ASTM F3047M-23
(Guide)Standard Guide for High Demand Hip Simulator Wear Testing of Hard-on-Hard Articulations
Standard Guide for High Demand Hip Simulator Wear Testing of Hard-on-Hard Articulations
SIGNIFICANCE AND USE
5.1 The current hip simulator wear test standards (ISO 14242-1 or ISO 14242-3) stipulate only one load waveform and one set of articulation motions. There is a need for more versatile and rigorous wear test regimes, but the knowledge of what represents realistic high demand wear test features is limited. More research is clearly needed before a standard that defines what a representative high demand wear test should include can be written. The objective of this guide is to advise researchers on the possible high demand wear test features that should be included in evaluation of hard-on-hard articulations.
5.2 This guide makes suggestions of what high demand test features may need to be added to an overall high demand wear test regime. The features described here are not meant to be all inclusive. Based on current knowledge they appear to be relevant to adverse conditions that can occur in clinical use.
5.3 All the test features, both conventional and high demand, could have interactive effects on the wear of the components.
SCOPE
1.1 The objective of this guide is to advise researchers on the possible high demand wear test features that should be included in evaluation of hard-on-hard articulations. This guide makes suggestions for high demand test features that may need to be added to an overall wear test regime. Device articulating components manufactured from other metallic alloys, ceramics, or with coated or elementally modified surfaces without significant clinical use could possibly be evaluated with this guide. However, such materials may include risks and failure mechanisms that are not addressed in this guide.
1.2 Hard-on-hard hip bearing systems include metal-on-metal (for example, Specifications F75, F799, and F1537; ISO 5832-4, ISO 5832-12), ceramic-on-ceramic (for example, ISO 6474-1, ISO 6474-2, ISO 13356), ceramic-on-metal, or any other bearing systems where both the head and cup components have high surface hardness. An argument has been made that the hard-on-hard THR articulation may be better for younger, more active patients. These younger patients may be more physically fit and expect to be able to perform more energetic activities. Consequently, new designs of hard-on-hard THR articulations may have some implantations subjected to more demanding and longer wear performance requirements.
1.3 Total Hip Replacement (THR) with metal-on-metal articulations have been used clinically for more than 50 years (1, 2).2 Early designs had mixed clinical results. Eventually they were eclipsed by THR systems using metal-on-polyethylene articulations. In the 1990s the metal-on-metal articulation again became popular with more modern designs (3), including surface replacement.
1.4 In the 1970s the first ceramic-on-ceramic THR articulations were used. In general, the early results were not satisfactory (4, 5). Improvement in alumina, and new designs in the 1990s improved the results for ceramic-on-ceramic articulations (6).
1.5 The values stated in SI units are to be regarded as the standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-May-2023
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.22 - Arthroplasty
Relations
- Effective Date
- 01-Mar-2020
- Effective Date
- 15-May-2019
- Effective Date
- 01-Jan-2019
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Oct-2016
- Effective Date
- 01-Sep-2013
- Refers
ASTM F86-12a - Standard Practice for Surface Preparation and Marking of Metallic Surgical Implants - Effective Date
- 01-Dec-2012
- Effective Date
- 15-May-2012
- Refers
ASTM F86-12 - Standard Practice for Surface Preparation and Marking of Metallic Surgical Implants - Effective Date
- 15-May-2012
- Effective Date
- 15-Nov-2011
- Effective Date
- 01-Jun-2011
- Effective Date
- 01-Sep-2010
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Dec-2009
- Effective Date
- 01-Oct-2008
Overview
ASTM F3047M-23: Standard Guide for High Demand Hip Simulator Wear Testing of Hard-on-Hard Articulations provides recommendations for designing robust wear test regimens to assess hard-on-hard hip bearing systems, such as metal-on-metal and ceramic-on-ceramic articulations. Unlike existing wear test standards (such as ISO 14242-1 and ISO 14242-3), which specify a single set of test parameters, ASTM F3047M-23 seeks to guide researchers in incorporating more versatile and clinically relevant “high demand” test features. This assists in evaluating device performance under conditions that may better represent the challenges faced by younger, more active patients or adverse clinical scenarios.
Key Topics
- Scope of Applicability: The guide is primarily intended for use with hip prostheses using hard bearing surfaces (metal-on-metal, ceramic-on-ceramic, ceramic-on-metal) where increased physical demand or non-ideal conditions are anticipated.
- Test Suggestions: It outlines possible high demand wear test features, such as elevated loads, altered cup angles, third-body particle introduction, stop-dwell-start (stiction) cycles, and microseparation events.
- Material Coverage: Devices manufactured from other metallic alloys, ceramics, or coated surfaces without significant clinical use may be evaluated using this guide, with caution regarding unaddressed risks.
- Interactivity of Test Modes: Highlights the potential for interactive effects between conventional and high demand test features, underscoring the complexity of real-world wear mechanisms.
- Clinical Relevance: Each test feature is linked to clinical experience or known mechanisms of increased wear or failure in total hip replacement (THR) systems.
Applications
The guide is valuable for:
- Medical Device Researchers and Developers: When evaluating the wear performance of new or modified hard-on-hard hip replacement designs, especially for younger, more active patient populations likely to subject implants to higher demands.
- Preclinical Testing Laboratories: As a resource for developing rigorous test protocols that emulate a broader range of in vivo conditions, thereby potentially improving the predictive power of preclinical assessments.
- Regulatory and Safety Reviews: Supporting documentation for regulatory submissions or risk analyses by justifying the inclusion of challenging test modes and reporting requirements.
- Quality Assurance: As a benchmark for manufacturers to validate the durability of materials and designs under conditions that extend beyond conventional standards.
Practical high demand wear test features covered include:
- Cup Inclination Angle: Investigating the effects of increased or non-optimal cup angles as can occur with surgical variability.
- Third-Body Wear: Introducing particulate matter (e.g., bone, cement, metal, or ceramic debris) to simulate intraoperative or in vivo contamination.
- High Load and Variable Waveforms: Implementing load regimens that reflect more energetic activities or patient-specific risks.
- Stop-Dwell-Start Cycles: Mimicking common, short walking episodes and the impact of joint start/stop activity.
- Microseparation: Evaluating wear due to temporary loss of contact between head and cup (e.g., from joint laxity or insufficient soft tissue constraint).
Related Standards
ASTM F3047M-23 operates in concert with multiple international and ASTM standards, including:
- ISO 14242-1 and ISO 14242-3 – Wear of total hip-joint prostheses: baseline protocols on load and motion.
- ISO 14242-4 – Testing under edge-loading conditions and variations in component positioning.
- ASTM F75, F799, F1537 – Specifications for cobalt-chromium-molybdenum alloys commonly used in hard-on-hard prostheses.
- ISO 6474-1, ISO 6474-2, ISO 13356 – Ceramic materials standards used in ceramic-on-ceramic articulations.
- ASTM F3018 – Assessment of hard-on-hard articulation hip devices.
- ISO 17853, ASTM F1877, ASTM F561 – Guidance on particle isolation, characterization, and retrieval analysis.
Keywords: hard-on-hard hip prosthesis, hip simulator testing, high demand wear, metal-on-metal, ceramic-on-ceramic, third-body wear, acetabular cup angle, microseparation, preclinical evaluation, orthopedic device testing.
Buy Documents
ASTM F3047M-23 - Standard Guide for High Demand Hip Simulator Wear Testing of Hard-on-Hard Articulations
REDLINE ASTM F3047M-23 - Standard Guide for High Demand Hip Simulator Wear Testing of Hard-on-Hard Articulations
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F3047M-23 is a guide published by ASTM International. Its full title is "Standard Guide for High Demand Hip Simulator Wear Testing of Hard-on-Hard Articulations". This standard covers: SIGNIFICANCE AND USE 5.1 The current hip simulator wear test standards (ISO 14242-1 or ISO 14242-3) stipulate only one load waveform and one set of articulation motions. There is a need for more versatile and rigorous wear test regimes, but the knowledge of what represents realistic high demand wear test features is limited. More research is clearly needed before a standard that defines what a representative high demand wear test should include can be written. The objective of this guide is to advise researchers on the possible high demand wear test features that should be included in evaluation of hard-on-hard articulations. 5.2 This guide makes suggestions of what high demand test features may need to be added to an overall high demand wear test regime. The features described here are not meant to be all inclusive. Based on current knowledge they appear to be relevant to adverse conditions that can occur in clinical use. 5.3 All the test features, both conventional and high demand, could have interactive effects on the wear of the components. SCOPE 1.1 The objective of this guide is to advise researchers on the possible high demand wear test features that should be included in evaluation of hard-on-hard articulations. This guide makes suggestions for high demand test features that may need to be added to an overall wear test regime. Device articulating components manufactured from other metallic alloys, ceramics, or with coated or elementally modified surfaces without significant clinical use could possibly be evaluated with this guide. However, such materials may include risks and failure mechanisms that are not addressed in this guide. 1.2 Hard-on-hard hip bearing systems include metal-on-metal (for example, Specifications F75, F799, and F1537; ISO 5832-4, ISO 5832-12), ceramic-on-ceramic (for example, ISO 6474-1, ISO 6474-2, ISO 13356), ceramic-on-metal, or any other bearing systems where both the head and cup components have high surface hardness. An argument has been made that the hard-on-hard THR articulation may be better for younger, more active patients. These younger patients may be more physically fit and expect to be able to perform more energetic activities. Consequently, new designs of hard-on-hard THR articulations may have some implantations subjected to more demanding and longer wear performance requirements. 1.3 Total Hip Replacement (THR) with metal-on-metal articulations have been used clinically for more than 50 years (1, 2).2 Early designs had mixed clinical results. Eventually they were eclipsed by THR systems using metal-on-polyethylene articulations. In the 1990s the metal-on-metal articulation again became popular with more modern designs (3), including surface replacement. 1.4 In the 1970s the first ceramic-on-ceramic THR articulations were used. In general, the early results were not satisfactory (4, 5). Improvement in alumina, and new designs in the 1990s improved the results for ceramic-on-ceramic articulations (6). 1.5 The values stated in SI units are to be regarded as the standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 The current hip simulator wear test standards (ISO 14242-1 or ISO 14242-3) stipulate only one load waveform and one set of articulation motions. There is a need for more versatile and rigorous wear test regimes, but the knowledge of what represents realistic high demand wear test features is limited. More research is clearly needed before a standard that defines what a representative high demand wear test should include can be written. The objective of this guide is to advise researchers on the possible high demand wear test features that should be included in evaluation of hard-on-hard articulations. 5.2 This guide makes suggestions of what high demand test features may need to be added to an overall high demand wear test regime. The features described here are not meant to be all inclusive. Based on current knowledge they appear to be relevant to adverse conditions that can occur in clinical use. 5.3 All the test features, both conventional and high demand, could have interactive effects on the wear of the components. SCOPE 1.1 The objective of this guide is to advise researchers on the possible high demand wear test features that should be included in evaluation of hard-on-hard articulations. This guide makes suggestions for high demand test features that may need to be added to an overall wear test regime. Device articulating components manufactured from other metallic alloys, ceramics, or with coated or elementally modified surfaces without significant clinical use could possibly be evaluated with this guide. However, such materials may include risks and failure mechanisms that are not addressed in this guide. 1.2 Hard-on-hard hip bearing systems include metal-on-metal (for example, Specifications F75, F799, and F1537; ISO 5832-4, ISO 5832-12), ceramic-on-ceramic (for example, ISO 6474-1, ISO 6474-2, ISO 13356), ceramic-on-metal, or any other bearing systems where both the head and cup components have high surface hardness. An argument has been made that the hard-on-hard THR articulation may be better for younger, more active patients. These younger patients may be more physically fit and expect to be able to perform more energetic activities. Consequently, new designs of hard-on-hard THR articulations may have some implantations subjected to more demanding and longer wear performance requirements. 1.3 Total Hip Replacement (THR) with metal-on-metal articulations have been used clinically for more than 50 years (1, 2).2 Early designs had mixed clinical results. Eventually they were eclipsed by THR systems using metal-on-polyethylene articulations. In the 1990s the metal-on-metal articulation again became popular with more modern designs (3), including surface replacement. 1.4 In the 1970s the first ceramic-on-ceramic THR articulations were used. In general, the early results were not satisfactory (4, 5). Improvement in alumina, and new designs in the 1990s improved the results for ceramic-on-ceramic articulations (6). 1.5 The values stated in SI units are to be regarded as the standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F3047M-23 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3047M-23 has the following relationships with other standards: It is inter standard links to ASTM F1537-20, ASTM F799-19, ASTM F561-19, ASTM F3018-17, ASTM F1877-16, ASTM F561-13, ASTM F86-12a, ASTM F75-12, ASTM F86-12, ASTM F1537-11, ASTM F799-11, ASTM F561-05a(2010), ASTM F1877-05(2010), ASTM F86-04(2009), ASTM F1537-08. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F3047M-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F3047M − 23
Standard Guide for
High Demand Hip Simulator Wear Testing of Hard-on-Hard
Articulations
This standard is issued under the fixed designation F3047M; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope satisfactory (4, 5). Improvement in alumina, and new designs
in the 1990s improved the results for ceramic-on-ceramic
1.1 The objective of this guide is to advise researchers on
articulations (6).
the possible high demand wear test features that should be
included in evaluation of hard-on-hard articulations. This guide 1.5 The values stated in SI units are to be regarded as the
makes suggestions for high demand test features that may need standard.
to be added to an overall wear test regime. Device articulating
1.6 This standard does not purport to address all of the
components manufactured from other metallic alloys,
safety concerns, if any, associated with its use. It is the
ceramics, or with coated or elementally modified surfaces
responsibility of the user of this standard to establish appro-
without significant clinical use could possibly be evaluated
priate safety, health, and environmental practices and deter-
with this guide. However, such materials may include risks and
mine the applicability of regulatory limitations prior to use.
failure mechanisms that are not addressed in this guide.
1.7 This international standard was developed in accor-
dance with internationally recognized principles on standard-
1.2 Hard-on-hard hip bearing systems include metal-on-
ization established in the Decision on Principles for the
metal (for example, Specifications F75, F799, and F1537; ISO
Development of International Standards, Guides and Recom-
5832-4, ISO 5832-12), ceramic-on-ceramic (for example, ISO
mendations issued by the World Trade Organization Technical
6474-1, ISO 6474-2, ISO 13356), ceramic-on-metal, or any
Barriers to Trade (TBT) Committee.
other bearing systems where both the head and cup compo-
nents have high surface hardness. An argument has been made
2. Referenced Documents
that the hard-on-hard THR articulation may be better for
younger, more active patients. These younger patients may be
2.1 ASTM Standards:
more physically fit and expect to be able to perform more
F75 Specification for Cobalt-28 Chromium-6 Molybdenum
energetic activities. Consequently, new designs of hard-on-
Alloy Castings and Casting Alloy for Surgical Implants
hard THR articulations may have some implantations subjected
(UNS R30075)
to more demanding and longer wear performance require-
F86 Practice for Surface Preparation and Marking of Metal-
ments.
lic Surgical Implants
F561 Practice for Retrieval and Analysis of Medical
1.3 Total Hip Replacement (THR) with metal-on-metal
Devices, and Associated Tissues and Fluids
articulations have been used clinically for more than 50 years
F799 Specification for Cobalt-28 Chromium-6 Molybdenum
(1, 2). Early designs had mixed clinical results. Eventually
Alloy Forgings for Surgical Implants (UNS R31537,
they were eclipsed by THR systems using metal-on-
R31538, R31539)
polyethylene articulations. In the 1990s the metal-on-metal
F1537 Specification for Wrought Cobalt-28Chromium-
articulation again became popular with more modern designs
6Molybdenum Alloys for Surgical Implants (UNS
(3), including surface replacement.
R31537, UNS R31538, and UNS R31539)
1.4 In the 1970s the first ceramic-on-ceramic THR articu-
F1877 Practice for Characterization of Particles
lations were used. In general, the early results were not
F2033 Specification for Total Hip Joint Prosthesis and Hip
Endoprosthesis Bearing Surfaces Made of Metallic,
Ceramic, and Polymeric Materials
This guide is under the jurisdiction of ASTM Committee F04 on Medical and
F3018 Guide for Assessment of Hard-on-Hard Articulation
Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.22 on Arthroplasty.
Current edition approved June 1, 2023. Published June 2023. Originally
approved in 2015. Last previous edition approved in 2015 as F3047M – 15. DOI: For referenced ASTM standards, visit the ASTM website, www.astm.org, or
10.1520/F3047M-23. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
The boldface numbers in parentheses refer to the list of references at the end of Standards volume information, refer to the standard’s Document Summary page on
this standard. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3047M − 23
Total Hip Replacement and Hip Resurfacing Arthroplasty ISO 17853 Wear of Implant Materials—Polymer and Metal
Devices Wear Particles—Isolation and Characterization
2.2 ISO Standards:
3. Terminology
ISO 5832-4 Implants for Surgery—Metallic Materials—Part
4: Cobalt-Chromium-Molybdenum Casting Alloy
3.1 Definitions:
ISO 5832-12 Implants for Surgery—Metallic Materials—
3.1.1 acetabular liner—portion of the modular acetabular
Part 12: Wrought Cobalt-Chromium-Molybdenum Alloy
device with an internal hemispherical socket intended to
ISO 6474-1 Implants for Surgery—Ceramic Materials—Part
articulate with the head of a femoral prosthesis. The external
1: Ceramic Materials Based on High Purity Alumina
geometry of this component interfaces with the acetabular shell
ISO 6474-2 Implants for Surgery—Ceramic Materials—Part
through a locking mechanism which may be integral to the
2: Composite Materials Based on a High-Purity Alumina
design of the liner and shell or may rely upon additional
Matrix with Zirconia Reinforcement
components (for example, metal ring, screws, and so forth).
ISO 7206-2 Implants for Surgery—Partial and Total Hip
3.1.2 acetabular shell—the metallic external, hollow struc-
Joint Prostheses—Part 2: Articulating Surfaces Made of
ture that provides additional mechanical support or reinforce-
Metallic, Ceramic and Plastics Materials
ment for an acetabular liner and whose external features
ISO 13356 Implants for Surgery—Ceramic Materials Based
interface directly with the bones of the pelvic socket (for
on Yttria-Stabilized Tetragonal Zirconia (Y-TZP)
example, through bone cement, intimate press-fit, coatings for
ISO 14242-1 Implants for Surgery—Wear of Total Hip-Joint
attachment to bone cement or tissue, integral screw threads,
Prostheses. Part 1: Loading and Displacement Parameters
anchoring screws, pegs, and so forth). The acetabular shell may
for Wear-Testing Machines and Corresponding Environ-
be solid or contain holes for fixation to the pelvis or attachment
mental Conditions for Test
of instrumentation.
ISO 14242-2 Implants for Surgery—Wear of Total Hip-Joint
3.1.3 alloy fabricated form—the raw material form of the
Prostheses. Part 2: Methods of Measurement
metallic alloy (such as Specifications F75, F799, and F1537;
ISO 14242-3 Implants for Surgery—Wear of Total Hip-Joint
ISO 5832-4, ISO 5832-12) and any processing techniques
Prostheses—Part 3: Loading and Displacement Param-
(such as Practice F86, Specification F2033, and ISO 7206-2)
eters for Orbital Bearing Type Wear Testing Machines and
used to fabricate the final form of the implant.
Corresponding Environmental Conditions for Test
ISO 14242-4 Implants for Surgery—Wear of Total Hip-Joint 3.1.4 breakaway wear—a ‘higher’ unexpected wear rate that
Prostheses—Part 4: Testing Hip Prostheses Under Varia- follows a period of steady-state wear as illustrated in Fig. 2.
tions in Component Positioning Which Results in Direct
3.1.5 breakaway wear with recovery—a breakaway wear
Edge Loading
rate that returns to the lower steady-state wear rates. The
breakaway/recovery phenomenon can be a single event or
multiple ‘episodic’ events during the otherwise steady-state
Available from American National Standards Institute (ANSI), 25 W. 43rd St.,
conditions as illustrated in Fig. 2.
4th Floor, New York, NY 10036, http://www.ansi.org.
FIG. 1 Illustration of Cup Articular Arc Angle
F3047M − 23
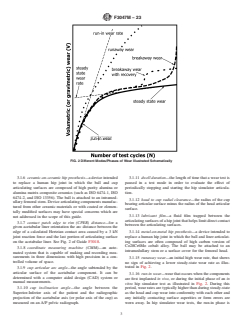
FIG. 2 Different Modes/Phases of Wear Illustrated Schematically
3.1.6 ceramic-on-ceramic hip prosthesis—a device intended 3.1.11 dwell duration—the length of time that a wear test is
to replace a human hip joint in which the ball and cup paused in a test mode in order to evaluate the effect of
articulating surfaces are composed of high purity alumina or periodically stopping and starting the hip simulator articula-
alumina matrix composite ceramics (such as ISO 6474-1, ISO tion.
6474-2, and ISO 13356). The ball is attached to an intramed-
3.1.12 head to cup radial clearance—the radius of the cup
ullary femoral stem. Device articulating components manufac-
bearing articular surface minus the radius of the head articular
tured from other ceramic materials or with coated or elemen-
surface.
tally modified surfaces may have special concerns which are
3.1.13 lubricant film—a fluid film trapped between the
not addressed in the scope of this guide.
articulating surfaces of a hip joint that helps limit direct contact
3.1.7 contact patch edge to rim (CPER) distance—for a
between the articulating surfaces.
given acetabular liner orientation the arc distance between the
edge of a calculated Hertzian contact area caused by a 3 kN 3.1.14 metal-on-metal hip prosthesis—a device intended to
replace a human hip joint in which the ball and liner articulat-
joint reaction force and the last portion of articulating surface
on the acetabular liner. See Fig. 2 of Guide F3018. ing surfaces are often composed of high carbon version of
Co28Cr6Mo cobalt alloy. The ball may be attached to an
3.1.8 coordinate measuring machine (CMM)—an auto-
intramedullary stem or a surface cover for the femoral head.
mated system that is capable of making and recording mea-
surements in three dimensions with high precision in a con-
3.1.15 runaway wear—an initial high wear rate, that shows
trolled volume of space.
no sign of achieving a lower steady-state wear rate as illus-
trated in Fig. 2.
3.1.9 cup articular arc angle—the angle subtended by the
articular surface of the acetabular component. It can be
3.1.16 run-in wear—wear that occurs when the components
determined with a computer aided design (CAD) system or
are first implanted in vivo, or during the initial phase of an in
manual measurements.
vitro hip simulator test as illustrated in Fig. 2. During this
3.1.10 cup inclination angle—the angle between the period, wear rates are typically higher than during steady-state
Superior-Inferior axis of the patient and the radiographic as the head and cup wear into conformity with each other and
projection of the acetabular axis (or polar axis of the cup) as any initially contacting surface asperities or form errors are
measured on an A/P pelvic radiograph. worn away. In hip simulator wear tests, the run-in phase is
F3047M − 23
often considered to be about 1 million cycles. The transition to inclusive. Based on current knowledge they appear to be
steady-state wear can be estimated graphically from the plot of relevant to adverse conditions that can occur in clinical use.
total wear versus number of cycles.
5.3 All the test features, both conventional and high
3.1.17 serum protein content—the concentration of protein
demand, could have interactive effects on the wear of the
molecules present in serum, usually expressed in grams per
components.
liter. The value is usually supplied by the commercial source
6. Test Samples
for the serum.
6.1 The materials and articulating geometry of the hard-on-
3.1.18 steady-state wear—wear rates that occur after a
hard system should be representative of the system intended for
transient run-in wear period as illustrated in Fig. 2. Typically,
clinical use. The acetabular components must have the same
the steady-state wear rate is less than the run-in wear rate. In
geometry as the acetabular system intended for clinical use
hip simulator wear tests, the steady-state rate typically is
because the stiffness of the acetabular system could affect the
reached after 1 million or more cycles.
response to loads and motions at the articulating surface.
3.1.19 third-body wear—the increased wear that occurs due
to particle(s) not permanently attached to the articulating
6.2 The test parts should receive all of the processing that is
surfaces being present in the articulation. The source of intended for product intended for clinical use, including
particle(s) can be external to the articulating surfaces or come
sterilization. There is no literature reporting any detrimental
from the articulating surfaces. effects of gamma sterilization or any other sterilization meth-
ods used for orthopedic devices on the physical or chemical
3.1.20 volumetric wear rate—the rate of material volume
properties of metallic alloys. However, it may be advisable to
lost from both articulating surfaces.
sterilize everything prior to definitive tests for preclinical
4. Summary of Guide
evaluation to make all parts as close to the clinical product as
possible. Coatings on non-articulating surfaces of the test parts
4.1 A conventional hip simulator wear test should be per-
could create problems with the handling of the parts and weight
formed according to ISO 14242-1 or ISO 14242-3 for five (5)
loss measurements during testing. It may be necessary to have
million cycles. This will be used as a basis for comparison of
test parts without the non-articulating surface coatings.
the results of any high demand test regime. Any high demand
However, any thermal processing the test parts would receive
wear test regime should use ISO 14242-1 or ISO 14242-3 as
as part of any coating process should still be performed.
the starting point and high demand parameters should be made
Particulate-based coating could be a source for third-body wear
as modifications to that standard. ISO 14242-3 may not be
particles, but random particle loss interferes with the repeat-
suitable for high demand wear tests that require modification of
ability of the test. Consideration should also be given to using
the articulating motion, because the motion cycle is built into
particulate from the coatings as controlled third-body particle
the test machine hardware and can’t be modified.
sources.
4.2 The high demand wear test can be performed as a
6.3 No preconditioning is required for the test samples other
continuation of the conventional ISO 14242-1 or ISO 14242-3
than careful handling to ensure that they remain clean and free
test or run as a separate test. High demand test features will be
of contamination prior to start of testing.
added to the high demand wear test and justified as clinically
relevant. This will require an understanding of the potential 6.4 The diameter and acetabular sizing must be justified as
interactions of the possible high demand modes which would
worst case for the wear tests. There are many possible factors
indicate a series of shorter duration tests. A final high demand
that could make a hard-on-hard couple a “worst case.” The
test(s) for the preclinical evaluation of a device shall include a
diameter of the articulation, head-to-cup radial clearance, the
test protocol of at least 5 million cycles. These high demand
thickness of material in the liner and the shell, the design of
wear test cycles will be in addition to the conventional 5 modularity of the liner and the shell, or the sphericity of the
million cycles of wear testing.
articulations could all potentially create a “worst case” com-
bination for wear. These factors and more should be considered
5. Significance and Use
in justifying a “worst case.”
5.1 The current hip simulator wear test standards (ISO
6.5 The usual small amount of material lost in hard-on-hard
14242-1 or ISO 14242-3) stipulate only one load waveform
wear tests combined with the larger mass of the components
and one set of articulation motions. There is a need for more
may make weight loss characterization of wear according to
versatile and rigorous wear test regimes, but the knowledge of
ISO 14242-2 more difficult. Another means of measuring loss
what represents realistic high demand wear test features is
of material from both the convex and concave surfaces of the
limited. More research is clearly needed before a standard that
metal-on-metal articulation is by measuring the change in the
defines what a representative high demand wear test should
surface geometries. For this measurement method, both articu-
include can be written. The objective of this guide is to advise
lating surfaces must be measured with enough precision before
researchers on the possible high demand wear test features that
testing to provide a baseline for estimating the volume of
should be included in evaluation of hard-on-hard articulations.
material lost from the surfaces due to the tests. This shall
5.2 This guide makes suggestions of what high demand test require a high precision coordinate measuring machine (CMM)
features may need to be added to an overall high demand wear or other high precision measurement devices. The volumetric
test regime. The features described here are not meant to be all measurement does have one advantage over the weight loss
F3047M − 23
method, because it can indicate the distribution of wear on a come from such sources as neck impingement (16, 17), porous
surface. Both methods require precise techniques that shall coatings, or tribocorrosion (18-20). The presence of such types
have validated procedures before they are used in an actual of third-body particulate may be of limited duration because
wear test. such particles could take time to form, then possibly be broken
down, and eventually be removed from the joint capsule.
6.6 The geometry of both articulating surfaces should be
7.3.2 There clearly needs to be a small particle size in order
characterized as to their original geometry and surface finish.
for there to be a potential that the particle could be entrapped
The same techniques should be used to characterize both
between the articulating surfaces and cause damage. Trying to
surfaces intermittently during testing and after completion of
standardize on small bone particles is not practical.
testing. These measurement results can be used to estimate the
7.3.3 The time and rate of third-body particle replenishment
amount of material lost, but the alternate weight loss method
should be justified.
should be used as a validation method for the volumetric
7.3.4 Care must be taken during the portion of the test with
measurement by making the alternative weight loss measure-
the third-body particles that they remain suspended in the
ments at the beginning and the end of the tests.
lubrication medium as much as possible to keep availability to
6.7 Additional characterization of the surfaces in a scanning
the articulating surface high. Additional agitation of the
electron microscope or three-dimensional digital optical mi-
lubricant, limiting crevice and corners in the test chamber, and
croscopes may also be desirable.
funnel-shaped collection areas at the bottom of the test
chamber where lubricant is collected for recirculation could
6.8 For all measurement and characterization methods, the
help keep third-body particles in circulation.
cleaning methods of ISO 14242-2 shall be used.
7.3.5 The orbital bearing hip wear simulator has an advan-
6.9 The serum protein content shall be the same as required
tage in the third-body wear evaluation, because the acetabular
by ISO 14242-1 and ISO 14242-3. If other serum protein
component(s) can be below the femoral component(s), letting
content is used it shall be justified.
gravity help keep the third-body particles in the area of the
articulation. However, it can also be argued that gravity
7. High Demand Features
keeping third-body particles permanently in the area of articu-
7.1 There may not be a single “worst case” high demand
lation would not be representative of an actual THA.
feature. Different high demand modes could possibly interact
7.4 Changing Load Parameters:
with each other to make the wear worse than would occur by
7.4.1 Higher demand tests may require some higher loads
the individual high demand f
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F3047M − 15 F3047M − 23
Standard Guide for
High Demand Hip Simulator Wear Testing of Hard-on-
hardHard-on-Hard Articulations
This standard is issued under the fixed designation F3047M; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 The objective of this guide is to advise researchers on the possible high demand wear test features that should be included in
evaluation of hard on hard hard-on-hard articulations. This guide makes suggestions of what for high demand test features that may
need to be added to an overall high demand wear test regime. Device articulating components manufactured from other metallic
alloys, ceramics, or with coated or elementally modified surfaces without significant clinical use could possibly be evaluated with
this guide. However, such materials may include risks and failure mechanisms whichthat are not adressedaddressed in this guide.
1.2 Hard-on-hard hip bearing systems include metal-on-metal, ceramic-on-ceramic, metal-on-metal (for example, Specifications
F75, F799, and F1537; ISO 5832-4, ISO 5832-12), ceramic-on-ceramic (for example, ISO 6474-1, ISO 6474-2, ISO 13356),
ceramic-on-metal, or any other bearing systems where both the head and cup components have high surface hardness. An argument
has been made that the hard-on-hard THR articulation may be better for younger, more active patients. These younger patients may
be more physically fit and expect to be able to perform more energetic activities. Consequently, new designs of hard-on-hard THR
articulations may have some implantations subjected to more demanding and longer wear performance requirements.
1.3 Total Hip Replacement (THR) with metal-on-metal articulations have been used clinically for more than 50 years (1, 2). Early
designs had mixed clinical results. Eventually they were eclipsed by THR systems using metal on polyethylene metal-on-
polyethylene articulations. In the 1990s the metal-on-metal articulation again became popular with more modern designs (3),
including surface replacement.
1.4 In the 1970s the first ceramic-on-ceramic THR articulations were used. In general, the early results were not satisfactory (4,
5). Improvement in alumina, and new designs in the 1990s improved the results for ceramic-on-ceramic articulations (6).
1.5 The values stated in SI units are to be regarded as the standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and healthsafety, health, and environmental practices and determine
the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
This guide is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee F04.22
on Arthroplasty.
Current edition approved March 15, 2015June 1, 2023. Published May 2015June 2023. Originally approved in 2015. Last previous edition approved in 2015 as
F3047M – 15. DOI: 10.1520/F3047M-15.10.1520/F3047M-23.
The boldface numbers in parentheses refer to the list of references at the end of this standard.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3047M − 23
2. Referenced Documents
2.1 ASTM Standards:
F75 Specification for Cobalt-28 Chromium-6 Molybdenum Alloy Castings and Casting Alloy for Surgical Implants (UNS
R30075)
F86 Practice for Surface Preparation and Marking of Metallic Surgical Implants
F561 Practice for Retrieval and Analysis of Medical Devices, and Associated Tissues and Fluids
F799 Specification for Cobalt-28 Chromium-6 Molybdenum Alloy Forgings for Surgical Implants (UNS R31537, R31538,
R31539)
F1537 Specification for Wrought Cobalt-28Chromium-6Molybdenum Alloys for Surgical Implants (UNS R31537, UNS
R31538, and UNS R31539)
F1814 Guide for Evaluating Modular Hip and Knee Joint Components
F1820 Test Method for Determining the Forces for Disassembly of Modular Acetabular Devices
F1877 Practice for Characterization of Particles
F2033 Specification for Total Hip Joint Prosthesis and Hip Endoprosthesis Bearing Surfaces Made of Metallic, Ceramic, and
Polymeric Materials
F3018 Guide for Assessment of Hard-on-Hard Articulation Total Hip Replacement and Hip Resurfacing Arthroplasty Devices
2.2 ISO Standards:
ISO 5832-4 Implants for Surgery—Metallic Materials—Part 4: Cobalt-Chromium-Molybdenum Casting Alloy
ISO 5832-12 Implants for Surgery—Metallic Materials—Part 12: Wrought Cobalt-Chromium-Molybdenum Alloy
ISO 6474-1 Implants for Surgery—Ceramic Materials—Part 1: Ceramic Materials Based on High Purity Alumina
ISO 6474-2 Implants for Surgery—Ceramic Materials—Part 2: Composite Materials Based on a High-Purity Alumina Matrix
with Zirconia Reinforcement
ISO 7206-2 Implants for Surgery—Partial and Total Hip Joint Prostheses—Part 2: Articulating Surfaces Made of Metallic,
Ceramic and Plastics Materials
ISO 13356 Implants for Surgery—Ceramic Materials Based on Yttria-Stabilized Tetragonal Zirconia (Y-TZP)
ISO 14242-1 Implants for Surgery—Wear of Total Hip-Joint Prostheses. Part 1: Loading and Displacement Parameters for
Wear-Testing Machines and Corresponding Environmental Conditions for Test
ISO 14242-2 Implants for Surgery—Wear of Total Hip-Joint Prostheses. Part 2: Methods of Measurement
ISO 14242-3:200914242-3 Implants for Surgery—Wear of Total Hip-Joint Prostheses—Part 3: Loading and Displacement
Parameters for Orbital Bearing Type Wear Testing Machines and Corresponding Environmental Conditions for Test
ISO 14242-4 Implants for Surgery—Wear of Total Hip-Joint Prostheses—Part 4: Testing Hip Prostheses Under Variations in
Component Positioning Which Results in Direct Edge Loading
ISO 17853 Wear of Implant Materials—Polymer and Metal Wear Particles—Isolation, Characterization and
QuantificationParticles—Isolation and Characterization
3. Terminology
3.1 Definitions:
3.1.1 acetabular liner—portion of the modular acetabular device with an internal hemispherical socket intended to articulate with
the head of a femoral prosthesis. The external geometry of this component interfaces with the acetabular shell through a locking
mechanism which may be integral to the design of the liner and shell or may rely upon additional components (for example, metal
ring, screws, and so forth).
3.1.2 acetabular shell—the metallic external, hollow structure that provides additional mechanical support or reinforcement for
an acetabular liner and whose external features interface directly with the bones of the pelvic socket (for example, through bone
cement, intimate press-fit, coatings for attachment to bone cement or tissue, integral screw threads, anchoring screws, pegs, and
so forth). The acetabular shell may be solid or contain holes for fixation to the pelvis or attachment of instrumentation.
3.1.3 acetabular liner/shell angle—the angle between the polar axis of the acetabular articulating surface and the horizontal (see
ISO 14242 Part 1 paragraph 7.4).
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
F3047M − 23
3.1.3 alloy fabricated form—the raw material form of the metallic alloy (such as Specifications F75, F799, and F1537; ISO
5832-4, ISO 5832-12) and any processing techniques (such as Practice F86, Specification F2033, and ISO 7206-2) used to fabricate
the final form of the implant.
3.1.4 breakaway wear—a ‘higher’ unexpected wear rate that follows a period of steady-state wear as illustrated in Fig. 2.
3.1.5 breakaway wear with recovery—a breakaway wear rate that returns to the lower steady state steady-state wear rates. The
breakaway/recovery phenomenon can be a single event or as multiple ‘episodic’ events during the otherwise steady-state
conditions as illustrated in Fig. 2.
3.1.6 ceramic-on-ceramic hip prosthesis—a device intended to replace a human hip joint in which the ball and cup articulating
surfaces are composed of high purity alumina or alumina matrix composite ceramics. ceramics (such as ISO 6474-1, ISO 6474-2,
and ISO 13356). The ball is attached to an intramedullary femoral stem. Device articulating components manufactured from other
ceramic materials or with coated or elementally modified surfaces may have special concerns which are not addressed in the scope
of this guide.
3.1.7 contact patch edge to rim (CPER) distance—for a given acetabular liner orientation the arc distance between the edge of a
calculated Hertzian contact area caused by a 3 kN joint reaction force and the last portion of articulating surface on the acetabular
liner as illustrated inliner. See Fig. 2 Fig. 1of Guide F3018.
3.1.8 coordinate measuring machine (CMM)—an automated system that is capable of making and recording measurements in
three dimensions with high precision in a controlled volume of space.
3.1.9 cup articular arc angle—the angle subtended by the articular surface of the acetabular component. It can be determined with
a computer aided design (CAD) system or manual measurements. With the head placed in the acetabular liner, it is the minimum
angle in a plane bisecting the head and the liner, formed by the last contact points between the bearing surfaces and the rotational
center of the head. It will be 180° or less. It is illustrated in Fig. 2.
3.1.10 cup inclination angle—the angle between the Superior-Inferior axis of the patient and the radiographic projection of the
acetabular axis (or polar axis of the cup) as measured on an A/P pelvic radiograph.
FIG. 1 Illustration of Cup Articular Arc Angle
F3047M − 23
FIG. 2 Different Modes/Phases of Wear Illustrated Schematically
3.1.11 dwell duration—the length of time that a wear test is paused in a test mode in order to evaluate the effect of periodically
stopping and starting the hip simulator articulation.
3.1.12 head to cup radial clearance—the radius of the cup bearing articular surface minus the radius of the head articular surface.
3.1.13 lubricant film—a fluid film trapped between the articulating surfaces of a hip joint that helps limit direct contact between
the articulating surfaces.
3.1.14 metal-on-metal hip prosthesis—a device intended to replace a human hip joint in which the ball and liner articulating
surfaces are often composed of high carbon version of Co28Cr6Mo cobalt alloy. The ball may be attached to an intramedullary
stem or a surface cover for the femoral head.
3.1.15 runaway wear—an initial high wear rate, that shows no sign of achieving a lower steady-state wear rate as illustrated in
Fig. 2.
3.1.16 run-in wear—wear that occurs when the components are first implanted in-vivo, in vivo, or during the initial phase of an
in-vitro in vitro hip simulator test as illustrated in Fig. 2. During this period, wear rates are typically higher than during steady-state
as the head and cup wear into conformity with each other and any initially contacting surface asperities or form errors are worn
away. In hip simulator wear tests, the run in run-in phase is often considered to be about 1 million cycles. The transition to
steady-state wear can be estimated graphically from the plot of total wear vs.versus number of cycles.
3.1.17 serum protein content—the concentration of protein molecules present in serum, usually expressed in grams per liter. The
value is usually supplied by the commercial source for the serum.
3.1.18 steady-state wear—wear rates that occur after a transient run-in wear period as illustrated in Fig. 2. Typically, the
F3047M − 23
steady-state wear rate is less than the run-in wear rate. In hip simulator wear tests the steady state tests, the steady-state rate
typically is reached after 1 million cycles and above.or more cycles.
3.1.19 third body third-body wear—the increased wear that occurs due to particle(s) not permanently attached to the articulating
surfaces being present in the articulation. The source of particle(s) can be external to the articulating surfaces or comingcome from
the articulating surfaces.
3.1.20 volumetric wear rate—the rate of material volume lost from both articulating surfaces.
4. Summary of PracticeGuide
4.1 A conventional hip simulator wear test should be performed according to ISO 14242 Part 1 or Part 3 for five 14242-1 or ISO
14242-3 for five (5) million cycles. This will be used as a basis for comparison of the results of any high demand test regime. Any
high demand wear test regime should use the ISO 14242 Part 1 or Part 3 standard ISO 14242-1 or ISO 14242-3 as the starting
point and high demand parameters should be made as modifications to that standard. The ISO 14242 Part 3 standard ISO 14242-3
may not be suitable for high demand wear tests that require modification of the articulating motion, because the motion cycle is
built into the test machine hardware and can’t be modified.
4.2 The high demand wear test can be performed as a continuation of the conventional ISO 14242 Part 1 14242-1 or ISO 14242-3
test or run as a separate test. High demand test features will be added to the high demand wear test and justified as clinically
relevant. This will require an understanding of the potential interactions of the possible high demand modes which would indicate
a series of shorter duration tests. A final high demand test(s) for the preclinical evaluation of a device shall include a test protocol
of at least 5 million cycles. These high demand wear test cycles will be in addition to the conventional 5 million cycles of wear
testing.
5. Significance and Use
5.1 The current hip simulator wear test standards (ISO 14242 Part 1 or Part 3)14242-1 or ISO 14242-3) stipulate only one load
wave form waveform and one set of articulation motions. There is a need for more versatile and rigorous wear test regimes, but
the knowledge of what represents realistic high demand wear test features is limited. More research is clearly needed before a
standard can be written that defines what a representative high demand wear test should include. include can be written. The
objective of this guide is to advise researchers on the possible high demand wear test features that should be included in evaluation
of hard-on-hard articulations.
5.2 This guide makes suggestions of what high demand test features may need to be added to an overall high demand wear test
regime. The features described here are not meant to be all inclusive. Based on current knowledge they appear to be relevant to
adverse conditions that can occur in clinical use.
5.3 All the test features, both conventional and high demand, could have interactive effects on the wear of the components.
6. Test Samples
6.1 The materials and articulating geometry of the hard-on-hard system should be representative of the system intended for clinical
use. The acetabular components must have the same geometry as the acetabular system intended for clinical use because the
stiffness of the acetabular system could affect the response to loads and motions at the articulating surface.
6.2 The test parts should receive all of the processing that is intended for product intended for clinical use, including sterilization.
There is no literature reporting any detrimental effects of gamma sterilization or any other sterilization methods used for orthopedic
devices on the physical or chemical properties of metallic alloys. However, it may be advisable to sterilize everything prior to
definitive tests for preclinical evaluation to make all parts as close to the clinical product as possible. Coatings on non-articulating
surfaces of the test parts could create problems with the handling of the parts and weight loss measurements during testing. It may
be necessary to have test parts without the non-articulating surface coatings. However, any thermal processing the test parts would
receive as part of any coating process should still be performed. Particulate based Particulate-based coating could be a source for
third body third-body wear particles, but random particle loss interferes with the repeatability of the test. Consideration should also
be given to using particulate from the coatings as controlled third body third-body particle sources.
F3047M − 23
6.3 No preconditioning is required for the test samples other than careful handling to assureensure that they remain clean and free
of contamination prior to start of testing.
6.4 The diameter and acetabular sizing must be justified as worst case for the wear tests. There are many possible factors that could
make a hard-on-hard couple a “Worst Case”.“worst case.” The diameter of the articulation, head to cup head-to-cup radial
clearance, the thickness of material in the liner and the shell, the design of modularity of the liner and the shell, or the sphericity
of the articulations could all potentially cause a “Worst Case” create a “worst case” combination for wear. These factors and more
should be considered in justifying a “Worst Case”.“worst case.”
6.5 The usual small amount of material lost in hard-on-hard wear tests combined with the larger mass of the components may
make weight loss characterization of wear according to ISO 14242 Part 2 14242-2 more difficult. Another means of measuring loss
of material from both the convex and concave surfaces of the metal-on-metal articulation is by measuring the change in the surface
geometries. For this measurement method, both articulating surfaces must be measured with enough precision before testing to
provide a baseline for estimating the volume of material lost from the surfaces due to the tests. This shall require a high precision
coordinate measuring machine (CMM) or other high precision measurement devices. The volumetric measurement does have one
advantage over the weight loss method, because it can indicate the distribution of wear on a surface. Both methods require precise
techniques that shall have validated procedures before they are used in an actual wear test.
6.6 The geometry of both articulating surfaces should be characterized as to their original geometry and surface finish. The same
techniques should be used to characterize both surfaces intermittently during testing and after completion of testing. These
measurement results can be used to estimate the amount of material lost, but the alternate weight loss method should be used as
a validation method for the volumetric measurement by making the alternatealternative weight loss measurements at the beginning
and the end of the tests.
6.7 Additional characterization of the surfaces in a scanning electron microscope or three dimensional three-dimensional digital
optical microscopes may also be desirable.
6.8 For all measurement and characterization methods, the cleaning methods of ISO 14242 Part 2 14242-2 shall be used.
6.9 The serum protein content shall be the same as required by ISO 14242 Part 1 and ISO 14242 Part 3. 14242-1 and ISO 14242-3.
If other serum protein content is used it shall be justified.
7. High Demand Features
7.1 There may not be a single “worst case” high demand feature. Different high demand modes could possibly interact with each
other to make the wear worse than would occur by the individual high demand feature. Investigation of the possible interactions
should be considered.
7.2 Acetabular Liner/ShellCup Inclination Angle:
7.2.1 Callanan et al. showed that for metal on polyethylene metal-on-polyethylene THR systems the acetabular component
abductor angle placement can be as much as 15º off optimal and still survive long term (7). With this cup positioning, the main
load axis is 15º closer to the equator of the acetabular component, reducing the effective contact area and consequently increasing
the contact stresses on the articulating surfaces. In fact, for metal-on-metal THRs, there are reports in the literature that higher
liner/shell cup inclination angles of the load axis in relation to optimal position do cause increases in wear (8, 9). There have been
reports (1010-12, 11, 12) of acetabular cupscup angles as high as 60º and 65º.
7.2.2 The CPER distance shall be determined for all cup inclination angles tested. The actual distance between the edge of the
contact patch and the end of the articulating surface of the cup shall be estimated after completion of the tests.
7.2.3 Since the acetabular liner/shell cup inclination angle is fixed for the life of the implant clinically, any high demand wear test
should have that liner/shell angle cup inclination angle fixed throughout the entire test. The choice of the high angle shall be
justified.
F3047M − 23
7.3 Third Body Third-Body Particles:
7.3.1 As a result of the intra-operativeintraoperative procedure it is possible that the joint space could have small bone chips or
particles of bone cement contamination. These particles could cause damage to the articulating surfaces (13, 14). There could be
other possible sources of ceramic particles such as hydroxyapatite (15) or zirconia radio-pacifierszirconia-based radio-opacifiers
from bone cement. Metallic particulateparticulates of titanium and CoCrMo could come from such sources as,as neck impingement
(16, 17)), porous coatings, or tribocorrosion (1818-20, 19, 20). The presence of such types of third body third-body particulate may
be of limited duration because such particles could take time to form, then possibly be broken down, and eventually be removed
from the joint capsule.
7.3.2 There clearly needs to be a small particle size in order for there to be a potential that the particle could be entrapped between
the articulating surfaces and cause damage. Trying to standardize on small bone particles is not practical.
7.3.3 The time and rate of 3rd body particle removalthird-body particle replenishment should be justified.
7.3.4 Care must be taken during the portion of the test with the third body third-body particles that they remain suspended in the
lubrication medium as much as possible to keep availability to the articulating surface high. Additional agitation of the lubricant,
limiting crevice and corners in the test chamber, and funnel shaped funnel-shaped collection areas at the bottom of the test chamber
where lubricant is collected for recirculation could help keep third body third-body particles in circulation.
7.3.5 The orbital bearing hip wear simulator has an advantage in the third body third-body wear evaluation, because the acetabular
component(s) can be below the femoral component(s), letting gravity help keep the third body third-body particles in the area of
the articulation. However, it can also be argued that gravity keeping 3rd body third-body particles permanently in the area of
articulation would not be representative of an actual THA.
7.4 Changing Load Parameters:
7.4.1 Higher demand tests may require some higher loads that could be representative of younger, more active patients. There is
literature that associates higher wear rates with higher loads (21, 22). These high loads could also come from activities with higher
cyclic frequencies. However, even those patients are not always in a higher demand activity. Consequently, a spectrum of different
higher load peaks might be included in the ISO 14242 Part 1 or Part 3 load wave forms 14242-1 or ISO 14242-3 waveforms or
even replace the standard wave forms.waveforms.
7.4.2 The number of cycles of each type of wave form, waveform, the cyclic frequency, and the amplitude of the peaks of the
higher demand wave forms waveforms shall be justified.
7.5 Stop-Dwell-Start (Stiction):
7.5.1 Hip simulator tests are normally run continuously. However, patients with implants in activities of daily living usually walk
relatively short distances,distances before stopping or performing another activity. In a study of activities of normal, healthy hip
patients on a typical day (23), the patients averaged walking periods of 10 seconds 10 s before pausing, sitting down, or changing
to a different activity like stair climbing. The highest frequency durations were 2 to 5 seco
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...