ASTM F451-21
(Specification)Standard Specification for Acrylic Bone Cement
Standard Specification for Acrylic Bone Cement
ABSTRACT
This specification covers self-curing resins used primarily for the fixation of internal orthopedic prostheses. While a variety of copolymers and comonomers may be incorporated, the composition of the set cement shall contain poly(methacrylic acid esters) as its main ingredient. The mixture may be used in either the predough or dough stage. This specification covers compositional, physical performance, and biocompatibility as well as packaging requirements. Materials shall be tested and shall conform to specified values of appearance, stability, sterility, viscosity, intrusion and compressive strength. The biocompatibility of acrylic bone cement as it has been traditionally formulated and used is also detailed.
SCOPE
1.1 This specification covers self-curing resins used primarily for the fixation of internal orthopedic prostheses. The mixture may be used in either the pre-dough or dough stage in accordance with the manufacturer’s recommendations.
1.2 Units of pre-measured powder and liquid are supplied in a form suitable for mixing. The mixture then sets in place.
1.3 While a variety of copolymers and comonomers may be incorporated, the composition of the set cement shall contain poly(methacrylic acid esters) as its main ingredient.
1.4 This specification covers compositional, physical performance, and biocompatibility as well as packaging requirements. The biocompatibility of acrylic bone cement as it has been traditionally formulated and used has been reported in the literature (1, 2).2
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Mar-2021
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.11 - Polymeric Materials
Relations
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Nov-2019
- Effective Date
- 01-Mar-2017
- Refers
ASTM F895-11(2016) - Standard Test Method for Agar Diffusion Cell Culture Screening for Cytotoxicity - Effective Date
- 01-Apr-2016
- Effective Date
- 01-Apr-2016
- Effective Date
- 01-Aug-2014
- Effective Date
- 01-Dec-2013
- Effective Date
- 01-Mar-2013
- Effective Date
- 01-Oct-2012
- Effective Date
- 01-Oct-2012
- Refers
ASTM F895-11 - Standard Test Method for Agar Diffusion Cell Culture Screening for Cytotoxicity - Effective Date
- 01-Oct-2011
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Jun-2010
- Effective Date
- 15-May-2010
Overview
ASTM F451-21: Standard Specification for Acrylic Bone Cement defines the quality and performance requirements for self-curing acrylic resins used mainly in the fixation of internal orthopedic prostheses. The standard ensures that acrylic bone cement - whose primary component is poly(methacrylic acid esters) - meets strict criteria for composition, sterility, physical performance, and packaging to support safe and effective application in medical settings. This international standard is developed under recognized guidelines to facilitate global harmonization.
Key Topics
- Composition: The set cement must predominantly comprise poly(methacrylic acid esters). Manufacturers can include various copolymers or comonomers, but the primary ingredient is non-negotiable.
- Physical Performance: Acrylic bone cement must fulfill requirements for appearance, viscosity, setting characteristics, extrusion, intrusion, and compressive strength, ensuring mechanical reliability in clinical use.
- Biocompatibility: The standard addresses the biocompatibility of traditionally formulated acrylic bone cement, referencing established literature and specialty test methods to ensure that these materials are safe for human use.
- Packaging & Labeling: Strict packaging guidelines help maintain sterility, prevent contamination, and facilitate safe handling in surgical environments. Labeling must communicate contents, usage, and safety warnings as per regulatory requirements.
- Testing Procedures: The standard describes specific methods for measuring critical properties such as doughing time, setting time, viscosity, radiopacity, residual monomer content, compressive strength, and stability.
Applications
Acrylic bone cements conforming to ASTM F451-21 are primarily used to anchor internal orthopedic prostheses such as hip and knee implants. The cement is mixed from pre-measured powder and liquid components, then applied during surgery to help stabilize prosthetic devices by creating a secure interface between the implant and living bone.
Practical applications include:
- Joint replacement surgeries: Ensures a stable fixation of prosthetic components.
- Revision surgeries: Used to repair or replace earlier implants, where reliable cement properties are crucial.
- Orthopedic trauma treatment: In select cases, bone cement is used for rapid stabilization in fracture repair.
The specification’s rigorous requirements for compressive strength, viscosity, and setting characteristics ensure the material can withstand biomechanical loads and operate safely within the body under normal physiological conditions.
Related Standards
Several international standards intersect with, or are referenced by, ASTM F451-21 to support comprehensive quality assurance:
- ASTM D638: Test method for tensile properties of plastics.
- ASTM D695: Measures compressive properties of rigid plastics.
- ASTM D3835: Determination of polymeric material flow characteristics.
- ASTM F748: Selecting biological test methods for medical devices.
- ASTM F619, F749, F763, F813, F895, F981: Biocompatibility and cytotoxicity testing procedures for materials used in surgical implants.
- ISO 5833: Implants for surgery - acrylic resin cements.
- ISO 80000-9: Physical chemistry and molecular physics (units).
- ANSI/ADA No. 15: Acrylic resin standards for dental applications.
Conclusion
ASTM F451-21 is a critical standard for manufacturers, regulators, and clinicians involved in orthopedic device production and application. By adhering to these requirements, stakeholders ensure that acrylic bone cement products are safe, effective, and consistent. This improves patient outcomes and supports global harmonization of orthopedic material standards.
Keywords: acrylic bone cement, ASTM F451-21, orthopedic prosthesis fixation, poly(methacrylic acid esters), biocompatibility, bone cement sterilization, compressive strength, medical device packaging, orthopedic implant standards
Buy Documents
ASTM F451-21 - Standard Specification for Acrylic Bone Cement
REDLINE ASTM F451-21 - Standard Specification for Acrylic Bone Cement
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F451-21 is a technical specification published by ASTM International. Its full title is "Standard Specification for Acrylic Bone Cement". This standard covers: ABSTRACT This specification covers self-curing resins used primarily for the fixation of internal orthopedic prostheses. While a variety of copolymers and comonomers may be incorporated, the composition of the set cement shall contain poly(methacrylic acid esters) as its main ingredient. The mixture may be used in either the predough or dough stage. This specification covers compositional, physical performance, and biocompatibility as well as packaging requirements. Materials shall be tested and shall conform to specified values of appearance, stability, sterility, viscosity, intrusion and compressive strength. The biocompatibility of acrylic bone cement as it has been traditionally formulated and used is also detailed. SCOPE 1.1 This specification covers self-curing resins used primarily for the fixation of internal orthopedic prostheses. The mixture may be used in either the pre-dough or dough stage in accordance with the manufacturer’s recommendations. 1.2 Units of pre-measured powder and liquid are supplied in a form suitable for mixing. The mixture then sets in place. 1.3 While a variety of copolymers and comonomers may be incorporated, the composition of the set cement shall contain poly(methacrylic acid esters) as its main ingredient. 1.4 This specification covers compositional, physical performance, and biocompatibility as well as packaging requirements. The biocompatibility of acrylic bone cement as it has been traditionally formulated and used has been reported in the literature (1, 2).2 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ABSTRACT This specification covers self-curing resins used primarily for the fixation of internal orthopedic prostheses. While a variety of copolymers and comonomers may be incorporated, the composition of the set cement shall contain poly(methacrylic acid esters) as its main ingredient. The mixture may be used in either the predough or dough stage. This specification covers compositional, physical performance, and biocompatibility as well as packaging requirements. Materials shall be tested and shall conform to specified values of appearance, stability, sterility, viscosity, intrusion and compressive strength. The biocompatibility of acrylic bone cement as it has been traditionally formulated and used is also detailed. SCOPE 1.1 This specification covers self-curing resins used primarily for the fixation of internal orthopedic prostheses. The mixture may be used in either the pre-dough or dough stage in accordance with the manufacturer’s recommendations. 1.2 Units of pre-measured powder and liquid are supplied in a form suitable for mixing. The mixture then sets in place. 1.3 While a variety of copolymers and comonomers may be incorporated, the composition of the set cement shall contain poly(methacrylic acid esters) as its main ingredient. 1.4 This specification covers compositional, physical performance, and biocompatibility as well as packaging requirements. The biocompatibility of acrylic bone cement as it has been traditionally formulated and used has been reported in the literature (1, 2).2 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F451-21 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F451-21 has the following relationships with other standards: It is inter standard links to ASTM F749-20, ASTM D5296-19, ASTM F756-17, ASTM F895-11(2016), ASTM F748-16, ASTM E169-04(2014), ASTM F756-13, ASTM F749-13, ASTM F813-07(2012), ASTM F749-98(2012), ASTM F895-11, ASTM F748-06(2010), ASTM F981-04(2010), ASTM F763-04(2010), ASTM D638-10. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F451-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:F451 −21
Standard Specification for
Acrylic Bone Cement
ThisstandardisissuedunderthefixeddesignationF451;thenumberimmediatelyfollowingthedesignationindicatestheyearoforiginal
adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.Asuperscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
1.1 This specification covers self-curing resins used primar- 2.1 ASTM Standards:
ily for the fixation of internal orthopedic prostheses. The D638 Test Method for Tensile Properties of Plastics
mixture may be used in either the pre-dough or dough stage in D695 Test Method for Compressive Properties of Rigid
accordance with the manufacturer’s recommendations. Plastics
D1193 Specification for Reagent Water
1.2 Unitsofpre-measuredpowderandliquidaresuppliedin
D3835 Test Method for Determination of Properties of
a form suitable for mixing. The mixture then sets in place.
Polymeric Materials by Means of a Capillary Rheometer
1.3 While a variety of copolymers and comonomers may be
D5296 Test Method for Molecular Weight Averages and
incorporated, the composition of the set cement shall contain
Molecular Weight Distribution of Polystyrene by High
poly(methacrylic acid esters) as its main ingredient.
Performance Size-Exclusion Chromatography
D5630 Test Method for Ash Content in Plastics
1.4 This specification covers compositional, physical
performance, and biocompatibility as well as packaging re- E169 PracticesforGeneralTechniquesofUltraviolet-Visible
Quantitative Analysis
quirements. The biocompatibility of acrylic bone cement as it
hasbeentraditionallyformulatedandusedhasbeenreportedin E275 Practice for Describing and Measuring Performance of
Ultraviolet and Visible Spectrophotometers
the literature (1, 2).
F619 Practice for Extraction of Materials Used in Medical
1.5 The values stated in SI units are to be regarded as
Devices
standard. No other units of measurement are included in this
F748 PracticeforSelectingGenericBiologicalTestMethods
standard.
for Materials and Devices
1.6 This standard does not purport to address all of the
F749 Practice for Evaluating Material Extracts by Intracuta-
safety concerns, if any, associated with its use. It is the
neous Injection in the Rabbit
responsibility of the user of this standard to establish appro-
F756 Practice for Assessment of Hemolytic Properties of
priate safety, health, and environmental practices and deter-
Materials
mine the applicability of regulatory limitations prior to use.
F763 Practice for Short-Term Screening of Implant Materi-
1.7 This international standard was developed in accor-
als
dance with internationally recognized principles on standard-
F813 Practice for Direct Contact Cell Culture Evaluation of
ization established in the Decision on Principles for the
Materials for Medical Devices
Development of International Standards, Guides and Recom-
F895 TestMethodforAgarDiffusionCellCultureScreening
mendations issued by the World Trade Organization Technical
for Cytotoxicity
Barriers to Trade (TBT) Committee.
F981 Practice for Assessment of Compatibility of Biomate-
rials for Surgical Implants with Respect to Effect of
This specification is under the jurisdiction of ASTM Committee F04 on Materials on Muscle and Insertion into Bone
Medical and Surgical Materials and Devices and is the direct responsibility of
Subcommittee F04.11 on Polymeric Materials.
Current edition approved March 15, 2021. Published April 2021. Originally
approved in 1976. Last previous edition approved in 2016 as F451 – 16. DOI: For referenced ASTM standards, visit the ASTM website, www.astm.org, or
10.1520/F0451-21. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
The boldface numbers in parentheses refer to the list of references at the end of Standards volume information, refer to the standard’s Document Summary page on
this standard. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F451−21
2.2 ANSI/ADA Standard: 4.2.2 Sterility—The powder, as poured from its package,
No. 15 Specification for Acrylic Resin Teeth shall pass the tests described in “Sterility Tests—Solids” (7.5)
(2).
2.3 ISO Standards:
ISO 5833 Implants for Surgery—Acrylic Resin Cements
4.3 Powder-Liquid Mixture—The material shall conform to
ISO 80000-9 Quantities and Units—Part 9: Physical Chem-
the properties given in Table 1.
istry and Molecular Physics
2.4 NIST Document:
TABLE 1 Requirements for Powder Liquid Mixture
Special Publication 811
Required Values or
Property
Ranges
3. Terminology
Max Dough Time, minutes 5.0
Setting Time Range, minutes 5 to 15
3.1 Definitions of Terms Specific to This Standard:
Temperature, maximum, °C 90
3.1.1 doughing time—the time after commencement of mix-
Intrusion, minimum, mm 2.0
ing at which the mixture ceases to adhere to a standard probe
(see 7.6).
3.1.1.1 Discussion—“Doughing time” and “dough time” are
4.4 Cured Cement—The material after setting shall conform
interchangeable in this standard.
to the properties given in Table 2.
3.1.2 exothermic or maximum temperature—the maximum
temperature of the mixture due to self-curing in a standard
TABLE 2 Requirements for Cured Polymer After Setting
mold (see 7.7).
Property Requirement
Compressive Strength, minimum, MPa 70
3.1.3 extrusion—the rate of flow of the material through a
standard orifice under load (see 7.9.1).
3.1.4 intrusion—the distance of flow of the mixture into a
5. Weights and Permissible Variations
standard mold under load (see 7.9.3).
5.1 Weight and volume measurements shall be made on the
3.1.5 setting time—the time after commencement of mixing
respective powder and liquid components of five units (see
at which the temperature of the curing mass equals the average
9.2.2). These units may be subsequently utilized in any of the
of the maximum and ambient temperatures (see 7.8).
nonsterile tests of this specification.
3.1.5.1 Discussion—“Setting time” and “set time” are inter-
changeable in this standard.
5.2 The weights, or volume of the powder and liquid
components, or both, shall not deviate by more than 5 % from
3.1.6 unit—one package or vial of pre-measured powder
those stated on the package (9.2.2), of each of five units.
component and one package or vial of pre-measured liquid
component.
6. Sampling
4. Physical Requirements
6.1 Units of powder and liquid shall be procured to provide
sufficient material for all the tests of this specification. The
4.1 Liquid—The liquid component includes the monomer,
unitsshallbeobtainedfromregularretaildistributionchannels.
inhibitors, accelerants, and, if applicable, colorants.
Provided no repeat tests are required, this will amount to
4.1.1 Appearance—The liquid shall be free of extraneous
between seven and ten units.
particulate matter or obvious visual contaminants in its con-
tainer.
6.2 It will only be necessary to maintain sterility in tests
4.1.2 Stability—After being heated for 48 h at 60 6 2 °C,
described in 7.5. All other tests described in this specification
the viscosity of the liquid shall not increase by more than 10 %
need not be conducted under sterile conditions.
of its original value (see 7.4).
7. Test Methods and Sample Size
4.1.3 Sterility—The liquid, as poured from its container,
shall pass the tests described in “Sterility Tests—Liquid and
7.1 Maintain all equipment, mixing surfaces, and materials
Ointments” (7.5) (3).
at23 61 °Cforatleast2hpriortotestingandconductalltests
at23 61 °Cand50 610 %relativehumidityunlessotherwise
4.2 Powder—The powder component includes the polymer
specified.
particles, initiator agents, the radio-opaque agent, and if
applicable, other additives such as antibiotics and colorants.
7.2 Inspection—Use visual inspection in determining com-
4.2.1 Appearance—The powder shall be pourable and free
pliance to the requirements outlined in 4.1.1, 4.2.1, 8.1, and
of extraneous materials, such as dirt or lint (7.2.2).
8.2.
7.2.1 The liquid component of two separate units shall
comply with the requirements of 4.1.1 and 8.1.
Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
7.2.2 The powder component of two separate units shall
4th Floor, New York, NY 10036, http://www.ansi.org.
comply with the requirements of 4.2.1 and 8.1.
Available from International Organization for Standardization (ISO), ISO
Central Secretariat, Chemin de Blandonnet 8, CP 401, 1214 Vernier, Geneva,
7.3 Radiopacifier Content in Powder Component—The ra-
Switzerland, https://www.iso.org.
diopacifier content in the powder component shall be assessed
Available from National Institute of Standards and Technology (NIST), 100
Bureau Dr., Stop 1070, Gaithersburg, MD 20899-1070, http://www.nist.gov. by net ash testing according to Test Method D5630, Procedure
F451−21
B. The radiopacifier content shall not vary from the nominal (UHMWPE) and be equipped with a No. 24 gage wire
content by more than 10 %. thermocouple, or similar device, positioned with its junction in
the center of the mold at a height of 3.0 mm in the internal
7.4 Liquid Component Viscosity Stability—Record the vis-
cavity.ImmediatelyseattheplungerwithaC-clamporsuitable
cosity change of two separate units (4.1.2) before and after the
press to produce the 6.0 mm specimen height. Upon producing
heatingexposurebytimingtheflowoftheliquidlevelbetween
plungerseating,removetheexcessmaterialandtheC-clampor
the 0 and 5 mL marks of a 10 mL measuring pipet. Calculate
press for the remainder of the procedure. Continuously record
the percent change as follows:
the temperature with respect to time from the onset of mixing
t 2 t
a b
theliquidandthepowderuntilcoolingisobserved(seeFig.2).
% Change 5 3100 (1)
t
b
Report the maximum temperature recorded to the nearest 1 °C.
This should not exceed the value given in Table 1.
where:
7.7.1 The average maximum temperature shall be the cal-
t = flow time before heating, and
b
culated average of two separate maximum temperature deter-
t = flowtimeafterheatingexposure(4.1.2)of60 62 °Cfor
a
minations reported to the nearest 1 °C.
48 h in the dark in a closed container.
7.7.2 If the difference between the maximum temperature
7.4.1 An alternative method for viscosity may be used if it
forthetwodeterminationsisgreaterthan5.0 °C,repeatthetest
can be demonstrated to yield similar results. Both shall comply
on two additional units and report the average of all four runs
to the less than 10 % change specified (4.1.2).
to the nearest 1 °C. Individual maximum and minimum values
7.5 The components of the two units shall be tested for for maximum temperature shall not differ by more than 64°C
of the average value of all determinations.
sterility in accordance with the test methods described in U.S.
Pharmacopoeia, “Sterility Tests” (3).
7.8 Setting Time—From the continuous time-versus-
temperature recording of 7.7, the setting time (T ) is the time
7.6 Doughing Time:
set
when the temperature of the polymerizing mass is as follows:
7.6.1 Environment—All equipment, mixing surfaces, and
material(unitsize)shallbemaintainedat23 61 °Cforatleast
T 1T /2 (2)
~ !
max amb
2 h prior to testing and tests shall be conducted at 23 6 1 °C.
where:
The relative humidity shall be 50 6 10 %.
T = maximum temperature, °C, and
7.6.2 Mix all the powder and liquid of a single unit together
max
T = ambient temperature of 23 6 1 °C.
as directed by the manufacturer’s instructions (see 8.2). Start a amb
stopwatch at the onset of combining the liquid and the powder
7.8.1 Report the setting time to the nearest 5 s.
and read all subsequent times from this stopwatch. Approxi-
7.8.2 Make two separate determinations of the setting time.
mately 1.5 min after the onset of mixing, gently probe the
7.8.3 The two values should agree within 1 minute of each
mixture with a non-powdered surgically gloved (latex) finger.
other; otherwise repeat the test on two additional units and
Take visual notice as to the formation of fibers between the
report the average of all runs.
surfaceofthemixandthefingerasitleavesthesurface.Repeat
7.8.4 Report the setting time to the nearest 15 s as the
this process from that time on at 15 s intervals with a clean
average of all determinations.
portion of the glove until the gloved finger separates cleanly.
7.9 Flow Properties and Viscosity Determination—The
Denote the time at which this is first observed as the doughing
manufacturer must specify whether the cement may be used in
time. Mix the mixture between determinations to expose fresh
its pre-dough or dough state, or both. The determination of its
material for each probing.
usage dictates which of the following tests the cement should
7.6.3 Determine the average doughing time from two sepa-
comply with. If the mixture is to be utilized in the pre-dough
rate units.
stage, use the extrusion viscosity test (7.9.1 and/or 7.9.2) and
7.6.4 The two values found shall agree within 30 s of each
Table 1. If the mixture is to be utilized in the dough stage, use
other; otherwise repeat the test on two additional units. Report
the intrusion test (7.9.3) and Table 1. If the mixture is to be
the average of all four tests and the range of values.
used as a dual usage cement, then both the extrusion (7.9.1
7.6.5 Report the doughing time to the nearest 15 s as the
and/or 7.9.2) and intrusion (7.9.3) tests shall be performed.
average of all determinations. Maximum and minimum values
7.9.1 Extrusion, Capillary Viscosity:
ofdoughingtimesmeasuredshallnotdifferbymorethan 61 ⁄2
7.9.1.1 Apparatus:
min from the average.
(1) Capillary Rheometer—Any capillary rheometer in
7.6.6 Report the brand of non-powdered surgical glove used
which acrylic bone cement can be forced from a reservoir
for dough time determinations. It is necessary that the type of
throughacapillarydieandinwhichtemperature,appliedforce,
glove be described in detail, including manufacturer, when the
output rate, and barrel and die dimensions can be controlled
dough time is reported.
and measured accurately is satisfactory. Equipment that pro-
7.7 Exothermic Temperature—Within 1 min after doughing videsaconstantshearratehasbeenshowntobeequallyuseful.
time,gentlypackapproximately25gofthedoughdescribedin The capillary die of the rheometer shall have a smooth, straight
7.6 into the mold described in Fig. 1. This mold shall be made borethatisheldwithin 60.0076mm(60.0003in.)indiameter
of polytetrafluoroethylene (PTFE), and shall be held to within 60.025 mm (60.001 in.) in length.
poly(ethyleneterephthalate), polyoxymethylene, high-density The bore and its finish are critical. It shall have no visible drill
polyethylene, or ultra-high molecular weight polyethylene or other tool marks and no detectable eccentricity.
F451−21
NOTE 1—Dimensions in millimetres and 60.2 unless otherwise specified. Material for all components: Polytetrafluoroethylene,
poly(ethyleneterephthalate), polyoxymethylene, high-density polyethylene, or ultra-high molecular weight polyethylene (UHMWPE).
FIG. 1Exothermic Heat Mold
FIG. 2Continuous Temperature Record
(2) Due to the extreme sensitivity of flow data to the sions are measured with precision and reported. The length-to-
capillary dimensions, it is important that the capillary dimen- diameter ratio shall normally be between 20 and 40. Larger
F451−21
ratios and ratios less than that suggested require applying large
F = force on ram in N,
corrections to the data (4, 5). In addition, the ratio of the
r = radius of capillary in m,
reservoir diameter to capillary diameter should be between 3
R = radius of barrel in m,
and 15. See Test Method D3835 for further details of capillary L = length of capillary in m,
Q = flow rate in m /s,
rheometers.
V = volume extruded in m , and
7.9.1.2 Calibration—Perform the test with a certified stan-
t = extrusion time in s.
dard viscosity fluid approximating that expected for bone
(2) These equations yield true shear rate and true viscosity
2 2
cement (50 N·s/m to 500 N·s/m ). Determine the viscosity of
for Newtonian fluids only; for non-Newtonian fluids, such as
thestandardfluidandthepercenterrorfromitsspecifiedvalue.
bone cement, the apparent shear rate and viscosity are ob-
Report this error along with the viscosity of the tested cements.
tained.
7.9.1.3 Corrections—Bone cement is a non-Newtonian
7.9.1.6 Report—The report of the flow properties of the
fluid; the data may be reported as corrected data. For example,
cement shall include:
true shear rates, corrected for non-Newtonian flow behavior,
(1) Description of the rheometer used.
and true shear stress, corrected for end effects or kinetic energy
(2) Temperature at which the data were obtained.
losses,maybecalculated.Insuchcases,theexactdetailsofthe
(3) The capillary diameter and length to diameter ratio of
mode of correction shall be reported. Some correction factors
the capillary.
which may apply are:
(4) The shear rate at which the test was performed.
(1) Piston friction,
(5) Viscosity versus observation time for three runs.
(2) Plunger back flow,
(6) Statement as to whether any correction factors (7.9.1.3)
(3) Cement compressibility,
were applied.
(4) Barrel back pressure,
7.9.2 Extrusion, Rotational Shear Viscosity:
(5) Capillary entrance effects (Bagley correction) (6), and
(6) Rabinowitsch shear rate correction (7). 7.9.2.1 Apparatus—Rotational Shear Rheometer—Any par-
allel plate rotational shear rheometer that can use 4 cm
7.9.1.4 Procedure:
diameter plates, a 1000 µm gap, and maintain a temperature of
(1) Select conditions of temperature and shear stress or
23 6 0.5 °C is satisfactory.
shear rate in accordance with expected usage so that the flow
7.9.2.2 Calibration—Calibrate the rheometer according to
rate will fall within the desired limits.
the manufacturer’s specifications.
(2) Inspect the rheometer and clean it if necessary. Ensure
that previous cleaning procedures and usage have not changed 7.9.2.3 Procedure:
(1) Mount a parallel plate geometry on the top fixture. A
thedimensionsorcausedscratchesordefectsinthecapillaryor
apparatus. Make the necessary measurements on the apparatus disposable plate system may be used. A stainless steel 4 cm
for future calculations. Prepare the apparatus for running the diameter plate is recommended.Aremovable bottom plate can
test. be added to facilitate sample removal.
(3) Mix one or more complete unit(s) of powder and liquid (2) Controlthetemperatureoftherheometersothatatleast
in the recommended manner. Start a stopwatch at the onset of one of the plates is at 23 6 0.5 °C.
mixing and read all subsequent times from this watch. After (3) Move the plates apart to allow sample loading.
complete mixing, transfer the cement to the thermally equili- (4) Mix the cement according to the manufacturer’s speci-
brated reservoir and eject any entrapped air or excess bone fications. Start a laboratory timer from the start of mixing.
cement. (5) After the requisite mixing procedure is complete, place
(4) Start the apparatus at a time not greater than 2 ⁄2 min a sufficient quantity of bone cement between the plates so as to
from the start of mixing and continue operating until the completely fill the gap between the plates and no bubbles in
estimated dough time or the viscosity exceeds 500 N·s/m . excess of 1 mm are visible. Reduce the gap height between the
(5) Disassemble the apparatus quickly before the cement plates to 1000 µm. Scrape away excess cement.
–1
sets and clean the apparatus of all remaining cement. (6) Start a steady shear experiment at 0.5 s , monitoring
the shear viscosity as a function of time at a sampling rate of
7.9.1.5 Calculations:
0.5 Hz or better. Note the elapsed time from the start of mixing
(1) Perform the calculation for viscosity of the cement at
to when the first data point is obtained.
timeintervalsof15sfromthestarttofinishoftestrun.Usethe
(7) Collect data until the viscosity reaches 1000 Pa·s. Stop
following equations:
the test and remove the cement before it completely hardens.
Pr Fr
(8) It is recommended that three runs are conducted per
Shear Stress, Pa 5 5 (3)
2L 2πR L
cement formulation.
4Q 4V
21 7.9.2.4 Report—The report of the flow properties of the
Shear Rate, s 5 5 (4)
3 3
πr πr t
cement shall include:
4 4
Pπr Fr t (1) Description of the rheometer used.
Viscosity, Pa·s 5 5 (5)
(2) The shear viscosity as a function of time from the start
8LQ 8R LV
of mixing. The reported instrument time points will need to be
where:
shifted by the elapsed time measured in 7.9.2.3(6).
P = pressure by ram in Pa,
7.9.3 Intrusion:
F451−21
7.9.3.1 The mold necessary for this test shall be made of
polytetrafluoroethylene (PTFE), poly(ethyleneterephthalate),
polyoxymethylene, high-density polyethylene, or ultra-high
molecular weight polyethylene (UHMWPE) and is shown in
Fig. 3.
7.9.3.2 Follow the procedure outlined in ISO 5833, section
D.4.3 for intrusion testing.
7.9.3.3 Following the set, remove the specimen and mea-
sure the average height of the intrusion into all four of the
1.0-mm diameter holes of the die to the nearest 0.5 mm.
7.9.3.4 Run this test once. If the requirement is not met, it
must be met in a repeat test.
7.10 Compressive Strength—The test specimens shall be
cylinders 12 mm high and 6 mm in diameter. The ends of the
specimens shall be flat and smooth and shall be parallel to each
other and at right angles to the long axis of the cylinder. An
apparatus found convenient for forming these test cylinders is
NOTE 1—Material for perforated plate: stainless steel, aluminum,
shown in Fig. 4. An apparatus containing additional or fewer
polytetrafluoroethylene, high-density polyethylene, or ultra-high molecu-
holes may be used as long as adequate spacing between the
lar weight polyethylene (UHMWPE).
holesismaintained.Amoldreleaseagentorsiliconespraymay
be sparingly applied to facilitate specimen removal.
FIG. 4Compression Specimens Mold
7.10.1 Place the specimen mold on a flat glass or smooth
metalplateandslightlyoverfillusingoneunitofmixedcement
of standard proportions at the commencement of dough time.
Press a second flat glass or smooth metal plate on top of the
mold. Hold the mold and plates firmly together with a small
C-clamp.Then, 1 h later, surface the ends of the cylinder plane
at right angles to the axis. The ends of the specimens may be
ground flat to the axis by use of a small amount of 240-mesh
silicon carbide powder and water. Draw the molds containing
the specimens back and forth across the plate coated with the
abrasive and water. After surfacing, remove the specimens
from the mold.The specimens should be visually examined for
surface defects. A surface defect is defined as a surface
discontinuity greater than 500 microns in major diameter.
Acceptable specimens for testing shall appear to be uniform
and meet the dimensional requirements of 7.10.Aminimum of
five specimens shall be selected from the remaining acceptable
specimens and tested. Report the results of all specimens
tested.
7.10.2 The time lapse between the start of mixing and the
measurement of the compressive strength testing shall be 24 6
2 h. Storage of the specimens before testing shall be at 23 6
2 °C and 50 6 10 % relative humidity. Run specimens on any
universal testing machine equipped to record load versus
deformation. Employ a deformation cross-head speed of 20 to
25.4 mm/min. Test the specimens without use of any type of
pad between the specimen and the platens of the machine. The
failure load shall be the load at the 2.0 % offset (2.0 % proof
stress), upper yield point, or at fracture, whichever occurs first
NOTE 1—Dimensions in millimetres; four holes in bottom to be 1.00 6
(Fig. 5).
0.05. Tolerance on all other dimensions 60.2. Material for all compo-
7.10.2.1 The load at 2.0 % offset is the load at the intersec-
nents: polytetrafluoroethylene, poly(ethyleneterephthalate),
tionoftheloaddeformationcurveandastraightlineparallelto
polyoxymethylene, high-density polyethylene, or ultra-high molecular
theHookeanportionofthecurve(seeFig.X1.1inTestMethod
weight polyethylene (UHMWPE).
D695)butoffsetalongthedeformationaxisby2.0 %ofthetest
FIG. 3Intrusion Mold specimen’s gauge length (specimen’s height).
F451−21
either polystyrene or polymethyl methacrylate standards shall
be reported for each aliquot of powder. Report the average and
standard deviation of the number-average, weight-average, and
z-average molar mass for the three samples.
7.12 Leachable Monomer:
7.12.1 The residual monomer during curing and post-curing
may be determined using the protocols described in AnnexA5
and Annex A6.
7.13 Stabilizer Concentration:
7.13.1 If a quinone-based stabilizer is used in the liquid
portion, the amount of hydroquinone or monomethyl ether
hydroquinonemaybedeterminedusingtheprotocolsdescribed
in Annex A3 or Annex A4. Alternative protocols may be used
if they can be shown to have the required sensitivity.
7.14 Benzoyl Peroxide Concentration:
7.14.1 Ifbenzoylperoxideisusedasaninitiator,theamount
FIG. 5Failure Load Criteria
of benzoyl peroxide in the powder portion may be determined
using the protocol described in Annex A1.
7.10.2.2 Calculate the compressive strength as the failure 7.15 N,N-dimethyl-p-toluidine Concentration:
load divided by the calculated cross-sectional area. 7.15.1 If N,N-dimethyl-p-toluidine is used as a reaction
7.10.2.3 Report the compressive strength of the material as accelerator in the liquid portion, its concentration may be
the average of the compression strengths of the specimens determined by high-performance liquid chromatography
tested in 7.10.2 to the nearest 1 MPa (145 psi).Aminimum of
(HPLC) or similar assays (8).
five specimens is required.
7.16 Stability Testing—The shelf life stability of bone ce-
7.11 Molar Mass by Gel Permeation Chromatography
ment powder-liquid systems shall be evaluated using the test
(GPC): methods listed in Table 3.
7.11.1 The molar mass (see Note 1) distribution and molar
7.17 Precision and Bias—Since 1976, the original Specifi-
mass averages (number-average molar mass, weight-average
cation F451 methodologies have reportedly been routinely
molar mass, and z-average molar mass) of the powder will be
utilized by the various manufacturers. With the exception of
determined by gel permeation chromatography with reference
the viscosity method of 7.9.1, which is based on another
to Test Method D5296. Tetrahydrofuran (THF) will be used to
accepted ASTM document (Test Method D3835), each test
dissolve the powder according to Test Method D5296.Ifthe
methodology in Section 7 contains its own statement of
powder contains radiopacifier, the mass of powder used to
reporting acceptable levels of performance, reproducibility,
make the solution should be increased to account for the
and precision. Therefore, no interlaboratory studies have been
radiopacifier. The solution should be filtered as suggested in
performed by Committee F04.
Test Method D5296 to remove any radiopacifier.
8. Packaging
NOTE 1—The term molecular weight (abbreviated MW) is obsolete and
should be replaced by the SI (Système Internationale) equivalent of either
8.1 Materials shall be supplied in properly sealed containers
relative molecular mass (M ), which reflects the dimensionless ratio of the
r
made of materials that shall not contaminate or permit con-
mass of a single molecule to an atomic mass unit (see ISO 80000-9), or
taminationofthecontents.Thecontainersshallbepackagedso
molar mass (M), which refers to the mass of a mole of a substance and is
typically expressed as grams/mole. For polymers and other as to prevent damage or leakage during shipping and storage.
macromolecules, use of the symbols M , M , and M continue, referring
w n z
Materials must be packaged to permit sterile transfer of
to mass-average molar mass, number-average molar mass, and z-average
contents to the surgical sterile field.
molar mass, respectively. For more information regarding proper utiliza-
tion of SI units, see NIST Special Publication SP811.
7.11.2 Poly(methyl methacrylate) molar mass standards
should be used to calibrate the GPC system. Poly(styrene) TABLE 3 Requirements for Stability Testing
standards may be used for the purpose of comparison to
Test Type Test Description Test Material
Viscosity 7.4 Liquid Component
historical results, but it should be understood that the results
Residual Peroxide Annex A1, Powder Component
will be relative and will not represent an absolute determina-
Annex A2,or
tion of the polymer’s molar mass distribution.
equivalent
Dough Time 7.6 Curing Cement
7.11.3 Three aliquots of powder shall be tested for each
Set Time 7.8 Curing Cement
bone cement.
Compressive Strength 7.10 Cured Cement
7.11.4 This method may also be used on cured bone cement Tensile Strength D638 Cured Cement
Leachable Monomer Annex A5, An- Cured and Curing Cement
to determine the molar mass distribution of the cured material.
nex A6,or
7.11.5 The number-average, weight-average, and z-average
equivalent
molar mass, along with the molar mass distribution relative to
F451−21
8.2 The contents shall be easily accessible, easy to open, versustemperatureshouldbeprovidedfortheallowedrangeof
and convenient to mix in the operating room. Entire package temperature (for example, 16 to 26 °C).
contents (both powder and liquid) must be mixed per the 9.3.5 The ranges of doughing and setting times as measured
manufacturer’s instructions for use. at 23 61°C(7.6 and 7.8) shall be clearly stated. If a range of
temperatures has been identified, then testing at the lower and
9. Labeling
upper temperature limits and any intermediate increments
should be performed and the results reported in the IFU.
9.1 Labeling on these cements must be in conformance with
the Federal Food, Drug, and Cosmetic Act, Code of Federal
10. Biocompatibility
Regulations, and other pertinent laws and regulations.
10.1 The biocompatibility of acrylic bone cement has been
9.2 The following minimal information shall appear on the
reported in the literature (1, 2).The material has been shown to
container label:
produce a well-characterized level of biological response
9.2.1 It shall be clearly stated or color coded, or both, if the
following long-term clinical use and laboratory studies. The
mixture is intended for usage in the pre-dough, dough, or dual
results of these studies and the clinical history indicate an
usage state.
acceptablelevelofbiologicalresponseinapplicationsinwhich
9.2.2 The weight or volume, or both, of the liquid and
the material has been utilized. When new applications of the
powder components shall be stated.
material, or significant modification to the material or its
9.2.3 Constituents of the powder and liquid shall be clearly
physical forms are being contemplated, the recommendations
stated in terms of weight or volume percent. This inform
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F451 − 16 F451 − 21
Standard Specification for
Acrylic Bone Cement
This standard is issued under the fixed designation F451; the number immediately following the designation indicates the year of original
adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A superscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This specification covers self-curing resins used primarily for the fixation of internal orthopedic prostheses. The mixture may
be used in either the predoughpre-dough or dough stage in accordance with the manufacturer’s recommendations.
1.2 Units of premeasuredpre-measured powder and liquid are supplied in a form suitable for mixing. The mixture then sets in
place.
1.3 While a variety of copolymers and comonomers may be incorporated, the composition of the set cement shall contain
poly(methacrylic acid esters) as its main ingredient.
1.4 This specification covers compositional, physical performance, and biocompatibility as well as packaging requirements. The
biocompatibility of acrylic bone cement as it has been traditionally formulated and used has been reported in the literature (1, 2).
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D638 Test Method for Tensile Properties of Plastics
D695 Test Method for Compressive Properties of Rigid Plastics
D1193 Specification for Reagent Water
D3835 Test Method for Determination of Properties of Polymeric Materials by Means of a Capillary Rheometer
D5296 Test Method for Molecular Weight Averages and Molecular Weight Distribution of Polystyrene by High Performance
Size-Exclusion Chromatography
This specification is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.11 on Polymeric Materials.
Current edition approved Oct. 1, 2016March 15, 2021. Published December 2016April 2021. Originally approved in 1976. Last previous edition approved in 20082016
as F451 – 08.F451 – 16. DOI: 10.1520/F0451-16.10.1520/F0451-21.
The boldface numbers in parentheses refer to the list of references at the end of this standard.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F451 − 21
D5630 Test Method for Ash Content in Plastics
E169 Practices for General Techniques of Ultraviolet-Visible Quantitative Analysis
E275 Practice for Describing and Measuring Performance of Ultraviolet and Visible Spectrophotometers
F619 Practice for Extraction of Materials Used in Medical Devices
F748 Practice for Selecting Generic Biological Test Methods for Materials and Devices
F749 Practice for Evaluating Material Extracts by Intracutaneous Injection in the Rabbit
F756 Practice for Assessment of Hemolytic Properties of Materials
F763 Practice for Short-Term Screening of Implant Materials
F813 Practice for Direct Contact Cell Culture Evaluation of Materials for Medical Devices
F895 Test Method for Agar Diffusion Cell Culture Screening for Cytotoxicity
F981 Practice for Assessment of Compatibility of Biomaterials for Surgical Implants with Respect to Effect of Materials on
Muscle and Insertion into Bone
2.2 ANSI/ADA Standard:
No. 15 Specification for Acrylic Resin Teeth
2.3 ISO Standards:
ISO 5833 Implants for Surgery—Acrylic Resin Cements
ISO 80000-9 Quantities and Units—Part 9: Physical Chemistry and Molecular Physics
2.4 NIST Document:
Special Publication 811
3. Terminology
3.1 Definitions of Terms Specific to This Standard:
3.1.1 doughing time—the time after commencement of mixing at which the mixture ceases to adhere to a standard probe (see
7.57.6).
3.1.1.1 Discussion—
“Doughing time” and “dough time” are interchangeable in this standard.
3.1.2 exothermic or maximum temperature—the maximum temperature of the mixture due to self-curing in a standard mold (see
7.67.7).
3.1.3 extrusion—the rate of flow of the material through a standard orifice under load (see 7.8.17.9.1).
3.1.4 intrusion—the distance of flow of the mixture into a standard mold under load (see 7.8.37.9.3).
3.1.5 setting time—the time after commencement of mixing at which the temperature of the curing mass equals the average of the
maximum and ambient temperatures (see 7.77.8).
3.1.5.1 Discussion—
“Setting time” and “set time” are interchangeable in this standard.
3.1.6 unit—one package or vial of premeasuredpre-measured powder component and one package or vial of premeasuredpre-
measured liquid component.
4. Physical Requirements
4.1 Liquid: Liquid—The liquid component includes the monomer, inhibitors, accelerants, and, if applicable, colorants.
4.1.1 Appearance—The liquid shall be free of extraneous particulate matter or obvious visual contaminants in its container.
4.1.2 Stability—After being heated for 48 h at 60 6 2°C,2 °C, the viscosity of the liquid shall not increase by more than 10 %
of its original value (see 7.37.4).
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
Available from International Organization for Standardization (ISO), ISO Central Secretariat, Chemin de Blandonnet 8, CP 401, 1214 Vernier, Geneva, Switzerland,
https://www.iso.org.
Available from National Institute of Standards and Technology (NIST), 100 Bureau Dr., Stop 1070, Gaithersburg, MD 20899-1070, http://www.nist.gov.
F451 − 21
4.1.3 Sterility—The liquid, as poured from its container, shall pass the tests described in “Sterility Tests—Liquid and Ointments”
(7.47.5) (3).
4.2 Powder: Powder—The powder component includes the polymer particles, initiator agents, the radio-opaque agent, and if
applicable, other additives such as antibiotics and colorants.
4.2.1 Appearance—The powder shall be pourable and free of extraneous materials, such as dirt or lint (7.2.2).
4.2.2 Sterility—The powder, as poured from its package, shall pass the tests described in “Sterility Tests—Solids” (7.47.5) (2).
4.3 Powder-Liquid Mixture: Mixture—
4.3.1 If the mixture is to be used in its predough stage, the material shall conform to the properties given in Table 1. The material
shall conform to the properties given in Table 1.
4.3.2 If the mixture is to be used in its dough stage, the material shall conform to the properties given in Table 1.
4.3.3 If the mixture can be used in either its predough or dough stages, separate units must be tested for compliance with 4.3.1
and 4.3.2.
4.4 Cured Polymer—Cement—The material after setting shall conform to the properties given in Table 2.
TABLE 2 Requirements for Cured Polymer After Setting
Property Requirement
Compressive Strength, min., MPa 70
Compressive Strength, minimum, MPa 70
5. Weights and Permissible Variations
5.1 Weight and volume measurements shall be made on the respective powder and liquid components of five units (see 3.19.2.2).
These units may be subsequently utilized in any of the nonsterile tests of this specification.
5.2 The weights, or volume of the powder and liquid components, or both, shall not deviate by more than 5 % from those stated
on the package (9.2.2), of each of five units.
6. Sampling
6.1 Units of powder and liquid shall be procured to provide sufficient material for all the tests of this specification. The units shall
be obtained from regular retail distribution channels. Provided no repeat tests are required, this will amount to between seven and
ten units.
TABLE 1 Requirements for Powder Liquid Mixture
Dough Usage,
Extrusion,
Property Intrusion
Viscosity Tests
Tests
Max Dough Time, min. 5.0 5.0
Setting Time Range, min. 5 to 15 5 to 15
Temperature, max., °C 90 90
Intrusion, min., mm . 2.0
TABLE 1 Requirements for Powder Liquid Mixture
Required
Property
Values or Ranges
Max Dough Time, minutes 5.0
Setting Time Range, minutes 5 to 15
Temperature, maximum, °C 90
Intrusion, minimum, mm 2.0
F451 − 21
6.2 It will only be necessary to maintain sterility in tests described in 7.47.5. All other tests described in this specification need
not be conducted under sterile conditions.
7. Test Methods and Sample Size
7.1 Maintain all equipment, mixing surfaces, and materials at 23 6 2°C 1 °C for at least 2 h prior to testing and conduct all tests
at 23 6 2°C1 °C and 50 6 10 % relative humidity unless otherwise specified.
7.2 Inspection—Use visual inspection in determining compliance to the requirements outlined in 4.1.1, 4.2.1, 8.1, and 8.2.
7.2.1 The liquid component of two separate units shall comply with the requirements of 4.1.1 and 8.1.
7.2.2 The powder component of two separate units shall comply with the requirements of 4.2.1 and 8.1.
7.3 Radiopacifier Content in Powder Component—The radiopacifier content in the powder component shall be assessed by net ash
testing according to Test Method D5630, Procedure B. The radiopacifier content shall not vary from the nominal content by more
than 10 %.
7.4 Liquid Component Viscosity—Viscosity Stability—Record the viscosity change of two separate units (4.1.2) before and after
the heating exposure by timing the flow of the liquid level between the 0 and 5 mL marks of a 10 mL measuring pipet. Calculate
the percent change as follows:
t 2 t
a b
% Change 5 3100 (1)
t
b
t 2 t
a b
% Change 5 3100 (1)
t
b
where:
t = flow time before heating, and
b
t = flow time after heating exposure (4.1.2) of 60 6 2°C for 48 h in the dark in a closed container.
a
t = flow time after heating exposure (4.1.2) of 60 6 2 °C for 48 h in the dark in a closed container.
a
7.4.1 An alternative method for viscosity may be used if it can be demonstrated to yield similar results. Both shall comply to the
less than 10 % change specified (4.1.2).
7.5 The components of the two units shall be tested for sterility in accordance with the test methods described in U.S.
Pharmacopoeia, “Sterility Tests” (3).
7.6 Doughing Time:
7.6.1 Environment—All equipment, mixing surfaces, and material (unit size) shall be maintained at 23 6 1°C 1 °C for at least 2
h prior to testing and tests shall be conducted at 23 6 1°C.1 °C. The relative humidity shall be 50 6 10 %.
7.6.2 Mix all the powder and liquid of a single unit together as directed by the manufacturer’s instructions (see 8.2). Start a stop
watch stopwatch at the onset of combining the liquid toand the powder and read all subsequent times from this stop watch.
stopwatch. Approximately 1.5 min after the onset of mixing, gently probe the mixture with a non-powdered surgically gloved
(latex) finger. Take visual notice as to the formation of fibers between the surface of the mix and the finger as it leaves the surface.
Repeat this process from that time on at 15 s intervals with a clean portion of the glove until the gloved finger separates cleanly.
Denote the time at which this is first observed as the doughing time. Mix the mixture between determinations to expose fresh
material for each probing.
7.6.3 Determine the average doughing time from two separate units.
7.6.4 The two values found shall agree within 30 s of each other,other; otherwise repeat the test on two additional units. Report
the average of all four tests and the range of values.
F451 − 21
7.6.5 Report the doughing time to the nearest 15 s as the average of all determinations. Maximum and minimum values of
doughing times measured shall not differ by more than 61 ⁄2 min from the average.
7.6.6 Report the brand of non-powdered surgical glove used for dough time determinations. It is necessary that the type of glove
be described in detail, including manufacturer, when the dough time is reported.
7.7 Exothermic Temperature—Within 1 min after doughing time, gently pack approximately 25 g of the dough described in 7.57.6
into the mold described in Fig. 1. This mold shall be made of polytetrafluoroethylene (PTFE), poly(ethyleneterephthalate),
polyoxymethylene, high density high-density polyethylene, or ultra-high molecular weight polyethylene (UHMWPE) and be
equipped with a No. 24 gage wire thermocouple, or similar device, positioned with its junction in the center of the mold at a height
of 3.0 mm in the internal cavity. Immediately seat the plunger with a C-clamp or suitable press to produce the 6.0 mm specimen
height. Upon producing plunger seating, remove the excess material and the C-clamp or press for the remainder of the procedure.
Continuously record the temperature with respect to time from the onset of mixing the liquid and the powder until cooling is
observed, observed (see Fig. 2.). Report the maximum temperature recorded to the nearest 1°C.1 °C. This should not exceed the
value given in Table 1.
7.7.1 The average maximum temperature shall be the calculated average of two separate maximum temperature determinations
reported to the nearest 1°C.1 °C.
NOTE 1—Dimensions in millimetres and 60.2 unless otherwise specified. Material for all components: Polytetrafluoroethylene,
poly(ethyleneterephthalate), polyoxymethylene, high density high-density polyethylene, or ultra-high molecular weight polyethylene (UHMWPE).
FIG. 1 Exothermic Heat Mold
F451 − 21
FIG. 2 Continuous Temperature Record
7.7.2 If the difference between the maximum temperature for the two determinations is greater than 5.0°C,5.0 °C, repeat the test
on two additional units and report the average of all four runs to the nearest 1°C.1 °C. Individual maximum and minimum values
for maximum temperature shall not differ by more than 64°C64 °C of the average value of all determinations.
7.8 Setting Time—From the continuous time versus temperature time-versus-temperature recording of 7.67.7, the setting time
(T ) is the time when the temperature of the polymerizing mass is as follows:
set
T 1T /2 (2)
~ !
max amb
where:
T = maximum temperature, °C, and
max
T = ambient temperature of 23 6 1°C.
amb
T = ambient temperature of 23 6 1 °C.
amb
7.8.1 Report the setting time to the nearest 5 s.
7.8.2 Make two separate determinations of the setting time.
7.8.3 The two values should agree within 1 minminute of each other,other; otherwise repeat the test on two additional units and
report the average of all runs.
7.8.4 Report the setting time to the nearest 15 s as the average of all determinations.
7.9 Flow Properties and Viscosity Determination—The manufacturer must specify whether the cement may be used in its
pre-dough or dough state, or both. The determination of its usage dictates which of the following tests the cement should comply
with. If the mixture is to be utilized in the pre-dough stage, use the extrusion viscosity test (7.8.17.9.1 and/or 7.8.27.9.2) and Table
1. If the mixture is to be utilized in the dough stage, use the intrusion test (7.8.37.9.3) and Table 1. If the mixture is to be used
as a dual usage cement, then both the extrusion (7.8.17.9.1 and/or 7.8.27.9.2) and intrusion (7.8.37.9.3) tests mustshall be
performed.
7.9.1 Extrusion, Capillary Viscosity:
7.9.1.1 Apparatus:
(1) Capillary Rheometer—Any capillary rheometer is satisfactory in which acrylic bone cement can be forced from a reservoir
through a capillary die and in which temperature, applied force, output rate, and barrel and die dimensions can be controlled and
measured accurately. accurately is satisfactory. Equipment that provides a constant shear rate has been shown to be equally useful.
The capillary die of the rheometer shall have a smooth, straight bore that is held within 60.0076 mm (60.0003 in.) in diameter
F451 − 21
and shall be held to within 60.025 mm (60.001 in.) in length. The bore and its finish are critical. It shall have no visible drill or
other tool marks and no detectable eccentricity.
(2) Due to the extreme sensitivity of flow data to the capillary dimensions, it is important that the capillary dimensions are
measured with precision and reported. The length to diameter length-to-diameter ratio shall normally be between 20 and 40. Larger
ratios and ratios less than that suggested require applying large corrections to the data (4, 5). In addition, the ratio of the reservoir
diameter to capillary diameter should be between 3 and 15. See Test Method D3835 for further details of capillary rheometers.
7.9.1.2 Calibration—Perform the test with a certified standard viscosity fluid approximating that expected for bone cement (50
2 2
N·s/m to 500 N·s/m ). Determine the viscosity of the standard fluid and the percent error from its specified value. Report this error
along with the viscosity of the tested cements.
7.9.1.3 Corrections—Bone cement is a non-Newtonian fluid,fluid; the data may be reported as corrected data. For example, true
shear rates, corrected for non-Newtonian flow behavior, and true shear stress, corrected for end effects or kinetic energy losses,
may be calculated. In such cases, the exact details of the mode of correction mustshall be reported. Some correction factors which
may apply are:
(1) Piston friction,
(2) Plunger back flow,
(3) Cement compressibility,
(4) Barrel back pressure,
(5) Capillary entrance effects (Bagley correction) (6), and
(6) Rabinowitsch shear rate correction (7).
7.9.1.4 Procedure:
(1) Select conditions of temperature and shear stress or shear rate in accordance with expected usage so that the flow rate will
fall within the desired limits.
(2) Inspect the rheometer and clean it if necessary. Ensure that previous cleaning procedures and usage have not changed the
dimensions or caused scratches or defects in the capillary or apparatus. Make the necessary measurements on the apparatus for
future calculations. Prepare the apparatus for running the test.
(3) Mix one or more complete unit(s) of powder and liquid in the recommended manner. Start a stopwatch at the onset of
mixing and read all subsequent times from this watch. After complete mixing, transfer the cement to the thermally equilibrated
reservoir and eject any entrapped air or excess bone cement.
(4) Start the apparatus at a time not greater than 2 ⁄2 min from the start of mixing and continue operating until the estimated
dough time or the viscosity exceeds 500 N·s/m .
(5) Disassemble the apparatus quickly before the cement sets and clean the apparatus of all remaining cement.
7.9.1.5 Calculations:
(1) Perform the calculation for viscosity of the cement at time intervals of 15 s from the start to finish of test run. Use the
following equations:
Pr Fr
Shear Stress, Pa 5 5 (3)
2L 2πR L
4Q 4V
Shear Rate, s 5 5 (4)
3 3
πr πr t
4 4
Pπr Fr t
Viscosity, Pa·s5 5 (5)
8LQ 8R LV
where:
P = pressure by ram, Pa,
F = force on ram, N,
r = radius of capillary, m,
R = radius of barrel, m,
L = length of capillary, m,
Q = flow rate, m /s,
V = volume extruded, m , and
t = extrusion time, s.
P = pressure by ram in Pa,
F = force on ram in N,
F451 − 21
r = radius of capillary in m,
R = radius of barrel in m,
L = length of capillary in m,
Q = flow rate in m /s,
V = volume extruded in m , and
t = extrusion time in s.
(2) These equations yield true shear rate and true viscosity for Newtonian fluids only; for non-Newtonian fluids, such as bone
cement, the apparent shear rate and viscosity are obtained.
7.9.1.6 Report—The report of the flow properties of the cement shall include:
(1) Description of the rheometer used.
(2) Temperature at which the data were obtained.
(3) The capillary diameter and length to diameter ratio of the capillary.
(4) The shear rate at which the test was performed.
(5) Viscosity versus observation time for three runs.
(6) Statement as to whether any correction factors (7.8.1.37.9.1.3) were applied.
7.9.2 Extrusion, Rotational Shear Viscosity:
7.9.2.1 Apparatus—Rotational Shear Rheometer—Any parallel plate rotational shear rheometer that can use 4 cm diameter plates,
a 1000 μm gap, and maintain a temperature of 23 6 0.5ºC0.5 °C is satisfactory.
7.9.2.2 Calibration—Calibrate the rheometer according to the manufacturer’s specifications.
7.9.2.3 Procedure:
(1) Mount a parallel plate geometry on the top fixture. A disposable plate system may be used. A stainless steel 4 cm diameter
plate is recommended. A removable bottom plate can be added to facilitate sample removal.
(2) Control the temperature of the rheometer so that at least one of the plates is at 23 6 0.5 ºC.°C.
(3) Move the plates apart to allow sample loading.
(4) Mix the cement according to the manufacturer’s specifications. Start a laboratory timer from the start of mixing.
(5) After the requisite mixing procedure is complete, place a sufficient quantity of bone cement between the plates so as to
completely fill the gap between the plates and no bubbles in excess of 1 mm are visible. Reduce the gap height between the plates
to 1000 μm. Scrape away excess cement.
-1–1
(6) Start a steady shear experiment at 0.5 s , monitoring the shear viscosity as a function of time at a sampling rate of 0.5
Hz or better. Note the elapsed time from the start of mixing to when the first data point is obtained.
(7) Collect data until the viscosity reaches 1000 Pa·s. Stop the test and remove the cement before it completely hardens.
(8) It is recommended that three runs are conducted per cement formulation.
7.9.2.4 Report—The report of the flow properties of the cement shall include:
(1) Description of the rheometer used.
(2) The shear viscosity as a function of time from the start of mixing. The reported instrument time points will need to be
shifted by the elapsed time measured in 7.8.2.37.9.2.3(6).
7.9.3 Intrusion:
7.9.3.1 The mold necessary for this test shall be made of polytetrafluoroethylene (PTFE), poly(ethyleneterephthalate),
polyoxymethylene, high density high-density polyethylene, or ultra-high molecular weight polyethylene (UHMWPE) and is shown
in Fig. 3.
7.9.3.2 Follow the procedure outlined in ISO 5833, section D.4.3 for intrusion testing.
7.9.3.3 Following the set, remove the specimen and measure the average height of the intrusion into all four of the 1.0-mm
diameter holes of the die to the nearest 0.5 mm.
7.9.3.4 Run this test once. If the requirement is not met, it must be met so in a repeat test.
7.10 Compressive Strength—The test specimens shall be cylinders 12 mm high and 6 mm in diameter. The ends of the specimens
shall be flat and smooth and shall be parallel to each other and at right angles to the long axis of the cylinder. An apparatus found
F451 − 21
NOTE 1—Dimensions in millimetres; 4four holes in bottom to be 1.00 6 0.05. Tolerance on all other dimensions 60.2. Material for all components:
Polytetrafluoroethylene,polytetrafluoroethylene, poly(ethyleneterephthalate), polyoxymethylene, high density high-density polyethylene, or ultra-high
molecular weight polyethylene (UHMWPE).
FIG. 3 Intrusion Mold
convenient for forming these test cylinders is shown in Fig. 4. An apparatus containing additional or fewer holes may be used as
NOTE 1—Material for Perforated Plate: Stainless Steel, Aluminum, Polytetrafluoroethylene, high density perforated plate: stainless steel, aluminum,
polytetrafluoroethylene, high-density polyethylene, or ultra-high molecular weight polyethylene (UHMWPE).
FIG. 4 Compression Specimens Mold
F451 − 21
long as adequate spacing between the holes is maintained. A mold release agent or silicone spray may be sparingly applied to
facilitate specimen removal.
7.10.1 Place the specimen mold on a flat glass or smooth metal plate and slightly overfill using one unit of mixed cement of
standard proportions at the commencement of dough time. Press a second flat glass or smooth metal plate on top of the mold. Hold
the mold and plates firmly together with a small C-clamp. Then, 1 h later, surface the ends of the cylinder plane at right angles
to the axis. The ends of the specimens may be ground flat to the axis by use of a small amount of 240-mesh silicon carbide powder
and water. Draw the molds containing the specimens back and forth across the plate coated with the abrasive and water. After
surfacing, remove the specimens from the mold. The specimens should be visually examined for surface defects. A surface defect
is defined as a surface discontinuity greater than 500 microns in major diameter. Acceptable specimens for testing shall appear to
be uniform and meet the dimensional requirements of 7.97.10. A minimum of five specimens shall be selected from the remaining
acceptable specimens and tested. Report the results of all specimens tested.
7.10.2 The time lapse between the start of mixing and the measurement of the compressive strength testing shall be 24 6 2 h.
Storage of the specimens before testing shall be at 23 6 2 °C 2 °C and 50 6 10 % relative humidity. Run specimens on any
universal testing machine equipped to record load versus deformation. Employ a deformation cross head cross-head speed of 20
to 25.4 mm/min. Test the specimens without use of any type of pad between the specimen and the platens of the machine. The
failure load shall be the load at the 2.0 % offset (2.0 % proof stress), upper yield point, or at fracture, whichever occurs first (Fig.
5).
7.10.2.1 The load at 2.0 % offset is the load at the intersection of the load deformation curve and a straight line parallel to the
Hookean portion of the curve (See(see Fig. X1.1 in Test Method D695) but offset along the deformation axis by 2.0 % of the test’s
test specimen’s gauge length (specimen’s height).
7.10.2.2 Calculate the compressive strength as the failure load divided by the calculated cross-sectional area.
7.10.2.3 Report the compressive strength of the material as the average of the compression strengths of the specimens tested in
7.9.27.10.2 to the nearest 1 MPa (145 psi). A minimum of five specimens is required.
7.11 Molar Mass by Gel Permeation Chromatography (GPC):
7.11.1 The molar mass (see Note 1) distribution and molar mass averages (number-average molar mass, weight-average molar
mass, and z-average molar mass) of the powder will be determined by gel permeation chromatography with reference to Test
Method D5296. Tetrahydrofuran (THF) will be used to dissolve the powder according to Test Method D5296. If the powder
contains radiopacifier, the mass of powder used to make the solution should be increased to account for the radiopacifier. The
solution should be filtered as suggested in Test Method D5296 to remove any radiopacifier.
NOTE 1—The term molecular weight (abbreviated MW) is obsolete and should be replaced by the SI (Système Internationale) equivalent of either relative
molecular mass (M ), which reflects the dimensionless ratio of the mass of a single molecule to an atomic mass unit (see ISO 80000-9), or molar mass
r
(M), which refers to the mass of a mole of a substance and is typically expressed as grams/mole. For polymers and other macromolecules, use of the
FIG. 5 Failure Load Criteria
F451 − 21
symbols M ,M , and M continue, referring to mass-average molar mass, number-average molar mass, and z-average molar mass, respectively. For more
w n z
information regarding proper utilization of SI units, see NIST Special Publication SP811.
7.11.2 Poly(methyl methacrylate) molar mass standards should be used to calibrate the GPC system. Poly(styrene) standards may
be used for the purpose of comparison to historical results, but it should be understood that the results will be relative and will
not represent an absolute determination of the polymer’s molar mass distribution.
7.11.3 Three aliquots of powder shall be tested for each bone cement.
7.11.4 This method may also be used on cured bone cement to determine the molar mass distribution of the cured material.
7.11.5 The number-average, weight-average, and z-average molar mass, along with the molar mass distribution relative to either
polystyrene or polymethyl methacrylate standards shall be reported for each aliquot of powder. Report the average and standard
deviation of the number-average, weight-average, and z-average molar mass for the three samples.
7.12 Leachable Monomer:
7.12.1 The residual monomer during curing and post-curing may be determined using the protocols described in Annex A5 and
Annex A6.
7.13 Stabilizer Concentration:
7.13.1 If a quinone-based stabilizer is used in the liquid portion, the amount of hydroquinone or monomethyl ether hydroquinone
may be determined using the protocols described in Annex A3 or Annex A4. Alternative protocols may be used if they can be
shown to have the required sensitivity.
7.14 Benzoyl Peroxide Concentration:
7.14.1 If benzoyl peroxide is used as an initiator, the amount of benzoyl peroxide in the powder portion may be determined using
the protocol described in Annex A1.
7.15 N,N-dimethyl-p-toluidine Concentration:
7.15.1 If N,N-dimethyl-p-toluidine is used as a reaction accelerator in the liquid portion, its concentration may be determined by
high-performance liquid chromatography (HPLC) or similar assays (8).
7.16 Stability Testing—The shelf life stability of bone cement powder-liquid systems shall be evaluated using the test methods
listed in Table 3.
7.17 Precision and Bias—Since 1976, the original Specification F451 methodologies have reportedly been routinely utilized by
TABLE 3 Requirements for Stability Testing
Test Type Test Description Test Material
Viscosity 7.3 Liquid Component
Viscosity 7.4 Liquid Component
Residual Peroxide Annex A1, Powder Component
Annex A2, or
equivalent
Dough Time 7.5 Curing Cement
Dough Time 7.6 Curing Cement
Set Time 7.7 Curing Cement
Set Time 7.8 Curing Cement
Compressive Strength 7.9 Cured Cement
Compressive Strength 7.10 Cured Cement
Tensile Strength D638 Cured Cement
Leachable Monomer Annex A2, or Cured and Curing Cement
equivalent
Leachable Monomer Annex A5, An- Cured and Curing Cement
nex A6, or
equivalent
F451 − 21
the various manufacturers. With the exception of the viscosity method of 7.8.17.9.1, which is based on another accepted ASTM
document (Test Method D3835), each test methodology in Section 7 contains its own statement of reporting acceptable levels of
performance, reproducibility, and precision. Therefore, no interlaboratory studies have been performed by the Committee F04.
8. Packaging
8.1 Materials shall be supplied in properly sealed containers made of materials that shall not contaminate or permit contamination
of the contents. The containers shall be packaged so as to prevent damage or leakage during shipping and storage. Materials must
be packaged to permit sterile transfer of contents to the surgical sterile field.
8.2 The contents shall be easily accessible, easy to open, and convenient to mix in the operating room. Entire package contents
(both powder and liquid) must be mixed to achieve recommended proportions.per the manufacturer’s instructions for use.
9. Labeling
9.1 Labeling on these cements must be in conformance with the Federal Food, Drug, and Cosmetic Act, Code of Federal
Regulations, and other pertinent laws and regulations.
9.2 The following minimal information mustshall appear on the container label.label:
9.2.1 It shall be clearly stated or color coded, or both, if the mixture is intended for usage in the pre-dough, dough, or dual usage
state.
9.2.2 The weight or volume, or both, of the liquid and powder components mustshall be stated.
9.2.3 Constituents of the powder and liquid shall be clearly stated in terms of weight or volume percent. This information shall
include the generic names of polymers, copolymers, chemical initiators, stabilizers, cross-linking agents, and any other ingredients,
such as radiopacify agents, gels, fillers, or antibiotics.
9.2.4 A statement that the contents are sterile and that the sterility shall be guaranteed only if the containers are undamaged.
Sterilization of the final polymerized cement is not applicable for this in-situ polymerization system. Sterilization has been
conducted on both the starting liquid and powder components.
9.2.5 The following warning shall appear on the label: (a) Flammable liquid; (b) Store below 25°C,25 °C, and (c) Protect from
light.
9.2.6 A statement to the effect that federal law restricts this device for sale by or on the order of a physician should be displayed.
9.2.7 The manufacturer and distributor shall be identified.
9.2.8 Each individual component of the package unit mustshall be clearly identified as to batch or lot number.
9.3 The following information shall appear on the product insert labeling accompanying each package.
9.3.1 Adequate and accurate instruction shall be given for handling the components and preparing the cement. Instructions shall
include a directive to mix all of the powder with all the liquid of a single unit. Procedures required to mix the materials, along
with recommended mixing utensils, shall be given.
9.3.2 Proper technique for administration and recommended procedures for using the cement, including any special precautions,
shall be indicated.
9.3.3 Toxic, hazardous, or irritating characteristics associated with the handling and use of the components and cement shall be
indicated.
9.3.4 A statement shall be included declaration that states that high temperatures of either the ambient surroundings or material
will cause shorter doughing and setting times, while low temperatures of either the ambient surroundings or material will increase
doughing and setting times. times shall be included. In addition, if the instructions for use (IFU) have an allowable temperature
F451 − 21
range, a chart showing handling (that is, doughing and setting) time versus temperature should be provided for the allowed range
of temperature (for example, 16 to 26°C).26 °C).
9.3.5 The ranges of doughing and setting times as measured at 23 6 1°C1 °C (7.57.6 and 7.77.8) shall be clearly stated. If a range
of temperatures has been identified, then testing at the lower and upper temperature limits and any intermediate increments should
be performed and the results reported in the IFU.
10. Biocompatibility
10.1 The biocompatibility of acrylic bone cement has been reported in the literature (1, 2). The material has been shown to produce
a well characterized well-characterized level of biological response following long-term clinical use and laboratory studies. The
results of these studies and the clinical history indicate an acceptable level of biological response in applications in which the
material has been utilized. When new applications of the material, or significant modification to the material,material or its physical
forms are being contemplated, the recommendations of Practice F748 and testing as described in Practices F619, F749, F756, F763,
F813, F895, and F981 should be considered. Any biocompatibility testing should be conducted on the final finished device (that
is, following mixing and complete cure of the liquid and powder in accordance with the instructions for use).
11. Report
11.1 Describe the composition of the liquid and powder fraction of the cement, along with the nominal concentrations.
11.2 Report the lot number of the cement.
11.3 Report any aging conditions applied to the components of the cement.
11.4 For the individual tests conducted, report the following:
11.4.1 Liquid Component Viscosity:
11.4.1.1 Describe the heating apparatus and 10 mL pipet.
11.4.1.2 Report the flow time for the two sets of runs before and after heating, along with percentage change. Indicate if the flow
time change is <10 %.
11.4.2 Doughing Time:
11.4.2.1 Describe the non-powdered surgical gloves in detail.
11.4.2.2 Report the dough time from all determinations and whether the minimum and maximum dough times differ by less than
1.5 minutes from the average.
11.4.3 Exothermic Temperature:
11.4.3.1 Describe the exotherm mold, including materials of construction, and the temperature measuring system used.
11.4.3.2 Report the average maximum temperature from the two runs, and whether the individual maximum and minimum values
for the maximum temperature are less than or equal to 64 °C from the average value.
11.4.4 Setting Time:
11.4.4.1 Report the setting time for the two runs, or four runs, if needed.
11.4.5 Flow Properties:
11.4.5.1 Describe in detail the rheometer used to measure the viscosity of the cement.
11.4.5.2 Report the test conditions, including test temperature, shear rate, and correction factors.
F451 − 21
11.4.5.3 Report the viscosity as a function of shear rate for the three repeated runs.
11.4.6 Intrusion:
11.4.6.1 Report the lengths of the four intrusion specimens along with the average.
11.4.7 Compression Strength:
11.4.7.1 Describe the mold used to prepare the specimens, including the materials of construction.
11.4.7.2 Describe the testing apparatus.
11.4.7.3 Report the compression strength for each of the specimens, along with the average and standard deviation.
11.4.7.4 Report the load-versus-displacement plots for each specimen.
11.4.8 Molar Mass Distribution:
11.4.8.1 Describe the chromatography system used.
11.4.8.2 Describe the calibration standards used.
11.4.8.3 Report the number-, weight-, and z-averaged molar mass for each run.
11.4.8.4 Report the molar mass distribution for each run.
11.4.9 Stability Testing:
11.4.9.1 Describe the stability testing conditions (temperature, time, light exposure), and the packaging used during the testing.
11.4.9.2 Describe the testing results pre- and post-stability testing.
12. Keywords
12.1 acrylic bone cement; compression strength; doughing time; exothermic temperature; extrusion; intrusion; poly(methacrylic
acid esters); setting time
F451 − 21
ANNEXES
(Mandatory Information)
A1. DETERMINATION OF TOTAL DIBENZOYL PEROXIDE (BPO) CONTENT BY TITRATION
A1.1 Weigh 2.0 6 0.1 g of polymer and transfer to a 250 mL 250 mL flask, recording the weight to the nearest 0.0001 g.
A1.2 Add 100 mL of reagent grade acetone to the flask, and mix the contents immediately.
A1.3 Add a magnetic stir bar and stir at room temperature for 1 h or until all polymer is dissolved. Cover or stopper the flask while
stirring.
A1.4 Prepare a 50 % (w ⁄w) aqueous solution of potassium iodide.
A1.5 Add 5 mLlmL of glacial acetic acid and 3 mL of the 50 % (w/w) aqueous potassium iodide solution and let stand (covered)
for 1 min.
A1.5.1 If the potassium iodide solution appears yellowish, a fresh solution should be prepared and used.
A1.6 Titrate the mixture with 0.01 N sodium thiosulfate solution to a yellow-free end point.
A1.7 Break up any precipitated clumps of polymer with a glass rod and continue the titration until the end point persists for 1 min.
A1.8 Record the volume of 0.01 N sodium thiosulfate used to the nearest 0.05 mL.
A1.8.1 Use a 25 mL burette for titrating when the anticipated BPO assay is less than 1.0 %.
A1.8.2 Use a 50 mL burette for titrating when the anticipated BPO assay is greater than 1.0 %.
A1.8.3 Process a blank sample through steps A1.2A1.2 – A1.7 to A1.7.
A1.9 Calculate the results as follows:
Test methods in these annexes have not been validated, but are provided as guidance.
F451 − 21
A1.9.1 Calculate the percent of dibenzoyl peroxide present in sample using the equation:
V 2 B 3N 312.11
~ !
% BPO 5 (A1.1)
W
where:
V = mL Na S O used for sample titration,
2 2 3
N = normality of Na S O ,
2 2 3
W = weight of sample (g), and
B = average mL of Na S O used for the blank titrations.
2 2 3
A2. DETERMINATION OF TOTAL DIBENZOYL PEROXIDE (BPO) CONTENT BY CHROMATOGRAPHY
A2.1 Required Apparatus
A2.1.1 Liquid Chromatograph, equipped with a multiple wavelength (see Practices E169 and E275) or photodiode array
ultraviolet detector, heated column compartment, and gradient elution capabilities. The liquid chromatograph should be equipped
with a means for a 10-μL injection such as a sample loop.
A2.1.2 Chromatographic Column, C8 or C18 reverse phase, 5 μm particle size, 15 cm by 4.6 mm or equivalent, capable of
separating degradation products.
A2.1.3 Data Acquisition/Handling System, providing the means for determining chromatographic peak areas and for handling and
reporting data. This is best accomplished using a computer with appropriate software.
A2.1.4 Vortex mixer.
A2.1.5 Analytical Balance, capable of weighing to 0.0001 g.
A2.2 Reagents and Materials
A2.2.1 Solvents:
A2.2.1.1 Acetonitrile—HPLC grade, spectro-quality or chromatography quality reagent (a reagent whose UV cutoff is about 190
nm).
A2.2.1.2 Water—HPLC, or UV quality reagent.
F451 − 21
A2.2.2 Buffer:
A2.2.2.1 Acetic Acid—HPLC grade.
A2.2.3 Additives:
A2.2.3.1 High-purity BPO.
A2.3 Preparation of Samples (including extraction of samples and calibration standards)
A2.3.1 Preparation of Samples:
A2.3.1.1 Weigh to the nearest 0.0001 g, approximately 4 g of the bone cement, that is, W , into a pre-weighed (to the nearest
sample
0.0001 g) 20-mL scintillation vial, that is, W .
vial
A2.3.1.2 Add approximately 20.0 mL of acetonitrile, close with the stopper, and weigh to the nearest 0.0001 g, that is, W
(vial +
sol).
A2.3.1.3 Vortex using a vortex mixer for a minimum of 2 h.
A2.3.1.4 Filter 1 mL solution through 0.45 μm PTFE syringe filter into an autosampler vials.
A2.3.2 Preparation of BPO Standards:
A2.3.2.1 Prepare stock solution of BPO in acetonitrile.
A2.3.2.2 Weigh into a 100 mL volumetric flask, to the nearest 0.1 mg, approximately 100 mg of BPO.
A2.3.2.3 Fill volumetric flask to 100 mL by addition of acetonitrile solvent.
A2.3.2.4 Prepare standards at concentrations in the range 100 to 600 ppm.
A2.3.2.5 Dilute stock solution by acetonitrile, weigh stock aliquots and acetonitrile diluent to the nearest 0.1 mg.
F451 − 21
A2.4 Performance Requirements
A2.4.1 Plate Count Number—A column is expected to have a plate count N in excess of what is specified when using column
calibration check standards:
t
R
N 5 16 (A2.1)
S D
W
where:
t = peak elution time in minutes, and
R
W = peak width in minutes.
A2.5 Preparation of Liquid Chromatograph
A2.5.1 Column: C8 or C18 reverse phase, 5 μm particle size, 150 mm by 4.6 mm or equivalent.
A2.5.2 Flow rate: 1 mL.
A2.5.3 Temperature: 30 °C.
A2.5.4 Injection volume: 10 μL.
A2.5.5 Detection diode array detector wavelength: 272 nm.
A2.5.6 Mobile phase A: Water with 0.1 % acetic acid. (See Table A2.1.)
A2.5.7 Mobile phase B: Acetonitrile. (See Table A2.1.)
A2.6 Calibration
A2.6.1 Inject 10 μL of each of the calibration standards.
A2.6.2 Measure the peak areas using a computer or an integrator.
TABLE A2.1 Liquid Chromatography Gradient Conditions
Mobile Phase A Mobile Phase B
Time (min)
(Water), % (Acetonitrile), %
0 80 20
15 80 20
35 0 100
40 0 100
41 80 20
F451 − 21
A2.6.3 Plot peak area versus concentration of BPO and fit the points to a linear regression line.
A2.7 Procedure
A2.7.1 Use liquid chromatographic conditions as prescribed in A2
...



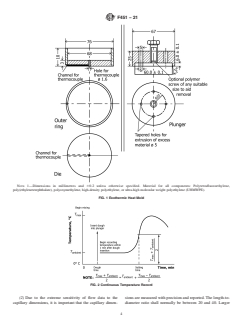




Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...