ASTM F2392-04(2015)
(Test Method)Standard Test Method for Burst Strength of Surgical Sealants
Standard Test Method for Burst Strength of Surgical Sealants
SIGNIFICANCE AND USE
4.1 Materials and devices that function at least in part by adhering to living tissues are finding increasing use in surgical procedures, either as adjuncts to sutures and staples or as frank replacements for those devices in a wide variety of medical procedures. While the nature and magnitude of the forces involved varies greatly with indication and with patient specific circumstances, all uses involve, to some extent, the ability of the material to resist imposed mechanical forces. Therefore, the mechanical properties of the materials, and in particular the adhesive and cohesive properties, are important parameters in evaluating their fitness for use. In addition, the mechanical properties of a given sealant composition can provide a useful means of determining product consistency for quality control, or as a means for determining the effects of various surface treatments on the substrate prior to use of the device.
4.2 The complexity and variety of individual applications for sealant, even within a single indicated use (surgical procedure), is such that the results of a burst test are not suitable for determining allowable design stresses without thorough analysis and understanding of the application and sealant behaviors.
4.3 This test method may be used for comparing sealants for susceptibility to environmental changes, but such comparisons must be made with great caution since different sealants may respond differently to varying conditions.
4.4 As the true sealant strength is strongly dependent on the strength of the sealant/substrate interface, the selection of a proper test substrate is critical. Care must be taken when extrapolating in vitro test results to in vivo expectations. In vitro sealant optimization may not translate to expected in vivo performance due to differences in substrate surface, strength, and elasticity.
SCOPE
1.1 This test method provides a means for comparison of the burst or rupture strength of sealants on soft tissue. This test method can be used as a clinically relevant model for quality assurance, development, and comparative testing of different adhesives or adherends.
1.2 This test method measures only burst strength or “cohesive strength” of an adhesive/adherend system, and not the adhesive strength.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2015
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.15 - Material Test Methods
Relations
- Effective Date
- 01-May-2015
- Effective Date
- 01-Jul-2012
- Effective Date
- 01-May-2012
- Effective Date
- 01-Dec-2011
- Effective Date
- 01-Jan-2011
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-Aug-2008
- Effective Date
- 01-Mar-2008
- Effective Date
- 01-Dec-2006
- Effective Date
- 01-Apr-2005
- Effective Date
- 01-Apr-2005
- Effective Date
- 01-Dec-2004
- Effective Date
- 01-Apr-2004
- Effective Date
- 10-May-2003
- Effective Date
- 10-Oct-2000
Overview
ASTM F2392-04(2015), titled Standard Test Method for Burst Strength of Surgical Sealants, is an internationally recognized standard developed by ASTM International. This standard provides a laboratory method to evaluate and compare the burst or rupture strength-also known as cohesive strength-of surgical sealants applied to soft tissue substrates. The principal goal is to support the development, quality assurance, and comparative testing of medical adhesives and sealants used as adjuncts or alternatives to sutures and staples in a variety of surgical procedures.
Understanding the mechanical integrity of surgical sealants is critical, as these materials must reliably resist mechanical forces when applied to living tissues. The test method described helps users assess factors such as product consistency, quality control, and the impact of surface treatments on sealant efficacy. ASTM F2392 applies only to SI units and does not address all safety aspects, emphasizing the need for users to establish appropriate safety and health practices.
Key Topics
Burst Strength Measurement
The core focus is on determining the burst (or rupture) strength of a surgical sealant-defined as the average pressure needed to cause cohesive or adhesive failure when the sealant is applied to a tissue or model substrate.Cohesive vs. Adhesive Strength
This method specifically measures the cohesive strength, or internal strength of the sealant, rather than the adhesive strength between the sealant and the tissue.Test Substrate Selection
Proper selection and preparation of the test substrate is crucial as the true performance of the sealant depends significantly on the sealant/substrate interface. Both model substrates (such as collagen casings) and animal tissues may be used, depending on the specific application and quality control requirements.Environmental Considerations
Environmental factors such as temperature and humidity are controlled during testing, as these can affect the outcome and comparability of results. Recommended conditions are provided for both comparative and quality control testing.Reporting and Documentation
The standard outlines specific data to be recorded in all test reports, including substrate and sealant details, application method, ambient conditions, burst strength results, and types of failure observed.
Applications
ASTM F2392-04(2015) is widely applied in:
Quality Assurance
Manufacturers use the method during production to verify product consistency and performance of surgical sealants before clinical use.Product Development
Research and development teams employ this standard to compare and optimize sealant formulations, ensuring adequate burst strength for various surgical scenarios.Comparative Testing
Laboratories and regulatory bodies utilize this test to benchmark new or competing sealant products, evaluating their mechanical integrity under controlled conditions.Assessment of Environmental Susceptibility
The method aids in evaluating how sealant performance changes under different conditions, helping inform materials selection and improvements.
It is important to note that while these burst strength results are useful for comparative assessments, they are not directly suitable for determining clinical design stresses without further application-specific analysis.
Related Standards
ASTM D907: Terminology of Adhesives
Provides definitions relevant to adhesives, many of which are referenced in ASTM F2392.American Association for Tissue Banks (AATB) Standards:
Cover best practices for tissue handling and storage, relevant for testing with biological substrates.Other Surgical Sealant Standards:
Additional ASTM standards relating to medical adhesive test methods and characterizations may also be applicable, depending on the specific requirements of the device or procedure.
By following ASTM F2392-04(2015), stakeholders can ensure a robust and standardized approach to evaluating the burst strength of surgical sealants, contributing to improved product reliability and patient safety in diverse surgical applications.
Buy Documents
ASTM F2392-04(2015) - Standard Test Method for Burst Strength of Surgical Sealants
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2392-04(2015) is a standard published by ASTM International. Its full title is "Standard Test Method for Burst Strength of Surgical Sealants". This standard covers: SIGNIFICANCE AND USE 4.1 Materials and devices that function at least in part by adhering to living tissues are finding increasing use in surgical procedures, either as adjuncts to sutures and staples or as frank replacements for those devices in a wide variety of medical procedures. While the nature and magnitude of the forces involved varies greatly with indication and with patient specific circumstances, all uses involve, to some extent, the ability of the material to resist imposed mechanical forces. Therefore, the mechanical properties of the materials, and in particular the adhesive and cohesive properties, are important parameters in evaluating their fitness for use. In addition, the mechanical properties of a given sealant composition can provide a useful means of determining product consistency for quality control, or as a means for determining the effects of various surface treatments on the substrate prior to use of the device. 4.2 The complexity and variety of individual applications for sealant, even within a single indicated use (surgical procedure), is such that the results of a burst test are not suitable for determining allowable design stresses without thorough analysis and understanding of the application and sealant behaviors. 4.3 This test method may be used for comparing sealants for susceptibility to environmental changes, but such comparisons must be made with great caution since different sealants may respond differently to varying conditions. 4.4 As the true sealant strength is strongly dependent on the strength of the sealant/substrate interface, the selection of a proper test substrate is critical. Care must be taken when extrapolating in vitro test results to in vivo expectations. In vitro sealant optimization may not translate to expected in vivo performance due to differences in substrate surface, strength, and elasticity. SCOPE 1.1 This test method provides a means for comparison of the burst or rupture strength of sealants on soft tissue. This test method can be used as a clinically relevant model for quality assurance, development, and comparative testing of different adhesives or adherends. 1.2 This test method measures only burst strength or “cohesive strength” of an adhesive/adherend system, and not the adhesive strength. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 4.1 Materials and devices that function at least in part by adhering to living tissues are finding increasing use in surgical procedures, either as adjuncts to sutures and staples or as frank replacements for those devices in a wide variety of medical procedures. While the nature and magnitude of the forces involved varies greatly with indication and with patient specific circumstances, all uses involve, to some extent, the ability of the material to resist imposed mechanical forces. Therefore, the mechanical properties of the materials, and in particular the adhesive and cohesive properties, are important parameters in evaluating their fitness for use. In addition, the mechanical properties of a given sealant composition can provide a useful means of determining product consistency for quality control, or as a means for determining the effects of various surface treatments on the substrate prior to use of the device. 4.2 The complexity and variety of individual applications for sealant, even within a single indicated use (surgical procedure), is such that the results of a burst test are not suitable for determining allowable design stresses without thorough analysis and understanding of the application and sealant behaviors. 4.3 This test method may be used for comparing sealants for susceptibility to environmental changes, but such comparisons must be made with great caution since different sealants may respond differently to varying conditions. 4.4 As the true sealant strength is strongly dependent on the strength of the sealant/substrate interface, the selection of a proper test substrate is critical. Care must be taken when extrapolating in vitro test results to in vivo expectations. In vitro sealant optimization may not translate to expected in vivo performance due to differences in substrate surface, strength, and elasticity. SCOPE 1.1 This test method provides a means for comparison of the burst or rupture strength of sealants on soft tissue. This test method can be used as a clinically relevant model for quality assurance, development, and comparative testing of different adhesives or adherends. 1.2 This test method measures only burst strength or “cohesive strength” of an adhesive/adherend system, and not the adhesive strength. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM F2392-04(2015) is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2392-04(2015) has the following relationships with other standards: It is inter standard links to ASTM F2392-04(2010), ASTM D907-12a, ASTM D907-12, ASTM D907-11a, ASTM D907-11, ASTM D907-08b, ASTM D907-08a, ASTM D907-08, ASTM D907-06, ASTM D907-05, ASTM D907-05e1, ASTM D907-04a, ASTM D907-04, ASTM D907-03, ASTM D907-00. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2392-04(2015) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2392 − 04 (Reapproved 2015)
Standard Test Method for
Burst Strength of Surgical Sealants
This standard is issued under the fixed designation F2392; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 3.2.2 burst strength—the average pressure required to cause
failure of the sealant, either by cohesive or adhesive mecha-
1.1 Thistestmethodprovidesameansforcomparisonofthe
nisms.
burst or rupture strength of sealants on soft tissue. This test
3.2.3 cohesive failure—failure of the sealant during burst
method can be used as a clinically relevant model for quality
testing.
assurance, development, and comparative testing of different
adhesives or adherends.
3.2.4 cohesive strength—the internal strength of the sealant,
sometimes referred to as the adhesive bulk strength.
1.2 This test method measures only burst strength or “co-
hesive strength” of an adhesive/adherend system, and not the
3.2.5 substrate failure—failure of the substrate during burst
adhesive strength.
testing.
1.3 The values stated in SI units are to be regarded as 3.2.6 tissue sealant—a surface coating to prevent leakage of
standard. No other units of measurement are included in this body fluids.
standard.
4. Significance and Use
1.4 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the 4.1 Materials and devices that function at least in part by
adhering to living tissues are finding increasing use in surgical
responsibility of the user of this standard to establish appro-
priate safety and health practices and determine the applica- procedures, either as adjuncts to sutures and staples or as frank
replacements for those devices in a wide variety of medical
bility of regulatory limitations prior to use.
procedures. While the nature and magnitude of the forces
2. Referenced Documents
involvedvariesgreatlywithindicationandwithpatientspecific
circumstances, all uses involve, to some extent, the ability of
2.1 ASTM Standards:
thematerialtoresistimposedmechanicalforces.Therefore,the
D907 Terminology of Adhesives
mechanical properties of the materials, and in particular the
2.2 American Association for Tissue Banks (AATB) Stan-
adhesive and cohesive properties, are important parameters in
dard:
evaluating their fitness for use. In addition, the mechanical
Standards for Tissue Banking
properties of a given sealant composition can provide a useful
3. Terminology means of determining product consistency for quality control,
or as a means for determining the effects of various surface
3.1 Definitions—Many terms in this test method are defined
treatments on the substrate prior to use of the device.
in Terminology D907.
4.2 The complexity and variety of individual applications
3.2 Definitions:
for sealant, even within a single indicated use (surgical
3.2.1 adhesive failure—failure of the sealant/substrate inter-
procedure), is such that the results of a burst test are not
face during burst testing.
suitable for determining allowable design stresses without
thorough analysis and understanding of the application and
sealant behaviors.
This test method is under the jurisdiction ofASTM Committee F04 on Medical
and Surgical Materials and Devicesand is the direct responsibility of Subcommittee
4.3 Thistestmethodmaybeusedforcomparingsealantsfor
F04.15 on Material Test Methods.
susceptibility to environmental changes, but such comparisons
CurrenteditionapprovedMay1,2015.PublishedJuly2015.Originallyapproved
in 2004. Last previous edition approved in 2010 as F2392 – 04 (2010). DOI:
must be made with great caution since different sealants may
10.1520/F2392-04R15.
respond differently to varying conditions.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
4.4 As the true sealant strength is strongly dependent on the
Standards volume information, refer to the standard’s Document Summary page on
strength of the sealant/substrate interface, the selection of a
the ASTM website.
proper test substrate is critical. Care must be taken when
Available from American Association for Tissue Banks (AATB), 1320 Old
Chain Bridge Rd., Suite 450, McLean, VA 22101. extrapolating in vitro test results to in vivo expectations. In
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2392 − 04 (2015)
vitro sealant optimization may not translate to expected in vivo that relatively large samples of tissue can be harvested from a
performance due to differences in substrate surface, strength, single source. Ideally, the tissue should be used within 24 h of
and elasticity. harvest and should be kept between 5 and 10°C prior to testing
if it cannot be used immediately after harvesting. Storage and
5. Apparatus
handling of tissue samples should be carried out according to
the guidelines set forth in Standards for Tissue Banking by the
5.1 Testing Machine—Atestingmachinefordeterminingthe
American Association of Tissue Banks. The specimens should
sealant strength and system failure mechanism and comprising
be brought to the test temperature or other prescribed tempera-
essentially the following:
ture (such as body temperature) prior to application of the
5.1.1 Test Fixture—A stationary fixture containing the test
sealant.
substrate and applied sealant. Fluid flows into the fixture at a
6.3.2 Fixed tissue should not be used since it has been
fixed rate, allowing for the pressurization of the sealed sub-
demonstrated that fixatives cause large alterations in the
strate.
mechanical properties of the tissue and it is probable that the
5.1.2 Positive Displacement Fluid Pump—A pump provid-
adhesive strength would be affected as well.
ing a constant flow of fluid to the test fixture. The pump must
6.3.3 Ifthetargetorganisofasizeorgeometry,orboth,that
be capable of constant flow at pressures of interest. Syringe
does not allow fabrication of test samples, a tissue of similar
pumps are particularly well suited for this type of testing since
origin but larger size should be used.
they do not cause pulsatile flow. Peristaltic pumps have also
6.3.4 The thickness of the tissue sample should be mini-
been used successfully since the pump tubing tends to dampen
mized and should not exceed 5 mm. Thicker samples will lead
pulsations.
to distortion of the substrate and may leak in the test fixture.
NOTE 1—Saline is the typical fluid of choice. When air is used, a
Also, thicker samples will lead to sealant adherence on the
reduction in pressurization rate is expected due to gas compressibility.
insides of the hole itself, possibly leading to different failure
5.1.3 Pressure gage—Consisting of a gage and method of
mechanisms. It is also important that the thickness be as
capturing peak pressures. System sampling rate should be
uniform as possible.
adequate to capture peak burst pressures. Sensitivity and
6.4 Substrates for Quality Control Testing:
precision should result in less than 1 % error. The burst test
6.4.1 For testing that is undertaken as part of a quality
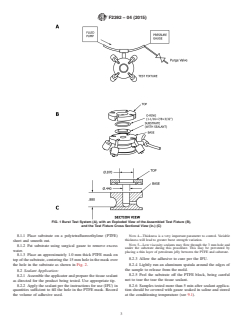
system is shown in Fig. 1. The system (A) consists of a fluid
control process in the manufacturing of a tissue sealant, the use
pump, a test fixture, and pressure gage connected by rigid
of freshly harvested tissue is highly inconvenient and may also
plastic tubing. The test fixture (B) consists of a base, O-ring,
lead to unacceptable variation in the test results, especially if
and top.
the failure occurs in the adherend (substrate failure). Since the
5.2 Temperature-controlling Equipment—Must be capable
purpose of quality control testing is to demonstrate consistency
of maintaining the test temperature to 62°C. If ambient
in the device, substitution of a model substrate is preferred so
laboratory conditions are employed, the same degree of control
long as it is demonstrated that the sealant does bond to the
is required.Awater bath or environmental chamber capable of
adherend.Sincethebursttestfailuremechanismcandependon
maint
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...