ASTM E1746-17a
(Test Method)Standard Test Method for Sampling and Analysis of Liquid Chlorine for Gaseous Impurities
Standard Test Method for Sampling and Analysis of Liquid Chlorine for Gaseous Impurities
SIGNIFICANCE AND USE
4.1 It is very difficult to exclude sample contamination by ambient air during the process of sampling. The levels of atmospheric contamination caused by poor sampling methods are often equal to or larger than the levels of the gaseous impurities present in the chlorine. This results in markedly elevated levels of detected impurities. As specifications become tighter, it becomes more important to measure the gaseous impurity levels in liquid chlorine correctly.
4.2 Additional problems are experienced in the sampling of liquefied gases for the gaseous impurities. The gaseous impurities reach an equilibrium between the liquid phase and vapor phase in a sample bomb. The quantity of gases measured in any particular sample containing both liquid and vapor will be a function of the amount of vapor space in the sample bomb. This test method avoids the presence of liquid in the sample bomb.
SCOPE
1.1 This test method covers sampling and analysis of liquid chlorine for the determination of oxygen (200 to 400 μg/g), nitrogen (400 to 800 μg/g), and carbon dioxide (800 to 1000 ppm) content at levels normally seen in liquid chlorine. Hydrogen and carbon monoxide concentrations in liquid chlorine are typically at or below the detection limit of this test method.
Note 1: The minimum detection limit of hydrogen using a 1 cm3 gas sample and argon carrier gas is 100 to 200 μg/g.2 The detection limit for the other components is significantly lower.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazards statements are given in Section 7.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Jun-2017
- Technical Committee
- D16 - Aromatic, Industrial, Specialty and Related Chemicals
- Drafting Committee
- D16.13 - Chlorine
Relations
- Effective Date
- 01-Nov-2016
- Effective Date
- 01-Jun-2007
- Effective Date
- 10-Jun-2002
Overview
ASTM E1746-17a: Standard Test Method for Sampling and Analysis of Liquid Chlorine for Gaseous Impurities provides an internationally recognized method for detecting and quantifying specific gaseous impurities in liquid chlorine. This standard, developed by ASTM International, details reliable procedures for sampling and analyzing levels of oxygen, nitrogen, and carbon dioxide commonly found as impurities in liquid chlorine. The standard emphasizes controlling atmospheric contamination during sampling and defines best practices for accurately determining impurity content, which is essential as product specifications tighten.
Key Topics
- Contamination Control: Ensures that sampling procedures minimize the risks of ambient air contamination, which can distort impurity readings.
- Sampling Liquefied Gases: Addresses the unique challenges posed by equilibrium between liquid and vapor phases in sample cylinders, and specifies methods for avoiding liquid contamination in gas samples.
- Impurity Analysis Ranges: Provides determination ranges for oxygen (200-400 μg/g), nitrogen (400-800 μg/g), and carbon dioxide (800-1000 ppm). Hydrogen and carbon monoxide are typically at or below the detection limits of this method.
- Safety Practices: Highlights the necessity of reviewing Safety Data Sheets (SDS) and maintaining rigorous safety and health practices when sampling and analyzing chlorine.
- Quality Assurance: Recommends robust laboratory quality control, referencing ASTM D6809 for guidelines where internal protocols are not established.
Applications
This test method is vital for:
- Producers and Users of Liquid Chlorine: Ensuring compliance with quality specifications by accurately measuring trace gaseous impurities.
- Industrial and Laboratory Settings: Facilitating quality control and regulatory compliance in sectors where liquid chlorine purity impacts product safety and process reliability.
- Regulatory Inspections: Supporting legal and regulatory requirements for liquid chlorine used or shipped across industries, such as water treatment, chemical manufacturing, and pharmaceuticals.
- Process Optimization: Enabling refinements in purification and handling processes by providing reliable impurity data.
- Safety Management: Assisting in risk assessment by identifying impurity levels that could affect toxicity or material compatibility.
By following ASTM E1746-17a, laboratories and facilities can standardize testing, minimize sample contamination risks, and ensure accurate reporting of gaseous impurity concentrations.
Related Standards
For comprehensive quality control and compliance in liquid chlorine analysis, the following standards and guidelines are relevant:
- ASTM D6809: Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials.
- 49 CFR 173: Code of Federal Regulations for Transportation, focusing on shippers' requirements for liquefied compressed gases.
- Chlorine Institute Pamphlet No. 1: Chlorine Basics: Essential industry guidance on chlorine safety and handling.
ASTM E1746-17a complements these standards by offering a dedicated, precise methodology for impurity analysis, supporting global quality and safety standards in the handling and use of liquid chlorine.
Practical Value
Using ASTM E1746-17a helps organizations:
- Ensure the reliability of impurity measurements in critical chemical processes.
- Meet stringent regulatory and industry requirements for chlorine purity.
- Enhance lab safety and operational procedures.
- Establish traceability and repeatability in analytical results, fostering confidence in reported data.
Adopting this standard is key for any operation where the integrity and quality of liquid chlorine are fundamental to process performance and product safety.
Buy Documents
ASTM E1746-17a - Standard Test Method for Sampling and Analysis of Liquid Chlorine for Gaseous Impurities
REDLINE ASTM E1746-17a - Standard Test Method for Sampling and Analysis of Liquid Chlorine for Gaseous Impurities
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E1746-17a is a standard published by ASTM International. Its full title is "Standard Test Method for Sampling and Analysis of Liquid Chlorine for Gaseous Impurities". This standard covers: SIGNIFICANCE AND USE 4.1 It is very difficult to exclude sample contamination by ambient air during the process of sampling. The levels of atmospheric contamination caused by poor sampling methods are often equal to or larger than the levels of the gaseous impurities present in the chlorine. This results in markedly elevated levels of detected impurities. As specifications become tighter, it becomes more important to measure the gaseous impurity levels in liquid chlorine correctly. 4.2 Additional problems are experienced in the sampling of liquefied gases for the gaseous impurities. The gaseous impurities reach an equilibrium between the liquid phase and vapor phase in a sample bomb. The quantity of gases measured in any particular sample containing both liquid and vapor will be a function of the amount of vapor space in the sample bomb. This test method avoids the presence of liquid in the sample bomb. SCOPE 1.1 This test method covers sampling and analysis of liquid chlorine for the determination of oxygen (200 to 400 μg/g), nitrogen (400 to 800 μg/g), and carbon dioxide (800 to 1000 ppm) content at levels normally seen in liquid chlorine. Hydrogen and carbon monoxide concentrations in liquid chlorine are typically at or below the detection limit of this test method. Note 1: The minimum detection limit of hydrogen using a 1 cm3 gas sample and argon carrier gas is 100 to 200 μg/g.2 The detection limit for the other components is significantly lower. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazards statements are given in Section 7. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 It is very difficult to exclude sample contamination by ambient air during the process of sampling. The levels of atmospheric contamination caused by poor sampling methods are often equal to or larger than the levels of the gaseous impurities present in the chlorine. This results in markedly elevated levels of detected impurities. As specifications become tighter, it becomes more important to measure the gaseous impurity levels in liquid chlorine correctly. 4.2 Additional problems are experienced in the sampling of liquefied gases for the gaseous impurities. The gaseous impurities reach an equilibrium between the liquid phase and vapor phase in a sample bomb. The quantity of gases measured in any particular sample containing both liquid and vapor will be a function of the amount of vapor space in the sample bomb. This test method avoids the presence of liquid in the sample bomb. SCOPE 1.1 This test method covers sampling and analysis of liquid chlorine for the determination of oxygen (200 to 400 μg/g), nitrogen (400 to 800 μg/g), and carbon dioxide (800 to 1000 ppm) content at levels normally seen in liquid chlorine. Hydrogen and carbon monoxide concentrations in liquid chlorine are typically at or below the detection limit of this test method. Note 1: The minimum detection limit of hydrogen using a 1 cm3 gas sample and argon carrier gas is 100 to 200 μg/g.2 The detection limit for the other components is significantly lower. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazards statements are given in Section 7. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E1746-17a is classified under the following ICS (International Classification for Standards) categories: 71.060.10 - Chemical elements. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E1746-17a has the following relationships with other standards: It is inter standard links to ASTM D6809-02(2016), ASTM D6809-02(2007), ASTM D6809-02. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E1746-17a is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E1746 − 17a
Standard Test Method for
Sampling and Analysis of Liquid Chlorine for Gaseous
Impurities
This standard is issued under the fixed designation E1746; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 2. Referenced Documents
2.1 ASTM Standards:
1.1 This test method covers sampling and analysis of liquid
D6809 Guide for Quality Control and Quality Assurance
chlorine for the determination of oxygen (200 to 400 µg/g),
Procedures for Aromatic Hydrocarbons and Related Ma-
nitrogen (400 to 800 µg/g), and carbon dioxide (800 to 1000
terials
ppm) content at levels normally seen in liquid chlorine.
2.2 Code of Federal Regulations:
Hydrogen and carbon monoxide concentrations in liquid chlo-
49 CFR 173, Code of Federal Regulations Title 49, Trans-
rine are typically at or below the detection limit of this test
portation: Shippers’General Requirements for Shipments
method.
and Packaging, including the following sections:
NOTE 1—The minimum detection limit of hydrogen usinga1cm gas
173.304 Charging of Cylinders with Liquefied Compressed
sample and argon carrier gas is 100 to 200 µg/g. The detection limit for
Gas
the other components is significantly lower.
173.314 Requirements for Compressed Gases in Tank Cars
1.2 The values stated in SI units are to be regarded as
173.315 Compressed Gases in Cargo Tanks and Portable
standard. No other units of measurement are included in this
Tank Containers
standard.
2.3 Other Document:
Chlorine Institute Pamphlet No. 1 Chlorine Basics
1.3 Review the current Safety Data Sheets (SDS) for de-
tailed information concerning toxicity, first aid procedures, and
3. Summary of Test Method
safety precautions.
3.1 Asampleofliquidchlorineistrappedinasamplingtube
1.4 This standard does not purport to address all of the
and vaporized into a steel bomb. The vaporized chlorine in the
safety concerns, if any, associated with its use. It is the
steel bomb is introduced into a gas chromatograph by a gas
responsibility of the user of this standard to establish appro-
sampling loop (1 cm ) using a ten-port gas sampling and
priate safety, health, and environmental practices and deter-
switching valve. The separations are made on a Porapak Q
mine the applicability of regulatory limitations prior to use.
column and on a 5Amolecular sieve column whose lengths are
Specific hazards statements are given in Section 7.
such that the peaks do not overlap.
1.5 This international standard was developed in accor-
3.2 Any component that co-elutes with the components of
dance with internationally recognized principles on standard-
interest may interfere with this analysis.
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom- 4. Significance and Use
mendations issued by the World Trade Organization Technical
4.1 It is very difficult to exclude sample contamination by
Barriers to Trade (TBT) Committee.
ambient air during the process of sampling. The levels of
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
This test method is under the jurisdiction of ASTM Committee D16 on contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsi- Standards volume information, refer to the standard’s Document Summary page on
bility of Subcommittee D16.13 on Chlorine. the ASTM website.
Current edition approved July 1, 2017. Published July 2017. Originally approved Available from DLA Document Services, Building 4/D, 700 Robbins Ave.,
in 1995. Last previous edition approved in 2017 as E1746 – 17. DOI: 10.1520/ Philadelphia, PA 19111-5094, http://quicksearch.dla.mil.
E1746-17a. Available from The Chlorine Institute, Inc., 1300 Wilson Blvd., Suite 525,
Thompson, B., Fundamentals of Gas Chromatography, Varian Instruments
Arlington, VA 22209.
Division, Sunnyvale, CA, p. 73. Porapak is a trademark of Waters Associates, Inc.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1746 − 17a
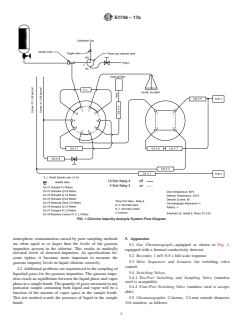
FIG. 1 Chlorine Impurity Analysis System Flow Diagram
atmospheric contamination caused by poor sampling methods 5. Apparatus
are often equal to or larger than the levels of the gaseous
5.1 Gas Chromatograph—equipped as shown in Fig. 1,
impurities present in the chlorine. This results in markedly
equipped with a thermal conductivity detector.
elevated levels of detected impurities. As specifications be-
5.2 Recorder, 1 mV, 0.5 s full-scale response.
come tighter, it becomes more important to measure the
5.3 Valve Sequencer and Actuator, for switching valve
gaseous impurity levels in liquid chlorine correctly.
control.
4.2 Additional problems are experienced in the sampling of
5.4 Switching Valves.
liquefied gases for the gaseous impurities. The gaseous impu-
5.4.1 Ten-Port Switching and Sampling Valve (stainless
rities reach an equilibrium between the liquid phase and vapor
steel is acceptable).
phaseinasamplebomb.Thequantityofgasesmeasuredinany
5.4.2 Four-Port Switching Valve (stainless steel is accept-
particular sample containing both liquid and vapor will be a
able).
function of the amount of vapor space in the sample bomb.
This test method avoids the presence of liquid in the sample 5.5 Chromatographic Columns, 3.2-mm outside diameter,
bomb. 316 stainless, as follows:
E1746 − 17a
5.5.1 2 m of 80/100 mesh Porapak N, 7.1.3 Donotallowthesamplecylindertobecomeliquidfull
5.5.2 0.8 m of 80/100 mesh Shimalite Q, if liquid samples are to be taken in cylinders. In accordance
5.5.3 1 m of 80/100 mesh Shimalite Q, with49CFR173.304,173.314,and173.315,agoodruleisthat
5.5.4 0.8 m of 80/100 mesh Shimalite Q, the weight of the chlorine in the cylinder should not be more
5.5.5 3 m of 45/60 mesh molecular sieve 5A, than 125 % of the weight of the water that the cylinder could
5.5.6 2 m of 80/100 mesh Porapak Q, contain.
5.5.7 2 m of 80/100 mesh Porapak N, and 7.1.4 When sampling and working with chlorine out of
5.5.8 1 m of 45/60 mesh molecular sieve 5A. doors, people downwind from such an operation should be
warned of the possible release of chlorine vapors.
5.6 Tantalum Tubing, 1.6-mm outside diameter, 0.57-mm
7.1.5 In the event that chlorine is inhaled, first aid should be
inside diameter.
summoned immediately and oxygen administered without
NOTE 2—Nickel tubing may be substituted for tantalum.
delay.
5.7 MonelSamplingTube, 9.5 by 140-mm long (volume 5.4 7.1.6 Store pressurized samples where involuntary release
3 9
cm ). would not cause excessive risk to people or property.
7.1.7 It is recommended that means be available for the
5.8 Electronic Integrator, or computer integration package.
disposal of excess chlorine in an environmentally safe and
5.9 TFE-Fluorocarbon Lined Flex Tubing, 6.35 mm.
acceptable manner. A chlorine absorption system should be
5.10 TFE-Fluorocarbon Tubing, 6.35 mm by 3.05 m. provided if the chlorine cannot be disposed of in a chlorine
10 consuming process. When the analysis and sampling regimen
5.11 Cajon VCR Fitting.
requires an initial purging of chlorine from a container, the
5.12 Two-Valves, 9.5 mm, Monel.
purged chlorine should be handled similarly. Purging to the
atmosphere should be avoided.
5.13 Four-Valves, 6.35-mm tubing to 6.35-mm pipe,
Monel.
8. Sampling
11 3 9
5.14 Hoke Sample Cylinder, 1000 cm , Monel, nickel,
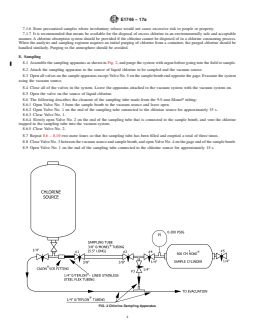
tantalum, or stainless steel. 8.1 Assemble the sampling apparatus as shown in Fig. 2,
and purge the system with argon before going into the field to
5.15 Pressure Gage, 91 kg, Monel.
sample.
5.16 Four-Pipe Tee, 6.35 mm, Monel.
8.2 Attach the sampling apparatus to the source of liquid
5.17 Vacuum Source, suitable for chlorine disposal.
chlorine to be sampled and the vacuum source.
6. Reagents
8.3 Open all valves on the sample apparatus except Valve
No. 5 on the sample bomb end opposite the gage. Evacuate the
6.1 Gas Standard, 500 µg/g H , 400 µg/g O , 800 µg/g N ,
2 2 2
system using the vacuum source.
50 µg/g CO, and 1000 µg/g CO in argon.
8.4 Close all of the valves in the system. Leave the
6.2 Argon Carrier Gas, chromatographic grade.
apparatus attached to the vacuum system with the vacuum
7. Hazards
system on.
7.1 Safety Precautions:
8.5 Open the valve on the source of liquid chlorine.
7.1.1 Chlorine is a corrosive and toxic material. A well-
8.6 The following describes the cleanout of the sampling
ventilated fume hood should be used to house all sample
tube made from the 9.5-mm Monel tubing:
handling and to vent the test equipment when this product is
8.6.1 Open Valve No. 3 from the sample bomb to the
analyzed in the laboratory.
vacuum source and leave open.
7.1.2 The analysis should be attempted only by individuals
8.6.2 Open Valve No. 1 on the end of the sampling tube
who are thoroughly familiar with the handling of chlorine, and
connected to the chlorine source for approximately 15 s.
even an experienced person should not work alone. The
8.6.3 Close Valve No. 1.
operator must be provided with adequate eye protection and
8.6.4 Slowly open Valve No. 2 on the end of the sampling
respirator. Splashes of liquid chlorine destroy clothing and will
tube that is connected to the sample bomb, and vent the
produce irritations and burns if such clothing is next to the
chlorine trapped in the sampling tube into the vacuum system.
skin.
8.6.5 Close Valve No. 2.
8.7 Repeat 8.6 – 8.10 two more times so that the sampling
Porapak materials, or their equivalent, have been found satisfactory for this
purpose. tube has been filled and emptied a total of three times.
Shimalite, a trademark of Shimadzu Seisakusho Ltd., Japan, materials or their
8.8 Close Valve No. 3 between the vacuum source and
equivalent, have been found satisfactory for this purpose.
Monel, a trademark of Special Metals Corporation, material or its equivalent, sample bomb, and open Valve No. 4 on the gage end of the
has been found satisfactory for this purpose.
sample bomb.
Cajon, a trademark of Swagelok Company, fittings or their equivalent, have
been found satisfactory for this purpose. 8.9 Open Valve No. 1 on the end of the sampling tube
Hoke, registered trademark of Hoke Inc., sample cylinders, or their equivalent,
connected to the chlorine source for approximately 15 s.
have been found satisfactory for this purpose.
This reagent is used for calibration only. 8.10 Close Valve No. 1 and open Valve No. 2 slowly.
E1746 − 17a
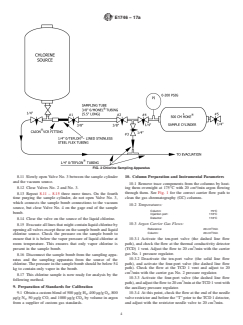
FIG. 2 Chlorine Sampling Apparatus
8.11 Slowly open Valve No. 3 between the sample cylinder 10. Column Preparation and Instrumental Parameters
and the vacuum source.
10.1 Remove trace components from the columns by heat-
8.12 Close Valves No. 2 and No. 3.
ing them overnight at 175°C with 20 cm /min argon flowing
through them. See Fig. 1 for the correct carrier flow path to
8.13 Repeat 8.11 – 8.15 three more times. On the fourth
clean the gas chromatography (GC) columns.
time purging the sample cylinder, do not open Valve No. 3,
which connects the sample bomb connections to the vacuum
10.2 Temperatures:
source, but close Valve No. 4 on the gage end of the sample
Column: 75°C
bomb.
Injection port: 110°C
Detector: 110°C
8.14 Close the valve on the source of the liquid chlorine.
10.3 Argon Carrier Gas Flows:
8.15 Evacuate all lines that might contain liquid chlorine by
opening all valves except those on the sample bomb and liquid Reference: 20 cm /min
Column: 20 cm /min
chlorine source. Check the pressure on the sample bomb to
ensure that it is below the vapor pressure of liqu
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E1746 − 17 E1746 − 17a
Standard Test Method for
Sampling and Analysis of Liquid Chlorine for Gaseous
Impurities
This standard is issued under the fixed designation E1746; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers sampling and analysis of liquid chlorine for the determination of oxygen (200 to 400 μg/g), nitrogen
(400 to 800 μg/g), and carbon dioxide (800 to 1000 ppm) content at levels normally seen in liquid chlorine. Hydrogen and carbon
monoxide concentrations in liquid chlorine are typically at or below the detection limit of this test method.
3 2
NOTE 1—The minimum detection limit of hydrogen using a 1 cm gas sample and argon carrier gas is 100 to 200 μg/g. The detection limit for the
other components is significantly lower.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first aid procedures, and safety
precautions.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use. Specific hazards statements are given in Section 7.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D6809 Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials
2.2 Code of Federal Regulations:
49 CFR 173, Code of Federal Regulations Title 49, Transportation: Shippers’ General Requirements for Shipments and
Packaging, including the following sections:
173.304 Charging of Cylinders with Liquefied Compressed Gas
173.314 Requirements for Compressed Gases in Tank Cars
173.315 Compressed Gases in Cargo Tanks and Portable Tank Containers
2.3 Other Document:
Chlorine Institute Pamphlet No. 1 Chlorine Basics
3. Summary of Test Method
3.1 A sample of liquid chlorine is trapped in a sampling tube and vaporized into a steel bomb. The vaporized chlorine in the
steel bomb is introduced into a gas chromatograph by a gas sampling loop (1 cm ) using a ten-port gas sampling and switching
valve. The separations are made on a Porapak Q column and on a 5A molecular sieve column whose lengths are such that the
peaks do not overlap.
This test method is under the jurisdiction of ASTM Committee D16 on Aromatic Hydrocarbons Aromatic, Industrial, Specialty and Related Chemicals and is the direct
responsibility of Subcommittee D16.16 on Industrial and Specialty Product Standards.
Current edition approved March 1, 2017July 1, 2017. Published March 2017July 2017. Originally approved in 1995. Last previous edition approved in 20082017 as E1746
– 08.17. DOI: 10.1520/E1746-17.10.1520/E1746-17a.
Thompson, B., Fundamentals of Gas Chromatography, Varian Instruments Division, Sunnyvale, CA, p. 73.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from DLA Document Services, Building 4/D, 700 Robbins Ave., Philadelphia, PA 19111-5094, http://quicksearch.dla.mil.
Available from The Chlorine Institute, Inc., 1300 Wilson Blvd., Suite 525, Arlington, VA 22209.
Porapak is a trademark of Waters Associates, Inc.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1746 − 17a
3.2 Any component that co-elutes with the components of interest may interfere with this analysis.
4. Significance and Use
4.1 It is very difficult to exclude sample contamination by ambient air during the process of sampling. The levels of atmospheric
contamination caused by poor sampling methods are often equal to or larger than the levels of the gaseous impurities present in
the chlorine. This results in markedly elevated levels of detected impurities. As specifications become tighter, it becomes more
important to measure the gaseous impurity levels in liquid chlorine correctly.
4.2 Additional problems are experienced in the sampling of liquefied gases for the gaseous impurities. The gaseous impurities
reach an equilibrium between the liquid phase and vapor phase in a sample bomb. The quantity of gases measured in any particular
sample containing both liquid and vapor will be a function of the amount of vapor space in the sample bomb. This test method
avoids the presence of liquid in the sample bomb.
5. Apparatus
5.1 Gas Chromatograph—Shimadzu GC-8AIT equipped as shown in Fig. 1, or equivalent, equipped with a thermal conductivity
FIG. 1 Chlorine Impurity Analysis System Flow Diagram
E1746 − 17a
detector.
5.2 Recorder, 1 mV, 0.5 s full-scale response.
5.3 Valve Sequencer and Actuator, for switching valve control.
5.4 Switching Valves.
5.4.1 Ten-Port Switching and Sampling Valve (stainless steel is acceptable).
5.4.2 Four-Port Switching Valve (stainless steel is acceptable).
5.5 Chromatographic Columns, 3.2-mm outside diameter, 316 stainless, as follows:
5.5.1 2 m of 80/100 mesh Porapak N,
5.5.2 0.8 m of 80/100 mesh Shimalite Q,
5.5.3 1 m of 80/100 mesh Shimalite Q,
5.5.4 0.8 m of 80/100 mesh Shimalite Q,
5.5.5 3 m of 45/60 mesh molecular sieve 5A,
5.5.6 2 m of 80/100 mesh Porapak Q,
5.5.7 2 m of 80/100 mesh Porapak N, and
5.5.8 1 m of 45/60 mesh molecular sieve 5A.
5.6 Tantalum Tubing, 1.6-mm outside diameter, 0.57-mm inside diameter.
NOTE 2—Nickel tubing may be substituted for tantalum.
3 9
5.7 Monel Sampling Tube, 9.5 by 140-mm long (volume 5.4 cm ).
5.8 Electronic Integrator, or computer integration package.
5.9 TFE-Fluorocarbon Lined Flex Tubing, 6.35 mm.
5.10 TFE-Fluorocarbon Tubing, 6.35 mm by 3.05 m.
5.11 Cajon VCR Fitting.
5.12 Two-Valves, 9.5 mm, Monel.
5.13 Four-Valves, 6.35-mm tubing to 6.35-mm pipe, Monel.
11 3 9
5.14 Hoke Sample Cylinder, 1000 cm , Monel, nickel, tantalum, or stainless steel.
5.15 Pressure Gage, 91 kg, Monel.
5.16 Four-Pipe Tee, 6.35 mm, Monel.
5.17 Vacuum Source, suitable for chlorine disposal.
6. Reagents
6.1 Gas Standard, 500 μg/g H , 400 μg/g O , 800 μg/g N , 50 μg/g CO, and 1000 μg/g CO in argon.
2 2 2 2
6.2 Argon Carrier Gas, chromatographic grade.
7. Hazards
7.1 Safety Precautions:
7.1.1 Chlorine is a corrosive and toxic material. A well-ventilated fume hood should be used to house all sample handling and
to vent the test equipment when this product is analyzed in the laboratory.
7.1.2 The analysis should be attempted only by individuals who are thoroughly familiar with the handling of chlorine, and even
an experienced person should not work alone. The operator must be provided with adequate eye protection and respirator. Splashes
of liquid chlorine destroy clothing and will produce irritations and burns if such clothing is next to the skin.
7.1.3 Do not allow the sample cylinder to become liquid full if liquid samples are to be taken in cylinders. In accordance with
49 CFR 173.304, 173.314, and 173.315, a good rule is that the weight of the chlorine in the cylinder should not be more than 125 %
of the weight of the water that the cylinder could contain.
7.1.4 When sampling and working with chlorine out of doors, people downwind from such an operation should be warned of
the possible release of chlorine vapors.
7.1.5 In the event that chlorine is inhaled, first aid should be summoned immediately and oxygen administered without delay.
Porapak materials, or their equivalent, have been found satisfactory for this purpose.
Shimalite, a trademark of Shimadzu Seisakusho Ltd., Japan, materials or their equivalent, have been found satisfactory for this purpose.
Monel, a trademark of Special Metals Corporation, material or its equivalent, has been found satisfactory for this purpose.
Cajon, a trademark of Swagelok Company, fittings or their equivalent, have been found satisfactory for this purpose.
Hoke, registered trademark of Hoke Inc., sample cylinders, or their equivalent, have been found satisfactory for this purpose.
This reagent is used for calibration only.
E1746 − 17a
7.1.6 Store pressurized samples where involuntary release would not cause excessive risk to people or property.
7.1.7 It is recommended that means be available for the disposal of excess chlorine in an environmentally safe and acceptable
manner. A chlorine absorption system should be provided if the chlorine cannot be disposed of in a chlorine consuming process.
When the analysis and sampling regimen requires an initial purging of chlorine from a container, the purged chlorine should be
handled similarly. Purging to the atmosphere should be avoided.
8. Sampling
8.1 Assemble the sampling apparatus as shown in Fig. 2, and purge the system with argon before going into the field to sample.
8.2 Attach the sampling apparatus to the source of liquid chlorine to be sampled and the vacuum source.
8.3 Open all valves on the sample apparatus except Valve No. 5 on the sample bomb end opposite the gage. Evacuate the system
using the vacuum source.
8.4 Close all of the valves in the system. Leave the apparatus attached to the vacuum system with the vacuum system on.
8.5 Open the valve on the source of liquid chlorine.
8.6 The following describes the cleanout of the sampling tube made from the 9.5-mm Monel tubing:
8.6.1 Open Valve No. 3 from the sample bomb to the vacuum source and leave open.
8.6.2 Open Valve No. 1 on the end of the sampling tube connected to the chlorine source for approximately 15 s.
8.6.3 Close Valve No. 1.
8.6.4 Slowly open Valve No. 2 on the end of the sampling tube that is connected to the sample bomb, and vent the chlorine
trapped in the sampling tube into the vacuum system.
8.6.5 Close Valve No. 2.
8.7 Repeat 8.6 – 8.10 two more times so that the sampling tube has been filled and emptied a total of three times.
8.8 Close Valve No. 3 between the vacuum source and sample bomb, and open Valve No. 4 on the gage end of the sample bomb.
8.9 Open Valve No. 1 on the end of the sampling tube connected to the chlorine source for approximately 15 s.
FIG. 2 Chlorine Sampling Apparatus
E1746 − 17a
FIG. 3 Chromatogram of the Gaseous Impurities in Chlorine
8.10 Close Valve No. 1 and open Valve No. 2 slowly.
8.11 Slowly open Valve No. 3 between the sample cylinder and the vacuum source.
8.12 Close Valves No. 2 and No. 3.
8.13 Repeat 8.11 – 8.15 three more times. On the fourth time purging the sample cylinder, do not open Valve No. 3, which
connects the sample bomb connections to the vacuum source, but close Valve No. 4 on the gage end of the sample bomb.
8.14 Close the valve on the source of the liquid chlorine.
8.15 Evacuate all lines that might contain liquid chlorine by opening all valves except those on the sample bomb and liquid
chlorine source. Check the pressure on the sample bomb to ensure that it is below the vapor pressure of liquid chlorine at room
temperature. This ensures that only vapor chlorine is present in the sample bomb.
8.16 Disconnect the sample bomb from the sampling apparatus and the sampling apparatus from the source of the chlorine. The
pressure in the sample bomb should be below 54
...




Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...