approach to mechanical testing of spinal implants, the following ASTM standards are often referenced alongside ASTM F1717-21:
- ASTM D638 - Test Method for Tensile Properties of Plastics
- ASTM E4 - Practices for Force Verification of Testing Machines
- ASTM F543 - Specification and Test Methods for Metallic Medical Bone Screws
- ASTM F1582 - Terminology Relating to Spinal Implants
- ASTM F1798 - Test Method for Evaluating Static and Fatigue Properties of Interconnection Mechanisms and Subassemblies Used in Spinal Arthrodesis Implants
- ASTM F2077 - Test Methods For Intervertebral Body Fusion Devices
Practical Value
Implementing ASTM F1717-21 ensures:
- Accurate, repeatable, and comparable mechanical test results for spinal implant constructs.
- Streamlined product development cycles by providing clear acceptance criteria for mechanical performance.
- Effective communication between manufacturers, regulators, clinicians, and researchers through common test protocols and shared terminology.
By adhering to these standardized test methods, stakeholders in the spinal implant field can better ensure device safety, promote innovation, and maintain compliance with international regulatory expectations.
Buy Documents
ASTM F1717-21 - Standard Test Methods for Spinal Implant Constructs in a Vertebrectomy Model
REDLINE ASTM F1717-21 - Standard Test Methods for Spinal Implant Constructs in a Vertebrectomy Model
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F1717-21 is a standard published by ASTM International. Its full title is "Standard Test Methods for Spinal Implant Constructs in a Vertebrectomy Model". This standard covers: SIGNIFICANCE AND USE 5.1 Spinal implants are generally composed of several components which, when connected together, form a spinal implant assembly. Spinal implant assemblies are designed to provide some stability to the spine while arthrodesis takes place. These test methods outline standard materials and methods for the evaluation of different spinal implant assemblies so that comparison between different designs may be facilitated. 5.2 These test methods are used to quantify the static and dynamic mechanical characteristics of different designs of spinal implant assemblies. The mechanical tests are conducted in vitro using simplified load schemes and do not attempt to mimic the complex loads of the spine. 5.3 The loads applied to the spinal implant assemblies in vivo will, in general, differ from the loading configurations used in these test methods. The results obtained here cannot be used directly to predict in-vivo performance. The results can be used to compare different component designs in terms of the relative mechanical parameters. 5.4 Fatigue testing in a simulated body fluid or saline may cause fretting, corrosion, or lubricate the interconnections and thereby affect the relative performance of tested devices. This test should be initially performed dry (ambient room conditions) for consistency. The effect of environment may be significant. Repeating all or part of these test methods in simulated body fluid, saline (9 g NaCl per 1000 mL water), a saline drip, water, or a lubricant should be considered. The maximum recommended frequency for this type of cyclic testing should be 5 Hz. 5.5 The location of the longitudinal elements is determined by where the anchors are clinically placed against bony structures. The perpendicular distance to the load direction (block moment arm) between the axis of a hinge pin and the anchor’s attachment points to a UHMWPE block is independent of anchor type. The distance between the anchor’s attachment point to the UHMWPE block an... SCOPE 1.1 These test methods cover the materials and methods for the static and fatigue testing of spinal implant assemblies in a vertebrectomy model. The test materials for most combinations of spinal implant components can be specific, depending on the intended spinal location and intended method of application to the spine. 1.2 These test methods are intended to provide a basis for the mechanical comparison among past, present, and future spinal implant assemblies. They allow comparison of spinal implant constructs with different intended spinal locations and methods of application to the spine. These test methods are not intended to define levels of performance, since sufficient knowledge is not available to predict the consequences of the use of a particular device. 1.3 These test methods set out guidelines for load types and methods of applying loads. Methods for three static load types and one fatigue test are defined for the comparative evaluation of spinal implant assemblies. 1.4 These test methods establish guidelines for measuring displacements, determining the yield load, and evaluating the stiffness and strength of the spinal implant assembly. 1.5 Some spinal constructs may not be testable in all test configurations. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World T...
SIGNIFICANCE AND USE 5.1 Spinal implants are generally composed of several components which, when connected together, form a spinal implant assembly. Spinal implant assemblies are designed to provide some stability to the spine while arthrodesis takes place. These test methods outline standard materials and methods for the evaluation of different spinal implant assemblies so that comparison between different designs may be facilitated. 5.2 These test methods are used to quantify the static and dynamic mechanical characteristics of different designs of spinal implant assemblies. The mechanical tests are conducted in vitro using simplified load schemes and do not attempt to mimic the complex loads of the spine. 5.3 The loads applied to the spinal implant assemblies in vivo will, in general, differ from the loading configurations used in these test methods. The results obtained here cannot be used directly to predict in-vivo performance. The results can be used to compare different component designs in terms of the relative mechanical parameters. 5.4 Fatigue testing in a simulated body fluid or saline may cause fretting, corrosion, or lubricate the interconnections and thereby affect the relative performance of tested devices. This test should be initially performed dry (ambient room conditions) for consistency. The effect of environment may be significant. Repeating all or part of these test methods in simulated body fluid, saline (9 g NaCl per 1000 mL water), a saline drip, water, or a lubricant should be considered. The maximum recommended frequency for this type of cyclic testing should be 5 Hz. 5.5 The location of the longitudinal elements is determined by where the anchors are clinically placed against bony structures. The perpendicular distance to the load direction (block moment arm) between the axis of a hinge pin and the anchor’s attachment points to a UHMWPE block is independent of anchor type. The distance between the anchor’s attachment point to the UHMWPE block an... SCOPE 1.1 These test methods cover the materials and methods for the static and fatigue testing of spinal implant assemblies in a vertebrectomy model. The test materials for most combinations of spinal implant components can be specific, depending on the intended spinal location and intended method of application to the spine. 1.2 These test methods are intended to provide a basis for the mechanical comparison among past, present, and future spinal implant assemblies. They allow comparison of spinal implant constructs with different intended spinal locations and methods of application to the spine. These test methods are not intended to define levels of performance, since sufficient knowledge is not available to predict the consequences of the use of a particular device. 1.3 These test methods set out guidelines for load types and methods of applying loads. Methods for three static load types and one fatigue test are defined for the comparative evaluation of spinal implant assemblies. 1.4 These test methods establish guidelines for measuring displacements, determining the yield load, and evaluating the stiffness and strength of the spinal implant assembly. 1.5 Some spinal constructs may not be testable in all test configurations. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World T...
ASTM F1717-21 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F1717-21 has the following relationships with other standards: It is inter standard links to ASTM F2077-17, ASTM F1582-98(2016), ASTM F2077-14, ASTM E4-14, ASTM E177-14, ASTM F543-13e1, ASTM F543-13, ASTM E691-13, ASTM E177-13, ASTM F1582-98(2011), ASTM E691-11, ASTM F2077-11, ASTM E739-10, ASTM E177-10, ASTM E4-10. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F1717-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F1717 − 21
Standard Test Methods for
Spinal Implant Constructs in a Vertebrectomy Model
This standard is issued under the fixed designation F1717; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
1.1 These test methods cover the materials and methods for
Barriers to Trade (TBT) Committee.
the static and fatigue testing of spinal implant assemblies in a
vertebrectomymodel.Thetestmaterialsformostcombinations
2. Referenced Documents
ofspinalimplantcomponentscanbespecific,dependingonthe
intended spinal location and intended method of application to
2.1 ASTM Standards:
the spine. D638Test Method for Tensile Properties of Plastics
E4Practices for Force Verification of Testing Machines
1.2 These test methods are intended to provide a basis for
E6Terminology Relating to Methods of MechanicalTesting
the mechanical comparison among past, present, and future
E177Practice for Use of the Terms Precision and Bias in
spinal implant assemblies. They allow comparison of spinal
ASTM Test Methods
implant constructs with different intended spinal locations and
E691Practice for Conducting an Interlaboratory Study to
methodsofapplicationtothespine.Thesetestmethodsarenot
Determine the Precision of a Test Method
intended to define levels of performance, since sufficient
E739PracticeforStatisticalAnalysisofLinearorLinearized
knowledge is not available to predict the consequences of the
Stress-Life (S-N) and Strain-Life (ε-N) Fatigue Data
use of a particular device.
E1150Definitions of Terms Relating to Fatigue (Withdrawn
1.3 These test methods set out guidelines for load types and 3
1996)
methods of applying loads. Methods for three static load types
F543Specification and Test Methods for Metallic Medical
and one fatigue test are defined for the comparative evaluation
Bone Screws
of spinal implant assemblies.
F1582Terminology Relating to Spinal Implants
1.4 These test methods establish guidelines for measuring F1798Test Method for Evaluating the Static and Fatigue
PropertiesofInterconnectionMechanismsandSubassem-
displacements, determining the yield load, and evaluating the
stiffness and strength of the spinal implant assembly. blies Used in Spinal Arthrodesis Implants
F2077TestMethodsForIntervertebralBodyFusionDevices
1.5 Some spinal constructs may not be testable in all test
configurations.
3. Terminology
1.6 The values stated in SI units are to be regarded as
3.1 Definitions:
standard. No other units of measurement are included in this
3.1.1 For definitions of terms relating to these test methods,
standard.
see Terminology E6, Terminology F1582, and Definitions
1.7 This standard does not purport to address all of the
E1150.
safety concerns, if any, associated with its use. It is the
3.2 Definitions of Terms Specific to This Standard:
responsibility of the user of this standard to establish appro-
3.2.1 active length of the longitudinal element—the straight
priate safety, health, and environmental practices and deter-
line distance between the center of attachment of the superior
mine the applicability of regulatory limitations prior to use.
anchor and the center of attachment of the inferior anchor.
1.8 This international standard was developed in accor-
3.2.2 angular displacement at 2 % offset yield (degrees)—
dance with internationally recognized principles on standard-
the angular displacement of a construct measured via the
ization established in the Decision on Principles for the
1 2
These test methods are under the jurisdiction of ASTM Committee F04 on For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Medical and Surgical Materials and Devices and are the direct responsibility of contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Subcommittee F04.25 on Spinal Devices. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved April 1, 2021. Published May 2021. Originally the ASTM website.
approved in 1996. Last previous edition approved in 2018 as F1717–18. DOI: The last approved version of this historical standard is referenced on
10.1520/F1717-21. www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1717 − 21
actuatorthatproducesapermanentangulardisplacementinthe left being positive when viewed posteriorly. The superior-
X-Y plane equal to 0.020 times the torsional aspect ratio (see inferior axis is Z with superior being positive.
Point A in Fig. 1).
3.2.8 displacement at 2 % offset yield (mm)—the displace-
3.2.3 block moment arm—the distance in the X direction in
ment of a construct measured via the actuator that produces a
the X-Y plane between the axis of the hinge pin and either (1)
permanent deformation equal to 0.020 times the active length
the center of the insertion point of an anchor (screws and
of the longitudinal element (see Point A in Fig. 1).
bolts), (2) the furthest point of contact between the block and
3.2.9 elastic angular displacement (degrees)—the angular
plate (anterior plates), or (3) the center point of attachment on
displacement at 2% offset yield (see PointAin Fig. 1) minus
the roll pin farthest from the hinge pin (hooks and wires).
the 2% offset angular displacement (see Point B in Fig. 1).
3.2.4 compressive or tensile bending stiffness (N/mm)—the
(The distance between Point A and Point B in Fig. 1.)
compressive or tensile bending yield force divided by elastic
3.2.10 elastic displacement (mm)—the displacement at 2%
displacement (see the initial slope of line BC in Fig. 1).
offset yield (see Point A in Fig. 1) minus the 2% offset
3.2.5 compressive or tensile bending ultimate load (N)—the
displacement (see Point B in Fig. 1). (The distance between
maximumcompressiveortensileforceintheX-Zplaneapplied
Point A and Point B in Fig. 1.)
toaspinalimplantassembly(seetheforceatPointEinFig.1).
3.2.11 failure—permanent deformation resulting from
Theultimateloadshouldbeafunctionofthedeviceandnotof
fracture, plastic deformation, or loosening beyond the ultimate
the load cell or testing machine.
displacement or loosening that renders the spinal implant
3.2.6 compressive or tensile bending yield load (N)—the
assembly ineffective or unable to adequately resist load.
compressive or tensile bending force in the X-Z plane neces-
sary to produce a permanent deformation equal to 0.020 times 3.2.12 fatigue life—the number of loading cycles, N,ofa
specified character that the spinal implant assembly sustains
the active length of the longitudinal element (see the force at
Point D in Fig. 1). before failure of a specified nature occurs (see Definitions
E1150).
3.2.7 coordinate system/axes—three orthogonal axes are
defined in Fig. 2 and Fig. 3. The anterior-posterior axis is X
3.2.13 insertion point of an anchor—the location where the
with positive being anterior. The medial-lateral axis is Y with
anchorisattachedtothetestblock.Theinsertionpointsshown
in Figs. 2-15 are to be adhered to if possible. In situations
where the design of the spinal implant assembly or the
manufacturer’s surgical instructions for installation dictate
otherwise, the attachment points may deviate from these
dimensions.
3.2.14 intended method of application—spinal implant as-
semblies contain different types of anchors. Each type of
anchor has an intended method of application to the spine.
3.2.15 intended spinal location—the anatomic region of the
spine intended for the application of the spinal implant
assembly.Spinalimplantassembliesaredevelopedforspecific
spinal locations such as the anterior cervical spine or the
posterior thoracolumbar, lumbar, and lumbosacral spine.
3.2.16 hingepin—thecylindricalrodconnectingatestblock
toasidesupport.Acervicalconstructissecuredwitha9.6mm
diameter pin and the thoracolumbar, lumbar, and lumbosacral
construct uses a 12.7 mm diameter pin.
3.2.17 longitudinal direction—the initial spatial orientation
parallel to the longitudinal element of the spinal implant
assembly. The longitudinal direction is generally in the
superior-inferior direction and, therefore, generally parallel to
the z axis.
3.2.18 maximum run-out load—the maximum load that can
be applied to a spinal implant assembly where all of the tested
constructs have withstood 5000000 cycles without a failure.
3.2.19 permanent deformation—the displacement (mm) or
angular displacement (degree) of the spinal implant construct
relative to the initial unloaded condition as measured via the
actuator after the applied load, moment, or torque has been
FIG. 1 Typical Load Displacement Curve or Torque Angulation
Curve removed.
F1717 − 21
FIG. 2 Typical Bilateral Construct Containing Bone Screws, Rods, and Set Screws
3.2.20 spinal implant assembly—a complete spinal implant 3.2.24 tightening torque—the specified torque that is ap-
configuration as intended for surgical use. A spinal implant plied to the various threaded fasteners of the spinal implant
assembly will contain anchors, interconnections, and longitu-
assembly.
dinalelementsandmaycontaintransverseelements(seeFig.4,
3.2.25 torsional aspect ratio—the active length of the lon-
Fig. 6, Fig. 8, Fig. 10, Fig. 12, and Fig. 14).
gitudinal element divided by the distance from the center of
3.2.21 spinal implant construct—a complete spinal implant
rotationtotheinsertionpointofananchor(forexample:inFig.
assembly attached to the appropriate test blocks.
41.14fora35-mmactivelength, X=30mmand Y=S⁄2mm,
3.2.22 test block—the component of the test apparatus for
whereS=12mm).
mountingthespinalimplantassembly.Aspecificdesignoftest
L L
blockisrequiredforeachintendedspinallocationandintended A 5 5 (1)
2 2 1/2
D ~x 1y !
method of application. Fig. 5, Fig. 7, Fig. 9, Fig. 11, Fig. 13,
where:
and Fig. 15 describe the recommended designs for the test
blocks; however, alternate designs can be used as long as
A = torsional aspect ratio,
equivalent performance is demonstrated. L = active length of longitudinal element,
D = distance to insertion point,
3.2.23 test block load point—the location on the test block
x = x distance to insertion point, and
at which the resultant load is transmitted from the test
y = y distance to insertion point.
apparatus.
F1717 − 21
FIG. 3 A Bilateral Hook, Rod, Screw, and Transverse Element Construct
3.2.26 torsional stiffness (N-m/degree)—the yield torque 3.2.30 ultimate displacement (mm)—the displacement asso-
(N-m) divided by elastic angular displacement (degrees) (the ciatedwiththeultimateload,ultimatebendingload,orultimate
initial slope of line BC in Fig. 1). torque (the displacement at Point F in Fig. 1).
3.2.27 torsional ultimate load (N-m)—the maximum torque 3.2.31 yield torque (N-m)—the torque in the X-Y plane
in the X-Y plane applied to a spinal implant assembly (the required to produce a permanent displacement of 0.020 times
torque at Point E in Fig. 1). The ultimate torque should be a the torsional aspect ratio (the torque at Point D in Fig. 1).
function of the device and not of the load cell or testing
3.2.32 zero displacement intercept (mm)—the intersection
machine.
of the straight line section of the load displacement curve and
3.2.28 two percent (2 %) offset angular displacement the zero load axis (the zero displacement reference Point 0 in
(degrees)—a permanent angular displacement in the X-Y plane Fig. 1).
measured via the actuator equal to 0.020 times the torsional
4. Summary of Test Methods
aspectratio(forexample:1.95°for1.70×0.02×180°⁄pi)(see
4.1 Similar test methods are proposed for the mechanical
Point B in Fig. 1).
evaluation of cervical spinal implant assemblies (see Fig. 4,
3.2.29 two percent (2 %) offset displacement (mm)—a per-
Fig.6,andFig.8)andthoracolumbar,lumbar,andlumbosacral
manent deformation measured via the actuator equal to 0.020
spinal implant assemblies (see Fig. 10, Fig. 12, and Fig. 14).
times the active length of the longitudinal element (for ex-
ample: 1.52 mm for a 76 mm active length of the longitudinal 4.2 Testing of the spinal implant assemblies will simulate a
element or 0.70 mm for 35 mm) (see Point B in Fig. 1). vertebrectomy model via a large gap between two Ultra High
F1717 − 21
FIG. 4 Cervical Unilateral Construct for Plates
Molecular Weight Polyethylene (UHMWPE) test blocks. The be evaluated with test configurations which simulate the
UHMWPE used to manufacture the test blocks should have a clinical requirements for the intended spinal location. The
tensile breaking strength equal to 40 6 3 MPa (see Test intended spinal locations are both anterior (see Fig. 4) and
Method D638). The UHMWPE test blocks (see Fig. 5, Fig. 7, posterior (see Fig. 6 and Fig. 8) surfaces of the cervical spine
Fig. 9, Fig. 11, Fig. 13, and Fig. 15) will eliminate the effects orbothanterior(seeFig.10)andposterior(seeFig.12andFig.
of the variability of bone properties and morphometry. Alter- 14) surfaces of the thoracolumbar, lumbar, and lumbosacral
nate designs of test blocks may be used as long as equivalent
spine.The block moment arm (see 6.6) for a test configuration
performance is demonstrated. depends on the intended spinal location. The cervical spine
configuration(seeFig.5,Fig.7,andFig.9)specifiesoneblock
4.3 Three static mechanical tests and one dynamic test will
moment arm, while a larger block moment arm (see Fig. 11,
evaluate the spinal implant assemblies. The three static me-
Fig.13,andFig.15)isspecifiedforthethoracolumbar,lumbar,
chanical tests are compression bending, tensile bending, and
and lumbosacral spine.
torsion.Thedynamictestisacompressionbendingfatiguetest.
It is the responsibility of the user of this standard to determine
4.5 The intended method of application of the spinal im-
which test(s) is (are) most appropriate for a particular spinal
plant assembly may vary for specific anatomic regions and
implant assembly.
clinicalindications.Spinalimplantassembliescontaindifferent
4.3.1 Assessment of cervical, thoracolumbar, lumbar, and
types of anchors. Each type of anchor has an intended method
lumbosacral spinal implant assemblies in torsion is not re-
of application to the spine. For example, one assembly may
quired for those assemblies consisting of hooks, cables, wires,
include anterior vertebral body screws and rods (see Fig. 2),
or rods in combination with vertebral body, pedicle, or sacral
while another assembly may contain posterior sacral screws,
screws.
hooks, rods, and transverse elements (see Fig. 3). The block
4.4 A specific clinical indication generally requires a spe- moment arm of a test configuration will be independent of the
cific spinal implant assembly. Spinal implant assemblies will intended method of application of a spinal implant assembly;
F1717 − 21
FIG. 5 Cervical Unilateral UHMWPE Block for Plates
therefore, the test data for different intended methods of saline drip, water, or a lubricant should be considered. The
application may be compared. maximum recommended frequency for this type of cyclic
testing should be 5 Hz.
5. Significance and Use
5.5 The location of the longitudinal elements is determined
5.1 Spinal implants are generally composed of several
by where the anchors are clinically placed against bony
components which, when connected together, form a spinal
structures. The perpendicular distance to the load direction
implant assembly. Spinal implant assemblies are designed to
(block moment arm) between the axis of a hinge pin and the
provide some stability to the spine while arthrodesis takes
anchor’s attachment points to a UHMWPE block is indepen-
place. These test methods outline standard materials and
dent of anchor type. The distance between the anchor’s
methods for the evaluation of different spinal implant assem-
attachment point to the UHMWPE block and the center of the
blies so that comparison between different designs may be
longitudinal element is a function of the interface design
facilitated.
betweenthescrew,hook,wire,cable,andsoforth,andtherod,
plate, and so forth.
5.2 These test methods are used to quantify the static and
dynamic mechanical characteristics of different designs of
5.6 During static torsion testing, the rotation direction
spinal implant assemblies. The mechanical tests are conducted
(clockwise or counter clockwise) may have an impact on the
in vitro using simplified load schemes and do not attempt to
results.
mimic the complex loads of the spine.
6. Apparatus
5.3 The loads applied to the spinal implant assemblies in
vivo will, in general, differ from the loading configurations 6.1 Test machines will conform to the requirements of
usedinthesetestmethods.Theresultsobtainedherecannotbe Practices E4.
useddirectlytopredict in-vivoperformance.Theresultscanbe
6.2 Thetestapparatusallowsmultipleloadingregimestobe
used to compare different component designs in terms of the
applied to all forms of spinal implant assemblies.Two pairs of
relative mechanical parameters.
side supports are mounted on the test machine (see Fig. 4, Fig.
5.4 Fatigue testing in a simulated body fluid or saline may 6, Fig. 8, Fig. 10, Fig. 12, and Fig. 14). One pair of side
cause fretting, corrosion, or lubricate the interconnections and supports attach to the actuator and the second to the load cell.
thereby affect the relative performance of tested devices. This A mounting plate for one of the sets of side support plates
test should be initially performed dry (ambient room condi- should be free to rotate about the Z axis for the compression
tions) for consistency. The effect of environment may be bending, tension bending, and fatigue tests. UHMWPE blocks
significant. Repeating all or part of these test methods in are connected to the side supports via hinge pins. All testing
simulated body fluid, saline (9 g NaCl per 1000 mL water), a will simulate a vertebrectomy model via a large gap between
F1717 − 21
FIG. 6 Cervical Bilateral Construct Test Setup for Screws or Bolts
FIG. 7 Cervical Bilateral UHMWPE Block for Screws or Bolts
F1717 − 21
FIG. 8 Cervical Bilateral Construct Test Setup for Hooks, Cables, or Wires
thetwoUHMWPEblocks.Selecttheappropriatedesignofthe contact an appropriate thickness of the UHMWPE. If screws
UHMWPE blocks (see Fig. 5, Fig. 7, Fig. 9, Fig. 11, Fig. 13, are used to mount the spinal construct to the test blocks (see
and Fig. 15) to facilitate testing of the spinal implant assembly Fig. 5, Fig. 7, Fig. 11, and Fig. 13), then the screws must be
inamannerthatsimulatesthespecificclinicalindicationatthe placed into UHMWPE inserts in the alternate design of test
intended spinal location. block.The diameter of the UHMWPE inserts must be equal to
or greater than three times the diameter of the screws.
6.3 The design of the UHMWPE blocks causes the plane
throughthespinalimplantassembliestobeparalleltotheplane 6.5 Ifthelocationsofthesuperioranchors,inferioranchors,
(the Y-Z plane) through the axes of the hinge pins. Align the orbothsetsofanchorsaredictatedbythelongitudinalelement
superior side supports and UHMWPE block with the inferior and are at different Z locations (a diagonal), then the set of
side supports and UHMWPE block. The center axis of each anchors should be centered above and below the standard
hinge pin should be perpendicular (60.5°) to and aligned location such that they maintain the average Z location. If the
(60.5 mm) with the load axis of the test machine. Center the anchors are secured into slots in the longitudinal element, then
testapparatusinthetestmachinesuchthatthelinethroughthe theyshouldbecentrallyplacedintheslotsandnotateitherend
mid-point (0, 0, Z1) of the inferior hinge pin’s axis and the to produce a worst-case scenario.
mid-point(0,0,Z2)ofthesuperiorhingepin’saxisiscollinear
6.6 The distance in the X direction between the axis of a
within 60.1 mm of the load and rotational axis of the test
hinge pin and the anchors’ attachment point should remain
machine’s actuator.
constant when comparing spinal implant assemblies. Spinal
6.4 Alternate designs of test blocks may be used as long as implant assemblies are designed for two intended spinal
equivalence is demonstrated. The solid UHMWPE test blocks locations having two unique block moment arms. The two
may be replaced with metal blocks with UHMWPE inserts of intendedspinallocationsarethecervicalspinalimplantsystem
appropriate size. Any surface or component of the spinal (see Fig. 4, Fig. 6, and Fig. 8) and the thoracolumbar, lumbar,
assemblywhichwouldcontactthesolidUHMWPEshouldalso and lumbosacral spinal implant system (see Fig. 10, Fig. 12,
F1717 − 21
FIG. 9 Cervical Bilateral UHMWPE Block for Hooks, Cables, or Wires
and Fig. 14). The test configurations for the cervical spinal wires, or cables around the steel roll pins means that the test
implant system have a block moment arm equal to 30.0 mm. configuration would be a mechanism. Therefore, the testing of
The thoracolumbar, lumbar, and lumbosacral test configura- hooks, wires, and cables necessitates that the modified UHM-
tions have a 40-mm block moment arm. WPE block must not rotate.
6.7 The UHMWPE blocks have been designed to provide
6.10 The relative location (X direction versus Z direction)
similar block moment arms regardless of the anchor being
between the hinge pin and the insertion point of an anchor
tested. Different spinal implant assemblies have different
produces minimal variation in the block moment arm. The
intended methods of application to the UHMWPE blocks. The
variation in the block moment arm is dependent on the
locations of the longitudinal elements are determined by the
direction of rotation of the UHMWPE blocks. The variation is
design of anchors and interconnections. The load capacity of
minimized by having the hinge pins in the UHMWPE blocks
the spinal construct would be a function of the designs of the
rotatepasttheanchorsasthetestprogresses.Positionthehinge
interconnections, anchors, and longitudinal elements but
pinsinternaltotheanchorsduringthetensionbendingtest(not
should not be a function of the test apparatus.
shown). Position the hinge pins external to the anchors during
the compression bending, torsion, and fatigue tests (see Fig. 4,
6.8 The hinge pin in the test configuration allows the same
Fig. 6, Fig. 8, Fig. 10, Fig. 12, and Fig. 14).
test apparatus to be used for the static compression bending
test,statictensilebendingtest,andstatictorsiontestaswellas
6.11 The thoracolumbar, lumbar, and lumbosacral test ap-
the compression bending fatigue test. The UHMWPE blocks
paratus have a recommended active length of the longitudinal
are allowed to rotate around the Y-axis of the hinge pin during
element equal to 76.0 mm and based on the work of Cunning-
the compression bending, tensile bending, and fatigue tests.
ham et al. (1). The recommended active length of the
longitudinal element for the cervical spinal implant system is
6.9 Modified bilateral UHMWPE blocks (see Fig. 8, Fig. 9,
35.0mm.Ifthelongitudinalelementhasfixedspacingsandthe
Fig. 14, and Fig. 15) have been developed for testing hooks,
recommendedactivelengthcannotbeachieved,thenselectthe
wires, or cables. Steel roll pins are placed into the modified
longitudinal element that is nearest the recommended active
blocks such that the outer surfaces of the roll pins are parallel
length. The active length should be constant for all constructs
to the front surfaces of the standard bilateral UHMWPE block
used in comparative testing.
(see Fig. 6, Fig. 7, Fig. 12, and Fig. 13). Hooks, wires, and
cables are not fully constrained (semi-rigid) fixation devices
because they cannot transfer bending moments in the three
axes. The combination of the rotation of the modified UHM-
The boldface numbers in parentheses refer to a list of references at the end of
WPE block on the hinge pin and the rotation of the hooks, this standard.
F1717 − 21
FIG. 10 Lumbar Unilateral Construct for Plates
6.12 The testing machine and the apparatus used in the 7. Sampling
static compression bending, static tension bending, and com-
7.1 All components in the spinal implant assembly shall be
pression bending fatigue tests apply load in the Z direction
previously unused parts only. Implants shall not be retested.
withoutconstrainingrotationinthe X-Yplane.Thehingepinin
7.2 Use the UHMWPE test blocks for only one test. The
the apparatus allows rotation in the X-Z plane during the static
UHMWPE used to manufacture the test blocks should have a
compression bending, static tension bending, and compression
tensile breaking strength equal to 40 6 3 MPa (see Test
bending fatigue tests. The compression bending fatigue test
MethodD638).Whenalternatedesignsoftestblocksareused,
will use the same test configuration as static compressive
then all UHMWPE inserts should be replaced after each test.
bending.
Alternate designs of test blocks which include steel roll pins
(see Fig. 9 and Fig. 15) should replace the steel roll pins and
6.13 The testing machine or the apparatus used in the static
UHMWPE inserts for the hinge after each test.
torsiontestappliestorqueabouttheZaxiswithoutconstraining
displacement in the Z direction. Aluminum blocks shall be
7.3 Labelandmaintainthetestconstructsaccordingtogood
placed in the apparatus to prevent rotation in the X-Z plane
laboratory practice. Do not disassemble the test construct after
during the static torsion tests. The total clearance between an
testing unless disassembly is necessary to evaluate failure
aluminumblock,anUHMWPEblock,andabaseplatewillnot
surfaces, interconnections, corrosion, or loosening surfaces.
exceed 0.10 mm. Photograph the construct prior to disassembly.
F1717 − 21
FIG. 11 Lumbar Unilateral UHMWPE Block for Plates
7.4 All static tests should have a minimum of five samples. 8. Procedure
Examination of each load-displacement curve may reveal a
8.1 Procedure for Static Tests—Evaluate only the load
laxityinthefixture.Afterthelaxityhasbeenremoved,thenthe
parameters in the relevant direction.
initial linear portion of the curve will define the straight line
8.1.1 Static Compression Bending Test:
sectionoftheload-displacementcurves.Theintersectionofthe
8.1.1.1 Select the appropriate UHMWPE blocks for the
straight line section and zero load axis is the zero load
spinal implant assembly as previously described.
displacement (Point 0).
8.1.1.2 Install the anchors according to the manufacturer’s
7.5 The results of the fatigue testing will provide a curve of instructions. When used as part of a rod/screw construct,
cyclical compression load or compression bending load versus
variable axis screw anchors shall be inserted in the UHMWPE
the number of cycles to failure and establish the endurance blocks in a manner that prevents the impingement of any
limitoftheconstruct.Initialfatigueloadingconditionsmaybe
potentially pivoting or rotating features of the anchor against
determinedprimarilybytheuser’sexperience.Intheabsence the test block. This may be achieved by inserting the anchor
of such experience, initial fatigue loads corresponding to 75, suchthat,atfullangulationofanyofthepotentiallypivotingor
50,and/or25%oftheyieldloadasdeterminedinthestatictest rotating features, clearance is always maintained with respect
may serve as a starting point for establishing the fatigue to the test block. If one modified bilateral UHMWPE block is
characteristics.Ifaspecimendoesnotfailby5000000cycles, used, then place an aluminum spacer block between the
then testing of that component should be considered run-out. modified UHMWPE block and the base plate to stop rotation
The precision of the endurance limit shall be established by around the hinge pin. A degree of freedom is eliminated in a
similar manner to the axial compression test. If the spinal
ensuringthatthelowestloadthatresultsinafailedconstructis
implant assembly requires two sets of modified bilateral
notgreaterthan1.25×thehighestestablishedrun-outload.For
UHMWPE blocks and aluminum spacer blocks, then it is
example,ifthehighestestablishedrun-outloadis100.0Nthen
equivalent to an axial compression test.
the lowest load that results in a failed construct shall not be
greater than 125.0 N. A minimum of two constructs shall be 8.1.1.3 Place the UHMWPE blocks into the test apparatus
tested at the highest established run-out load. The final sample such that the position of the hinge pins are external to the
size is recommended by Practice E739. anchors (the hinge pin in the superior block is more superior
F1717 − 21
FIG. 12 Lumbar Bilateral Construct for Screws or Bolts
than the screw, hook, and so forth). Secure the UHMWPE variable axis screw anchors shall be inserted in the UHMWPE
blocks with hinge pins. If one modified bilateral UHMWPE blocks in a manner that prevents the impingement of any
block is used to test hooks, wires, or cables, then place it
potentially pivoting or rotating features of the anchor against
superiorly.
the test block. This may be achieved by inserting the anchor
8.1.1.4 Complete the spinal implant assembly in a standard
suchthat,atfullangulationofanyofthepotentiallypivotingor
construct (see Fig. 4, Fig. 6, Fig. 8, Fig. 10, Fig. 12, and Fig.
rotating features, clearance is always maintained with respect
14) or a hybrid construct (see Fig. 3). Set the active length of
to the test block. If one modified bilateral UHMWPE block is
thelongitudinalelementfortheintendedspinallocation.Apply
used, then place an aluminum spacer block between the
alltightening,crimping,orlockingmechanismsasspecifiedby
modified UHMWPE block and the base plate to stop rotation
the manufacturer.
around the hinge pin. A degree of freedom is eliminated in a
8.1.1.5 Loadthetestapparatusatarateuptoamaximumof
similar manner to the axial compression test. If the spinal
25 mm/min.
implant assembly requires two sets of modified bilateral
8.1.1.6 Record the load displacement curves. Establish the
UHMWPE blocks and aluminum spacer blocks, then it is
displacement at 2% offset yield (mm), elastic displacement
equivalent to an axial tension test.
(mm), compressive bending yield load (N), compressive bend-
8.1.2.3 Place the UHMWPE blocks into the test apparatus
ing stiffness (N/mm), compressive bending ultimate displace-
such that the positions of the hinge pins are internal to the
ment (mm), and compressive bending ultimate load (N).
anchors (the hinge pin in the superior block is more inferior
8.1.2 Static Tension Bending Test:
than the screw, hook, and so forth). Secure the UHMWPE
8.1.2.1 Select the appropriate UHMWPE blocks for the
blocks with hinge pins. If one modified bilateral UHMWPE
spinal implant assembly as previously described.
block is used to test hooks, wires, or cables, then place it
8.1.2.2 Install the anchors according to the manufacturer’s
instr
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F1717 − 18 F1717 − 21
Standard Test Methods for
Spinal Implant Constructs in a Vertebrectomy Model
This standard is issued under the fixed designation F1717; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 These test methods cover the materials and methods for the static and fatigue testing of spinal implant assemblies in a
vertebrectomy model. The test materials for most combinations of spinal implant components can be specific, depending on the
intended spinal location and intended method of application to the spine.
1.2 These test methods are intended to provide a basis for the mechanical comparison among past, present, and future spinal
implant assemblies. They allow comparison of spinal implant constructs with different intended spinal locations and methods of
application to the spine. These test methods are not intended to define levels of performance, since sufficient knowledge is not
available to predict the consequences of the use of a particular device.
1.3 These test methods set out guidelines for load types and methods of applying loads. Methods for three static load types and
one fatigue test are defined for the comparative evaluation of spinal implant assemblies.
1.4 These test methods establish guidelines for measuring displacements, determining the yield load, and evaluating the stiffness
and strength of the spinal implant assembly.
1.5 Some spinal constructs may not be testable in all test configurations.
1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D638 Test Method for Tensile Properties of Plastics
E4 Practices for Force Verification of Testing Machines
These test methods are under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and are the direct responsibility of Subcommittee
F04.25 on Spinal Devices.
Current edition approved June 1, 2018April 1, 2021. Published August 2018May 2021. Originally approved in 1996. Last previous edition approved in 20152018 as
F1717F1717 – 18.–15. DOI: 10.1520/F1717-18.10.1520/F1717-21.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1717 − 21
E6 Terminology Relating to Methods of Mechanical Testing
E177 Practice for Use of the Terms Precision and Bias in ASTM Test Methods
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E739 Practice for Statistical Analysis of Linear or Linearized Stress-Life (S-N) and Strain-Life (ε-N) Fatigue Data
E1150 Definitions of Terms Relating to Fatigue (Withdrawn 1996)
F543 Specification and Test Methods for Metallic Medical Bone Screws
F1582 Terminology Relating to Spinal Implants
F1798 Test Method for Evaluating the Static and Fatigue Properties of Interconnection Mechanisms and Subassemblies Used
in Spinal Arthrodesis Implants
F2077 Test Methods For Intervertebral Body Fusion Devices
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms relating to these test methods, see Terminology E6, Terminology F1582, and Definitions E1150.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 active length of the longitudinal element—the straight line distance between the center of attachment of the superior anchor
and the center of attachment of the inferior anchor.
3.2.2 angular displacement at 2 % offset yield (degrees)—the angular displacement of a construct measured via the actuator that
produces a permanent angular displacement in the X-Y plane equal to 0.020 times the torsional aspect ratio (see Point A in Fig.
1).
FIG. 1 Typical Load Displacement Curve or Torque Angulation Curve
The last approved version of this historical standard is referenced on www.astm.org.
F1717 − 21
3.2.3 block moment arm—the distance in the X direction in the XYX-Y plane between the axis of the hinge pin and either (1) the
center of the insertion point of an anchor (screws and bolts), (2) the furthest point of contact between the block and plate (anterior
plates), or (3) the center point of attachment on the roll pin farthest from the hinge pin (hooks and wires).
3.2.4 compressive or tensile bending stiffness (N/mm)—the compressive or tensile bending yield force divided by elastic
displacement (see the initial slope of line BC in Fig. 1).
3.2.5 compressive or tensile bending ultimate load (N)—the maximum compressive or tensile force in the X-Z plane applied to
a spinal implant assembly (see the force at Point E in Fig. 1). The ultimate load should be a function of the device and not of the
load cell or testing machine.
3.2.6 compressive or tensile bending yield load (N)—the compressive or tensile bending force in the X-Z plane necessary to
produce a permanent deformation equal to 0.020 times the active length of the longitudinal element (see the force at Point D in
Fig. 1).
3.2.7 coordinate system/axes—three orthogonal axes are defined in Fig. 2 and Fig. 3. The anterior-posterior axis is X with positive
being anterior. The medial-lateral axis is Y with left being positive when viewed posteriorly. The superior-inferior axis is Z with
superior being positive.
3.2.8 displacement at 2 % offset yield (mm)—the displacement of a construct measured via the actuator that produces a permanent
deformation equal to 0.020 times the active length of the longitudinal element (see Point A in Fig. 1).
3.2.9 elastic angular displacement (degrees)—the angular displacement at 2 % offset yield (see Point A in Fig. 1) minus the 2 %
offset angular displacement (see Point B in Fig. 1). (The distance between Point A and Point B in Fig. 1.)
3.2.10 elastic displacement (mm)—the displacement at 2 % offset yield (see Point A in Fig. 1) minus the 2 % offset displacement
(see Point B in Fig. 1). (The distance between Point A and Point B in Fig. 1.)
3.2.11 failure—permanent deformation resulting from fracture, plastic deformation, or loosening beyond the ultimate displace-
ment or loosening that renders the spinal implant assembly ineffective or unable to adequately resist load.
3.2.12 fatigue life—the number of loading cycles, N, of a specified character that the spinal implant assembly sustains before
failure of a specified nature occurs (see Definitions E1150).
3.2.13 insertion point of an anchor—the location where the anchor is attached to the test block. The insertion points shown in Figs.
2-15 are to be adhered to if possible. In situations where the design of the spinal implant assembly or the manufacturer’s surgical
instructions for installation dictate otherwise, the attachment points may deviate from these dimensions.
3.2.14 intended method of application—spinal implant assemblies contain different types of anchors. Each type of anchor has an
intended method of application to the spine.
3.2.15 intended spinal location—the anatomic region of the spine intended for the application of the spinal implant assembly.
Spinal implant assemblies are developed for specific spinal locations such as the anterior cervical spine or the posterior
thoracolumbar, lumbar, and lumbosacral spine.
3.2.16 hinge pin—the cylindrical rod connecting a test block to a side support. A cervical construct is secured with a 9.6 mm
diameter pin and the thoracolumbar, lumbar, and lumbosacral construct uses a 12.7 mm diameter pin.
3.2.17 longitudinal direction—the initial spatial orientation parallel to the longitudinal element of the spinal implant assembly. The
longitudinal direction is generally in the superior-inferior direction and, therefore, generally parallel to the z axis.
3.2.18 maximum run-out load—the maximum load that can be applied to a spinal implant assembly where all of the tested
constructs have withstood 5 000 000 cycles without a failure.
F1717 − 21
FIG. 2 Typical Bilateral Construct Containing Bone Screws, Rods, and Set Screws
3.2.19 permanent deformation—the displacement (mm) or angular displacement (degree) of the spinal implant construct relative
to the initial unloaded condition as measured via the actuator after the applied load, moment, or torque has been removed.
3.2.20 spinal implant assembly—a complete spinal implant configuration as intended for surgical use. A spinal implant assembly
will contain anchors, interconnections, and longitudinal elements and may contain transverse elements (see Fig. 4, Fig. 6, Fig. 8,
Fig. 10, Fig. 12, and Fig. 14).
3.2.21 spinal implant construct—a complete spinal implant assembly attached to the appropriate test blocks.
3.2.22 test block—the component of the test apparatus for mounting the spinal implant assembly. A specific design of test block
is required for each intended spinal location and intended method of application. Fig. 5, Fig. 7, Fig. 9, Fig. 11, Fig. 13, and Fig.
15 describe the recommended designs for the test blocks; however, alternate designs can be used as long as equivalent performance
is demonstrated.
3.2.23 test block load point—the location on the test block at which the resultant load is transmitted from the test apparatus.
F1717 − 21
FIG. 3 A Bilateral Hook, Rod, Screw, and Transverse Element Construct
3.2.24 tightening torque—the specified torque that is applied to the various threaded fasteners of the spinal implant assembly.
3.2.25 torsional aspect ratio—the active length of the longitudinal element divided by the distance from the center of rotation to
the insertion point of an anchor (for example: in Fig. 24 1.701.14 for a 76-mm35-mm active length, X = 40 = 30 mm and
Y = 40 = S ⁄2 mm). ⁄2 mm, where S = 12 mm).
L L
A 5 5 (1)
2 2 1/2
D ~x 1y !
where:
A = torsional aspect ratio,
L = active length of longitudinal element,
D = distance to insertion point,
x = x distance to insertion point, and
y = y distance to insertion point.
3.2.26 torsional stiffness (N-m/degree)—the yield torque (N-m) divided by elastic angular displacement (degrees) (the initial slope
of line BC in Fig. 1).
F1717 − 21
FIG. 4 Cervical Unilateral Construct for Plates
3.2.27 torsional ultimate load (N-m)—the maximum torque in the X-Y plane applied to a spinal implant assembly (the torque at
Point E in Fig. 1). The ultimate torque should be a function of the device and not of the load cell or testing machine.
3.2.28 two percent (2 %) offset angular displacement (degrees)—a permanent angular displacement in the X-Y plane measured via
the actuator equal to 0.020 times the torsional aspect ratio (for example: 1.95° for 1.70 × 0.02 × 180° ⁄pi) (see Point B in Fig. 1).
3.2.29 two percent (2 %) offset displacement (mm)—a permanent deformation measured via the actuator equal to 0.020 times the
active length of the longitudinal element (for example: 1.52 mm for a 76 mm active length of the longitudinal element or 0.70 mm
for 35 mm) (see Point B in Fig. 1).
3.2.30 ultimate displacement (mm)—the displacement associated with the ultimate load, ultimate bending load, or ultimate torque
(the displacement at Point F in Fig. 1).
3.2.31 yield torque (N-m)—the torque in the X-Y plane required to produce a permanent displacement of 0.020 times the torsional
aspect ratio (the torque at Point D in Fig. 1).
3.2.32 zero displacement intercept (mm)—the intersection of the straight line section of the load displacement curve and the zero
load axis (the zero displacement reference Point 0 in Fig. 1).
F1717 − 21
FIG. 5 Cervical Unilateral UHMWPE Block for Plates
4. Summary of Test Methods
4.1 Similar test methods are proposed for the mechanical evaluation of cervical spinal implant assemblies (see Fig. 4, Fig. 6, and
Fig. 8) and thoracolumbar, lumbar, and lumbosacral spinal implant assemblies (see Fig. 10, Fig. 12, and Fig. 14).
4.2 Testing of the spinal implant assemblies will simulate a vertebrectomy model via a large gap between two Ultra High
Molecular Weight Polyethylene (UHMWPE) test blocks. The UHMWPE used to manufacture the test blocks should have a tensile
breaking strength equal to 40 6 3 MPa (see Specification Test Method D638). The UHMWPE test blocks (see Fig. 5, Fig. 7, Fig.
9, Fig. 11, Fig. 13, and Fig. 15) will eliminate the effects of the variability of bone properties and morphometry. Alternate designs
of test blocks may be used as long as equivalent performance is demonstrated.
4.3 Three static mechanical tests and one dynamic test will evaluate the spinal implant assemblies. The three static mechanical
tests are compression bending, tensile bending, and torsion. The dynamic test is a compression bending fatigue test. It is the
responsibility of the user of this standard to determine which test(s) is (are) most appropriate for a particular spinal implant
assembly.
4.3.1 Assessment of cervical, thoracolumbar, lumbar, and lumbosacral spinal implant assemblies in torsion is not required for
those assemblies consisting of hooks, cables, wires, or rods in combination with vertebral body, pedicle, or sacral screws.
4.4 A specific clinical indication generally requires a specific spinal implant assembly. Spinal implant assemblies will be evaluated
with test configurations which simulate the clinical requirements for the intended spinal location. The intended spinal locations are
both anterior (see Fig. 4) and posterior (see Fig. 6 and Fig. 8) surfaces of the cervical spine or both anterior (see Fig. 10) and
posterior (see Fig. 12 and Fig. 14) surfaces of the thoracolumbar, lumbar, and lumbosacral spine. The block moment arm (see 6.6)
for a test configuration depends on the intended spinal location. The cervical spine configuration (see Fig. 5, Fig. 7, and Fig. 9)
specifies one block moment arm, while a larger block moment arm (see Fig. 11, Fig. 13, and Fig. 15) is specified for the
thoracolumbar, lumbar, and lumbosacral spine.
4.5 The intended method of application of the spinal implant assembly may vary for specific anatomic regions and clinical
indications. Spinal implant assemblies contain different types of anchors. Each type of anchor has an intended method of
F1717 − 21
FIG. 6 Cervical Bilateral Construct Test Setup for Screws or Bolts
FIG. 7 Cervical Bilateral UHMWPE Block for Screws or Bolts
F1717 − 21
FIG. 8 Cervical Bilateral Construct Test Setup for Hooks, Cables, or Wires
application to the spine. For example, one assembly may include anterior vertebral body screws and rods (see Fig. 2), while another
assembly may contain posterior sacral screws, hooks, rods, and transverse elements (see Fig. 3). The block moment arm of a test
configuration will be independent of the intended method of application of a spinal implant assembly; therefore, the test data for
different intended methods of application may be compared.
5. Significance and Use
5.1 Spinal implants are generally composed of several components which, when connected together, form a spinal implant
assembly. Spinal implant assemblies are designed to provide some stability to the spine while arthrodesis takes place. These test
methods outline standard materials and methods for the evaluation of different spinal implant assemblies so that comparison
between different designs may be facilitated.
5.2 These test methods are used to quantify the static and dynamic mechanical characteristics of different designs of spinal implant
assemblies. The mechanical tests are conducted in vitro using simplified load schemes and do not attempt to mimic the complex
loads of the spine.
5.3 The loads applied to the spinal implant assemblies in vivo will, in general, differ from the loading configurations used in these
test methods. The results obtained here cannot be used directly to predict in vivoin-vivo performance. The results can be used to
compare different component designs in terms of the relative mechanical parameters.
5.4 Fatigue testing in a simulated body fluid or saline may cause fretting, corrosion, or lubricate the interconnections and thereby
affect the relative performance of tested devices. This test should be initially performed dry (ambient room conditions) for
F1717 − 21
FIG. 9 Cervical Bilateral UHMWPE Block for Hooks, Cables, or Wires
consistency. The effect of environment may be significant. Repeating all or part of these test methods in simulated body fluid, saline
(9 g NaCl per 1000 mL water), a saline drip, water, or a lubricant should be considered. The maximum recommended frequency
for this type of cyclic testing should be 5 Hz.
5.5 The location of the longitudinal elements is determined by where the anchors are clinically placed against bony structures. The
perpendicular distance to the load direction (block moment arm) between the axis of a hinge pin and the anchor’s attachment-points
attachment points to a UHMWPE block is independent of anchor-type. anchor type. The distance between the anchor’s attachment
point to the UHMWPE block and the center of the longitudinal element is a function of the interface design between the screw,
hook, wire, cable, and so forth, and the rod, plate, and so forth.
5.6 During static torsion testing, the rotation direction (clockwise or counter clockwise) may have an impact on the results.
6. Apparatus
6.1 Test machines will conform to the requirements of Practices E4.
6.2 The test apparatus allows multiple loading regimes to be applied to all forms of spinal implant assemblies. Two pairs of side
supports are mounted on the test machine (see Fig. 4, Fig. 6, Fig. 8, Fig. 10, Fig. 12, and Fig. 14). One pair of side supports attach
to the actuator and the second to the load cell. A mounting plate for one of the sets of side support plates should be free to rotate
about the Z axis for the compression bending, tension bending, and fatigue tests. UHMWPE blocks are connected to the side
supports via hinge pins. All testing will simulate a vertebrectomy model via a large gap between the two UHMWPE blocks. Select
the appropriate design of the UHMWPE blocks (see Fig. 5, Fig. 7, Fig. 9, Fig. 11, Fig. 13, and Fig. 15) to facilitate testing of the
spinal implant assembly in a manner that simulates the specific clinical indication at the intended spinal location.
6.3 The design of the UHMWPE blocks causes the plane through the spinal implant assemblies to be parallel to the plane (the
Y-Z plane) through the axes of the hinge pins. Align the superior side supports and UHMWPE block with the inferior side supports
and UHMWPE block. The center axis of each hinge pin should be perpendicular (60.5°) to and aligned (60.5 mm) with the load
axis of the test machine. Center the test apparatus in the test machine such that the line through the mid-point (0, 0, Z1) of the
F1717 − 21
FIG. 10 Lumbar Unilateral Construct for Plates
inferior hinge pin’s axis and the mid-point (0, 0, Z2) of the superior hinge pin’s axis is collinear within 60.1 mm of the load and
rotational axis of the test machine’s actuator.
6.4 Alternate designs of test blocks may be used as long as equivalence is demonstrated. The solid UHMWPE test blocks may
be replaced with metal blocks with UHMWPE inserts of appropriate size. Any surface or component of the spinal assembly which
would contact the solid UHMWPE should also contact an appropriate thickness of the UHMWPE. If screws are used to mount
the spinal construct to the test blocks (see Fig. 5, Fig. 7, Fig. 11, and Fig. 13), then the screws must be placed into UHMWPE
inserts in the alternate design of test block. The diameter of the UHMWPE inserts must be equal to or greater than three times the
diameter of the screws.
6.5 If the locations of the superior anchors, inferior anchors, or both sets of anchors are dictated by the longitudinal element and
are at different Z locations (a diagonal), then the set of anchors should be centered above and below the standard location such
that they maintain the average Z location. If the anchors are secured into slots in the longitudinal element, then they should be
centrally placed in the slots and not at either end to produce a worst-case scenario.
6.6 The distance in the X direction between the axis of a hinge pin and the anchors’ attachment point should remain constant when
comparing spinal implant assemblies. Spinal implant assemblies are designed for two intended spinal locations having two unique
block moment arms. The two intended spinal locations are the cervical spinal implant system (see Fig. 4, Fig. 6, and Fig. 8) and
F1717 − 21
FIG. 11 Lumbar Unilateral UHMWPE Block for Plates
the thoracolumbar, lumbar, and lumbosacral spinal implant system (see Fig. 10, Fig. 12, and Fig. 14). The test configurations for
the cervical spinal implant system have a block moment arm equal to 30.0 mm. The thoracolumbar, lumbar, and lumbosacral test
configurations have a 40-mm block moment arm.
6.7 The UHMWPE blocks have been designed to provide similar block moment arms regardless of the anchor being tested.
Different spinal implant assemblies have different intended methods of application to the UHMWPE blocks. The locations of the
longitudinal elements are determined by the design of anchors and interconnections. The load capacity of the spinal construct
would be a function of the designs of the interconnections, anchors, and longitudinal elements but should not be a function of the
test apparatus.
6.8 The hinge pin in the test configuration allows the same test apparatus to be used for the static compression bending test, static
tensile bending test, and static torsion test as well as the compression bending fatigue test. The UHMWPE blocks are allowed to
rotate around the Y-axis of the hinge pin during the compression bending, tensile bending, and fatigue tests.
6.9 Modified bilateral UHMWPE blocks (see Fig. 8, Fig. 9, Fig. 14, and Fig. 15) have been developed for testing hooks, wires,
or cables. Steel roll pins are placed into the modified blocks such that the outer surfaces of the roll pins are parallel to the front
surfaces of the standard bilateral UHMWPE block (see Fig. 6, Fig. 7, Fig. 12, and Fig. 13). Hooks, wires, and cables are not fully
constrained (semi-rigid) fixation devices because they cannot transfer bending moments in the three axes. The combination of the
rotation of the modified UHMWPE block on the hinge pin and the rotation of the hooks, wires, or cables around the steel roll pins
means that the test configuration would be a mechanism. Therefore, the testing of hooks, wires, and cables necessitates that the
modified UHMWPE block must not rotate.
6.10 The relative location (X direction versus Z direction) between the hinge pin and the insertion point of an anchor produces
minimal variation in the block moment arm. The variation in the block moment arm is dependent on the direction of rotation of
the UHMWPE blocks. The variation is minimized by having the hinge pins in the UHMWPE blocks rotate past the anchors as the
F1717 − 21
FIG. 12 Lumbar Bilateral Construct for Screws or Bolts
test progresses. Position the hinge pins internal to the anchors during the tension bending test (not shown). Position the hinge pins
external to the anchors during the compression bending, torsion, and fatigue tests (see Fig. 4, Fig. 6, Fig. 8, Fig. 10, Fig. 12, and
Fig. 14).
6.11 The thoracolumbar, lumbar, and lumbosacral test apparatus have a recommended active length of the longitudinal element
equal to 76.0 mm and based on the work of Cunningham,Cunningham et alal. (1). The recommended active length of the
longitudinal element for the cervical spinal implant system is 35.0 mm. If the longitudinal element has fixed spacings and the
recommended active length cannot be achieved, then select the longitudinal element that is nearest the recommended active length.
The active length should be constant for all constructs used in comparative testing.
6.12 The testing machine and the apparatus used in the static compression bending, static tension bending, and compression
bending fatigue tests apply load in the Z direction without constraining rotation in the X-Y plane. The hinge pin in the apparatus
allows rotation in the X-Z plane during the static compression bending, static tension bending, and compression bending fatigue
tests. The compression bending fatigue test will use the same test configuration as static compressive bending.
6.13 The testing machine or the apparatus used in the static torsion test applies torque about the Z axis without constraining
displacement in the Z direction. Aluminum blocks shall be placed in the apparatus to prevent rotation in the X-Z plane during the
static torsion tests. The total clearance between an aluminum block, an UHMWPE block, and a base plate will not exceed 0.10
mm.
The boldface numbers in parentheses refer to a list of references at the end of this standard.
F1717 − 21
FIG. 13 Lumbar Bilateral UHMWPE Block for Screws or Bolts
7. Sampling
7.1 All components in the spinal implant assembly shall be previously unused parts only. Implants shall not be retested.
7.2 Use the UHMWPE test blocks for only one test. The UHMWPE used to manufacture the test blocks should have a tensile
breaking strength equal to 40 6 3 MPa (see Test Method D638). When alternate designs of test blocks are used, then all UHMWPE
inserts should be replaced after each test. Alternate designs of test blocks which include steel roll pins (see Fig. 9 and Fig. 15)
should replace the steel roll pins and UHMWPE inserts for the hinge after each test.
7.3 Label and maintain the test constructs according to good laboratory practice. Do not disassemble the test construct after testing
unless disassembly is necessary to evaluate failure surfaces, interconnections, corrosion, or loosening surfaces. Photograph the
construct prior to disassembly.
7.4 All static tests should have a minimum of five samples. Examination of each load-displacement curve may reveal a laxity in
the fixture. After the laxity has been removed, then the initial linear portion of the curve will define the straight line section of the
load-displacement curves. The intersection of the straight line section and zero load axis is the zero load displacement (Point 0).
7.5 The results of the fatigue testing will provide a curve of cyclical compression load or compression bending load versus the
number of cycles to failure and establish the endurance limit of the construct. Initial fatigue loading conditions may be determined
primarily by the user’s experience. In the absence of such experience, initial fatigue loads corresponding to 75, 50, and/or 25 %
of the yield load as determined in the static test may serve as a starting point for establishing the fatigue characteristics. If a
specimen does not fail by 5 000 000 cycles, then testing of that component should be considered run-out. The precision of the
endurance limit shall be established by ensuring that the lowest load that results in a failed construct is not greater than 1.25× the
highest established run-out load. For example, if the highest established run-out load is 100.0 N then the lowest load that results
in a failed construct shall not be greater than 125.0 N. A minimum of two constructs shall be tested at the highest established
run-out load. The final sample size is recommended by Practice E739.
8. Procedure
8.1 Procedure for Static Tests—Evaluate only the load parameters in the relevant direction.
F1717 − 21
FIG. 14 Lumbar Bilateral Construct for Hooks, Cables, or Wires
8.1.1 Static Compression Bending Test:
8.1.1.1 Select the appropriate UHMWPE blocks for the spinal implant assembly as previously described.
8.1.1.2 Install the anchors according to the manufacturer’s instructions. When used as part of a rod/screw construct, variable axis
screw anchors shall be inserted in the UHMWPE blocks in a manner that prevents the impingement of any potentially pivoting
or rotating features of the anchor against the test block. This may be achieved by inserting the anchor such that, at full angulation
of any of the potentially pivoting or rotating features, clearance is always maintained with respect to the test block. If one modified
bilateral UHMWPE block is used, then place an aluminum spacer block between the modified UHMWPE block and the base plate
to stop rotation around the hinge pin. A degree of freedom is eliminated in a similar manner to the axial compression test. If the
spinal implant assembly requires two sets of modified bilateral UHMWPE blocks and aluminum spacer blocks, then it is equivalent
to an axial compression test.
8.1.1.3 Place the UHMWPE blocks into the test apparatus such that the position of the hinge pins are external to the anchors (the
hinge pin in the superior block is more superior than the screw, hook, and so forth). Secure the UHMWPE blocks with hinge pins.
If one modified bilateral UHMWPE block is used to test hooks, wires, or cables, then place it superiorly.
8.1.1.4 Complete the spinal implant assembly in a standard construct (see Fig. 4, Fig. 6, Fig. 8, Fig. 10, Fig. 12, and Fig. 14) or
a hybrid construct (see Fig. 3). Set the active length of the longitudinal element for the intended spinal location. Apply all
tightening, crimping, or locking mechanisms as specified by the manufacturer.
F1717 − 21
FIG. 15 Lumbar Bilateral UHMWPE Block for Hooks, Cables, or Wires
8.1.1.5 Load the test apparatus at a rate up to a maximum of 25 mm/min.
8.1.1.6 Record the load displacement curves. Establish the displacement at 2 % offset yield (mm), elastic displacement (mm),
compressive bending yield load (N), compressive bending stiffness (N/mm), compressive bending ultimate displacement
(mm)(mm), and compressive bending ultimate load (N).
8.1.2 Static Tension Bending Test:
8.1.2.1 Select the appropriate UHMWPE blocks for the spinal implant assembly as previously described.
8.1.2.2 Install the anchors according to the manufacturer’s instructions. When used as part of a rod/screw construct, variable axis
screw anchors shall be inserted in the UHMWPE blocks in a manner that prevents the impingement of any potentially pivoting
or rotating features of the anchor against the test block. This may be achieved by inserting the anchor such that, at full angulation
of any of the potentially pivoting or rotating features, clearance is always maintained with respect to the test block. If one modified
bilateral UHMWPE block is used, then place an aluminum spacer block between the modified UHMWPE block and the base plate
to stop rotation around the hinge pin. A degree of freedom is eliminated in a similar manner to the axial compression test. If the
spinal implant assembly requires two sets of modified bilateral UHMWPE blocks and aluminum spacer blocks, then it is equivalent
to an axial tension test.
8.1.2.3 Place the UHMWPE blocks into the test apparatus such that the positionpositions of the hinge pins are internal to the
anchors (the hinge pin in the superior block is more inferior than the screw, hook, and so forth). Secure the UHMWPE blocks with
hinge pins. If one modified bilateral UHMWPE block is used to test hooks, wires, or cables, then place it superiorly.
8.1.2.4 Complete the spinal implant assembly in a standard construct (see Fig. 4, Fig. 6, Fig. 8, Fig. 10, Fig. 12, and Fig. 14 except
the UHMWPE blocks are inverted) or a hybrid construct (see Fig. 3 except the UHMWPE block are inverted). Set the active length
of the longitudinal element for the intended spinal location. Apply all tightening, crimping, or locking mechanisms as specified by
the manufacturer.
8.1.2.5 Load the test apparatus at a rate up to a maximum of 25 mm/min.
F1717 − 21
8.1.2.6 Record the load displacement curves. Establish the displacement at 2 % offset yield (mm), elastic displacement (mm),
tensile bending yield load (N), tensile bending stiffness (N/mm), tensile bending ultimate displacement (mm)(mm), and tensile
bending ultimate load (N).
8.1.3 Static Torsional Test:
8.1.3.1 Select the appropriate UHMWPE blocks for the spinal implant assembly as previously described.
8.1.3.2 Install the anchors according to the manufacturer’s instructions. When used as part of a rod/screw construct, variable axis
screw anchors shall be inserted in the UHMWPE blocks in a manner that prevents the impingement of any potentially pivoting
or rotating features of the anchor against the test block. This may be achieved by inserting the anchor such that, at full angulation
of any of the potentially pivoting or rotating features, clearance is always maintained with respect to the test block. If the spinal
implant assembly contains only hooks, wires, or cables then the system may not be able to resist torsional moments and need not
be tested; however, this should be verified by testing.
8.1.3.3 Place the UHMWPE blocks in the test apparatus such that the positions of the hinge pins are external to the anchors. The
hinge pin in the superior block is more superior than the screw, hook, and so forth, and the hinge pin in the inferior block is more
inferior than the screw, hook, and so forth. Secure the UHMWPE blocks with hinge pins. If only one modified bilateral UHMWPE
block is used to test hooks, wires, or cables, then place it superiorly. Attach UHMWPE blocks to the side supports via hinge pins.
8.1.3.4 Place the aluminum blocks between the UHMWPE blocks and the base plates to stop rotation around the hinge pin.
8.1.3.5 Complete the spinal implant assembly in a standard construct (Fig. 4, Fig. 6,and Fig. 8, Fig. 10, Fig. 12, and Fig. 14) or
a hybrid construct (Fig. 3). Set the active length of the longitudinal element for the intended spinal location. Apply all tightening,
crimping, or locking mechanisms as specified by the manufacturer.
8.1.3.6 Load the test apparatus at a maximum rate up to 60°/min. An axial load of approximately zero (N) should be maintained
during testing.
8.1.3.7 Record the torque-angular displacement curves. Determine the angular displacement at 2 % offset yield (degrees), elastic
angular displacement (degrees), yield torque (N-m), and torsional stiffness (N-m/degree).
8.2 Procedure for Fatigue Testing:
8.2.1 Select the appropriate UHMWPE blocks for the spinal implant assembly as previously described. Use unilateral UHMWPE
blocks (see Fig. 5 and Fig. 11) for singular longitudinal element constructs. Use bilateral UHMWPE blocks (see Fig. 7 and Fig.
13) for the testing of screws, bolts, and so forth. Use modified bilateral UHMWPE blocsk (see Fig. 9 and Fig. 15) for the testing
of hooks, wires, or cables.
8.2.2 Install the anchors according to the manufacturer’s instructions. When used as part a of rod/screw construct, variable axis
screw anchors shall be inserted in the UHMWPE blocks in a manner that prevents the impingement of any potentially pivoting
or rotating features of the anchor against the test block. This may be achieved by inserting the anchor such that, at full angulation
of any of the potentially pivoting or rotating features, clearance is always maintained with respect to the test block. If one modified
bilateral UHMWPE block for hooks, wires, cables, and so forth, is used then place an aluminum spacer block between the modified
UHMWPE block and the base plate to stop rotation about the hinge pin. The extra degree of freedom is eliminated in a manner
similar to the axial compression test. If the spinal implant assembly requires two sets of modified bilateral UHMWPE blocks and
aluminum spacer blocks, then the testing mode becomes an axial compression fatigue test.
8.2.3 Place UHMWPE blocks in the test apparatus such that the positions of the hinge pins are external to the anchors. The hinge
pin in the superior block is more superior than the anchor and the hinge pin in the inferior block is more inferior than the screw,
hook, and so forth. Secure UHMWPE blocks with hinge pins. If only one modified bilateral UHMWPE block is used to test hooks,
wires, cables, and so forth, then place it superiorly.
8.2.4 Complete the spinal implant assembly in a standard construct (Fig. 4, Fig. 6, Fig. 8, Fig. 10, Fig. 12, and Fig. 14) or a hybrid
construct (Fig. 3). Set the active length of the longitudinal element for the intended spinal location. Apply all tightening, crimping,
or locking mechanisms as specified by the manufacturer. Record the distance from the center of the superior hinge pin to that of
the inferior hinge pin (center-to-center hinge pin distance).
F1717 − 21
FIG. 16 Alternate Lumbar Bilateral UHMWPE Block for Screws and Bolts
8.2.5 The fatigue test applies a sinusoidal load to the spinal construct. The loading should be maintained via a constant sinusoidal
load amplitude control. A constant load ratio (R) for all tests should be established and should be equal to or greater than 10:
minimum load
R 5 $ 10 (2)
maximum load
Example: if minimum load = −200 N and maximum load = −10 N then R = 20. The maximum cycle rate is five cycles per second
for the fatigue test. The end of the test occurs when the spinal construct has a failure or reaches run-out.
8.2.6 Evaluate specimens at the initial fatigue loads as described in Section 7 and establish the endurance limit. A semi-log fatigue
curve of the compression bending load versus number of cycles at failure shall be plotted.
8.2.7 Note the initial and secondary failures, modes of failure, and deformations of components prior to removing the spinal
construct from the test apparatus. Evaluate all surface changes.
9. Report
9.1 The report should specify the spinal implant components, the spinal implant assembly, the intended spinal location, and the
numbers of specimens tested. Describe all relevant information about the components including name, lot number, manufacturer,
material, part number, and so forth. Also include any specific information necessary to produce the assembly, including the
tightening torque.
9.2 Include an illustration of the exact loading configurations. Describe the similarities and differences to relevant figures
contained herein. Report the active length. Report the block moment arm and the distance in the X direction between the centerline
of the longitudinal element and the insertion point of the anchors on the UHMWPE blocks. For constructs using variable axis screw
anchors, describe the measure employed to prevent impingement of pivoting or rotating features against the test block. Note any
deviations from the recommended test procedure. State the rate of loading.
F1717 − 21
9.3 The report of the static mechanical testing shall include a complete description of all failures, modes of failure, or deformations
of the spinal implant assembly or test apparatus. Include any noticeable fretting or surface texturing. The static mechanical test
report shall:
9.3.1 Show the load-displacement curves for all static compression bending tests. Report all static compression bending test data,
the mean and standard deviation for the displacement at 2 % offset yield (mm), elastic displacement (mm), compressive bending
yield load (N), compressive bending stiffness (N/mm), compressive bending ultimate displacement (mm), and compressive bending
ultimate load (N).
9.3.2 Show all load-displacement curves for the static tension bending test. Report all static tension bending test data, the mean
and standard deviation for the displacement at 2 % offset yield (mm), elastic displacement (mm), tensile bending yield load (N),
tensile bending stiffness (N/mm), tensile bending ultimate displacement (mm), and tensile bending ultimate load (N).
9.3.3 Show the torque-angular displacement curves for all static torsional tests. Report all static torsional test data, the mean and
standard deviation for the angular displacement at 2 % offset yield (degrees), elastic angular displacement (degrees), yield torque
(N-m), and torsional stiffness (N-m/degree). Report the direction of rotation for the tests along with the reference point.
9.3.4 Show the reason for each test being halted (for example, device fracture, screws pulling out of UHWMPE blocks, UHWMPE
blocks touching, clevis fixtures touching, and so forth).
9.4 The report of the dynamic mechanical testing shall:
9.4.1 State the fatigue test environment, load wave form, and test frequency. State the final sample sizes and load versus number
of cycles at failure for all fatigue tests. State the load levels for the specimens enduring 5 000 000 cycles and the maximum run-out
load. Report the constant load ratio (R).
9.4.2 Report all initial and secondary failures, modes of failure, and deformations of components for the spinal implant assembly
and the test apparatus. Fatigue failures should include a description of the failure initiation site, propagation zone, and ultimate
failure zone. Describe all surface changes, any fretting of interfaces, or loosening of interconnections. Include pictures of failure
surfaces and surface texturing from fretting.
9.4.3 For any fatigue test of a rod/screw construct that incorporates variable axis screw anchors and has achieved full
runoutrun-out without apparent failure, report the change in center-to-center hinge pin distance, and describe the causal mechanism
if a change is noted (for example, permanent deformation of one or more components, slippage of the polyaxial head, or
deformation/failure of the UHMWPE block).
9.4.4 Plot a semi-log fatigue curve of the compression or compression bending load versus number of cycles at failure. Indicate
specimens that have not failed before 5 000 000 cycles.
9.4.5 Report a regression analysis of the compression load or compression bending load versus number of cycles for only failed
constructs.
10. Precision and Bias
10.1 Precision—The precision of these test methods is based on two interlaboratory studies (ILS) conducted in 2008 and 2011.
Six laboratories participated in this study. Not all six laboratories were used for the two interlaboratory studies. Each of the six
labs was asked to report five replicate test results of a uniform assembly of screws, set screws, rods, and cross connectors, for ten
different measurement parameters. Every “test result” reported represents an individual determination. Except for the use of only
six laboratories and a single material, Practice E691 was followed for the design and analysis of the data; the details are given in
5 6
ASTM Research Report No. F04–1012Nos. F04-1012 and F04–1013.F04-1013.
10.1.1 Repeatability Limit (r)—Two test results obtained within one laboratory shall be judged not equivalent if they differ by more
Supporting data have been filed at ASTM International Headqua
...




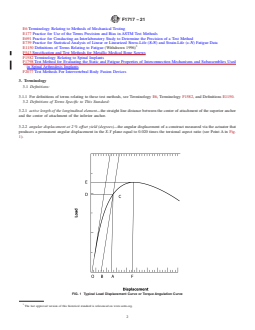


Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...