ASTM D6042-23
(Test Method)Standard Test Method for Determination of Phenolic Antioxidants and Erucamide Slip Additives in Polypropylene Homopolymer Formulations Using Liquid Chromatography (LC)
Standard Test Method for Determination of Phenolic Antioxidants and Erucamide Slip Additives in Polypropylene Homopolymer Formulations Using Liquid Chromatography (LC)
SIGNIFICANCE AND USE
5.1 Separation and identification of stabilizers used in the manufacture of polypropylene is necessary in order to correlate performance properties with polymer composition. This test method provides a means to determine erucamide slip, Vitamin E, Irgafos 168, Irganox 3114, Irganox 1010, and Irganox 1076 levels in polypropylene samples. This test method is also applicable for the determination of other antioxidants, such as Ultranox 626, Ethanox 330, Santanox R, and BHT, but the applicability of this test method has not been investigated for these antioxidants.
5.2 The additive-extraction procedure is made effective by the insolubility of the polymer sample in solvents generally used for liquid chromatographic analysis.
5.3 Under optimum conditions, the lowest level of detection for a phenolic antioxidant is approximately 2 ppm.
Note 2: Other methods that have been used successfully to remove additives from the plastics matrix include thin film, microwave, ultrasonic, and supercritical fluid extractions. Other methods have been used successfully to separate additives including SFC and capillary GC.
5.4 Irgafos 168 is a phosphite antioxidant. Phosphites are known to undergo both oxidation and hydrolysis reactions. Less Irgafos 168 will be determined in the polymer when oxidation occurs during processing. The HPLC separation is capable of separating the phosphite, phosphate (oxidation product), and hydrolysis product and quantify them if standards are obtained. No significant breakdown of the phosphite antioxidant has been seen due to either extraction technique or the separation presented in this standard.
SCOPE
1.1 This test method covers a liquid-chromatographic procedure for the separation of some additives currently used in polypropylene. These additives are extracted with a cyclohexane:methylene chloride mixture using either reflux or ultrasonic bath prior to liquid-chromatographic separation. The ultraviolet absorbance (200 nm) of the compound(s) is measured, and quantitation is performed using the internal standard method.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific precautionary statements are given in Section 9.
Note 1: There is no known ISO equivalent to this test method.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Oct-2023
- Technical Committee
- D20 - Plastics
- Drafting Committee
- D20.70 - Analytical Methods
Relations
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Apr-2022
- Effective Date
- 01-Apr-2022
- Refers
ASTM D1600-18 - Standard Terminology for Abbreviated Terms Relating to Plastics (Withdrawn 2024) - Effective Date
- 01-Jan-2018
Overview
ASTM D6042-23 specifies the standard test method for the determination of phenolic antioxidants and erucamide slip additives in polypropylene homopolymer formulations using liquid chromatography (LC). This test method provides a reliable and efficient procedure to separate and quantify various stabilizers, antioxidants, and slip agents added to polypropylene (PP) to enhance performance characteristics. Accurate identification and quantification of these additives help ensure product quality and tailor polymer formulations according to specific end-use requirements.
Key Topics
Scope and Applicability
- Covers the extraction and quantification of selected additives in polypropylene using LC with ultraviolet (UV) detection at 200 nm.
- Designed for the determination of erucamide slip additive, vitamin E, Irgafos 168, Irganox 3114, Irganox 1010, and Irganox 1076.
- Also potentially suitable for other antioxidants such as Ultranox 626, Ethanox 330, Santanox R, and BHT, though not formally investigated for these compounds.
Sample Preparation
- Polypropylene samples are ground and extracted using a 75:25 methylene chloride:cyclohexane mixture via reflux or ultrasonic bath.
- The extract is analyzed by liquid chromatography, with quantitation performed using the internal standard method to assure accuracy.
Analytical Detection and Quantification
- The method achieves precise additive extraction due to the insolubility of polypropylene in chromatographic solvents.
- Under optimal conditions, the method can detect phenolic antioxidant levels as low as 2 ppm.
Precision and Sensitivity
- Results have shown high repeatability and reproducibility across multiple laboratories, emphasizing the method's reliability for both intra- and inter-laboratory analysis.
Applications
Quality Control in Polypropylene Manufacturing
- Enables manufacturers and quality control laboratories to monitor and ensure the correct concentration of antioxidants and slip agents in polypropylene homopolymer products.
- Facilitates correlation between additive concentration and desired material properties, such as oxidative stability and processability.
Polymer Formulation Development
- Essential for R&D activities in the plastics industry, supporting optimization of stabilizer packages to meet performance requirements for specific applications.
Regulatory Compliance
- Supports compliance with industry standards and customer specifications by providing traceable, reproducible test results for polypropylene additives.
Troubleshooting and Product Development
- Useful for investigating performance issues related to degradation, slippage, or discoloration potentially linked to additive concentration during polymer processing.
Related Standards
- ASTM D883 - Terminology Relating to Plastics
- ASTM D1600 - Terminology for Abbreviated Terms Relating to Plastics
- ASTM E131 - Terminology Relating to Molecular Spectroscopy
- ASTM E456 - Terminology Relating to Quality and Statistics
- ASTM E691 - Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
- IEEE/ASTM SI-10 - Practice for Use of the International System of Units (SI)
- Note: There is no ISO equivalent for ASTM D6042-23 at this time.
By following ASTM D6042-23, laboratories and manufacturers in the plastics industry achieve accurate, reproducible, and sensitive quantification of critical stabilizers and slip additives in polypropylene materials. This supports robust quality assurance, process optimization, and product innovation, reinforcing the value of standardized test methods in polymer analytics.
Buy Documents
ASTM D6042-23 - Standard Test Method for Determination of Phenolic Antioxidants and Erucamide Slip Additives in Polypropylene Homopolymer Formulations Using Liquid Chromatography (LC)
REDLINE ASTM D6042-23 - Standard Test Method for Determination of Phenolic Antioxidants and Erucamide Slip Additives in Polypropylene Homopolymer Formulations Using Liquid Chromatography (LC)
Get Certified
Connect with accredited certification bodies for this standard

Smithers Quality Assessments
US management systems and product certification.

DIN CERTCO
DIN Group product certification.
Sponsored listings
Frequently Asked Questions
ASTM D6042-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Phenolic Antioxidants and Erucamide Slip Additives in Polypropylene Homopolymer Formulations Using Liquid Chromatography (LC)". This standard covers: SIGNIFICANCE AND USE 5.1 Separation and identification of stabilizers used in the manufacture of polypropylene is necessary in order to correlate performance properties with polymer composition. This test method provides a means to determine erucamide slip, Vitamin E, Irgafos 168, Irganox 3114, Irganox 1010, and Irganox 1076 levels in polypropylene samples. This test method is also applicable for the determination of other antioxidants, such as Ultranox 626, Ethanox 330, Santanox R, and BHT, but the applicability of this test method has not been investigated for these antioxidants. 5.2 The additive-extraction procedure is made effective by the insolubility of the polymer sample in solvents generally used for liquid chromatographic analysis. 5.3 Under optimum conditions, the lowest level of detection for a phenolic antioxidant is approximately 2 ppm. Note 2: Other methods that have been used successfully to remove additives from the plastics matrix include thin film, microwave, ultrasonic, and supercritical fluid extractions. Other methods have been used successfully to separate additives including SFC and capillary GC. 5.4 Irgafos 168 is a phosphite antioxidant. Phosphites are known to undergo both oxidation and hydrolysis reactions. Less Irgafos 168 will be determined in the polymer when oxidation occurs during processing. The HPLC separation is capable of separating the phosphite, phosphate (oxidation product), and hydrolysis product and quantify them if standards are obtained. No significant breakdown of the phosphite antioxidant has been seen due to either extraction technique or the separation presented in this standard. SCOPE 1.1 This test method covers a liquid-chromatographic procedure for the separation of some additives currently used in polypropylene. These additives are extracted with a cyclohexane:methylene chloride mixture using either reflux or ultrasonic bath prior to liquid-chromatographic separation. The ultraviolet absorbance (200 nm) of the compound(s) is measured, and quantitation is performed using the internal standard method. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific precautionary statements are given in Section 9. Note 1: There is no known ISO equivalent to this test method. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Separation and identification of stabilizers used in the manufacture of polypropylene is necessary in order to correlate performance properties with polymer composition. This test method provides a means to determine erucamide slip, Vitamin E, Irgafos 168, Irganox 3114, Irganox 1010, and Irganox 1076 levels in polypropylene samples. This test method is also applicable for the determination of other antioxidants, such as Ultranox 626, Ethanox 330, Santanox R, and BHT, but the applicability of this test method has not been investigated for these antioxidants. 5.2 The additive-extraction procedure is made effective by the insolubility of the polymer sample in solvents generally used for liquid chromatographic analysis. 5.3 Under optimum conditions, the lowest level of detection for a phenolic antioxidant is approximately 2 ppm. Note 2: Other methods that have been used successfully to remove additives from the plastics matrix include thin film, microwave, ultrasonic, and supercritical fluid extractions. Other methods have been used successfully to separate additives including SFC and capillary GC. 5.4 Irgafos 168 is a phosphite antioxidant. Phosphites are known to undergo both oxidation and hydrolysis reactions. Less Irgafos 168 will be determined in the polymer when oxidation occurs during processing. The HPLC separation is capable of separating the phosphite, phosphate (oxidation product), and hydrolysis product and quantify them if standards are obtained. No significant breakdown of the phosphite antioxidant has been seen due to either extraction technique or the separation presented in this standard. SCOPE 1.1 This test method covers a liquid-chromatographic procedure for the separation of some additives currently used in polypropylene. These additives are extracted with a cyclohexane:methylene chloride mixture using either reflux or ultrasonic bath prior to liquid-chromatographic separation. The ultraviolet absorbance (200 nm) of the compound(s) is measured, and quantitation is performed using the internal standard method. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific precautionary statements are given in Section 9. Note 1: There is no known ISO equivalent to this test method. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D6042-23 is classified under the following ICS (International Classification for Standards) categories: 83.080.20 - Thermoplastic materials. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D6042-23 has the following relationships with other standards: It is inter standard links to ASTM D6042-09(2016), ASTM D883-24, ASTM D883-23, ASTM E456-13a(2022)e1, ASTM E456-13a(2022), ASTM D1600-18. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D6042-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D6042 − 23
Standard Test Method for
Determination of Phenolic Antioxidants and Erucamide Slip
Additives in Polypropylene Homopolymer Formulations
Using Liquid Chromatography (LC)
This standard is issued under the fixed designation D6042; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* E131 Terminology Relating to Molecular Spectroscopy
E456 Terminology Relating to Quality and Statistics
1.1 This test method covers a liquid-chromatographic pro-
E691 Practice for Conducting an Interlaboratory Study to
cedure for the separation of some additives currently used in
Determine the Precision of a Test Method
polypropylene. These additives are extracted with a cyclo-
IEEE/ASTM SI-10 Practice for Use of the International
hexane:methylene chloride mixture using either reflux or
System of Units (SI) (the Modernized Metric System)
ultrasonic bath prior to liquid-chromatographic separation. The
ultraviolet absorbance (200 nm) of the compound(s) is
3. Terminology
measured, and quantitation is performed using the internal
standard method.
3.1 For definitions of plastic terms used in this test method,
see Terminologies D883 and D1600.
1.2 The values stated in SI units are to be regarded as
standard. No other units of measurement are included in this
3.2 For the units, symbols, and abbreviations used in this
standard.
test method, refer to Terminology E131 or Practice IEEE/
ASTM SI-10.
1.3 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
3.3 Abbreviations:
responsibility of the user of this standard to establish appro-
3.3.1 LC—liquid chromatography.
priate safety, health, and environmental practices and deter-
3.3.2 PP—polypropylene.
mine the applicability of regulatory limitations prior to use.
3.4 Vitamin E—α-Tocopherol, or 3,4-dihydro-2,5,7,8-
Specific precautionary statements are given in Section 9.
tetramethyl-2-(4,8,12-trimethyltridecyl)-2H-1-benzopyran-6-
NOTE 1—There is no known ISO equivalent to this test method.
ol.
1.4 This international standard was developed in accor-
3.5 Irgafos 168—Tris(2,4 di-tert-butylphenyl) phosphite.
dance with internationally recognized principles on standard-
3.6 Irganox 3114—Tris(3,5-di-t-butyl-4-hydroxybenzyl)
ization established in the Decision on Principles for the
isocyanurate.
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
3.7 Kemamide-E—cis-13-docosenamide or erucamide.
Barriers to Trade (TBT) Committee.
3.8 Irganox 1010—tetrakis[methylene(3,5-di-t-butyl-4-
hydroxy hydrocinnamate)]methane.
2. Referenced Documents
3.9 Irganox 1076—octadecyl-3,5-di-t-butyl-4-hydroxy hy-
2.1 ASTM Standards:
drocinnamate.
D883 Terminology Relating to Plastics
D1600 Terminology for Abbreviated Terms Relating to Plas- 3.10 Tinuvin P—2(2'-hydroxy-5'-methyl phenyl)benzotriaz-
tics ole.
4. Summary of Test Method
This test method is under the jurisdiction of ASTM Committee D20 on Plastics
and is the direct responsibility of Subcommittee D20.70 on Analytical Methods.
4.1 The PP sample is ground to a 20-mesh particle size (850
Current edition approved Nov. 1, 2023. Published December 2023. Originally
microns) and extracted by refluxing with a mixture of 75:25
approved in 1996. Last previous edition approved in 2016 as D6042 - 09 (2016).
methylene chloride:cyclohexane or placing in an ultrasonic
DOI: 10.1520/D6042-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or bath with the same mixture.
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
4.2 The solvent extract is examined by liquid chromatogra-
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. phy.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6042 − 23
4.3 Additive concentrations are determined relative to an
internal standard (contained in the solvent) using reverse-phase
chromatography (C-18 column) with ultraviolet (UV) detection
at 200 nm.
5. Significance and Use
5.1 Separation and identification of stabilizers used in the
manufacture of polypropylene is necessary in order to correlate
performance properties with polymer composition. This test
method provides a means to determine erucamide slip, Vitamin
E, Irgafos 168, Irganox 3114, Irganox 1010, and Irganox 1076
levels in polypropylene samples. This test method is also
applicable for the determination of other antioxidants, such as
Ultranox 626, Ethanox 330, Santanox R, and BHT, but the
applicability of this test method has not been investigated for
these antioxidants.
5.2 The additive-extraction procedure is made effective by
the insolubility of the polymer sample in solvents generally
used for liquid chromatographic analysis.
5.3 Under optimum conditions, the lowest level of detection
for a phenolic antioxidant is approximately 2 ppm.
NOTE 2—Other methods that have been used successfully to remove
additives from the plastics matrix include thin film, microwave, ultrasonic,
and supercritical fluid extractions. Other methods have been used success-
fully to separate additives including SFC and capillary GC.
5.4 Irgafos 168 is a phosphite antioxidant. Phosphites are
known to undergo both oxidation and hydrolysis reactions.
Less Irgafos 168 will be determined in the polymer when
oxidation occurs during processing. The HPLC separation is
capable of separating the phosphite, phosphate (oxidation
product), and hydrolysis product and quantify them if stan-
dards are obtained. No significant breakdown of the phosphite
antioxidant has been seen due to either extraction technique or
the separation presented in this standard.
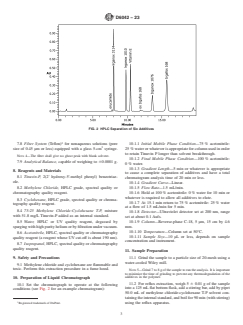
6. Interferences FIG. 1 Sample Extraction
6.1 Any material eluting at or near the same retention time
as the additive has been known to cause erroneous results.
7.2 Chromatographic Column, RP-18, 5-μm particle size,
Examining a polymer-solvent-extract solution containing no
15 cm by 4.6 mm.
internal standard is recommended to minimize the possibility
NOTE 3—A Zorbax RX C-18 Column by Agilent was used for this test
of interferences.
method. The gradient described in 10.1 provides complete separation of
6.2 Solvent impurity is a major source of interference. It is
antioxidants using this C-18 column.
a good practice to examine the solvents prior to use by
7.3 Computer System or Integrator, coupled with the chro-
injecting a sample of solvent on the HPLC system and
matograph is recommended to measure peak area.
analyzing as in Section 10.
7.4 Wiley Mill, equipped with a 20-mesh screen and water-
6.3 The oxidation product of Irgafos 168 sometimes over-
cooled jacket to prevent thermodegradation of antioxidants.
laps with other additives with retention times of components
7.5 Recorder, mV scale dependent upon the output of the
that elute between Irganox 1010 and Irgafos 168. In these
detector.
cases, run known standards to ensure that components of
interest do not coelute with the oxidation product of Irgafos 7.6 Reflux Extraction Apparatus, consisting of a condenser
168.
(24/40 ground-glass joint), a flat bottom 125 mL flask having
a 24/40 ground-glass joint, and a hot plate with magnetic stirrer
7. Apparatus
(see Fig. 1).
7.1 Liquid Chromatograph, equipped with a sample 7.7 Ultrasonic Bath Apparatus, consisting of the ultrasonic
injector, variable-wavelength ultraviolet detector, heated bath, a flat bottom 125-mL flask having a 24/40 ground-glass
column, and gradient-elution capabilities. joint, and a 24/40 ground-glass stopper.
D6042 − 23
FIG. 2 HPLC Separation of Six Additives
7.8 Filter System (Teflon) for nonaqueous solutions (pore 10.1.1 Initial Mobile Phase Condition—75 % acetonitrile:
size of 0.45 μm or less) equipped with a glass 5-cm syringe. 25 % water or whatever is appropriate for column used in order
to retain Tinuvin P longer than solvent breakthrough.
NOTE 4—The filter shall give no ghost peak with blank solvent.
10.1.2 Final Mobile Phase Condition—100 % acetonitrile:
7.9 Analytical Balance, capable of weighing to 60.0001 g.
0 % water.
10.1.3 Gradient Length—5 min or whatever is appropriate
8. Reagents and Materials
to cause a complete separation o
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D6042 − 09 (Reapproved 2016) D6042 − 23
Standard Test Method for
Determination of Phenolic Antioxidants and Erucamide Slip
Additives in Polypropylene Homopolymer Formulations
Using Liquid Chromatography (LC)
This standard is issued under the fixed designation D6042; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Scope*
1.1 This test method covers a liquid-chromatographic procedure for the separation of some additives currently used in
polypropylene. These additives are extracted with a cyclohexane:methylene chloride mixture using either reflux or ultrasonic bath
prior to liquid-chromatographic separation. The ultraviolet absorbance (200 nm) of the compound(s) is measured, and quantitation
is performed using the internal standard method.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and healthsafety, health, and environmental practices and determine
the applicability of regulatory limitations prior to use. Specific precautionary statements are given in Section 9.
NOTE 1—There is no known ISO equivalent to this test method.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D883 Terminology Relating to Plastics
D1600 Terminology for Abbreviated Terms Relating to Plastics
E131 Terminology Relating to Molecular Spectroscopy
E456 Terminology Relating to Quality and Statistics
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
IEEE/ASTM SI-10 Practice for Use of the International System of Units (SI) (the Modernized Metric System)
3. Terminology
3.1 For definitions of plastic terms used in this test method, see Terminologies D883 and D1600.
This test method is under the jurisdiction of ASTM Committee D20 on Plastics and is the direct responsibility of Subcommittee D20.70 on Analytical Methods
(D20.70.02).
Current edition approved Sept. 1, 2016Nov. 1, 2023. Published September 2016December 2023. Originally approved in 1996. Last previous edition approved in 20092016
as D6042 - 09.D6042 - 09 (2016). DOI: 10.1520/D6042-09R16.10.1520/D6042-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6042 − 23
3.2 For the units, symbols, and abbreviations used in this test method, refer to Terminology E131 or Practice IEEE/ASTM SI-10.
3.3 Abbreviations:
3.3.1 LC—liquid chromatography.
3.3.2 PP—polypropylene.
3.4 Trade Names:
3.4 Vitamin E—α-Tocopherol, or 3,4-dihydro-2,5,7,8-tetramethyl-2-(4,8,12-trimethyltridecyl)-2H-1-benzopyran-6- ol.
3.5 Irgafos 168—Tris(2,4 di-tert-butylphenyl) phosphite.
3.6 Irganox 3114—Tris(3,5-di-t-butyl-4-hydroxybenzyl) isocyanurate.
3.7 Kemamide-E—cis-13-docosenamide or erucamide.
3.8 Irganox 1010—tetrakis[methylene(3,5-di-t-butyl-4-hydroxy hydrocinnamate)]methane.
3.9 Irganox 1076—octadecyl-3,5-di-t-butyl-4-hydroxy hydrocinnamate.
3.10 Tinuvin P—2(2'-hydroxy-5'-methyl phenyl)benzotriazole.
4. Summary of Test Method
4.1 The PP sample is ground to a 20-mesh particle size (850 microns) and extracted by refluxing with a mixture of 75:25
methylene chloride:cyclohexane or placing in an ultrasonic bath with the same mixture.
4.2 The solvent extract is examined by liquid chromatography.
4.3 Additive concentrations are determined relative to an internal standard (contained in the solvent) using reverse-phase
chromatography (C-18 column) with ultraviolet (UV) detection at 200 nm.
5. Significance and Use
5.1 Separation and identification of stabilizers used in the manufacture of polypropylene is necessary in order to correlate
performance properties with polymer composition. This test method provides a means to determine erucamide slip, Vitamin E,
Irgafos 168, Irganox 3114, Irganox 1010, and Irganox 1076 levels in polypropylene samples. This test method is also applicable
for the determination of other antioxidants, such as Ultranox 626, Ethanox 330, Santanox R, and BHT, but the applicability of this
test method has not been investigated for these antioxidants.
5.2 The additive-extraction procedure is made effective by the insolubility of the polymer sample in solvents generally used for
liquid chromatographic analysis.
5.3 Under optimum conditions, the lowest level of detection for a phenolic antioxidant is approximately 2 ppm.
NOTE 2—Other methods that have been used successfully to remove additives from the plastics matrix include thin film, microwave, ultrasonic, and
supercritical fluid extractions. Other methods have been used successfully to separate additives including SFC and capillary GC.
5.4 Irgafos 168 is a phosphite antioxidant. Phosphites are known to undergo both oxidation and hydrolysis reactions. Less Irgafos
168 will be determined in the polymer when oxidation occurs during processing. The HPLC separation is capable of separating
D6042 − 23
the phosphite, phosphate (oxidation product), and hydrolysis product and quantify them if standards are obtained. No significant
breakdown of the phosphite antioxidant has been seen due to either extraction technique or the separation presented in this
standard.
6. Interferences
6.1 Any material eluting at or near the same retention time as the additive has been known to cause erroneous results. Examining
a polymer-solvent-extract solution containing no internal standard is recommended to minimize the possibility of interferences.
6.2 Solvent impurity is a major source of interference. It is a good practice to examine the solvents prior to use by injecting a
sample of solvent on the HPLC system and analyzing as in Section 10.
6.3 The oxidation product of Irgafos 168 sometimes overlaps with other additives with retention times of components that elute
between Irganox 1010 and Irgafos 168. In these cases, run known standards to ensure that components of interest do not coelute
with the oxidation product of Irgafos 168.
7. Apparatus
7.1 Liquid Chromatograph, equipped with a sample injector, variable-wavelength ultraviolet detector, heated column, and
gradient-elution capabilities.
7.2 Chromatographic Column, RP-18, 5-μm particle size, 15 cm by 4.6 mm.
NOTE 3—A Zorbax RX C-18 Column by Agilent was used for this test method. The gradient described in 10.1 provides complete separation of
antioxidants using this C-18 column.
7.3 Computer System or Integrator, coupled with the chromatograph is recommended to measure peak area.
7.4 Wiley Mill, equipped with a 20-mesh screen and water-cooled jacket to prevent thermodegradation of antioxidants.
7.5 Recorder, mV scale dependent upon the output of the detector.
7.6 Reflux Extraction Apparatus, consisting of a condenser (24/40 ground-glass joint), a flat bottom 125 mL flask having a 24/40
ground-glass joint, and a hot plate with magnetic stirrer (see Fig. 1).
7.7 Ultrasonic Bath Apparatus, consisting of the ultrasonic bath, a flat bottom 125-mL flask having a 24/40 ground-glass joint,
and a 24/40 ground-glass stopper.
3 3
7.8 Filter System (Teflon) for nonaqueous solutions (pore size of 0.45 μm or less) equipped with a glass 5-cm syringe.
NOTE 4—The filter shall give no ghost peak with blank solvent.
7.9 Analytical Balance, capable of weighing to 60.0001 g.
8. Reagents and Materials
8.1 Tinuvin-P, 2(2' hydroxy-5'-methyl phenyl) benzotriazole.
8.2 Methylene Chloride, HPLC grade, spectral quality or chromatography quality reagent.
8.3 Cyclohexane, HPLC grade, spectral quality or chromatography quality reagent.
Registered trademark of DuPont.
D6042 − 23
FIG. 1 Sample Extraction
8.4 75:25 Methylene Chloride:Cyclohexane T-P, mixture with 51.8 mg/L Tinuvin-P added as an internal standard.
8.5 Water, HPLC or UV quality reagent, degassed by sparging with high purity helium or by filtration under vacuum.
8.6 Acetonitrile, HPLC, spectral quality or chromatography quality reagent (a reagent whose UV cut-off is about 190 nm).
8.7 Isopropanol, HPLC, spectral quality or chromatography quality reagent.
9. Safety and Precautions
9.1 Methylene chloride and cyclohexane are flammable and toxic. Perform this extraction procedure in a fume hood.
10. Preparation of Liqui
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...