ASTM F2102-17
(Guide)Standard Guide for Evaluating the Extent of Oxidation in Polyethylene Fabricated Forms Intended for Surgical Implants
Standard Guide for Evaluating the Extent of Oxidation in Polyethylene Fabricated Forms Intended for Surgical Implants
ABSTRACT
This guide covers method for evaluating the relative extent of oxidation in ultra-high-molecular-weight polyethylene fabricated forms intended for surgical implants. Equipments for this method shall include an infrared spectrometer, specimen holder, and microtome. Specimen shall be tested with infrared spectrometer in accordance to the procedure of test specimen preparation, spectrometer setup, and test specimen configuration. Oxidation peak area, normalization peak area, oxidation index, oxidation index depth locator, oxidation index profile, surface oxidation index, bulk oxidation index, and maximum oxidation index shall be calculated from the spectra. Report shall include material information, sample information, spectrometer information, and data analysis information.
SCOPE
1.1 This guide covers a method for the measurement of the relative extent of oxidation present in HDPE homopolymers and ultra-high-molecular-weight polyethylene (UHMWPE) intended for use in medical implants. The material is analyzed by infrared spectroscopy. The intensity (area) of the carbonyl absorptions (>C=O) centered near 1720 cm-1 is related to the amount of chemically bound oxygen present in the material. Other forms of chemically bound oxygen (C-O-C, C-O-O-C, C-O-H, and so forth) are not captured by this guide.
1.2 Although this guide may give the investigator a means to compare the relative extent of carbonyl oxidation present in various UHMWPE samples, it is recognized that other forms of chemically bound oxygen may be important contributors to these materials' characteristics.
1.3 The applicability of the infrared method has been demonstrated by many literature reports. This particular method, using the intensity (area) of the C-H absorption centered near 1370 cm-1 to normalize for the sample’s thickness, has been validated by an Interlaboratory Study (ILS) conducted according to Practice E691.
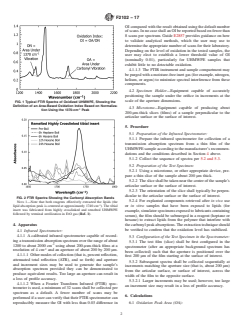
FIG. 1 Typical FTIR Spectra of Oxidized UHMWPE, Showing the Definition of an Area-Based Oxidation Index Based on Normalization Using the 1370-cm-1 Peak
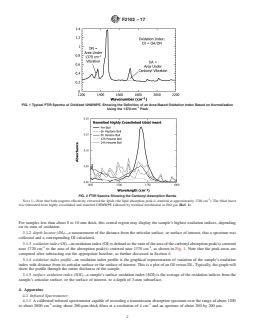
FIG. 2 FTIR Spectra Showing the Carbonyl Absorption Bands
Note 1: Note that both reagents effectively extracted the lipids (the lipid absorption peak is centered at approximately 1740 cm-1). The tibial insert was fabricated from highly crosslinked and remelted UHMWPE followed by terminal sterilization in EtO gas (Ref. 1).
1.4 The following precautionary caveat pertains only to the test method portion, Section 5, of this specification: This standard may involve hazardous materials, operations, and equipment. This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory requirements prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Aug-2017
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.15 - Material Test Methods
Relations
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-May-2013
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Nov-2005
- Effective Date
- 10-May-1999
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Sep-2017
Overview
ASTM F2102-17 is a standard guide developed by ASTM International for evaluating the extent of oxidation in polyethylene fabricated forms, particularly ultra-high-molecular-weight polyethylene (UHMWPE), intended for surgical implants. The standard details a method using infrared spectroscopy-specifically, Fourier Transform Infrared (FTIR) analysis-to determine the relative extent of oxidation by quantifying carbonyl absorption in the polyethylene material. This quantitative assessment is critical, as the oxidation of UHMWPE can affect its mechanical integrity and clinical performance in applications such as orthopedic implants.
Key Topics
Scope of the Standard
- Applies to both high-density polyethylene (HDPE) homopolymers and UHMWPE for surgical implant use.
- Focuses on the measurement of carbonyl oxidation, identified by IR absorption near 1720 cm⁻¹.
- Normalizes oxidation measurement via the C-H absorption band at 1370 cm⁻¹.
Equipment and Sample Preparation
- Requires a calibrated infrared spectrometer, specimen holder, and a microtome for preparing thin sample slices.
- Methods accommodate a variety of spectrometry collection modes, with recommendations for aperture size and scan number to ensure data consistency.
- Test specimens must be carefully prepared, sometimes including lipid extraction for explanted or in vitro exposed samples.
Calculation and Data Reporting
- Calculation of oxidation peak area, normalization peak area, oxidation index (OI), and related metrics such as surface oxidation index (SOI), bulk oxidation index (BOI), and maximum oxidation index (MOI).
- Oxidation profiles are plotted as Oxidation Index versus depth to map oxidation throughout the thickness of the sample.
Interpretation and Limitations
- This method provides a comparative measure of carbonyl-based oxidation but does not account for other forms of chemically bound oxygen.
- The guide emphasizes the importance of consistent sample preparation and instrument settings for reliable, reproducible results.
Applications
Medical Device Manufacturing
- Used by manufacturers of UHMWPE components for hip and knee implants to assure the oxidative stability of medical-grade polyethylene.
- Assists in quality control by enabling the comparison of batch-to-batch oxidation levels and assessing the effects of sterilization, cross-linking, and post-processing treatments.
Research and Development
- Supports comparative studies on the aging, durability, and performance of polyethylene implants under different storage and clinical conditions.
- Employed in validating shelf life and accelerated aging protocols in compliance with regulatory and safety standards.
Implant Retrieval Analysis
- Facilitates assessment of explanted devices to investigate in vivo oxidative degradation, guiding improvements in materials and manufacturing processes.
Related Standards
- ASTM E691: Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method. Referenced for validation of the reproducibility and precision of the FTIR method.
- ASTM E2857: Guide for Validating Analytical Methods. Provides guidance for validating FTIR analysis.
- ISO and ASTM Polyethylene Standards: ASTM F2102-17 complements other standards focused on medical polymers, aging, and performance testing for implantable devices.
Practical Value
Adhering to ASTM F2102-17 ensures reproducible and standardized evaluation of oxidation in UHMWPE medical implants, which is vital for patient safety and device longevity. The guide’s rigorous approach to FTIR-based oxidation indexing supports regulatory compliance, product improvement, and contributes to the body of knowledge on the durability of orthopedic implant materials.
Keywords: ASTM F2102-17, polyethylene, UHMWPE, oxidation index, surgical implants, FTIR spectroscopy, orthopedic implants, oxidation evaluation, medical device standards.
Buy Documents
ASTM F2102-17 - Standard Guide for Evaluating the Extent of Oxidation in Polyethylene Fabricated Forms Intended for Surgical Implants
REDLINE ASTM F2102-17 - Standard Guide for Evaluating the Extent of Oxidation in Polyethylene Fabricated Forms Intended for Surgical Implants
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2102-17 is a guide published by ASTM International. Its full title is "Standard Guide for Evaluating the Extent of Oxidation in Polyethylene Fabricated Forms Intended for Surgical Implants". This standard covers: ABSTRACT This guide covers method for evaluating the relative extent of oxidation in ultra-high-molecular-weight polyethylene fabricated forms intended for surgical implants. Equipments for this method shall include an infrared spectrometer, specimen holder, and microtome. Specimen shall be tested with infrared spectrometer in accordance to the procedure of test specimen preparation, spectrometer setup, and test specimen configuration. Oxidation peak area, normalization peak area, oxidation index, oxidation index depth locator, oxidation index profile, surface oxidation index, bulk oxidation index, and maximum oxidation index shall be calculated from the spectra. Report shall include material information, sample information, spectrometer information, and data analysis information. SCOPE 1.1 This guide covers a method for the measurement of the relative extent of oxidation present in HDPE homopolymers and ultra-high-molecular-weight polyethylene (UHMWPE) intended for use in medical implants. The material is analyzed by infrared spectroscopy. The intensity (area) of the carbonyl absorptions (>C=O) centered near 1720 cm-1 is related to the amount of chemically bound oxygen present in the material. Other forms of chemically bound oxygen (C-O-C, C-O-O-C, C-O-H, and so forth) are not captured by this guide. 1.2 Although this guide may give the investigator a means to compare the relative extent of carbonyl oxidation present in various UHMWPE samples, it is recognized that other forms of chemically bound oxygen may be important contributors to these materials' characteristics. 1.3 The applicability of the infrared method has been demonstrated by many literature reports. This particular method, using the intensity (area) of the C-H absorption centered near 1370 cm-1 to normalize for the sample’s thickness, has been validated by an Interlaboratory Study (ILS) conducted according to Practice E691. FIG. 1 Typical FTIR Spectra of Oxidized UHMWPE, Showing the Definition of an Area-Based Oxidation Index Based on Normalization Using the 1370-cm-1 Peak FIG. 2 FTIR Spectra Showing the Carbonyl Absorption Bands Note 1: Note that both reagents effectively extracted the lipids (the lipid absorption peak is centered at approximately 1740 cm-1). The tibial insert was fabricated from highly crosslinked and remelted UHMWPE followed by terminal sterilization in EtO gas (Ref. 1). 1.4 The following precautionary caveat pertains only to the test method portion, Section 5, of this specification: This standard may involve hazardous materials, operations, and equipment. This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory requirements prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ABSTRACT This guide covers method for evaluating the relative extent of oxidation in ultra-high-molecular-weight polyethylene fabricated forms intended for surgical implants. Equipments for this method shall include an infrared spectrometer, specimen holder, and microtome. Specimen shall be tested with infrared spectrometer in accordance to the procedure of test specimen preparation, spectrometer setup, and test specimen configuration. Oxidation peak area, normalization peak area, oxidation index, oxidation index depth locator, oxidation index profile, surface oxidation index, bulk oxidation index, and maximum oxidation index shall be calculated from the spectra. Report shall include material information, sample information, spectrometer information, and data analysis information. SCOPE 1.1 This guide covers a method for the measurement of the relative extent of oxidation present in HDPE homopolymers and ultra-high-molecular-weight polyethylene (UHMWPE) intended for use in medical implants. The material is analyzed by infrared spectroscopy. The intensity (area) of the carbonyl absorptions (>C=O) centered near 1720 cm-1 is related to the amount of chemically bound oxygen present in the material. Other forms of chemically bound oxygen (C-O-C, C-O-O-C, C-O-H, and so forth) are not captured by this guide. 1.2 Although this guide may give the investigator a means to compare the relative extent of carbonyl oxidation present in various UHMWPE samples, it is recognized that other forms of chemically bound oxygen may be important contributors to these materials' characteristics. 1.3 The applicability of the infrared method has been demonstrated by many literature reports. This particular method, using the intensity (area) of the C-H absorption centered near 1370 cm-1 to normalize for the sample’s thickness, has been validated by an Interlaboratory Study (ILS) conducted according to Practice E691. FIG. 1 Typical FTIR Spectra of Oxidized UHMWPE, Showing the Definition of an Area-Based Oxidation Index Based on Normalization Using the 1370-cm-1 Peak FIG. 2 FTIR Spectra Showing the Carbonyl Absorption Bands Note 1: Note that both reagents effectively extracted the lipids (the lipid absorption peak is centered at approximately 1740 cm-1). The tibial insert was fabricated from highly crosslinked and remelted UHMWPE followed by terminal sterilization in EtO gas (Ref. 1). 1.4 The following precautionary caveat pertains only to the test method portion, Section 5, of this specification: This standard may involve hazardous materials, operations, and equipment. This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory requirements prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2102-17 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2102-17 has the following relationships with other standards: It is inter standard links to ASTM F2102-13, ASTM E691-13, ASTM E2857-11, ASTM E691-11, ASTM E691-08, ASTM E691-05, ASTM E691-99, ASTM F2381-19, ASTM F561-19, ASTM F2759-19, ASTM F2565-21, ASTM F2977-20, ASTM F3336-22. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2102-17 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:F2102 −17
Standard Guide for
Evaluating the Extent of Oxidation in Polyethylene
Fabricated Forms Intended for Surgical Implants
This standard is issued under the fixed designation F2102; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
1.1 This guide covers a method for the measurement of the
2.1 ASTM Standards:
relative extent of oxidation present in HDPE homopolymers
E691 Practice for Conducting an Interlaboratory Study to
and ultra-high-molecular-weight polyethylene (UHMWPE) in-
Determine the Precision of a Test Method
tendedforuseinmedicalimplants.Thematerialisanalyzedby
E2857 Guide for Validating Analytical Methods
infrared spectroscopy. The intensity (area) of the carbonyl
-1
absorptions (>C=O) centered near 1720 cm is related to the
3. Terminology
amount of chemically bound oxygen present in the material.
3.1 Definitions:
Other forms of chemically bound oxygen (C-O-C, C-O-O-C,
3.1.1 bulkoxidationindex(BOI)—asample’sbulkoxidation
C-O-H, and so forth) are not captured by this guide.
index (BOI) is the average of the oxidation indices collected
1.2 Although this guide may give the investigator a means
over a 500-µm section at the center of the sample.
to compare the relative extent of carbonyl oxidation present in
3.1.1.1 Discussion—Typically, this is a plateau region with
variousUHMWPEsamples,itisrecognizedthatotherformsof
the smallest oxidation indices.
chemically bound oxygen may be important contributors to
3.1.1.2 Discussion—Forsampleslessthanabout8to10mm
these materials’ characteristics.
thick, this central region may display the sample’s highest
oxidation indices, depending on its state of oxidation.
1.3 The applicability of the infrared method has been
demonstrated by many literature reports. This particular
3.1.2 depth locator (DL)—a measurement of the distance
method, using the intensity (area) of the C-H absorption
fromthearticularsurface,orsurfaceofinterest,thataspectrum
-1
centered near 1370 cm to normalize for the sample’s
was collected and a corresponding OI calculated.
thickness,hasbeenvalidatedbyanInterlaboratoryStudy(ILS)
3.1.3 oxidation index (OI)—an oxidation index (OI) is
conducted according to Practice E691.
defined as the ratio of the area of the carbonyl absorption
-1
1.4 The following precautionary caveat pertains only to the
peak(s) centered near 1720 cm to the area of the absorption
-1
test method portion, Section 5, of this specification: This
peak(s) centered near 1370 cm , as shown in Fig. 1. Note that
standard may involve hazardous materials, operations, and
the peak areas are computed after subtracting out the appro-
equipment. This standard does not purport to address all of the
priate baseline, as further discussed in Section 6.
safety concerns, if any, associated with its use. It is the
3.1.4 oxidation index profile—an oxidation index profile is
responsibility of the user of this standard to establish appro-
the graphical representation of variation of the sample’s
priate safety and health practices and determine the applica-
oxidation index with distance from its articular surface or the
bility of regulatory requirements prior to use.
surface of interest.This is a plot of an OI versus DL.Typically,
1.5 This international standard was developed in accor-
the graph will show the profile through the entire thickness of
dance with internationally recognized principles on standard-
the sample.
ization established in the Decision on Principles for the
3.1.5 surface oxidation index (SOI)—a sample’s surface
Development of International Standards, Guides and Recom-
oxidation index (SOI) is the average of the oxidation indices
mendations issued by the World Trade Organization Technical
fromthesample’sarticularsurface,orthesurfaceofinterest,to
Barriers to Trade (TBT) Committee.
a depth of 3-mm subsurface.
This guide is under the jurisdiction of ASTM Committee F04 on Medical and
Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.15 on Material Test Methods. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Sept. 1, 2017. Published September 2017. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2001. Last previous edition approved in 2013 as F2102 – 13. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/F2102-17. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2102−17
OI compared with the result obtained using the default number
ofscans.InnocaseshallanOIbereportedbasedonfewerthan
8 scans per spectrum. Guide E2857 provides guidance on how
to validate analytical methods, which the user may use to
determine the appropriate number of scans for their laboratory.
Depending on the level of oxidation in the tested samples, the
user may elect to establish a lower threshold value of OI
(nominally 0.01), particularly for UHMWPE samples that
exhibit little to no detectable oxidation.
4.1.1.3 The FTIR instrument and sample compartment may
bepurgedwithamoisture-freeinertgas(forexample,nitrogen,
helium, or argon) to minimize spectral interference from these
components.
4.2 Specimen Holder—Equipment capable of accurately
positioning the sample under the orifice in increments at the
scale of the aperture dimensions.
FIG. 1Typical FTIR Spectra of Oxidized UHMWPE, Showing the
Definition of an Area-Based Oxidation Index Based on Normaliza-
4.3 Microtome—Equipment capable of producing about
-1
tion Using the 1370-cm Peak
200-µm-thick slices (films) of a sample perpendicular to the
articular surface or the surface of interest.
5. Procedure
5.1 Preparation of the Infrared Spectrometer:
5.1.1 Prepare the infrared spectrometer for collection of a
transmission absorption spectrum from a thin film of the
UHMWPEsampleaccordingtothemanufacturer’srecommen-
dations and the conditions described in Section 4 above.
5.1.2 Collect the sequence of spectra per 5.2 and 5.3.
5.2 Preparation of the Test Specimen:
5.2.1 Using a microtome, or other appropriate device, pre-
pare a thin slice of the sample about 200 µm thick.
5.2.2 The slice shall be taken near the center of the sample’s
articular surface or the surface of interest.
5.2.3 The orientation of the slice shall typically be perpen-
dicular to the articular surface or the surface of interest.
FIG. 2FTIR Spectra Showing the Carbonyl Absorption Bands
5.2.4 For explanted components retrieved after in vivo use
NOTE 1—Note that both reagents effectively extracted the lipids (the
-1
lipid absorption peak is centered at approximately 1740 cm ). The tibial or in vitro samples that have been exposed to lipids (for
insert was fabricated from highly crosslinked and remelted UHMWPE
example,simulatorspecimensexposedtolubricantscontaining
followed by terminal sterilization in EtO gas (Ref. 1).
serum), the film should be submerged in a reagent (heptane or
hexane) to extract lipids from the polymer that interfere with
4. Apparatus
thecarbonylpeakabsorptions.Theextractiontechniqueshould
be verified to confirm that the oxidation level has stabilized.
4.1 Infrared Spectrometer:
4.1.1 A calibrated infrared spectrometer capable of record-
5.3 Configuration of the Test Specimen in the Spectrometer:
ing a transmission absorption spectrum over the range of about
5.3.1 The test film (slice) shall be first configured in the
-1
1200 to about 2000 cm using about 200-µsm-thick films at a
spectrometer (after an appropriate background spectrum has
-1
resolution of 4 cm and an aperture of about 200 by 200 µm.
been collected) such that the aperture is positioned over the
4.1.1.1 Othermodesofcollection(thatis,percentreflection,
first 200 µm of the film starting at the surface of interest.
attenuated total reflection (ATR), and so forth) and aperture
5.3.2 Subsequent spectra shall be collected sequentially at
and increment sizes may be used to generate the sample’s
increments matching the aperture size (that is, about 200 µm)
absorption spectrum provided they can be demonstrated to
from the articular surface, or surface of interest, across the
produce equivalent results. Too large an aperture can result in
width of the film to the opposite surface.
a loss of profile accuracy.
5.3.2.1 Larger increments may be used; however, too large
4.1.1.2 When a Fourier Transform Infrared (FTIR) spec-
an increment size may result in a loss of profile accuracy.
trometer is used, a minimum of 32 scans shall be collected per
spectrum as a default. A fewer number of scans may be
6. Calculations
performed if a user can verify that their FTIR spectrometer can
reproducibly measure the OI with less than 0.03 difference in 6.1 Oxidation Peak Area (OA):
F2102−17
6.1.1 For each absorbance spectrum, calculate the total area 7. Report
-1
of the carbonyl peak absorptions centered near 1720 cm (Fig.
7.1 The report shall contain at least the following experi-
1).
mental details and results:
6.1.1.1 This is the area below the sample’s carbonyl absorp-
7.1.1 Material Information:
tion curve and above the straight line baseline drawn between
7.1.1.1 Resin type and resin lot number.
the starting and ending points.
7.1.1.2 Consolidation method and manufacturer and manu-
facturer lot number.
6.2 Normalization Peak Area (ON):
7.1.1.3 Any special post-consolidation treatments, for
6.2.1 For each absorbance spectrum, calculate the total area
-1
example, shot isostatic pressing (HIPing), annealing,
of the peak absorptions centered near 1370 cm (Fig. 1).
sterilization, cross-linking, stabilization, accelerated aging, and
6.2.1.1 This is the area below the sample’s absorption curve
storage conditions.
and above the straight line baseline drawn between the same
7.1.2 Sample Information:
starting and ending points.
7.1.2.1 Orthopedic implant or laboratory test specimen.
6.3 Oxidation Index (OI):
7.1.2.2 Time elapsed between sample preparation and test-
6.3.1 For each absorbance spectrum, calculate its OI by
ing in the FTIR.
dividing the area of its oxidation peak (6.1) by the area of its
7.1.2.3 Articular surface or non-articulator surface.
normalization peak (6.2), as shown in Fig. 1.
7.1.2.4 Test sample’s original dimensions.
6.4 Oxidation Index Depth Locator (DL): 7.1.2.5 Any special post-treatments of the original test
6.4.1 Calculate the distance from the articular surface, or sample, for example, annealing, sterilization, cross-linking,
surface of interest (DL), for each spectrum and its correspond- stabilization, accelerated aging, and storage conditions.
ing OI from the following equation. 7.1.2.6 Test film thickness and total width.
7.1.2.7 Any special post-treatments of the test films, for
DL 5 0.5~A!1n~S!
example, annealing, sterilization, cross-linking, stabilization,
where:
accelerated aging, and storage condition.
A = the size of the aperture in micrometres in the step
7.1.2.8 Describe sample fixturing (for example, pressed
direction, between KBr plates).
n = the number of steps (increments) the aperture had been
7.1.3 Spectrometer Information:
moved from its initial location at the articular surface or
7.1.3.1 Manufacturer and model number.
surface of interest, and
7.1.3.2 Analogue or Fourier Transform spectrometer.
S = the step (increment) size in micrometres.
7.1.3.3 Aperture dimensions, profile step size, spectral
resolution, and number of scans per spect
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2102 − 13 F2102 − 17
Standard Guide for
Evaluating the Extent of Oxidation in Polyethylene
Fabricated Forms Intended for Surgical Implants
This standard is issued under the fixed designation F2102; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This guide covers a method for the measurement of the relative extent of oxidation present in HDPE homopolymers and
ultra-high-molecular-weight polyethylene (UHMWPE) intended for use in medical implants. The material is analyzed by infrared
-1
spectroscopy. The intensity (area) of the carbonyl absorptions (>C=O) centered near 1720 cm is related to the amount of
chemically bound oxygen present in the material. Other forms of chemically bound oxygen (C-O-C, C-O-O-C, C-O-H, and so
forth) are not captured by this guide.
1.2 Although this guide may give the investigator a means to compare the relative extent of carbonyl oxidation present in
various UHMWPE samples, it is recognized that other forms of chemically bound oxygen may be important contributors to these
materials’ characteristics.
1.3 The applicability of the infrared method has been demonstrated by many literature reports. This particular method, using
-1
the intensity (area) of the C-H absorption centered near 1370 cm to normalize for the sample’s thickness, has been validated by
an Interlaboratory Study (ILS) conducted according to Practice E691.
1.4 The following precautionary caveat pertains only to the test method portion, Section 5, of this specification: This standard
may involve hazardous materials, operations, and equipment. This standard does not purport to address all of the safety concerns,
if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices
and determine the applicability of regulatory requirements prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E2857 Guide for Validating Analytical Methods
3. Terminology
3.1 Definitions:
3.1.1 bulk oxidation index (BOI)—a sample’s bulk oxidation index (BOI) is the average of the oxidation indices collected over
a 500-μm section at the center of the sample.
This guide is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee F04.15
on Material Test Methods.
Current edition approved Nov. 1, 2013Sept. 1, 2017. Published December 2013September 2017. Originally approved in 2001. Last previous edition approved in 20062013
ε1
as F2102 – 06F2102 – 13. . DOI: 10.1520/F2102-13.10.1520/F2102-17.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
3.1.1.1 Discussion—
Typically, this is a plateau region with the smallest oxidation indices.
3.1.1.2 Discussion—
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2102 − 17
FIG. 1 Typical FTIR Spectra of Oxidized UHMWPE, Showing the Definition of an Area-Based Oxidation Index Based on Normalization
-1
Using the 1370-cm Peak
FIG. 2 FTIR Spectra Showing the Carbonyl Absorption Bands
-1
NOTE 1—Note that both reagents effectively extracted the lipids (the lipid absorption peak is centered at approximately 1740 cm ). The tibial insert
was fabricated from highly crosslinked and remelted UHMWPE followed by terminal sterilization in EtO gas (Ref. 1).
For samples less than about 8 to 10 mm thick, this central region may display the sample’s highest oxidation indices, depending
on its state of oxidation.
3.1.2 depth locator (DL)—a measurement of the distance from the articular surface, or surface of interest, that a spectrum was
collected and a corresponding OI calculated.
3.1.3 oxidation index (OI)—an oxidation index (OI) is defined as the ratio of the area of the carbonyl absorption peak(s) centered
-1 -1
near 1720 cm to the area of the absorption peak(s) centered near 1370 cm , as shown in Fig. 1. Note that the peak areas are
computed after subtracting out the appropriate baseline, as further discussed in Section 6.
3.1.4 oxidation index profile—an oxidation index profile is the graphical representation of variation of the sample’s oxidation
index with distance from its articular surface or the surface of interest. This is a plot of an OI versus DL. Typically, the graph will
show the profile through the entire thickness of the sample.
3.1.5 surface oxidation index (SOI)—a sample’s surface oxidation index (SOI) is the average of the oxidation indices from the
sample’s articular surface, or the surface of interest, to a depth of 3-mm subsurface.
4. Apparatus
4.1 Infrared Spectrometer:
4.1.1 A calibrated infrared spectrometer capable of recording a transmission absorption spectrum over the range of about 1200
-1 -1
to about 2000 cm using about 200-μsm-thick films at a resolution of 4 cm and an aperture of about 200 by 200 μm.
F2102 − 17
4.1.1.1 Other modes of collection (that is, percent reflection, attenuated total reflection (ATR), and so forth) and aperture and
increment sizes may be used to generate the sample’s absorption spectrum provided they can be demonstrated to produce
equivalent results. Too large an aperture can result in a loss of profile accuracy.
4.1.1.2 When a Fourier Transform Infrared (FTIR) spectrometer is used, a minimum of 32 scans shall be collected per
spectrum.spectrum as a default. A fewer number of scans may be performed if a user can verify that their FTIR spectrometer can
reproducibly measure the OI with less than 0.03 difference in OI compared with the result obtained using the default number of
scans. In no case shall an OI be reported based on fewer than 8 scans per spectrum. Guide E2857 provides guidance on how to
validate analytical methods, which the user may use to determine the appropriate number of scans for their laboratory. Depending
on the level of oxidation in the tested samples, the user may elect to establish a lower threshold value of OI (nominally 0.01),
particularly for UHMWPE samples that exhibit little to no detectable oxidation.
4.1.1.3 The FTIR instrument and sample compartment may be purged with a moisture-free inert gas (for example, nitrogen,
helium, or argon) to minimize spectral interference from these components.
4.2 Specimen Holder—Equipment capable of accurately positioning the sample under the orifice in increments at the scale of
the aperture dimensions.
4.3 Microtome—Equipment capable of producing about 200-μm-thick slices (films) of a sample perpendicular to the articular
surface or the surface of interest.
5. Procedure
5.1 Preparation of the Infrared Spectrometer:
5.1.1 Prepare the infrared spectrometer for collection of a transmission absorption spectrum from a thin film of the UHMWPE
sample according to the manufacturer’s recommendations and the conditions described in Section 4 above.
5.1.2 Collect the sequence of spectra per 5.2 and 5.3.
5.2 Preparation of the Test Specimen:
5.2.1 Using a microtome, or other appropriate device, prepare a thin slice of the sample about 200 μm thick.
5.2.2 The slice shall be taken near the center of the sample’s articular surface or the surface of interest.
5.2.3 The orientation of the slice shall typically be perpendicular to the articular surface or the surface of interest.
5.2.4 For explanted components retrieved after in vivo use or in vitro samples that have been exposed to lipids (for example,
simulator specimens exposed to lubricants containing serum), the film should be submerged in a reagent (heptane or hexane) to
extract lipids from the polymer that interfere with the carbonyl peak absorptions. The extraction technique should be verified to
confirm that the oxidation level has stabilized.
5.3 Configuration of the Test Specimen in the Spectrometer:
5.3.1 The test film (slice) shall be first configured in the spectrometer (after an appropriate background spectrum has been
collected) such that the aperture is positioned over the first 200 μm of the film starting at the surface of interest.
5.3.2 Subsequent spectra shall be collected sequentially at increments matching the aperture size (that is, about 200 μm) from
the articular surface, or surface of interest, across the width of the film to the opposite surface.
5.3.2.1 Larger increments may be used; however, too large an increment size may result in a loss of profile accuracy.
6. Calculations
6.1 Oxidation Peak Area (OA):
-1
6.1.1 For each absorbance spectrum, calculate the total area of the carbonyl peak absorptions centered near 1720 cm (Fig. 1).
6.1.1.1 This is the area below the sample’s carbonyl absorption curve and above the straight line baseline drawn between the
starting and ending points.
6.2 Normalization Peak Area (ON):
-1
6.2.1 For each absorbance spectrum, calculate the total area of the peak absorptions centered near 1370 cm (Fig. 1).
6.2.1.1 This is the area below the sample’s absorption curve and above the straight line baseline drawn between the same
starting and ending points.
6.3 Oxidation Index (OI):
6.3.1 For each absorbance spectrum, calculate its OI by dividing the area of its oxidation peak (6.1) by the area of its
normalization peak (6.2), as shown in Fig. 1.
6.4 Oxidation Index Depth Locator (DL):
6.4.1 Calculate the distance from the articular surface, or surface of interest (DL), for each spectrum and its corresponding OI
from the following equation.
DL 5 0.5~A!1n~S!
where:
A = the size of the aperture in micrometres in the step direction,
F2102 − 17
n = the number of steps (increments) the aperture had been moved from its initial location at the articular surface or surface of
interest, and
S = the step (increment) size in micrometres.
6.5 Sample’s Oxidation Index Profile—Construct a plot of a sample’s oxidation indices (OI) versus the corresponding depth
locators (DLs).
6.6 Surface Oxidation Index (SOI)—Calculate a sample’s SOI by calculating the average of the sample’s oxidation indices (OI)
with depth locator (DL) values between 0 and 3000.
6.7 Bulk Oxidation Index (BOI)—Calculate a sample’s BOI by calculating the average of the sample‘s oxidation indices (OIs)
corresponding to the center 500 mm of material.
6.8 Maximum Oxidation Index (MOI)—Calculate the sample’s MOI index observed between depth locator (DL) values of 0 and
3000.
7. Report
7.1 The report shall contain at least the following experimental details and results:
7.1.1 Material Information:
7.1.1.1 Resin type and resin lot number.
7.1.1.2 Consolidation method and manufacturer and manufacturer lot number.
7.1.1.3 Any special post-consolidation treatments, for example, shot isostatic pressing (HIPing), annealing, sterilization,
cross-linking, stabilization, accelerated aging, and storage conditions.
7.1.2 Sample Information:
7.1.2.1 Orthopedic implant or laboratory test specimen.
7.1.2.2 Time elapsed between sample preparation and testing in the FTIR.
7.1.2.3 Articular surface or non-articulator surface.
7.1.2.4 Test sample’s original dimension
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...